Effect of heat treatment temperature on the Pt3Co binary metal catalysts for oxygen reduced reaction and DFT calculations
-
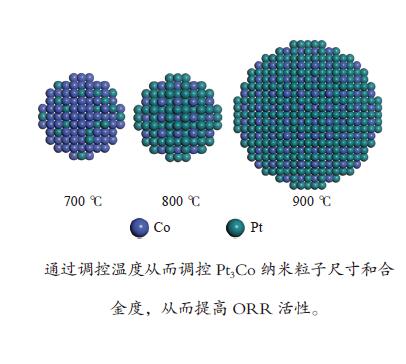
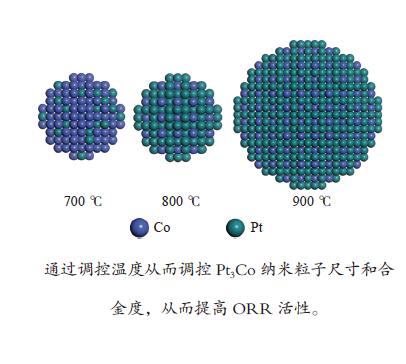
摘要: 制备低成本、高活性、高稳定性的铂(Pt)基氧还原反应(ORR)催化剂是质子交换燃料电池(PEMFC)大规模商业化应用的关键。以钴(Co)等非贵金属与Pt掺杂制备二元合金PtM催化剂不仅可以减少Pt用量,还可以获得高于Pt金属催化剂的ORR催化活性和稳定性。本研究使用浸渍还原法制备碳载铂钴ORR催化剂,通过控制热处理还原温度来控制纳米颗粒的结构、晶相、尺寸等,从而改善催化剂的ORR性能。XRD、TEM和电化学分析结果综合表明,热处理温度对纳米颗粒合金度和平均粒径有显著的影响,平均粒径和合金度随着热处理温度升高而增大。通过控制热处理温度可以获得粒径与合金度之间的最优值从而提高催化剂氧还原活性,实验表明,800 ℃是低粒径和高合金度的平衡点,在所有制备的催化剂中有最高的质量活性(0.41 A/mgPt)和稳定性。进一步的密度泛函理论(DFT)计算表明高合金度的Pt3Co结构表面可以降低速控步反应势垒,提高ORR活性。Abstract: Synthesis of low-cost, high-activity and high-stability Pt-based catalysts is of great importance to the large commercialization of proton exchange membrane fuel cell (PEMFC). Doping non-precious metals such as cobalt (Co) with Pt is attractive due to the reduced depletion of Pt and, more importantly, the enhanced activity on the oxygen reduction reaction (ORR) compared with pure Pt. In this work, carbon-supported platinum-cobalt nanoparticles (NPs) were prepared by the impregnation reduction method for the ORR catalyst. By changing the heat treatment temperature, the structure, the crystal phase and the size of the Pt3Co nanoparticles could be controlled. TEM and XRD characterizations show that larger size NPs with higher alloying degree are obtained at higher temperature. The electrochemical results demonstrate that the Pt3Co NPs at 800 ℃ have the highest mass activity (0.41 A/mgPt) and the best stability among all the samples due to their lower particle size and higher alloying degree. Further Density functional theory (DFT) calculation shows that the surface of the Pt3Co structure with high alloying degree can reduce the rate-determining step barrier and improve the ORR activity.
-
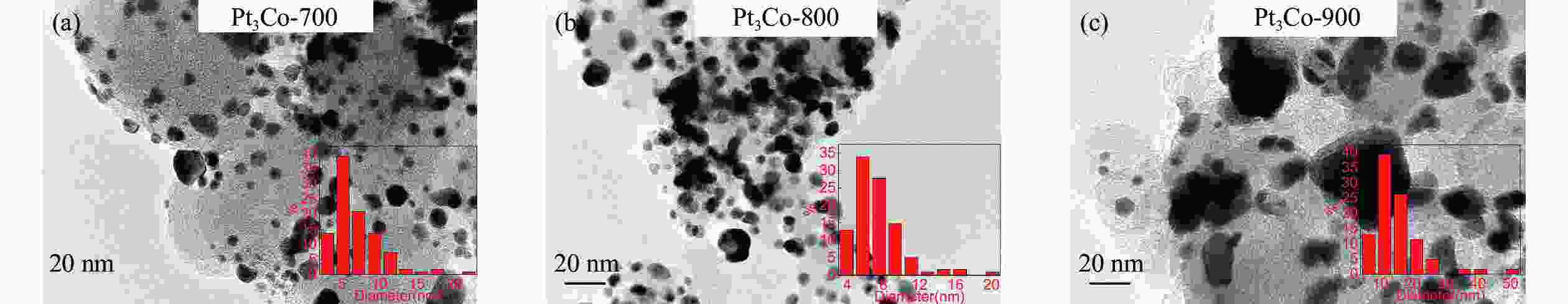
图 2 Pt3Co-700、Pt3Co-800和Pt3Co-900的高分辨TEM照片 (a)、(d)和(g),元素线性扫描图(b)、(e)和(h)(红色为 Pt 分布曲线,绿色为 Co 分布曲线)和元素分布图(c)、(f)和(i)
Figure 2 Characterization of Pt3Co-700, Pt3Co-800 and Pt3Co-900 (a), (d) and (g): high-resolution TEM image; (b), (e) and (h): linear scan of a single particle, red line is the Pt atom distribution curve, green is the Co atom distribution curve; (c), (f) and (i): the nanoparticle element distribution diagram
图 3 (a)Pt3Co-700、Pt3Co-800和Pt3Co-900的XRD谱图,蓝色线为Pt3Co的标准卡片(ICSD #102624)衍射峰,(b)为图(a)的XRD谱图在36°−43°附近的局部放大图,(c)为Pt3Co-700、Pt3Co-800和Pt3Co-900的Pt 4f XPS谱图,(d)为Co 2p XPS谱图
Figure 3 (a) XRD patterns of Pt3Co-700, Pt3Co-800 and Pt3Co-900, the blue line is the diffraction peak of Pt3Co standard card (ICSD #102624), (b) is the partial enlarged view XRD curve of Figure (a) around 37°− 44°, (c) is Pt 4f XPS spectra of Pt3Co-700, Pt3Co-800 and Pt3Co-900, (d) is Co 2p XPS spectra
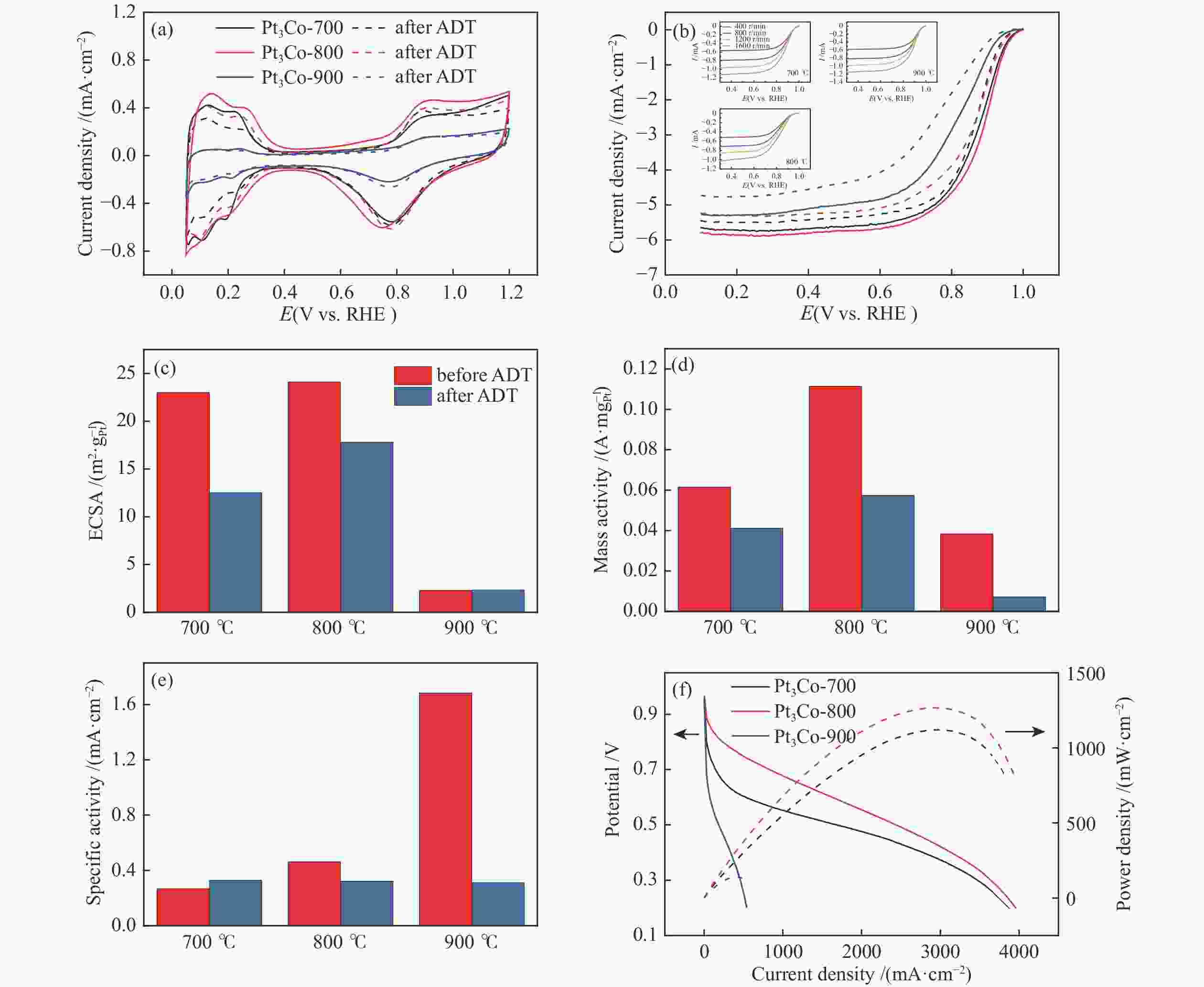
图 4 Pt3Co-700、Pt3Co-800和Pt3Co-900 ADT前后的(a)CV曲线,(b)LSV曲线,内嵌有Pt3Co-700、Pt3Co-800和Pt3Co-900在不同转速下LSV曲线,(c)ECSA,(d)0.9 V vs. RHE下质量活性,(e)0.9 V vs. RHE下本征活性,(f)极化曲线,实线为电压随电流密度变化的极化曲线,虚线为功率随电流密度变化的功率曲线
Figure 4 For Pt3Co-700, Pt3Co-800 and Pt3Co-900 (a) CV curve, (b) LSV curve, embedded with Pt3Co-700, Pt3Co-800 and Pt3Co-900 LSV curve at different rotation rates, (c) ECSA, (d) 0.9 V vs. RHE mass activity and (e) specific surface area activity before and after the ADT (f) Polarization curve, which the solid line is the polarization curve of voltage changing with current density, and the dashed line is the power curve of power changing with current density
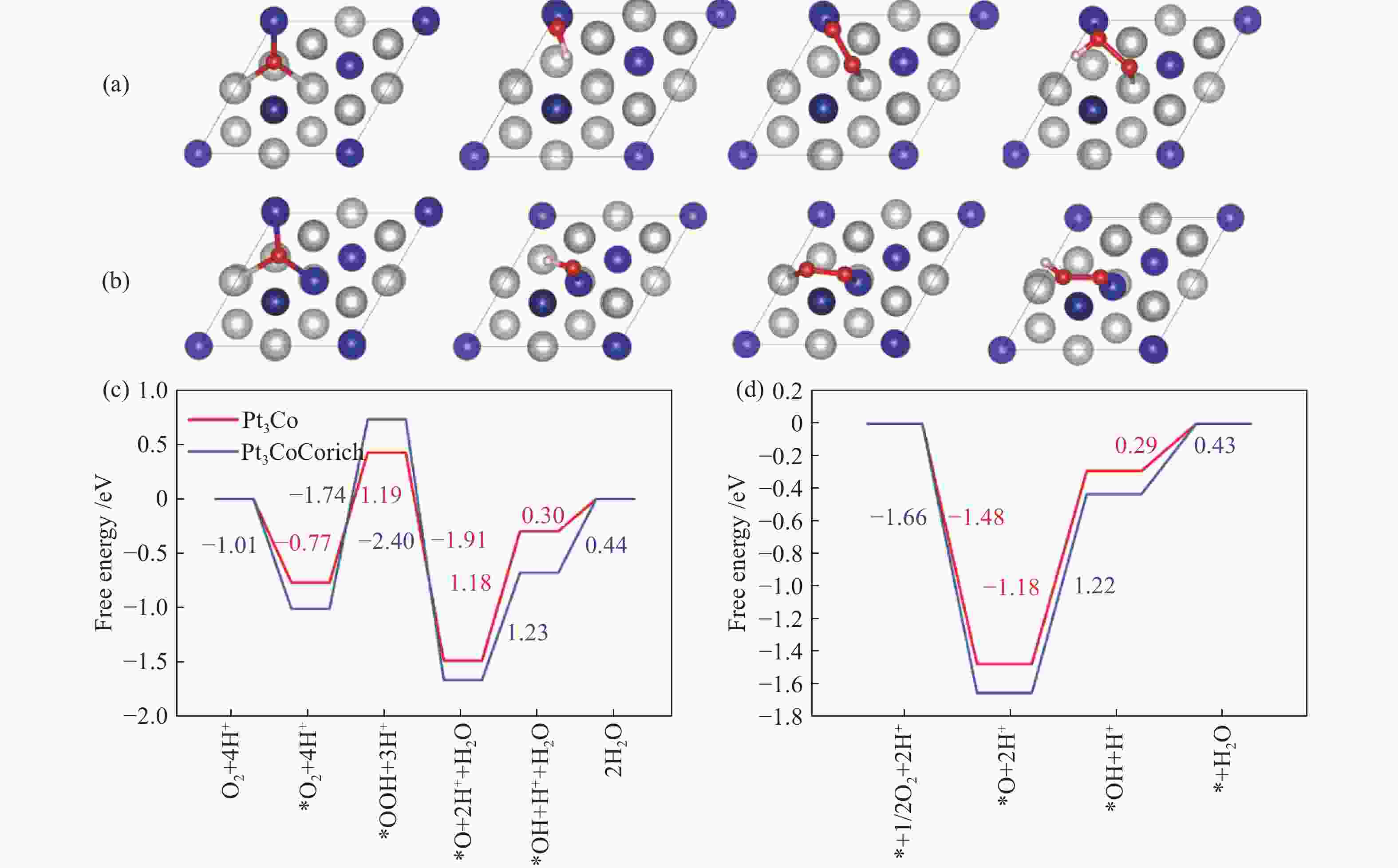
图 5 (a)Pt3Co表面O、OH、OO和OOH吸附结构示意图;(b)Pt3CoCorich表面O、OH、OO和OOH吸附结构示意图其中银色为Pt原子,蓝色为Co原子,红色为O原子,白色为H原子; (c)为Pt3Co和Pt3CoCorich在U = 1.23 V下associative机理反应自由能对比;(d)为dissociative机理反应自由能对比
Figure 5 (a) Schematic diagram of the adsorption structure of O, OH, OO and OOH on the surface of Pt3Co, (b) Schematic diagram of the adsorption structure of O, OH, OO and OOH on the surface of Pt3CoCorich. Silver ball corresponds to Pt atom, blue ball corresponds to Co atom, red ball corresponds to O atom , and white ball corresponds to H atom, (c) is the comparison of the associative mechanism reaction free energy diagram of Pt3Co and Pt3CoCorich at U = 1.23 V, (d) is the comparison of the dissociative mechanism reaction free energy diagram
表 1 ADT前后LSV曲线半波电位
Table 1 Half wave potential before and after ADT test.
Pt3Co-700 before ADT Pt3Co-700 after ADT Pt3Co-800 before ADT Pt3Co-800 after ADT Pt3Co-900 before ADT Pt3Co-800 after ADT Half wave potential
(V vs. RHE)0.89 0.86 0.89 0.86 0.81 0.76 表 2 ORR反应机理步骤
Table 2 Reaction steps of ORR mechanisms
ORR mechanism Reaction steps Associative O2 (g) + * →*O2 *O2 + H+ + e− →*OOH *OOH + H+ + e− → H2O(l) + *O *O + H+ + e−→ *OH *OH + H+ + e−→ H2O(l) + * Dissociative 1/2O2 (g) + * →*O *O + H+ + e−→*OH *OH + H+ + e−→ H2O(l) + * -
[1] SHAO M, CHANG Q, DODELET, CHENITZ R. Recent advances in electrocatalysts for oxygen reduction reaction[J]. Chem Rev,2016,116(6):3594−3657. doi: 10.1021/acs.chemrev.5b00462 [2] JIA Q, CALDWELL K, STRICKLAND K, ZIEGELBAUER J M, LIU Z, YU Z, RAMAKER D E, MUKERJEE S. Improved oxygen reduction activity and durability of dealloyed PtCox catalysts for proton exchange membrane fuel cells: Strain, ligand, and particle size effects[J]. ACS Catal,2015,5(1):176−186. doi: 10.1021/cs501537n [3] YU Y, YANG W, SUN X, ZHU W, LI X, SELLMYER D J, SUN S. Monodisperse MPt (M = Fe, Co, Ni, Cu, Zn) nanoparticles prepared from a facile oleylamine reduction of metal salts[J]. Nano Lett,2014,14(5):2778−2782. doi: 10.1021/nl500776e [4] ANTOLINI E, SALGADO JRC, GIZ M J, GONZALEZ E R. Effects of geometric and electronic factors on ORR activity of carbon supported Pt-Co electrocatalysts in PEM fuel cells[J]. Int J Hydrogen Energy,2005,30(11):1213−1220. doi: 10.1016/j.ijhydene.2005.05.001 [5] LI Q, WU L, WU G, SU D, LV H, ZHANG S, ZHU W, CASIMMR A, ZHU H, GARCIA A M, SUN S. New approach to fully ordered fct-FePt nanoparticles for much enhanced electrocatalysis in acid[J]. Nano Lett,2015,15(4):2468−2473. doi: 10.1021/acs.nanolett.5b00320 [6] LI J, SHARMA S, LIU X, PAN Y, SPENDELOW J S, CHI M, JIA Y, ZHANG P, CULLEN D A, XI Z, LIN H, YIN Z, SHEN B, MUZZIO M, YU C, KIM Y S, PETERSON A A, MORE K L, SUN S. Hard-magnet L10-CoPt nanoparticles advance fuel cell catalysis[J]. Joule,2019,3(1):124−135. doi: 10.1016/j.joule.2018.09.016 [7] WANG D, XIN H L, HOVDEN R, WANG H, YU Y, MULLER D A, DISALVO F J, ABRUNA H D. Structurally ordered intermetallic platinum-cobalt core-shell nanoparticles with enhanced activity and stability as oxygen reduction electrocatalysts[J]. Nat Mater,2013,12(1):81−87. doi: 10.1038/nmat3458 [8] ZHANG B, FU G, LI Y, LIANG L, GRUNDISH N S, TANG Y, GOODENOUGH J B, CUI Z. General strategy for synthesis of ordered Pt3M intermetallics with ultrasmall particle size[J]. Angew Chem Int Ed Eng,2020,59(20):7857−7863. doi: 10.1002/anie.201916260 [9] 吕银荣, 孙维艳, 王峰. 用于直接甲醇燃料电池的高活性PtCo-CNT@TiO2复合纳米阳极催化剂[J]. 燃料化学学报,2019,47(12):1522−1528. doi: 10.3969/j.issn.0253-2409.2019.12.012LÜ Ying-rong, SUN Wei-yan, WANG Feng. Highly active PtCo-CNT@TiO2 composite nanoanode catalyst for direct methanol fuel cells[J]. J Fuel Chem Technol,2019,47(12):1522−1528. doi: 10.3969/j.issn.0253-2409.2019.12.012 [10] 赵海东, 卢珍, 刘锐, 李作鹏, 郭永. 铂银合金的制备及其对甲醇电氧化反应的催化性能[J]. 燃料化学学报,2020,48(8):1015−1024. doi: 10.3969/j.issn.0253-2409.2020.08.014ZHAO Hai-dong, LU Zhen, LIU Rui, Li Zuo-peng, Guo yong. Preparation of platinum-silver alloy nanoparticles and their catalytic performance in methanol electro-oxidation[J]. J Fuel Chem Technol,2020,48(8):1015−1024. doi: 10.3969/j.issn.0253-2409.2020.08.014 [11] 杨改秀, 王可欣, 张泽珍, 甄峰, 孙永明. 电沉积制备MnO2催化剂及其在微生物燃料电池中的应用[J]. 燃料化学学报,2020,48(7):889−896. doi: 10.3969/j.issn.0253-2409.2020.07.015YANG Gai-xiu, WANG Ke-xin, ZHANG Ze-zhen, ZHEN Feng, SUN Yong-ming. Preparation of MnO2 catalyst by electrochemical deposition and its application in the microbial fuel cells[J]. J Fuel Chem Technol,2020,48(7):889−896. doi: 10.3969/j.issn.0253-2409.2020.07.015 [12] GUO S, ZHANG S, SUN S. Tuning nanoparticle catalysis for the oxygen reduction reaction[J]. Angew Chem Int Ed Eng,2013,52(33):8526−8544. doi: 10.1002/anie.201207186 [13] CHOI D S, ROBERTSON A W, WARNER J H, KIM S Q, KIM H. Low-temperature chemical vapor deposition synthesis of Pt-Co alloyed nanoparticles with enhanced oxygen reduction reaction catalysis[J]. Adv Mater Weinheim,2016,28(33):7115−7122. doi: 10.1002/adma.201600469 [14] JUNG W S, POPOV B N. New method to synthesize highly active and durable chemically ordered fct-PtCo cathode catalyst for PEMFCs[J]. ACS Appl Mater Interfaces,2017,9(28):23679−23686. doi: 10.1021/acsami.7b04750 [15] WANG C, MARKOVIC N M, STAMENKOVIC V R. Advanced platinum alloy electrocatalysts for the oxygen reduction reaction[J]. ACS Catal,2012,2(5):891−898. doi: 10.1021/cs3000792 [16] XIA B Y, WU H B, LI N, YAN Y, LOU X W, WANG X. One-pot synthesis of Pt-Co alloy nanowire assemblies with tunable composition and enhanced electrocatalytic properties[J]. Angew Chem,2015,127(12):3868−3872. doi: 10.1002/ange.201411544 [17] KNUPP S L, LI W, PASCHOS O, MURRAY T M, SNYDER J, HALDAR P. The effect of experimental parameters on the synthesis of carbon nanotube/nanofiber supported platinum by polyol processing techniques[J]. Carbon,2008,46(10):1276−1284. doi: 10.1016/j.carbon.2008.05.007 [18] LOUKRAKPAM R, LUO J, HE T, CHEN Y, XU Z, NJOKI P N, WANJALA B N, FANG B, MOTT D, YIN J, KLAR J, POWELL B, ZHONG C. Nanoengineered PtCo and PtNi catalysts for oxygen reduction reaction: An assessment of the structural and electrocatalytic properties[J]. J Phys Chem C,2011,115(5):1682−1694. doi: 10.1021/jp109630n [19] SCHMIES H, HORNBERGER E, ANKE B, JURZINSKY T, NONG H N, DIONIGI F, KUHL S, DRNEC J, LERCH M, CREMERS C, STRASSER P. Impact of carbon support functionalization on the electrochemical stability of Pt fuel cell catalysts[J]. Chem Mater,2018,30(20):7287−7295. doi: 10.1021/acs.chemmater.8b03612 [20] CAI Y, GAO P, WANG F, ZHU H. Carbon supported chemically ordered nanoparicles with stable Pt shell and their superior catalysis toward the oxygen reduction reaction[J]. Electrochimica Acta,2017,245:924−933. doi: 10.1016/j.electacta.2017.04.173 [21] WEI C, RAO R R, PENG J, HUANG B, STEPHENS I E L, RISCH M, XU Z J, HORN Y S. Recommended practices and benchmark activity for hydrogen and oxygen electrocatalysis in water splitting and fuel cells[J]. Adv Mater Weinheim,2019,31(31):e1806296. doi: 10.1002/adma.201806296 [22] BAHN S R, JACOBSEN K W. An object-oriented scripting interface to a legacy electronic structure code[J]. Comput Sci Eng,2002,4(3):56−66. doi: 10.1109/5992.998641 [23] HAMMER B, HANSEN L B, NøRSKOV J K. Improved adsorption energetics within density-functional theory using revised Perdew-Burke-Ernzerhof functionals[J]. Phys Rev B,1999,59(11):7413−7421. doi: 10.1103/PhysRevB.59.7413 [24] SETHURAMAN V A, VAIRAVAPANDIAN D, LAFOURESSE M C, MAARK T A, KARAN N, SUN S, BERTOCCI U, PETERSON A A, STAFFORD G R, GUDURU P R. Role of elastic strain on electrocatalysis of oxygen reduction reaction on Pt[J]. J Phys Chem C,2015,119(33):19042−19052. doi: 10.1021/acs.jpcc.5b06096 [25] FROST K, KAMINSKI D, KIRWAN G, LASCARIS E, SHANKS R. Crystallinity and structure of starch using wide angle X-ray scattering[J]. Carbohydrate Polymers,2009,78(3):543−548. doi: 10.1016/j.carbpol.2009.05.018 -




 下载:
下载: