Effect of calcination temperature on the structure and performance of molybdenum-tin catalyst for DME oxidation
-
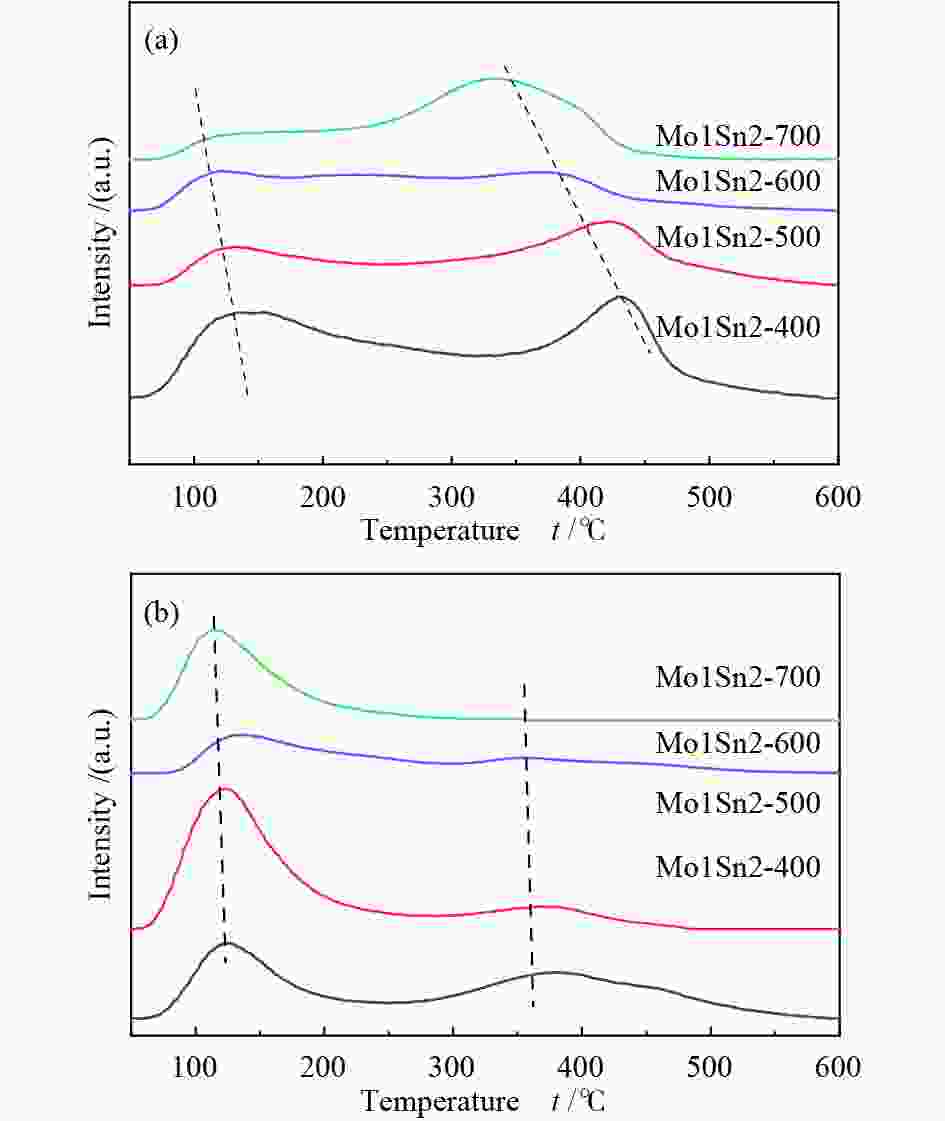
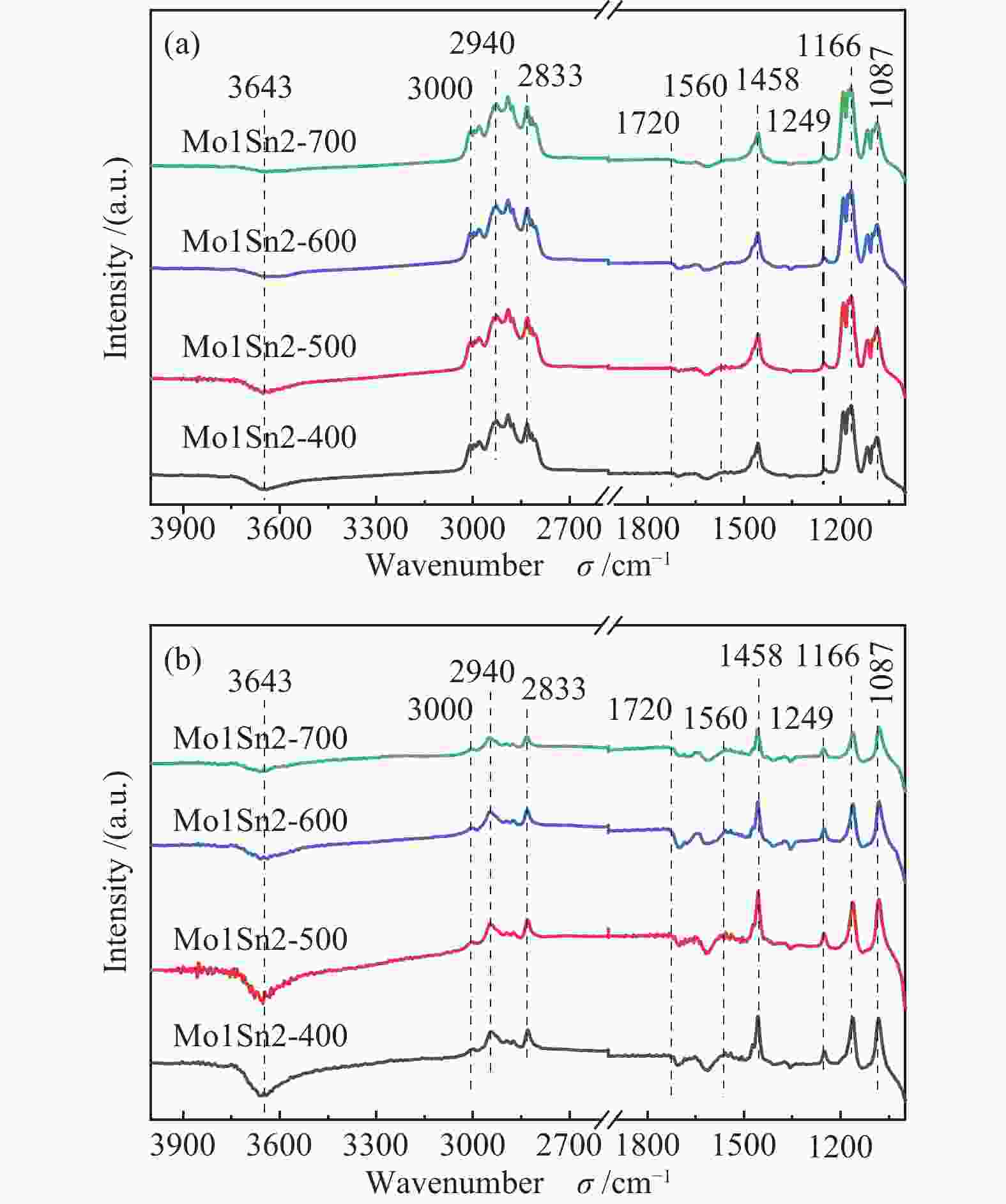
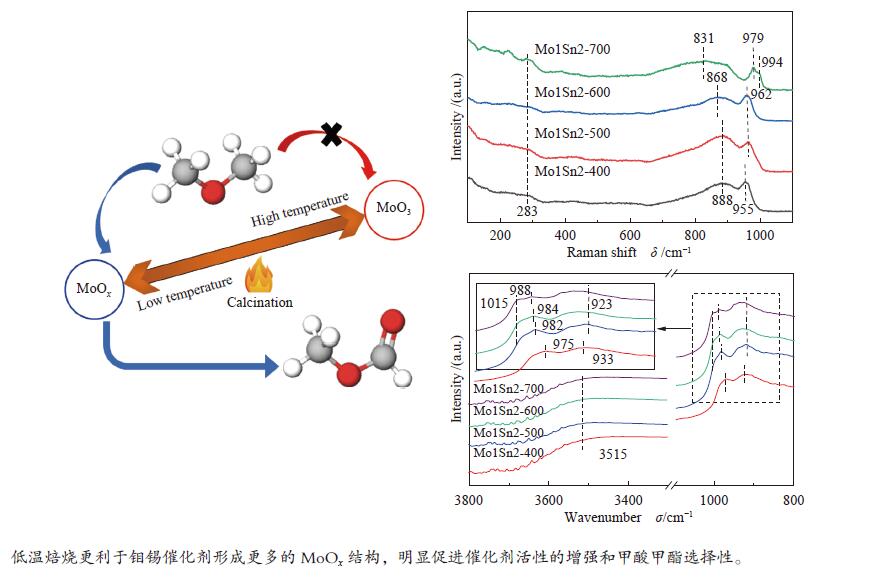
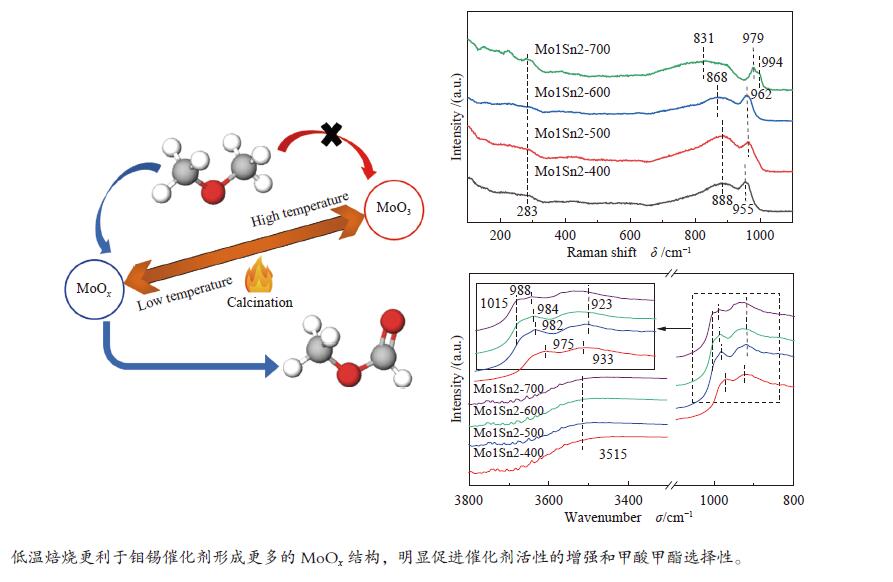
摘要: 采用水热法制备了Mo/Sn物质的量比为1∶2的Mo1Sn2催化剂,通过改变焙烧温度(400−700 ℃),调控了钼锡催化剂的结构,并研究了催化剂结构变化对二甲醚(DME)选择氧化制甲酸甲酯(MF)性能的影响。发现400 ℃焙烧的Mo1Sn2催化剂具有良好的催化氧化二甲醚生成甲酸甲酯的性能,在110 ℃、常压条件下,DME转化率为9.2%,MF选择性可达86.9%,并且无COx生成。采用XRD、Raman、XPS、TPD、H2-TPR和in-situ FT-IR等表征手段对催化剂的结构和表面性质进行了系统研究。结果表明,低温焙烧更利于钼锡催化剂表面形成更多的MoOx结构和Mo5+物种,由此引起的催化剂的酸性、氧化还原性的增强和中强碱性位的增多可明显促进催化剂活性的增强和甲酸甲酯的生成。Abstract: The Mo1Sn2 catalysts with a Mo/Sn molar ratio of 1∶2 were prepared by the hydrothermal method, and their structure was regulated by changing the calcination temperature (400–700 ℃). The effect of the structural transformation of catalysts on the performance of selective oxidation of dimethyl ether (DME) to methyl formate (MF) was studied. It was found that the Mo1Sn2 catalyst calcined at 400 ℃ exhibited good performance in the oxidation of DME to methyl formate, showing a DME conversion of 9.2% and the MF selectivity of 86.9% at 110 ℃ and under atmospheric pressure without the generation of COx. The catalysts were systematically characterized by XRD, Raman, XPS, TPD, H2-TPR and in-situ FT-IR. The results showed that the low-temperature calcination was favorable for the formation of MoOx
structure and more Mo5+ species on the catalyst surface, resulting in the enhanced acidity and redox ability of the catalyst, and the increase of medium to strong basic sites on the catalysts. In such case, the activity and methyl formate production of the catalyst were significantly promoted. -
Key words:
- dimethyl ether oxidation /
- Mo1Sn2 catalyst /
- calcination temperature /
- structure /
- methyl formate
-
表 1 不同焙烧温度的Mo1Sn2催化剂对二甲醚选择氧化制备甲酸甲酯的影响
Table 1 Effect of calcination temperatures on the selective oxidation of DME to MF over the Mo1Sn2 catalysts
Catalyst DME conversion
x /%Cmol-selectivity s /% MF FA CH3OH CO DMM Mo1Sn2-400 9.2 86.9 0 13.1 0 0 Mo1Sn2-500 9.7 77.5 0 22.5 0 0 Mo1Sn2-600 10.6 72.9 0 27.1 0 0 Mo1Sn2-700 7.2 65.4 0 34.6 0 0 Reaction conditions: tR = 110 ℃, atmospheric pressure, DME/O2 = 1, GHSV = 1800 h−1 表 2 反应温度对Mo1Sn2-400催化剂上二甲醚氧化制备甲酸甲酯的影响
Table 2 Effect of the reaction temperature on the oxidation of DME to MF over the Mo1Sn2-400 catalyst
tR/℃ DME conversion
x/%Cmol-selectivity s/% MF FA CH3OH CO DMM 90 5.5 89.8 0 10.2 0 0 100 8.4 83.4 0 16.6 0 0 110 9.2 86.9 0 13.1 0 0 120 11.3 83.4 0 14.5 2.1 0 130 11.5 82.4 0 11.2 6.4 0 140 14.5 82.5 0 7.0 10.5 0 Reaction conditions:atmospheric pressure, DME/O2 = 1, GHSV = 1800 h−1 表 3 不同焙烧温度Mo1Sn2催化剂的织构性质
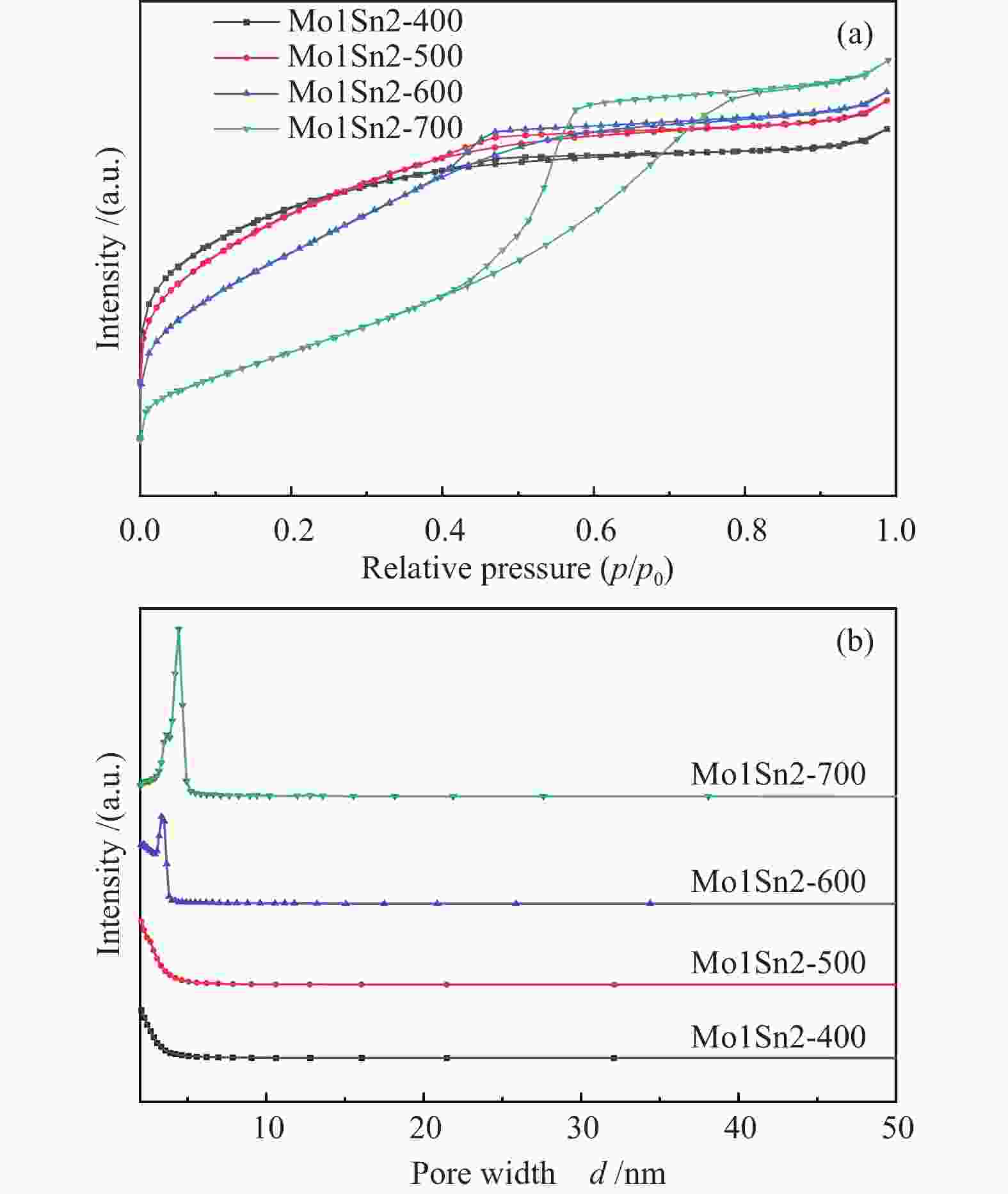
Table 3 Physical properties of Mo1Sn2 catalysts calcined at different temperatures
Catalyst BET surface area/
(m2·g−1)Micropore area/
(m2·g−1)External surface area/
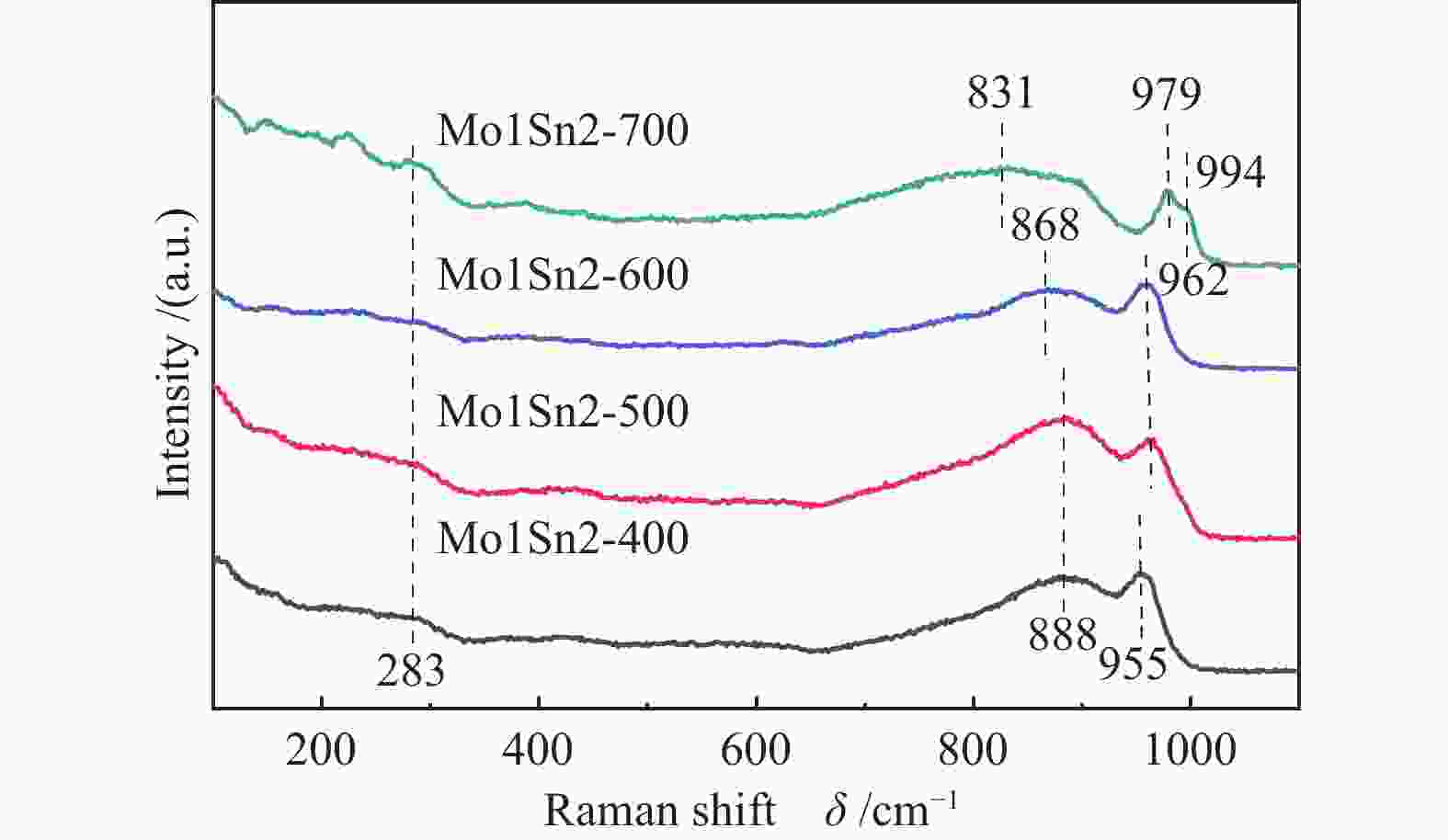
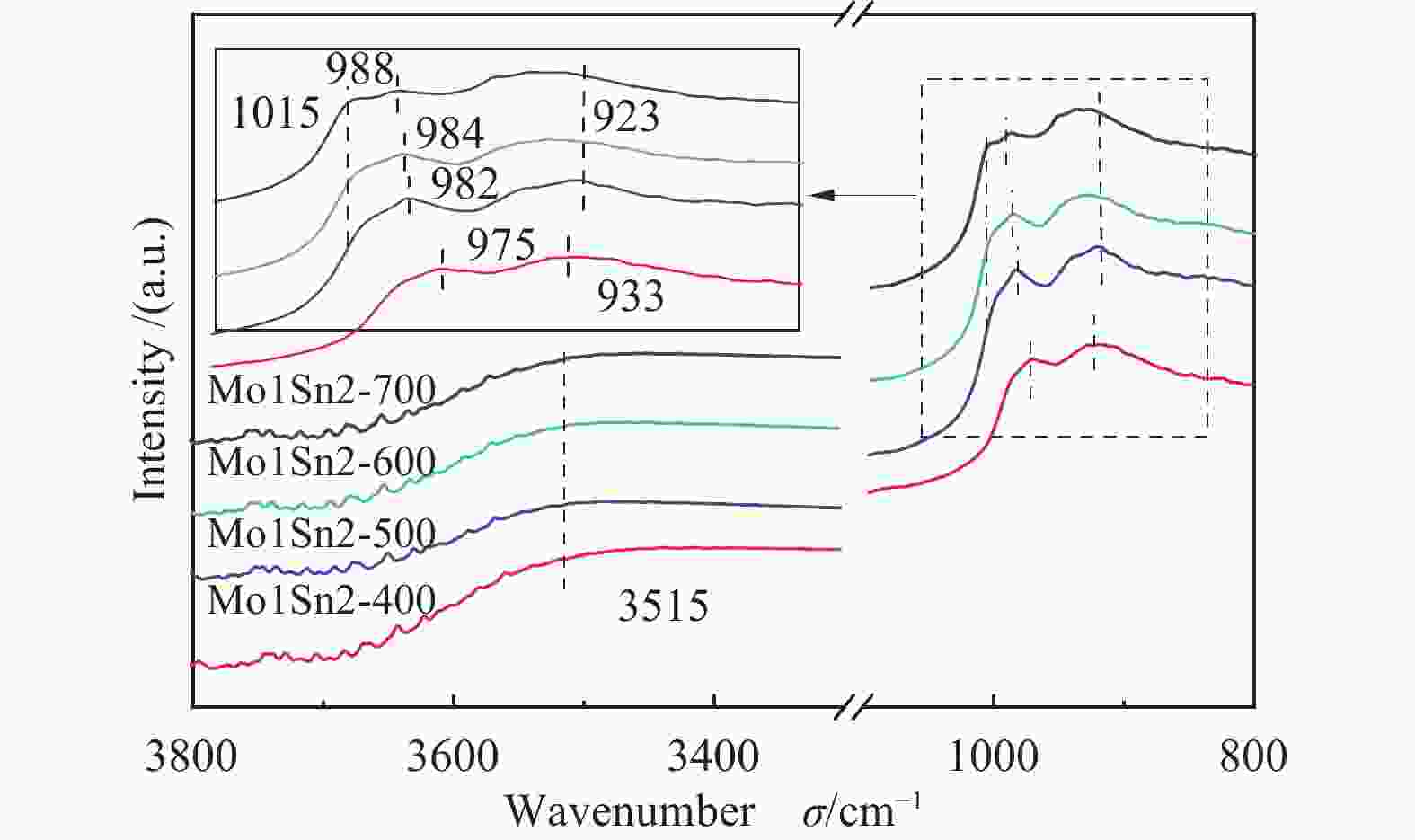
(m2·g−1)Pore volume/
(cm3·g−1)Average pore
diameter/nmMo1Sn2-400 163 117 46 0.09 2.3 Mo1Sn2-500 167 90 77 0.10 2.4 Mo1Sn2-600 152 15 137 0.10 2.7 Mo1Sn2-700 92 0 92 0.11 4.8 表 4 不同焙烧温度Mo1Sn2催化剂XPS-Mo 3d 谱图分析
Table 4 Mo 3d XPS spectra analysis of Mo1Sn2 catalysts calcined at different temperatures
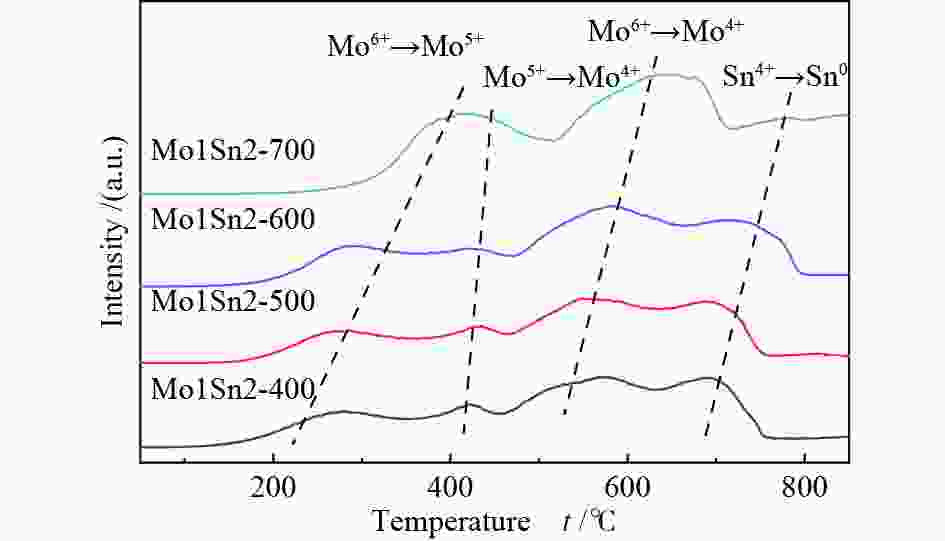
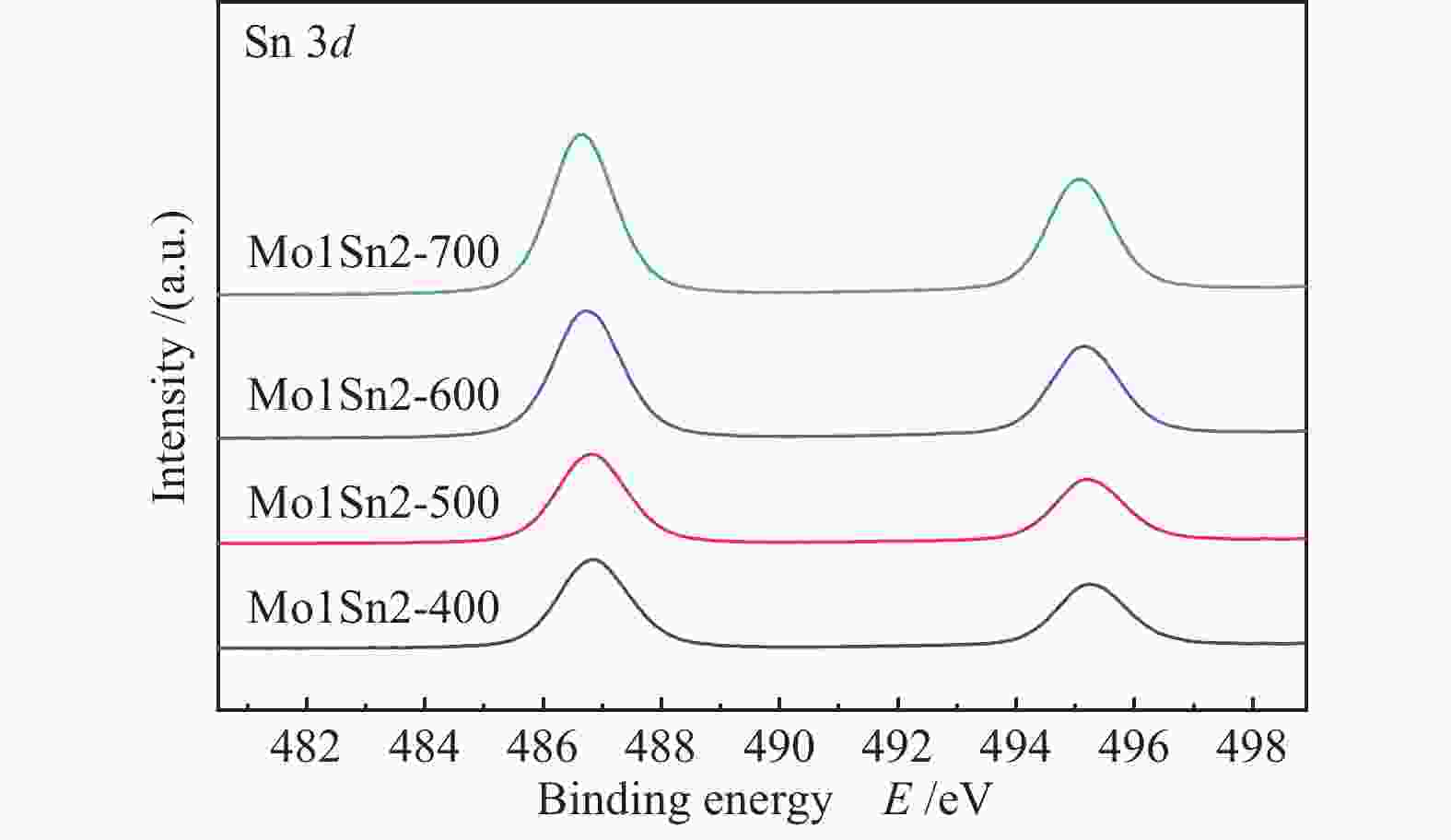
Catalyst Mo6+ 3d3/2 Mo6+ 3d5/2 Mo5+ 3d3/2 Mo5+ 3d5/2 Mo5+/% Mo1Sn2-400 236.19 233.05 235.11 232.04 10.02 Mo1Sn2-500 236.15 233.02 235.10 232.01 8.51 Mo1Sn2-600 236.10 232.95 235.11 231.90 7.06 Mo1Sn2-700 236.11 232.95 235.10 231.91 6.24 -
[1] SEMELSBERGER T A, BORUP R L, GREENE H L. Dimethyl ether (DME) as an alternative fuel[J]. J Power Sources,2006,156(2):497−511. doi: 10.1016/j.jpowsour.2005.05.082 [2] SUN J, YANG G H, YONEYAMA Y, TSUBAKI N. Catalysis chemistry of dimethyl ether synthesis[J]. ACS Catal,2014,4(10):3346−3356. doi: 10.1021/cs500967j [3] TAN Y S, XIE H J, CUI H T, HAN Y Z, ZHONG B. Modification of Cu-based methanol synthesis catalyst for dimethyl ether synthesis from syngas in slurry phase[J]. Catal Today,2005,104(1):25−29. doi: 10.1016/j.cattod.2005.03.033 [4] XU M T, LUNSFORD J H, GOODMAN D W, BHATTACHARYYA A. Synthesis of dimethyl ether (DME) from methanol over solid-acid catalysts[J]. Appl Catal A: Gen,1997,149(2):289−301. doi: 10.1016/S0926-860X(96)00275-X [5] 孙明, 余林, 孙长勇, 宋一兵, 孙建. 二甲醚的应用及下游产品开发[J]. 精细化工,2003,20(11):695−699. doi: 10.3321/j.issn:1003-5214.2003.11.017SUN Ming, YU Lin, SUN Chang-yong, SONG Yi-bing, SUN Jian. Application of dimethyl ether and development of its downstream products[J]. Fine Chem,2003,20(11):695−699. doi: 10.3321/j.issn:1003-5214.2003.11.017 [6] 常雁红, 韩怡卓, 王心葵. 二甲醚的生产、应用及下游产品的开发[J]. 天然气化工,2000,25(3):45−49.CHANG Yan-hong, HAN Yi-zhuo, WANG Xin-kui. Production, application and development of downstream products of dimethyl ether[J]. Nat Gas Chem Ind,2000,25(3):45−49. [7] GAO X J, WANG W F, GU Y Y, ZHANG Z Z, ZHANG J F, ZHANG Q D, TSUBAKI N, HAN Y Z, TAN Y S. Synthesis of polyoxymethylene dimethyl ethers from dimethyl ether direct oxidation over carbon-based catalysts[J]. ChemCatChem,2018,10(1):273−279. doi: 10.1002/cctc.201701213 [8] 周寿祖. 甲酸甲酯的生产技术和应用前景[J]. 化工科技市场,2003,26(2):13−18.ZHOU Shou-zu. Production technology and application foreground of methyl formate[J]. Chem Technol Market,2003,26(2):13−18. [9] 陈文龙, 刘海超. 甲醇脱氢和选择氧化制备甲酸甲酯的催化剂与反应路径[J]. 科学通报,2015,60(16):1502−1512. doi: 10.1360/N972014-01349CHEN Wen-long, LIU Hai-chao. Catalysts and reaction pathways for dehydrogenation and selective oxidation methanol to methyl formate[J]. Chin Sci Bull,2015,60(16):1502−1512. doi: 10.1360/N972014-01349 [10] AI M. The production of methyl formate by the vapor-phase oxidation of methanol[J]. J Catal,1982,77(1):279−288. doi: 10.1016/0021-9517(82)90168-3 [11] LIU G B, ZHANG Q D, HAN Y Z, TSUBAKI N, TAN Y S. Effects of the MoO3 structure of Mo-Sn catalysts on dimethyl ether oxidation to methyl formate under mild conditions[J]. Green Chem,2015,17(2):1057−1064. doi: 10.1039/C4GC01591F [12] LIU G B, ZHANG Q D, HAN Y Z, TSUBAKI N, TAN Y S. Selective oxidation of dimethyl ether to methyl formate over trifunctional MoO3-SnO2 catalyst under mild conditions[J]. Green Chem,2013,15(6):1501−1504. doi: 10.1039/c3gc40279g [13] CHEUNG P, LIU H C, IGLESIA E. Kinetics and mechanism of dimethyl ether oxidation to formaldehyde on supported molybdenum oxide domains[J]. J Phys Chem B,2004,108(48):18650−18658. doi: 10.1021/jp0477405 [14] LIU H C, CHEUNG P, IGLESIA E. Zirconia-supported MoOx, catalysts for the selective oxidation of dimethyl ether to formaldehyde: Structure, redox properties, and reaction pathways[J]. J Phys Chem B,2003,107(17):4118−4127. doi: 10.1021/jp0221744 [15] LIU H C, CHEUNG P, IGLESIA E. Structure and support effects on the selective oxidation of dimethyl ether to formaldehyde catalyzed by MoOx domains[J]. J Catal,2003,217(1):222−232. [16] ZHANG Z Z, ZHANG Q D, JIA L Y, WANG W F, ZHANG T, HAN Y Z, TSUBAKI N, TAN Y S. Effects of tetrahedral molybdenum oxide species and MoOx domains on the selective oxidation of dimethyl ether under mild conditions[J]. Catal Sci Technol,2016,6(9):2975−2983. doi: 10.1039/C5CY01569C [17] ZHANG Z Z, ZHANG Q D, JIA L Y, WANG W F, TIAN S P, WANG P, XIAO H, HAN Y Z, TSUBAKI N, TAN Y S. The effects of the Mo-Sn contact interface on the oxidation reaction of dimethyl ether to methyl formate at a low reaction temperature[J]. Catal Sci Technol,2016,6(15):6109−6117. doi: 10.1039/C6CY00460A [18] 杨奇, 高秀娟, 冯茹, 李明杰, 张俊峰, 张清德, 韩怡卓, 谭猗生. 水热合成的MoO3-SnO2催化剂催化氧化二甲醚的性能研究[J]. 燃料化学学报,2019,47(8):934−941. doi: 10.3969/j.issn.0253-2409.2019.08.005(YANG Qi, GAO Xiu-juan, FENG Ru, LI Ming-jie, ZHANG Jun-feng, ZHANG Qing-de, HAN Yi-zhuo, TAN Yi-sheng. MoO3-SnO2 catalyst prepared by hydrothermal synthesis method for dimethyl ether catalytic oxidation[J]. J Fuel Chem Technol,2019,47(8):934−941. doi: 10.3969/j.issn.0253-2409.2019.08.005 [19] LI A, LONG H, ZHANG H, LI H. High-yield synthesis of Ce modified Fe-Mn composite oxides benefitting from catalytic destruction of chlorobenzene[J]. Rsc Adv,2020,10(17):10030−10037. doi: 10.1039/C9RA10489E [20] TAO Z, YANG Y, DING M, LI T, XIANG H, LI Y. Effect of calcination behaviors on precipitated iron-manganese Fischer-Tropsch synthesis catalyst[J]. Catal Lett,2007,117(3/4):130−135. doi: 10.1007/s10562-007-9118-5 [21] 张建荣, 高濂. 水热法合成纳米SnO2粉体[J]. 无机材料学报,2004,19(5):1177−1180. doi: 10.3321/j.issn:1000-324X.2004.05.034ZHANG Jian-rong, GAO Lian. Hydrothermal synthesis of tin oxide nanoparticles[J]. J Inorg Mater,2004,19(5):1177−1180. doi: 10.3321/j.issn:1000-324X.2004.05.034 [22] LAKSHMI L J, ALYEA E C. ESR, FT-Raman spectroscopic and ethanol partial oxidation studies on MoO3/SnO2 catalysts made by metal oxide vapor synthesis[J]. Catal Lett,1999,59(1):73−77. doi: 10.1023/A:1019099900418 [23] HERRMANN J M, VILLAIN F, APPEL L G. Characterization of Mo-Sn-O system by means of Raman spectroscopy and electrical conductivity measurements[J]. Appl Catal A: Gen,2003,240(1/2):177−182. doi: 10.1016/S0926-860X(02)00419-2 [24] GONCALVES F, MEDEIROS P R S, EON J G, APPEL L G. Active sites for ethanol oxidation over SnO2-supported molybdenum oxides[J]. Appl Catal A: Gen,2000,193(1/2):195−202. doi: 10.1016/S0926-860X(99)00430-5 [25] HABER J, LALIK E. Catalytic properties of MoO3 revisited[J]. Catal Today,1997,33(1/3):119−137. doi: 10.1016/S0920-5861(96)00107-1 [26] Zhang Q D, TAN Y S, LIU G B, ZHANG J F, HAN Y Z. Rhenium oxide-modified H3PW12O40/TiO2 catalysts for selective oxidation of dimethyl ether to dimethoxy dimethyl ether[J]. Green Chem,2014,16(11):4708−4715. doi: 10.1039/C4GC01373E [27] 陶东平, 杨显万. 氧化亚锡歧化还原动力学和二氧化锡还原机理[J]. 中国有色金属学报,1998,8(1):126−130. doi: 10.3321/j.issn:1004-0609.1998.01.027TAO Dong-ping, YANG Xian-wan. Kinetics of disproportionation and reduction of stannous oxide and mechanism of reduction of stannic oxide[J]. Chin J Nonferrous Met,1998,8(1):126−130. doi: 10.3321/j.issn:1004-0609.1998.01.027 [28] 杜英辉, 许国基. 氢气还原金属氧化物[J]. 原子能科学技术,1999,33(4):360−362. doi: 10.3969/j.issn.1000-6931.1999.04.020DU Ying-hui, XU Guo-ji. Hydrogen reduction of metal oxides to metals[J]. Atom Energy Sci Technol,1999,33(4):360−362. doi: 10.3969/j.issn.1000-6931.1999.04.020 [29] TAN X J, WANG L Z, CHENG C, YAN X F, SHEN B, ZHANG J L. Plasmonic MoO3−x @MoO3 nanosheets for highly sensitive SERS detection through nanoshell-isolated electromagnetic enhancement[J]. Chem Commun,2016,52(14):2893−2896. doi: 10.1039/C5CC10020H [30] SWIATOWSKA-MROWIECKA J, DE DIESBACH S, MAURICE V, ZANNA S, KLEIN L, BRIAND E, VICKRIDGE I, MARCUS P. Li-ion intercalation in thermal oxide thin films of MoO3 as studied by XPS, RBS, and NRA[J]. J Phys Chem C,2008,112(29):11050−11058. doi: 10.1021/jp800147f [31] 杨奇. 钼锡催化剂上二甲醚低温氧化机理研究[D]. 北京: 中国科学院大学, 2019.YANG Qi. Study on the mechanism of the low-temperature oxidation of dimethyl ether over MoO3-SnO2 catalyst[D]. Beijing: University of Chinese Academy of Sciences, 2019. [32] LOCHAR V. FT-IR study of methanol, formaldehyde and methyl formate adsorption on the surface of Mo/Sn oxide catalyst[J]. Appl Catal A: Gen,2006,309(1):33−36. doi: 10.1016/j.apcata.2006.04.030 -




 下载:
下载: