The structure and hydrogenation performance for methyl benzoate of MnOx/ZrO2-Cr2O3 catalyst
-
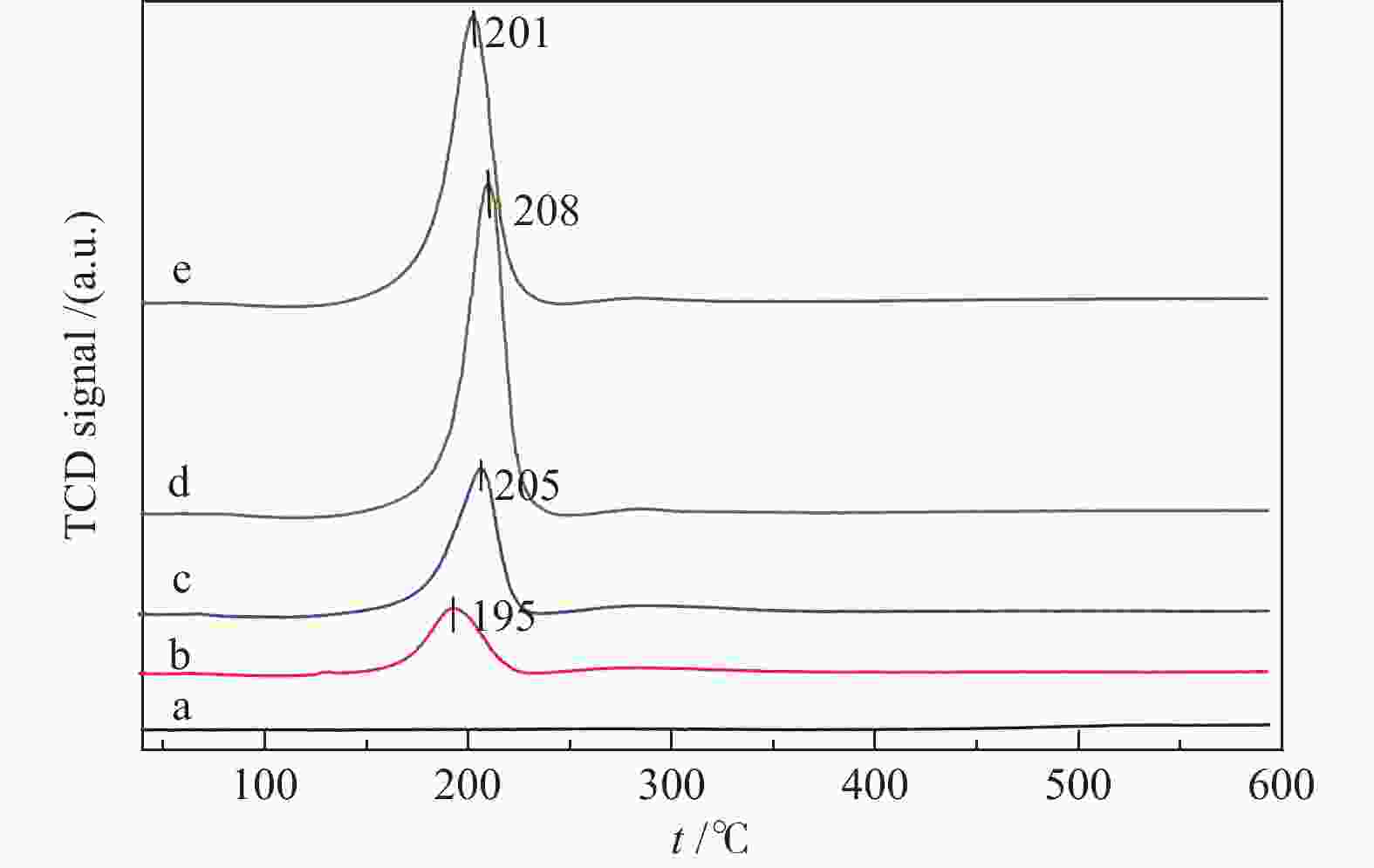
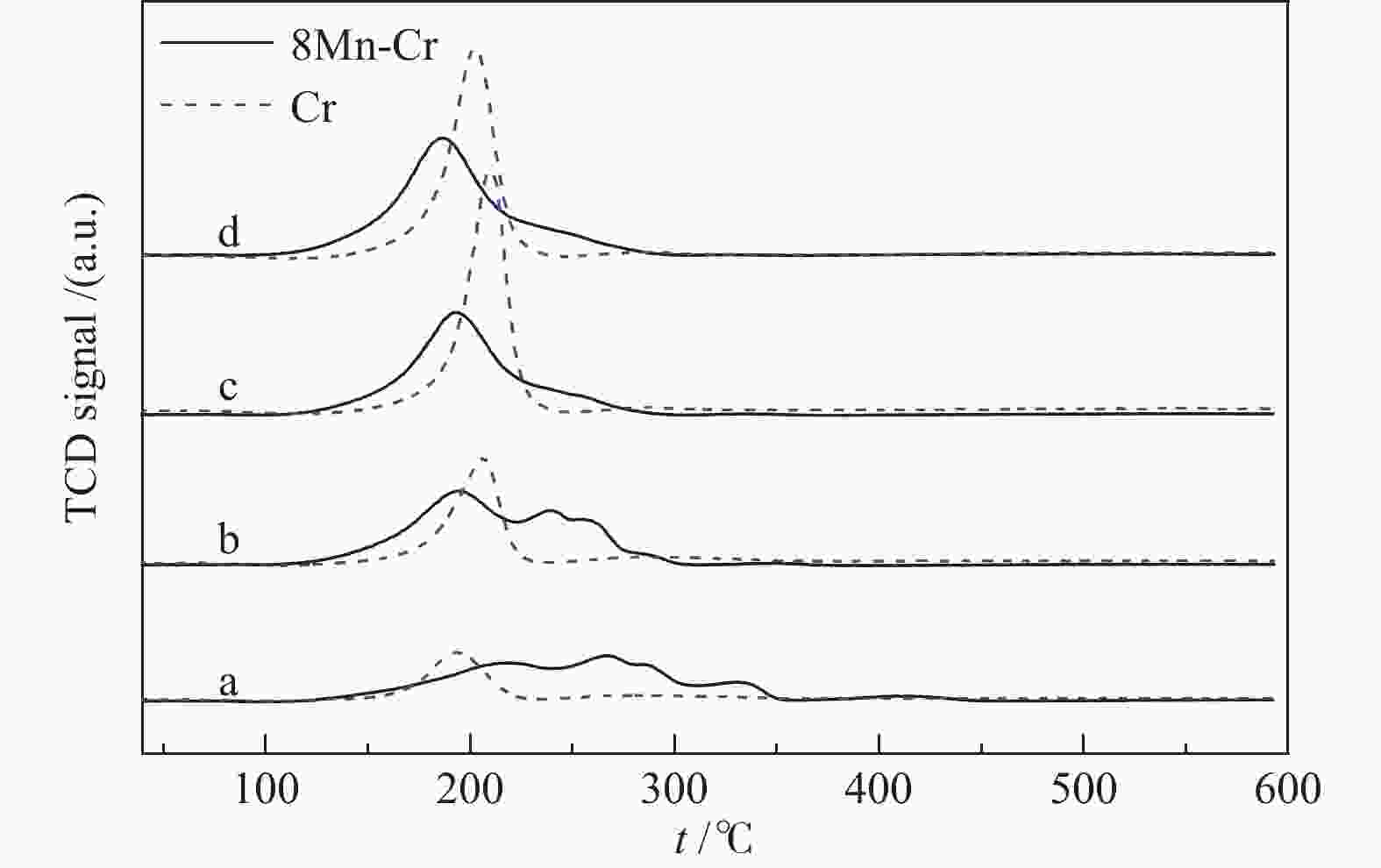
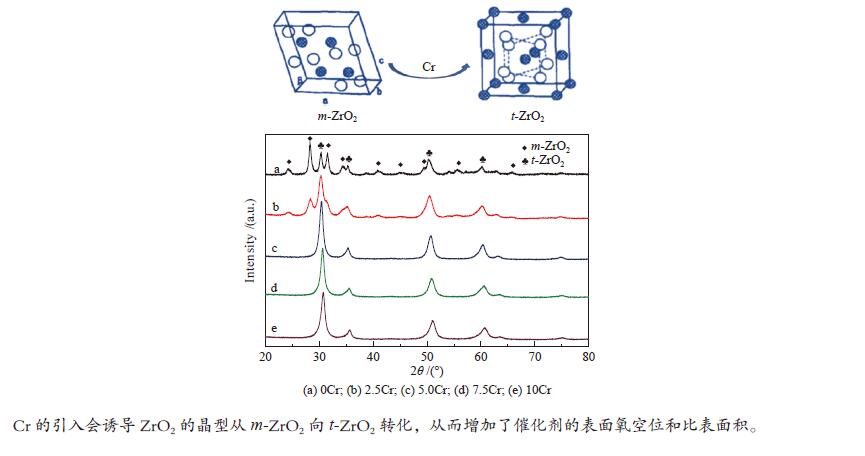
摘要: 采用并流共沉淀和浸渍法制备了MnOx/ZrO2-Cr2O3催化剂。利用X射线衍射(XRD)、N2物理吸附(BET)、氢气程序还原(H2-TPR)、X射线光电子能谱(XPS)等手段对催化剂的结构和表面性质进行了表征。结果表明,Cr的引入导致ZrO2晶型由m-ZrO2向t-ZrO2转变,随着Cr含量的增加,ZrO2-Cr2O3复合氧化物的比表面积逐渐增大,平均孔径逐渐降低,孔结构也发生变化;Mn的引入会造成ZrO2-Cr2O3复合氧化物中Cr的迁移,同时也有利于ZrO2晶型由m-ZrO2向t-ZrO2转变,从而引起催化剂比表面积、孔结构的变化;含4% Mn和2.5% Cr的MnOx/ZrO2-Cr2O3催化剂对苯甲酸甲酯加氢反应的单程转化率和选择性分别达到93.86%和86.05%。Abstract: MnOx/ZrO2-Cr2O3 catalysts were prepared by cocurrent coprecipitation and impregnation. The structure and surface properties of the catalyst were characterized by X-ray diffraction (XRD), N2 physical adsorption (BET), hydrogen temperature programmed reduction (H2-TPR), and X-ray photoelectron spectroscopy (XPS). These results showed that the addition of Cr led to the transformation of ZrO2 crystal form from m-ZrO2 to t-ZrO2. With the increase of Cr content, the BET of the composite oxide ZrO2-Cr2O3 increased gradually, the average pore size decreased and the pore structure also changed. The impregnation of Mn caused the migration of Cr in the ZrO2-Cr2O3 composite oxide, and promoted the transformation of the ZrO2 crystal form from m-ZrO2 to t-ZrO2, the BET and pore structure of the catalyst changed also. The conversion and selectivity of MnOx/ZrO2-Cr2O3 catalysts containing 4% Mn and 2.5% Cr for methyl benzoate hydrogenation were 93.86% and 86.05%, respectively.
-
Key words:
- methyl benzoate /
- benzaldehyde /
- benzyl alcohol /
- selective hydrogenation /
- catalyst
-
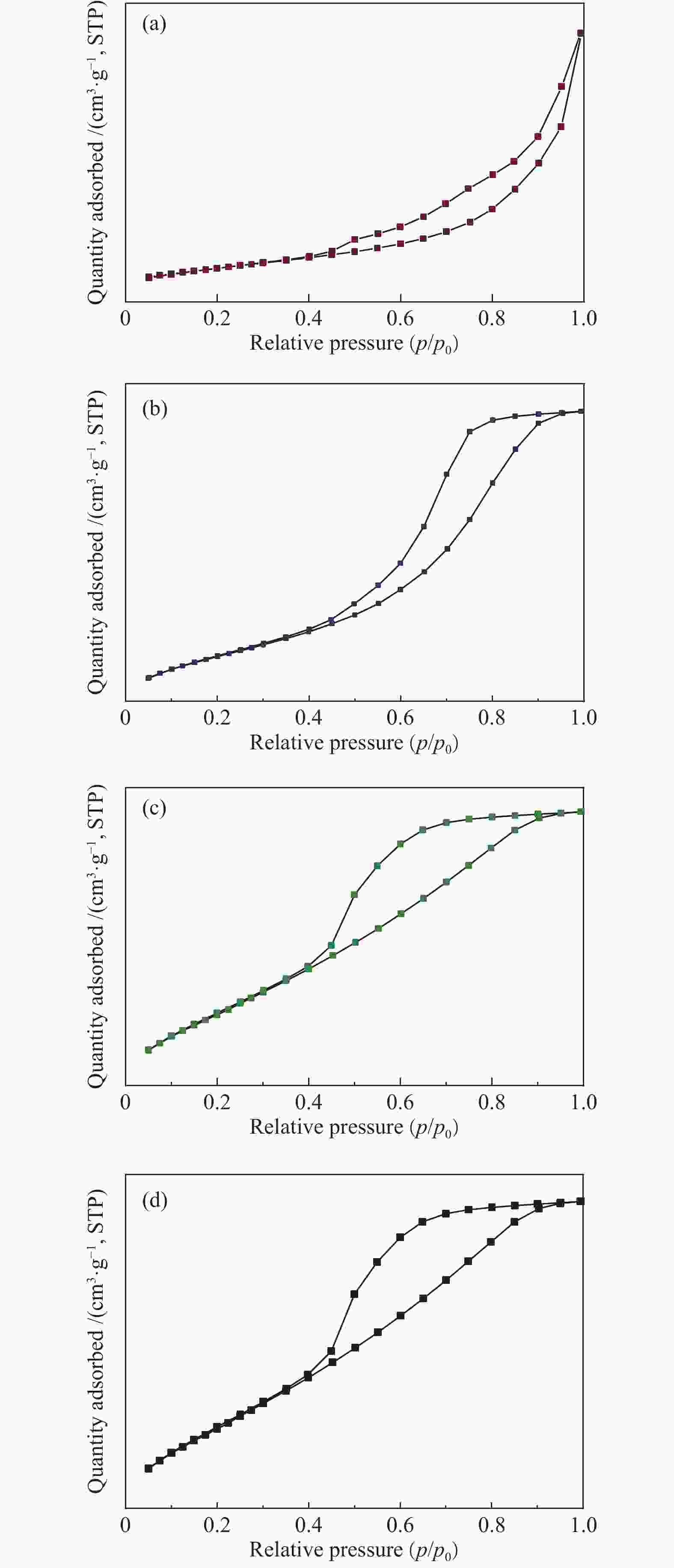
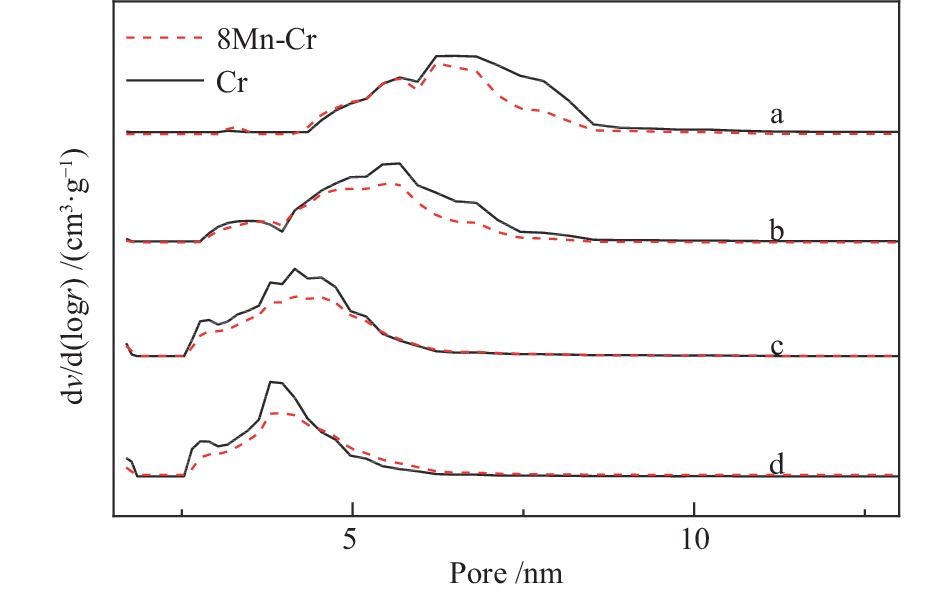
表 1 ZrO2-Cr2O3复合氧化物的孔径及比表面积
Table 1 Textual properties of ZrO2-Cr2O3
Cr content w/% SBET/(m2·g−1) v/(cm3·g−1) d/nm 0 28.6 0.097 13.7 2.5 86.1 0.201 9.3 5.0 109.1 0.194 7.1 7.5 144.1 0.182 5.0 10 135.1 0.152 4.5 表 2 bMnOx/ZrO2-aCr2O3的孔径及比表面积
Table 2 Textual properties of bMnOx/ZrO2-aCr2O3
Mn content
w/%Cr content
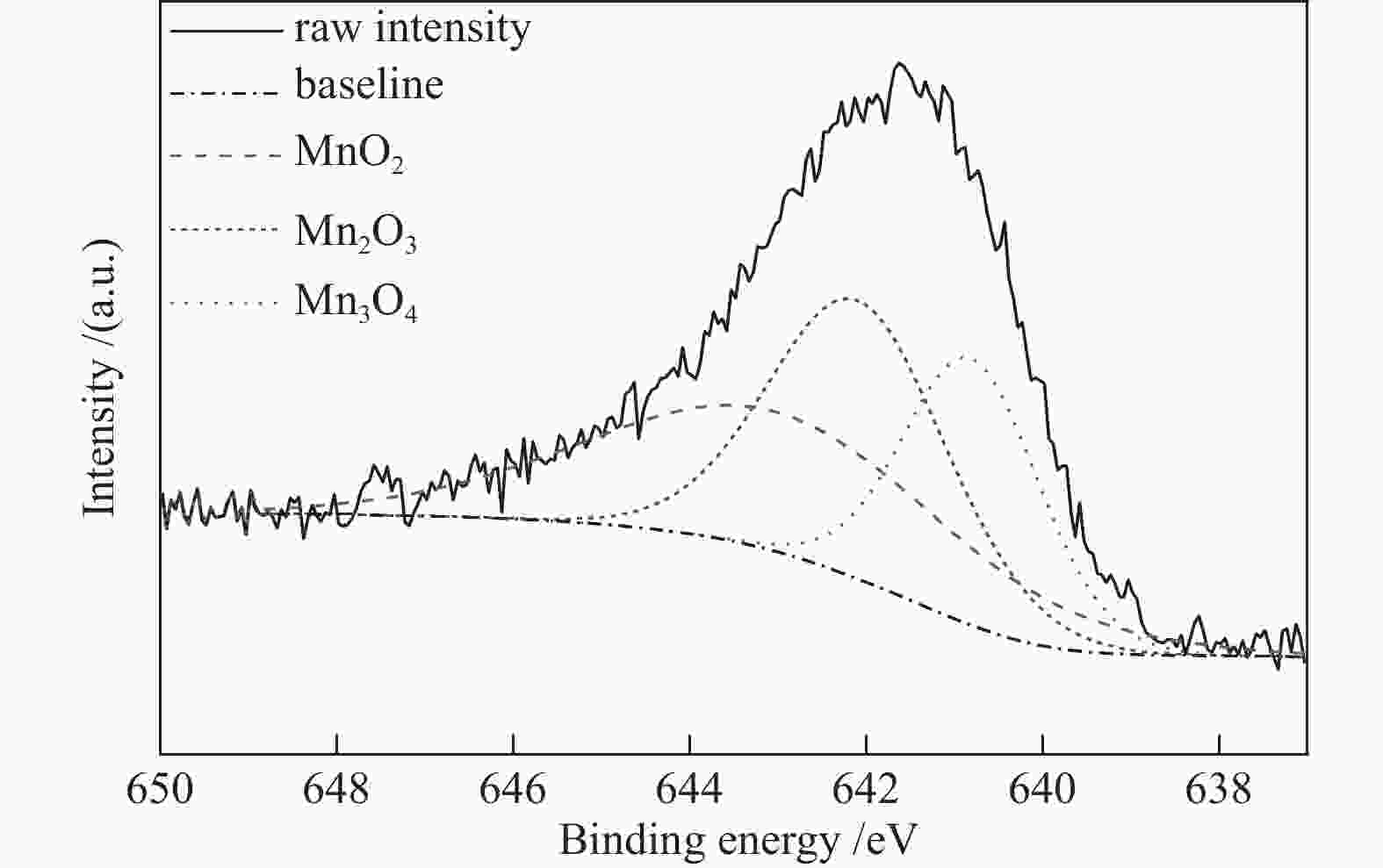
w/%SBET/(m2·g−1) v /(cm3·g−1) d/nm 0 2.5 86.1 0.20 9.3 4 2.5 82.1 0.18 8.6 8 2.5 72.2 0.16 8.9 10 2.5 63.0 0.14 8.9 8 2.5 72.2 0.16 8.9 8 5.0 84.9 0.14 7.1 8 10 87.6 0.12 5.3 表 3 bMn/2.5Cr-Zr的Mn元素结合能
Table 3 Binding energy of Mn 2p3/2 determined from XPS
Sample Mn 2p3/2 /eV Mn 2p1/2 /eV ΔEB
/eVRelative ratio of Mn oxide w/% MnO2 Mn2O3 Mn3O4 8Mn/
2.5Cr-Zr641.6 653.3 11.7 37.37 36.04 26.58 表 4 bMnOx/ZrO2-aCr2O3催化剂的苯甲酸甲酯加氢性能
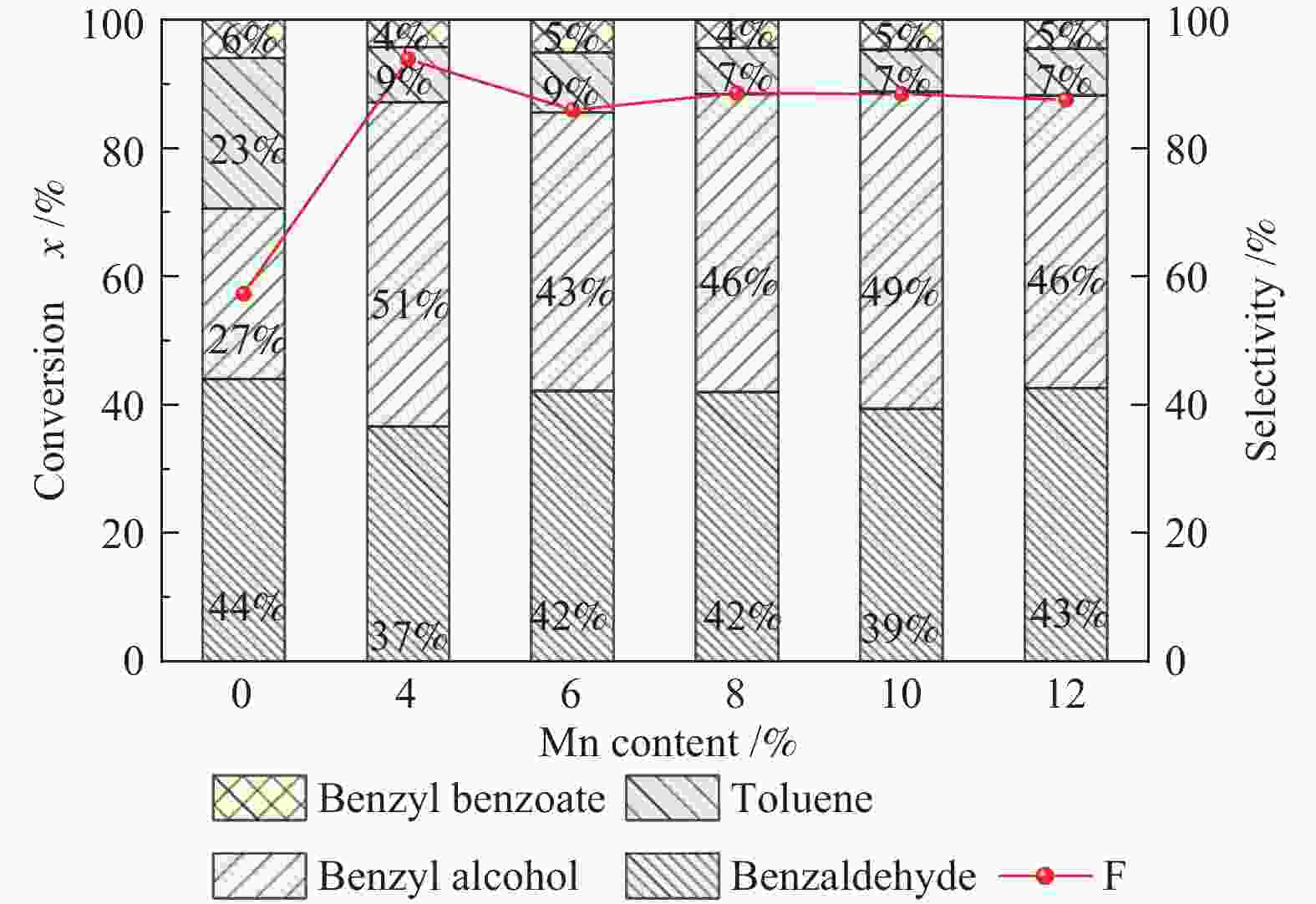
Table 4 Performance of bMnOx/ZrO2-aCr2O3 catalysts
Mn content w/% Cr content w/% Methyl benzoate x/% s/% Selectivity s/% benzaldehyde benzyl alcohol toluene benzyl benzoate 0 0 19.84 81.29 48.43 32.87 3.85 11.60 0 2.5 57.27 67.23 41.92 25.31 22.37 5.68 0 5.0 92.68 52.12 27.45 24.67 38.66 3.11 4 2.5 93.86 86.05 36.14 49.91 8.49 4.21 8 2.5 89.47 87.71 40.89 46.81 6.91 4.29 8 5.0 87.66 83.32 38.99 44.33 10.47 4.47 8 10 86.76 82.44 40.04 42.40 12.00 4.04 10 2.5 88.43 87.54 38.82 48.72 6.52 4.56 *reaction conditions: t = 390 ℃, p = 1.0 MPa, weight hourly space velocity of methyl benzoate = 0.5 h−1, n(H2)/ n(MB) = 10 -
[1] NURHIKMAH N, CAHYANA A H, LIANDI A R. Utilization of cinnamaldehyde for synthesis of nitrogen-containing 2-hydroxy-1, 4-naphthoquinone derivatives as antioxidant agent[J]. AIP Publishing, 2020, 2242(1): 928. [2] WANG M. Production technology and market of benzaldehyde[J]. Fine Chem Ind Raw Mater Interm,2005,(6):15−18. [3] YU Z Y. Synthesis and application of benzaldehyde and its derivatives[J]. Chem Interm,2003,(4):10−11+15. [4] YANG S J, XIA J, SUN J T. Synthesis of benzaldehyde 1,2-propanediol acetal catalyzed by using TiSiW(12)O(40)/TiO2[J]. Chem React Eng Technol,2003,(1):9−13. [5] KUNJAPUR A M, YEKATERINA T, PRATHER K L J. Synthesis and accumulation of aromatic aldehydes in an engineered strain of escherichia coli[J]. J Am Chem Soc,2014,136(33):44−54. [6] WANG Y, QIAO X. Preparation of benzaldehyde from benzyl chloride by-product mixture[J]. Fine Chem Interm,2005,(1):44−46+49. [7] DENG C S, XU M X, DONG Z, LI L, YANG J Y, GUO X F, PENG L M, XUE N H, ZHU Y, DING W P. Exclusively catalytic oxidation of toluene to benzaldehyde in an O/W emulsion stabilized by hexadecylphosphate acid terminated mixed-oxide nanoparticles[J]. Chin J Catal,2020,41(2):341−349. doi: 10.1016/S1872-2067(19)63417-0 [8] ZHANG W X, SHAO X G, ZHANG H C, LIN X Q. Review on the electrosynthesis of benzaldehyde and its derivatives[J]. Chem Ind Eng Prog,2000,(4):24−27. [9] LI G X, YU H, TENG Z J, LU Y S, TANG Q. Oxidation of benzyl alcohol to benzaldehyde over keggin type multicomponent heteropoly compounds[J]. Chem Ind Eng Prog,2010,29(1):71−75. [10] BAI Z J, SHEN S, CHEN L, QU Z T, YIN S F. Recent progress of selective toluene oxidation via heterogeneous photocatalyst[J]. Sci Sin Chim,2020,50(2):223−234. doi: 10.1360/SSC-2019-0144 [11] WILDE C A, RYABENKOVA Y L, FIRTH I M, PRATT L, RAILTON J, BRAVO S M, SANON, CUMPSON P J, COATES P D, LIU X, CONTE M. Novel rhodium on carbon catalysts for the oxidation of benzyl alcohol to benzaldehyde: A study of the modification of metal/support interactions by acid pre-treatments[J]. Appl Catal A: Gen,2019,570:271−282. doi: 10.1016/j.apcata.2018.11.006 [12] MAHMOUD N, MOJTABA B, HIRBOD K. Preparation, characterization and catalytic activity of CoFe2O4 nanoparticles as a magnetically recoverable catalyst for selective oxidation of benzyl alcohol to benzaldehyde and reduction of organic dyes[J]. J Colloid Interf Sci,2016,465:271−278. doi: 10.1016/j.jcis.2015.11.074 [13] LIU J H, WANG F, GU Z G, XU X L. Vanadium phosphorus oxide catalyst modified by silver doping for mild oxidation of styrene to benzaldehyde[J]. J Chem Eng,2009,151(1):319−323. [14] 张岳娇. 苯甲酸甲酯加氢合成苯甲醛催化剂的制备研究[D]. 北京: 北京化工大学, 2017.ZHANG Yue-jiao. Study on catalysts for methyl benzoate hydrogenation[D]. Beijing: Beijing University of Chemical Technology, 2017. [15] CHEN X H, WANG Z Y, DALY H, MORGAN R, MANYAR H, BYRNE C, WALTON A S, TAYLOR S. F R, SMITH M, BURCH R, HU P J, HARDACRE C. Hydrogenation of benzoic acid to benzyl alcohol over Pt/SnO2[J]. Appl Catal A: Gen,2020,593:117420. [16] DE LANGE M W, VAN OMMEN J G, LEFFERTS L. Deoxygenation of benzoic acid on metal oxides[J]. Appl Catal A: Gen,2002,231(1):17−26. [17] YOKOYAMA T, SETOYAMA T, FUJITA N, MAKI T. Novel direct hydrogenation process of aromatic carboxylic acids to the corresponding aldehydes with zirconia catalyst[J]. Appl Catal A: Gen,1992,88(2):149−161. doi: 10.1016/0926-860X(92)80212-U [18] MAKI T, YOKOYAMA T. Process for producing aromatic aldehydes: US, 4613700A[P]. 1986-09-23. [19] LV C X. The study of crystallization proeess of ZrO2[D]. Lanzhou: Lanzhou University of Technology, 2010. [20] 刘森, 黄锐, 孙培永, 张胜红, 姚志龙. Mn/Ti-Zr复合氧化物催化苯甲酸甲酯选择性加氢[J]. 精细化工,2021,38(4):782−789.LIU Sen, HUANG rui, SUN Pei-yong, ZHANG Sheng-hong, YAO Zhi-long. Selective hydrogenation of methyl benzoate catalyzed by Mn/Ti-Zr mixed oxides[J]. Fine Chem,2021,38(4):782−789. [21] IWANOWSKI R J, HEINONEN M H, PASZKOWICZ W, MINIKAEV R, WITKOWSKA B. X-ray photoelectron study of Sn1−xMnxTe semimagnetic semiconductors[J]. Appl Surf Sci,2006,252(10):3632−3641. doi: 10.1016/j.apsusc.2005.05.056 -





 下载:
下载: