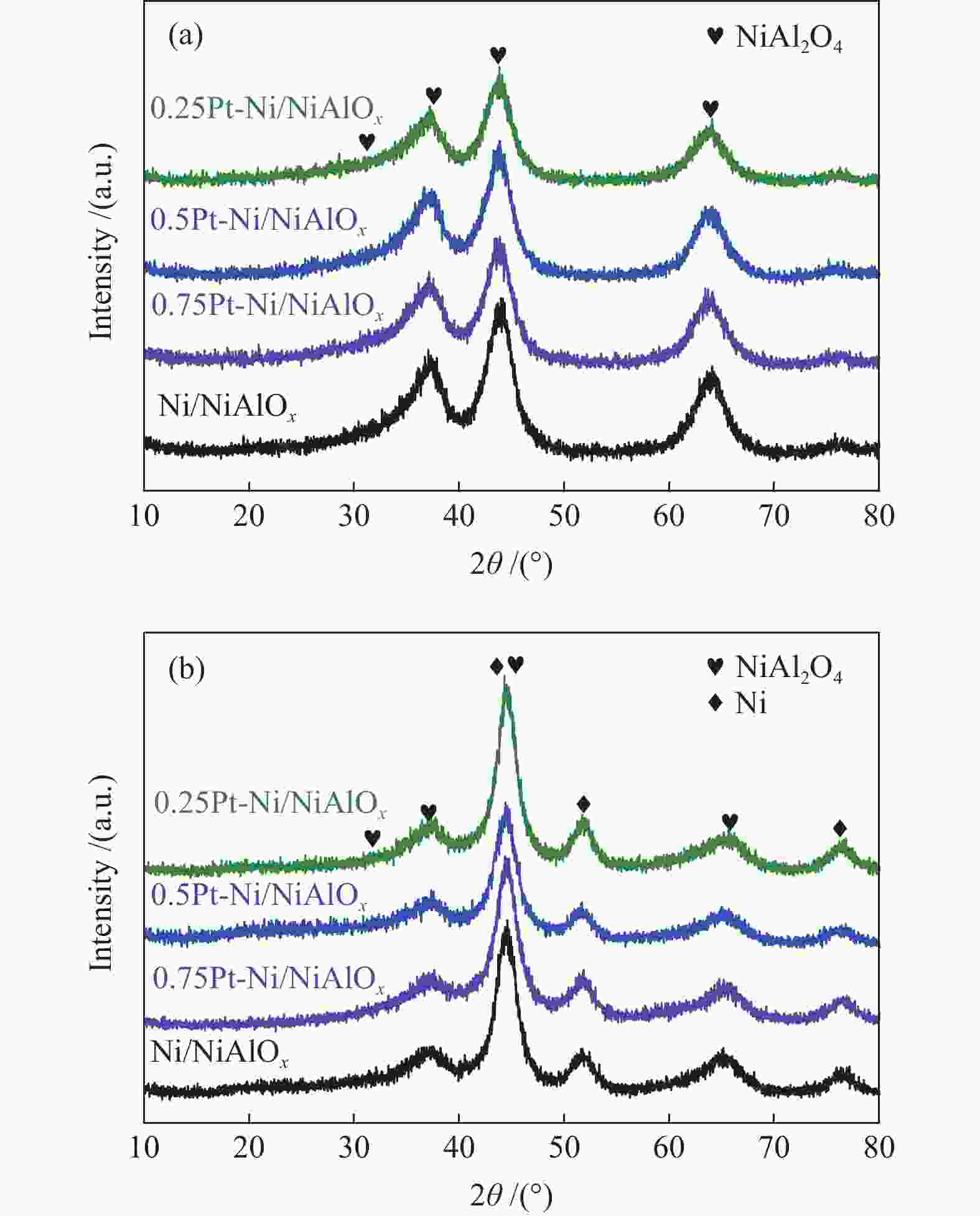
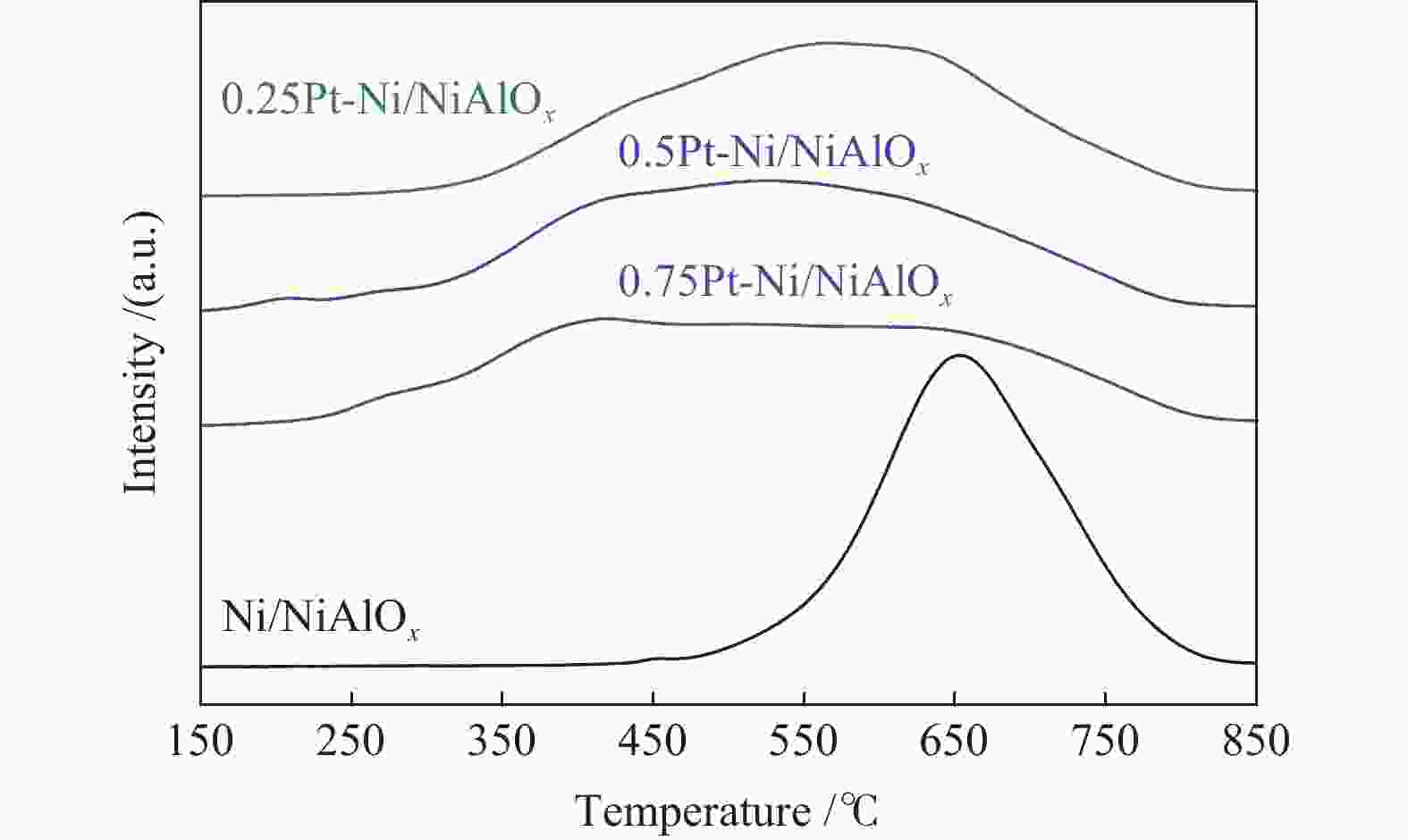
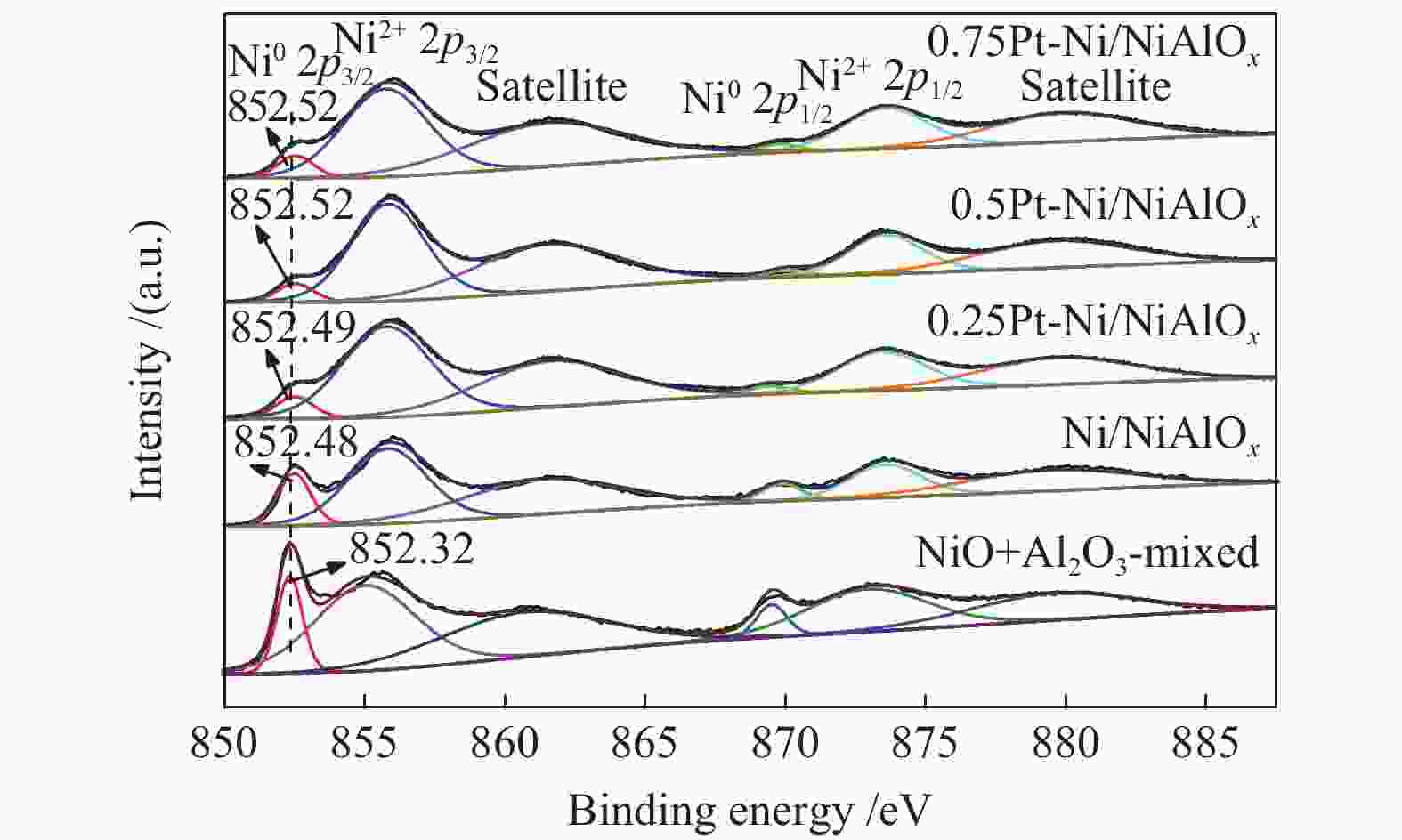
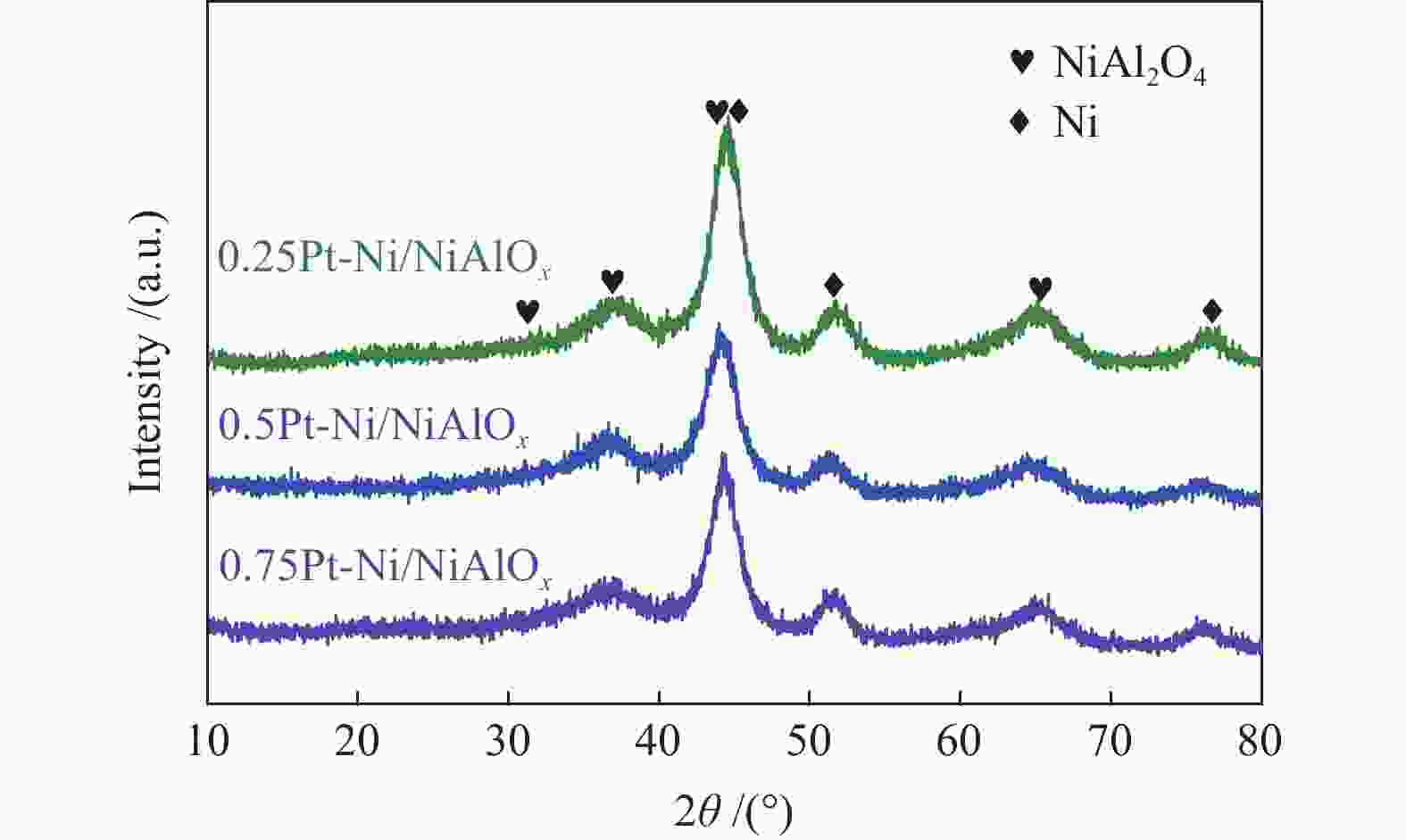
Performance of Pt-doped Ni/NiAlOx catalysts for phenanthrene hydrogenation saturation
-
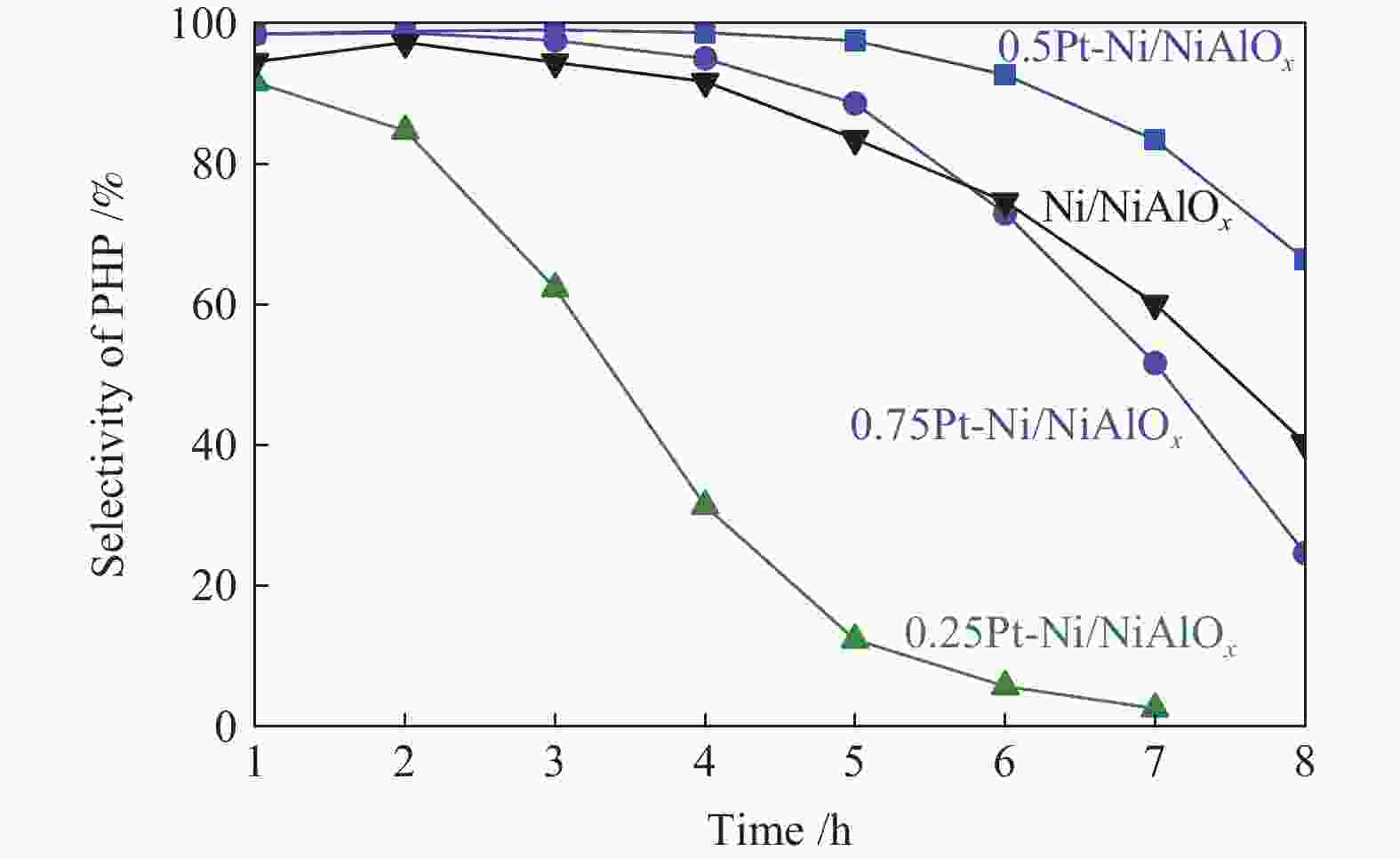
摘要: 本研究选取菲为模型化合物,以强化芳烃吸附为目标,采用等体积浸渍法制备了系列Pt-Ni/NiAlOx催化剂,系统考察了Pt掺杂量对催化剂结构和菲加氢饱和性能的影响。结果表明,当反应温度300 ℃、氢气压力5 MPa、重时空速52 h−1时,相比Ni/NiAlOx催化剂,掺杂0.5%Pt的0.5Pt-Ni/NiAlOx催化剂上全氢菲选择性在反应8 h后由40%提升至67%,且表观反应速率和转化频率分别由1.53 × 10−3 mol·kg−1·s−1和14.64 × 10−3 s−1提升至1.81 × 10−3 mol·kg−1·s−1和22.16 × 10−3 s−1。这主要归因于金属Pt适宜的掺杂量提高了金属Ni缺电子结构的稳定性,促进芳烃吸附,提升了菲加氢饱和性能。Abstract: Limited by the steric hindrance, hydrogenation of the final unsaturated ring in polycyclic aromatic hydrocarbons remains a challenge. In this work, a series of Pt-Ni/NiAlOx catalysts were synthesized by impregnation method to enhance the adsorption of aromatics, and phenanthrene was served as the model compound. The effects of Pt content on the structure and saturation performance of Pt-Ni/NiAlOx catalysts were systematically investigated. When the saturation reaction was performed at 300 ℃, 5 MPa and a weight hourly space velocity of 52 h−1, the selectivity of perhydrophenanthrene could be enhanced from 40% over Ni/NiAlOx catalysts to 67% over 0.5Pt-Ni/NiAlOx catalysts with 0.5% Pt loading. Meanwhile, the obvious reaction rate and turnover frequency were also improved from 1.53×10−3 mol·kg−1·s−1 and 14.64×10−3 s−1 to 1.81×10−3 mol·kg−1·s−1 and 22.16×10−3 s−1 respectively. This is related to the modified stability of metallic electron-deficient structure of Ni by Pt introduction in phenanthrene hydrogenation, which can promote the adsorption of aromatic hydrocarbons as well as the hydrogenation activity.
-
Key words:
- phenanthrene /
- hydrogenation saturation /
- Pt-Ni bimetal /
- Pt loadings
-
表 1 不同Pt负载量Pt-Ni/NiAlOx催化剂的比表面积和平均孔径
Table 1 Surface area and average pore size of Pt-Ni/NiAlOx catalysts with different Pt loadings
Catalyst $ {{S}}_{{\rm{BET}}}^{\rm{a}}$/(m2·g−1) $ {{S}}_{{\rm{meso }}}^{\rm{b}}$/(m2·g−1) $ {{S}}_{{\rm{mic}}}^{\rm{b}}$/(m2·g−1) Average pore size/ nm 0.25Pt-Ni/NiAlOx 177.5 177.5 − 5.2 0.5Pt-Ni/NiAlOx 163.4 163.4 − 4.4 0.75Pt-Ni/NiAlOx 162.1 162.1 − 4.8 Ni/NiAlOx 178.0 178.0 − 4.5 a: BET surface area, b: calculated by t-plot, all of the samples are tableted at 15 MPa 表 2 系列Pt-Ni/NiAlOx催化剂金属分散度及本征动力学
Table 2 Metal dispersion and intrinsic activity of Pt-Ni/NiAlOx catalysts with different Pt loadings
Catalyst Metal dispersiona/% Hydrogenation
conversion of PHE/%robs × 103/(mol·kg−1·s−1) TOF × 103/s−1 Ref. 0.25Pt-Ni/NiAlOx 0.94 10.91 b 1.20 18.68 b this work 0.5Pt-Ni/NiAlOx 1.14 15.68 b 1.81 22.16 b this work 0.75Pt-Ni/NiAlOx 1.76 14.31 b 1.63 13.48 b this work Ni/NiAlOx 1.99 18.50 c 1.53 14.64 c this work 0.5Pt/Al2O3 99.12 6.24 d 0.05 0.58 d this work NiMoS/MZSM-5 − − 0.74 1.80 [9] Ni2P/HZSM-5-M − − 1.32 8.20 [26] a: calculated by CO pulse adsorption; b: determined at 300 ℃, H2 pressure of 5 MPa, 1% PHE in decalin, WHSV of 720 h−1; c: determined at 300 ℃, H2 pressure of 5 MPa, 1% PHE in decalin, WHSV of 520 h−1; d: determined at 300 ℃, H2 pressure of 5 MPa, 1% PHE in decalin, WHSV of 52 h−1 -
[1] JING J Y, WANG J Z, LIU D C, QIE Z Q, BAI H C, LI W Y. Naphthalene hydrogenation saturation over Ni2P/Al2O3 catalysts synthesized by thermal decomposition of hypophosphite[J]. ACS Omega,2020,5(48):31423−31431. doi: 10.1021/acsomega.0c05019 [2] YOON E M, SELVARAJ L, SONG C, STALLMAN J B, COLEMAN M M. High-temperature stabilizers for jet fuels and similar hydrocarbon mixtures. 1. Comparative studies of hydrogen donors[J]. Energy Fuels,1996,10(3):806−811. doi: 10.1021/ef950228l [3] ZHANG X W, PAN L, WANG L, ZOU J J. Review on synthesis and properties of high-energy-density liquid fuels: Hydrocarbons, nanofluids and energetic ionic liquids[J]. Chem Eng Sci,2018,180:95−125. doi: 10.1016/j.ces.2017.11.044 [4] JIA T H, ZHANG X W, LIU Y, GONG S, DENG C, PAN L, ZOU J J. A comprehensive review of the thermal oxidation stability of jet fuels[J]. Chem Eng Sci,2021,229:116157. doi: 10.1016/j.ces.2020.116157 [5] LI C S, SUZUKI K. Resources, properties and utilization of tar[J]. Resour Conserv Recycl,2010,54(11):905−915. doi: 10.1016/j.resconrec.2010.01.009 [6] HAYAKAWA K, MURAHASHI T, AKUTSU K, KANDA T, TANG N, KAKIMOTO H, TORIBA A, KIZU R. Comparison of polycyclic aromatic hydrocarbons and nitropolycyclic aromatic hydrocarbons in airborne and automobile exhaust particulates[J]. Polycycl Aromat Compd,2000,20(1/4):179−190. doi: 10.1080/10406630008034784 [7] HAYAKAWA K, MURAHASHI T, AKUTSU K, KANDA T, TANG N, KAKIMOTO H, TORIBA A, KIZU R. Simultaneous hydrogenation of multiring aromatic compounds over NiMo catalyst[J]. Ind Eng Chem Res,2008,47(19):7161−7166. doi: 10.1021/ie8004258 [8] QIAN W H, YODA Y, HIRAI Y, ISHIHARA A, KABE T. Hydrodesulfurization of dibenzothiophene and hydrogenation of phenanthrene on alumina-supported Pt and Pd catalysts[J]. Appl Catal A: Gen,1999,184(1):81−88. doi: 10.1016/S0926-860X(99)00083-6 [9] FU W Q, ZHANG L, WU D F, XIANG M, ZHUO Q, HUANG K, TAO Z D, TANG T D. Mesoporous zeolite-supported metal sulfide catalysts with high activities in the deep hydrogenation of phenanthrene[J]. J Catal,2015,330:423−433. doi: 10.1016/j.jcat.2015.07.026 [10] WANG D G, LI J H, ZHENG A D, MA H J, PAN Z D, QU W, WANG L, HAN J Q, WANG C X, TIAN Z J. Quasi-single-layer MoS2 on MoS2/TiO2 nanoparticles for anthracene hydrogenation[J]. ACS Appl Nano Mater,2019,2(8):5096−5107. doi: 10.1021/acsanm.9b01001 [11] WANG D G, LI J H, ZHENG A D, MA H J, PAN Z D, QU W, WANG L, HAN J Q, WANG C X, TIAN Z J. Designing MoS2 nanocatalysts with increased exposure of active edge sites for anthracene hydrogenation reaction[J]. Catal Sci Technol,2017,7:2998−3007. doi: 10.1039/C7CY01026E [12] LUO W Q, SHI H, SCHACHTL E, GUTIéRREZ O T, LERCHER J A. Active sites on nickel-promoted transition-metal sulfides that catalyze hydrogenation of aromatic compounds[J]. Angew Chem-Int Ed,2018,57:14555−14559. doi: 10.1002/anie.201808428 [13] GHADAMI YAZDI M, MOUD P H, MARKS K, PISKORZ W, ÖSTRÖM H, HANSSON T, KOTARBA A, ENGVALL K, GÖTHELID M. Naphthalene on Ni(111): Experimental and theoretical insights into adsorption, dehydrogenation, and carbon passivation[J]. J Mater Chem C,2017,121(40):22199−22207. [14] 刘道诚, 王九占, 荆洁颖, 杨志奋, 冯杰, 李文英. 稠环芳烃加氢饱和催化剂研究进展[J]. 化工进展,2021,40(2):835−844.LIU Dao-cheng, WANG Jiu-zhan, JING Jie-ying, YANG Zhi-feng, FENG Jie, LI Wen-ying. Research progress on the catalysts for saturated hydrogenation of polycyclic aromatic hydrocarbons[J]. Chem Ind Eng Prog,2021,40(2):835−844. [15] LIU D C, JING J Y, WANG J Z, FENG J, LI W Y. Phenanthrene hydrogenation saturation over Ni/NiAl2O4 catalyst prepared by modified sol-gel method[C]. 37th Annual International Pittsburgh Coal Conference, 2020. [16] KITCHIN J R, NØRSKOV J K, BARTEAU M A, CHEN J G. Role of strain and ligand effects in the modification of the electronic and chemical properties of bimetallic surfaces[J]. Phys Rev Lett,2004,93(15):156801. doi: 10.1103/PhysRevLett.93.156801 [17] MAVRIKAKIS M, HAMMER B, NøRSKOV J K. Effect of strain on the reactivity of metal surfaces[J]. Phys Rev Lett,1998,81(13):2819−2822. doi: 10.1103/PhysRevLett.81.2819 [18] BERTOLINI J-C. Surface stress and chemical reactivity of Pt and Pd overlayers[J]. Appl Catal A: Gen,2000,191(1):15−21. [19] MORALES-MARíN A, AYASTUY J L, IRIARTE-VELASCO U, GUTIéRREZ-ORTIZ M A. Nickel aluminate spinel-derived catalysts for the aqueous phase reforming of glycerol: Effect of reduction temperature[J]. Appl Catal B: Environ,2019,244:931−945. doi: 10.1016/j.apcatb.2018.12.020 [20] RAAB C, LERCHER J A, GOODWIN J G, SHYU J Z. Preparation and characterization of silica-supported Ni/Pt catalysts[J]. J Catal,1990,122(2):406−414. doi: 10.1016/0021-9517(90)90294-T [21] 方洁, 李娜, 成浪, 陆江银. Ni-Cu作用对于1,4-丁炔二醇一步法低压加氢的影响[J]. 燃料化学学报,2019,47(6):725−736. doi: 10.3969/j.issn.0253-2409.2019.06.010FANG Jie, LI Na, CHENG Lang, LU Jiang-yin. Effect of Ni-Cu on the one-step mild pressure hydrogenation of 1,4-butynediol[J]. J Fuel Chem Technol,2019,47(6):725−736. doi: 10.3969/j.issn.0253-2409.2019.06.010 [22] VAN DEELEN T W, HERNÁNDEZ MEJÍA C, DE JONG K P. Control of metal-support interactions in heterogeneous catalysts to enhance activity and selectivity[J]. Nat Catal,2019,2(11):955−970. doi: 10.1038/s41929-019-0364-x [23] LEE Y-K, OYAMA S T. Bifunctional nature of a SiO2-supported Ni2P catalyst for hydrotreating: EXAFS and FTIR studies[J]. J Catal,2006,239(2):376−389. doi: 10.1016/j.jcat.2005.12.029 [24] SRIFA A, KAEWMEESRI R, FANG C, ITTHIBENCHAPONG V, FAUNGNAWAKIJ K. NiAl2O4 spinel-type catalysts for deoxygenation of palm oil to green diesel[J]. Chem Eng J,2018,345:107−113. doi: 10.1016/j.cej.2018.03.118 [25] TUO Y X, MENG Y, CHEN C, LIN D, FENG X, PAN Y, LI P, CHEN D, LIU Z N, ZHOU Y, ZHANG J. Partial positively charged Pt in Pt/MgAl2O4 for enhanced dehydrogenation activity[J]. Appl Catal B: Environ,2021,288:119996. doi: 10.1016/j.apcatb.2021.119996 [26] FU W Q, ZHANG L, WU D F, YU Q Y, TANG T, TANG T D. Mesoporous zeolite ZSM-5 supported Ni2P catalysts with high activity in the hydrogenation of phenanthrene and 4, 6-dimethyldibenzothiophene[J]. Ind Eng Chem Res,2016,55(26):7085−7095. doi: 10.1021/acs.iecr.6b01583 -




 下载:
下载: