Study on size effect of γ-Fe2O3 nanoparticles and gas atmosphere on carburization process
-
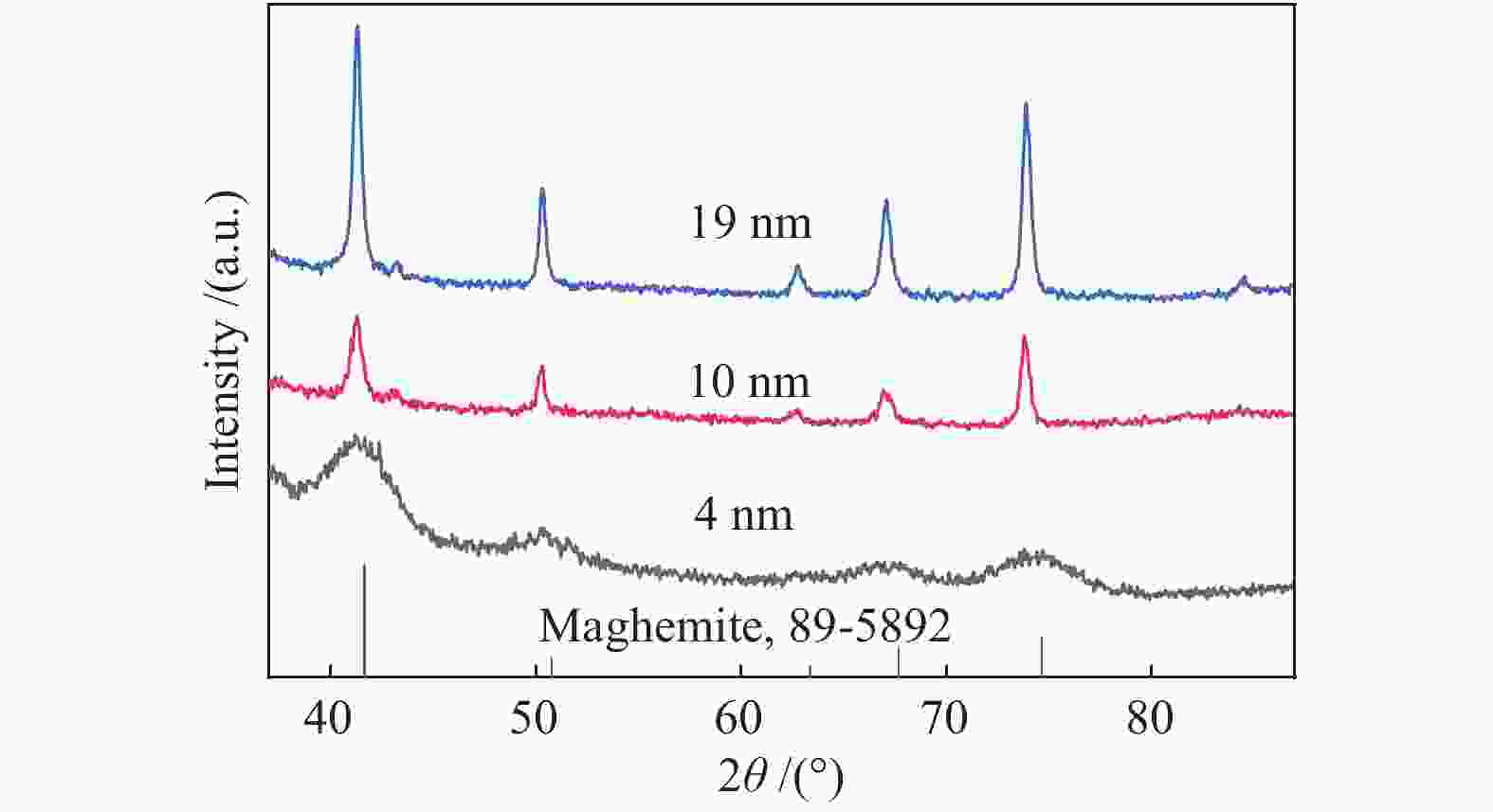
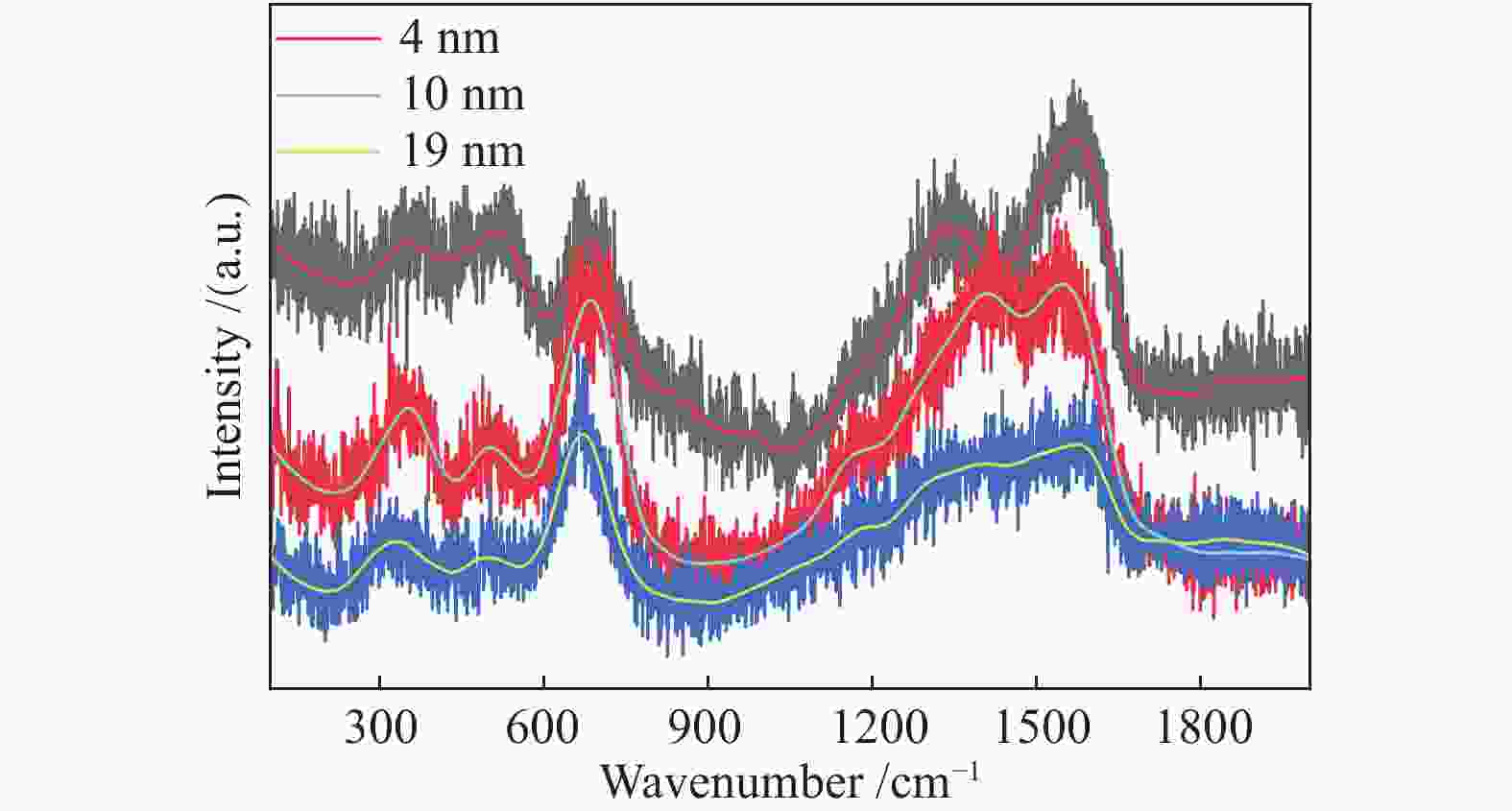
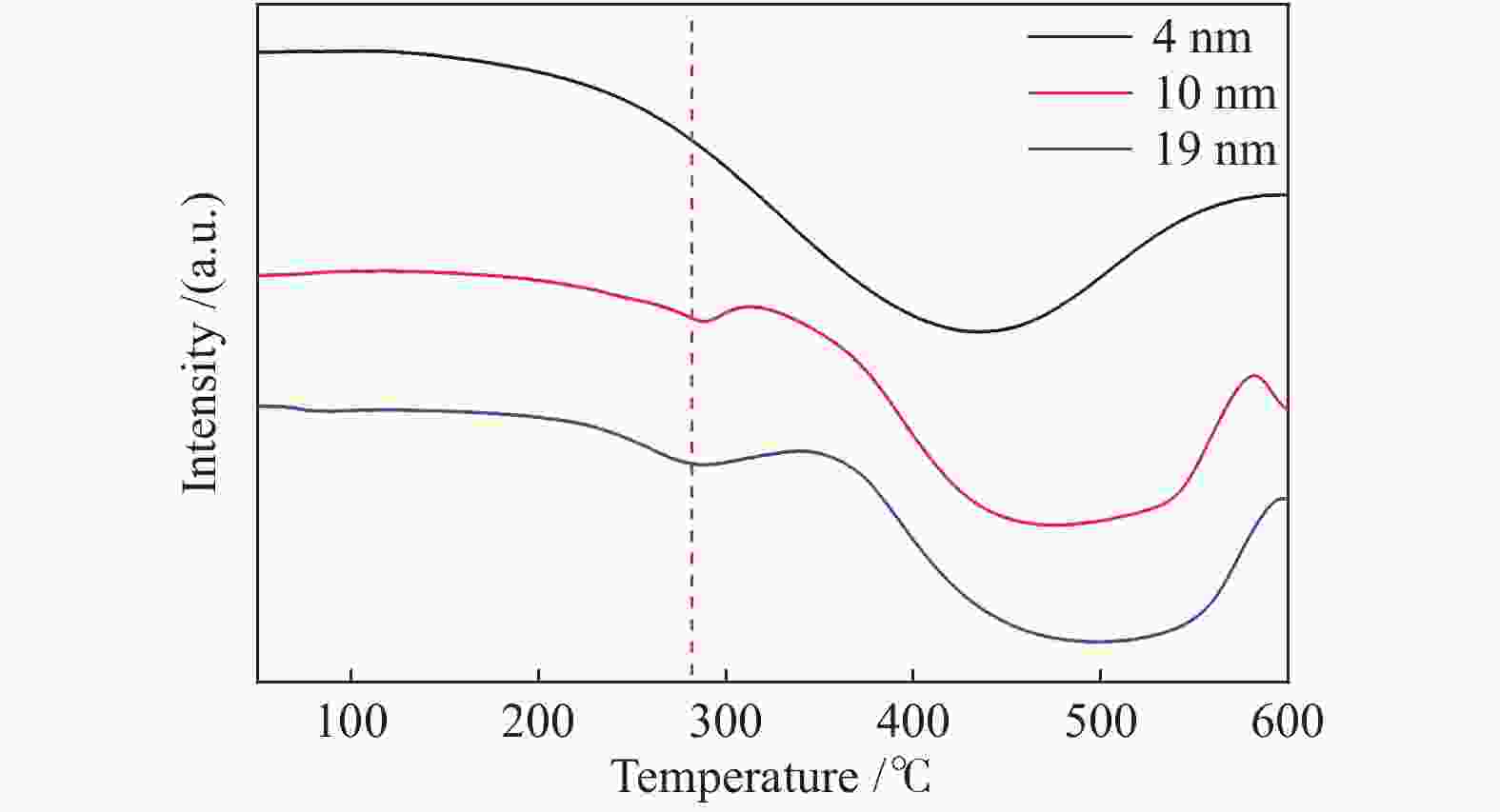
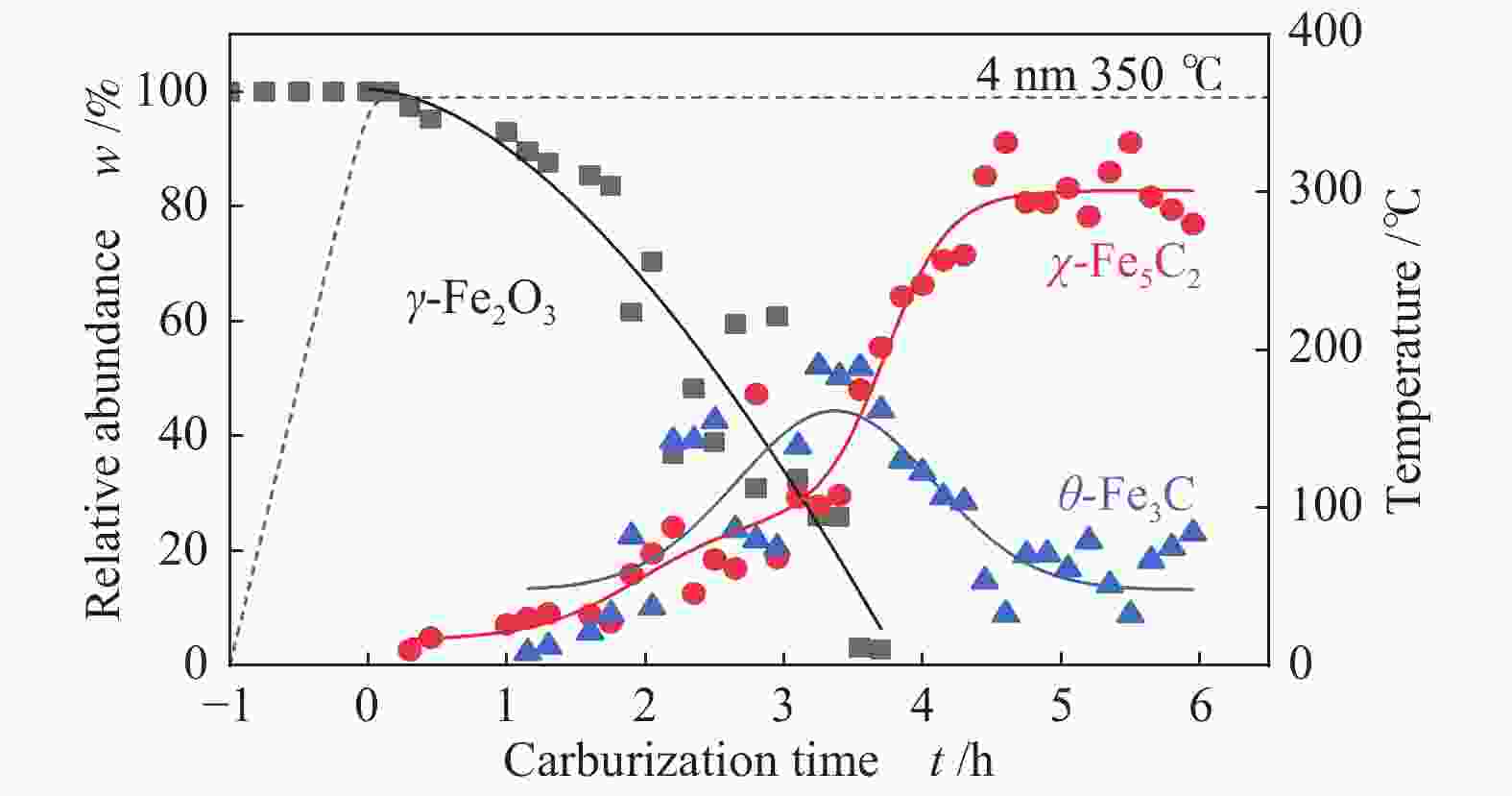
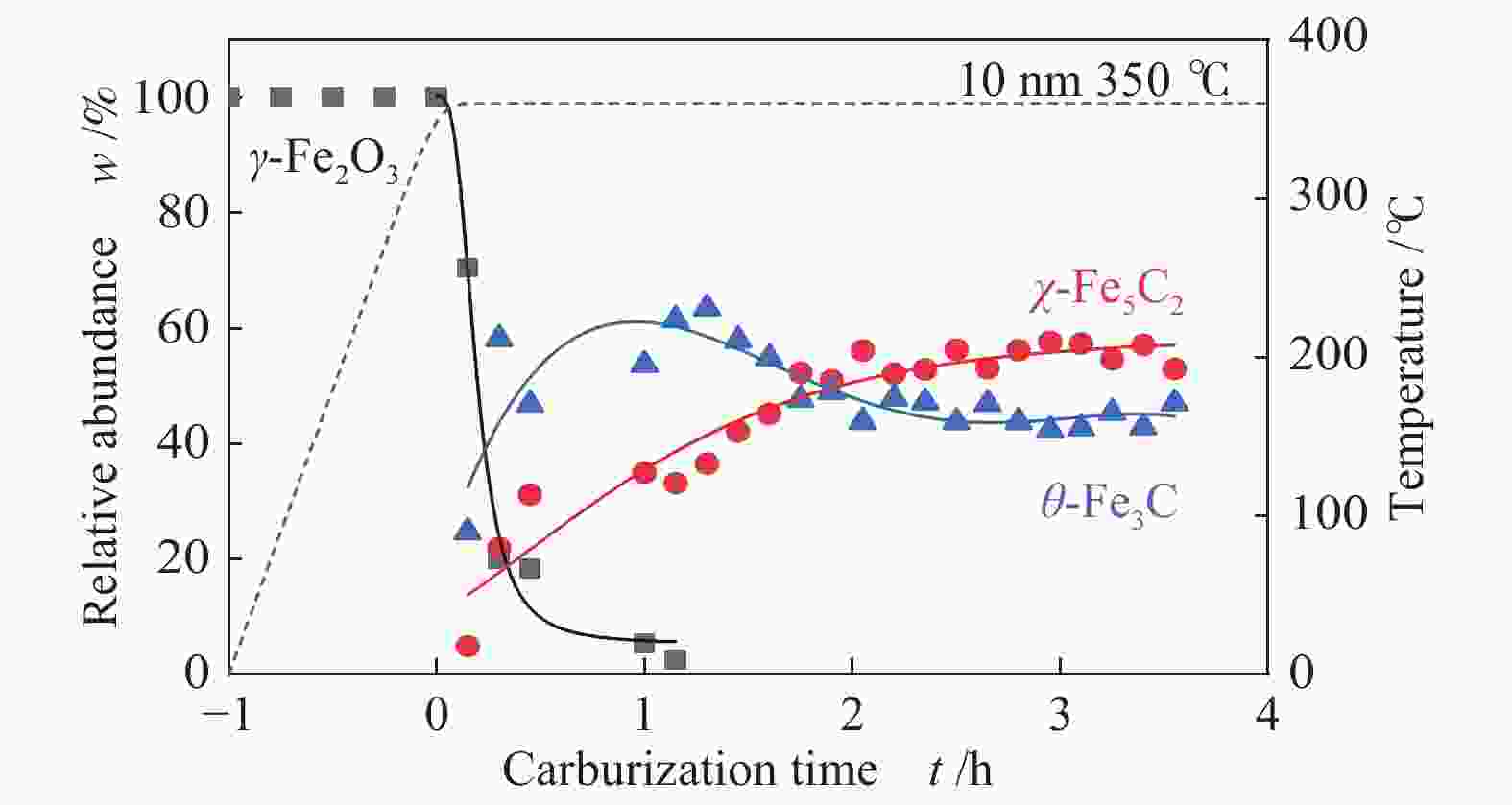
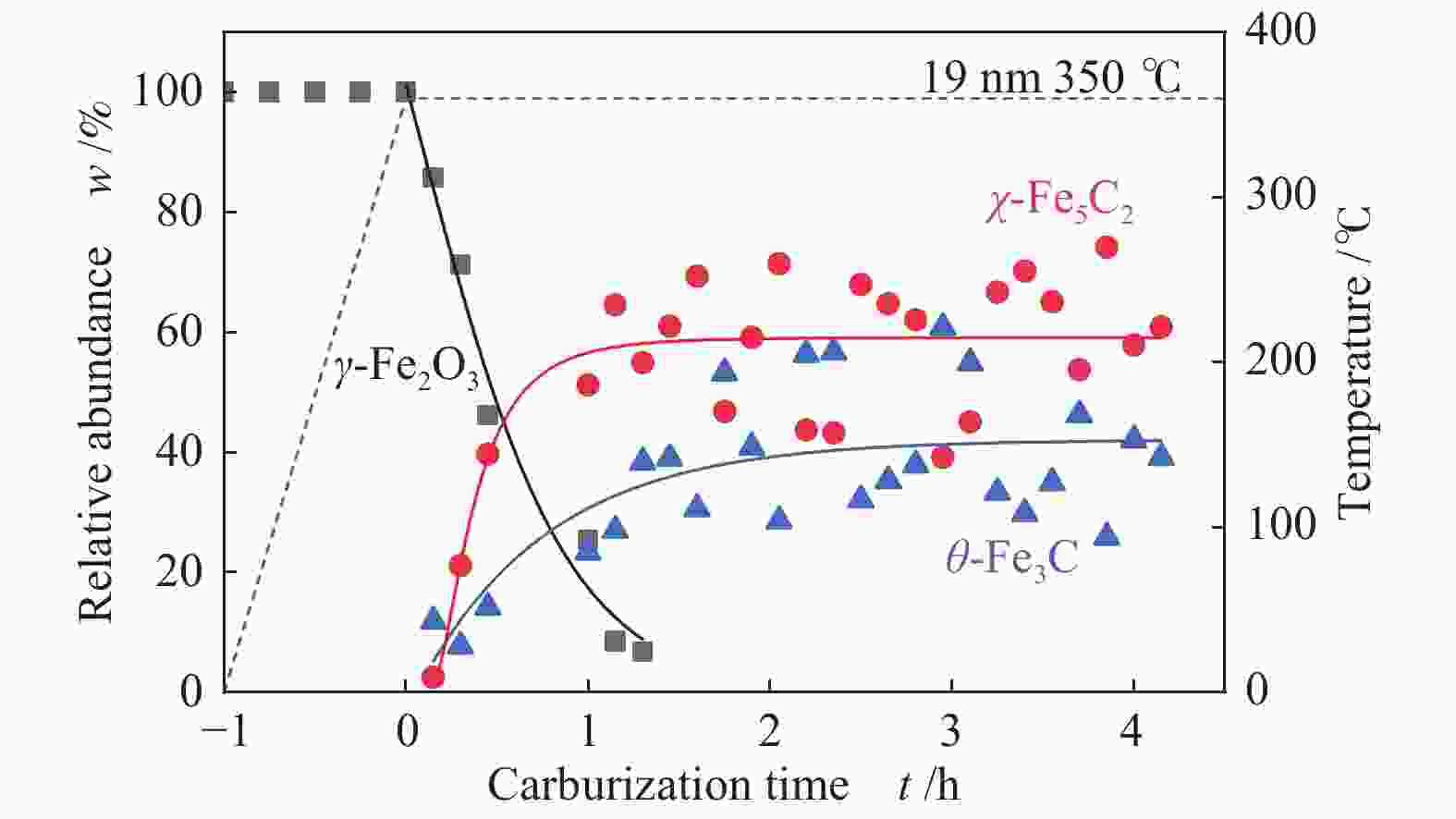
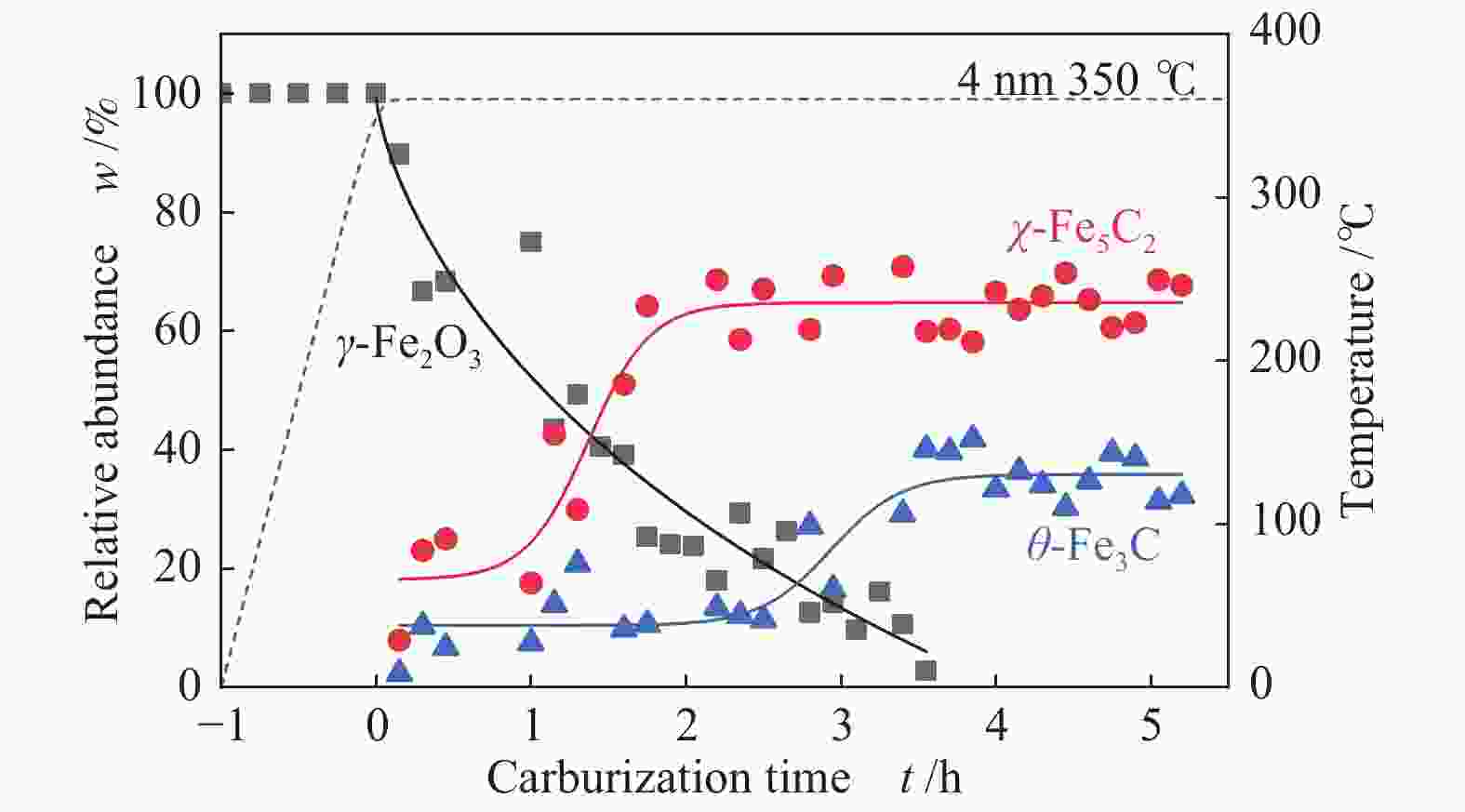
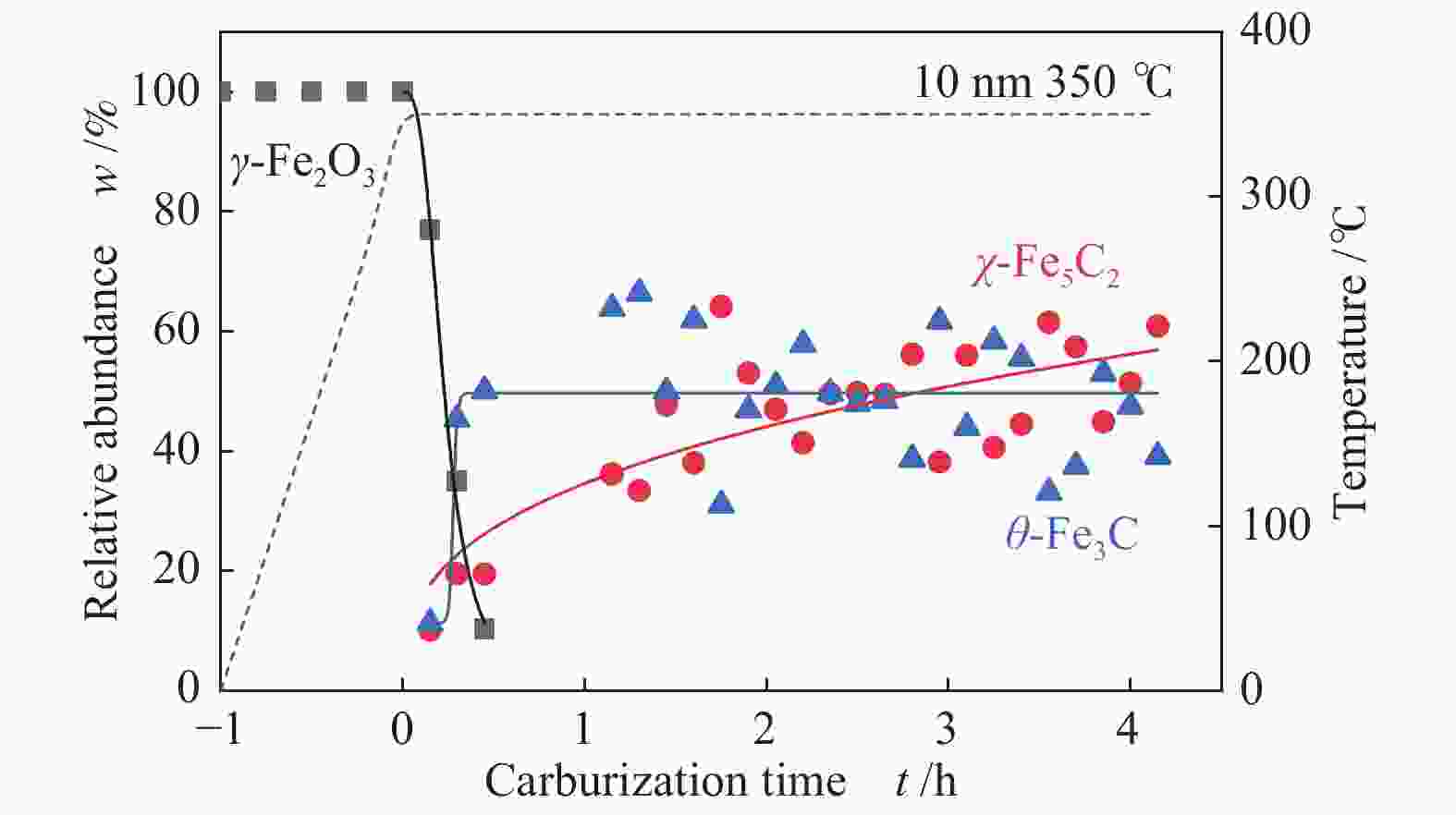
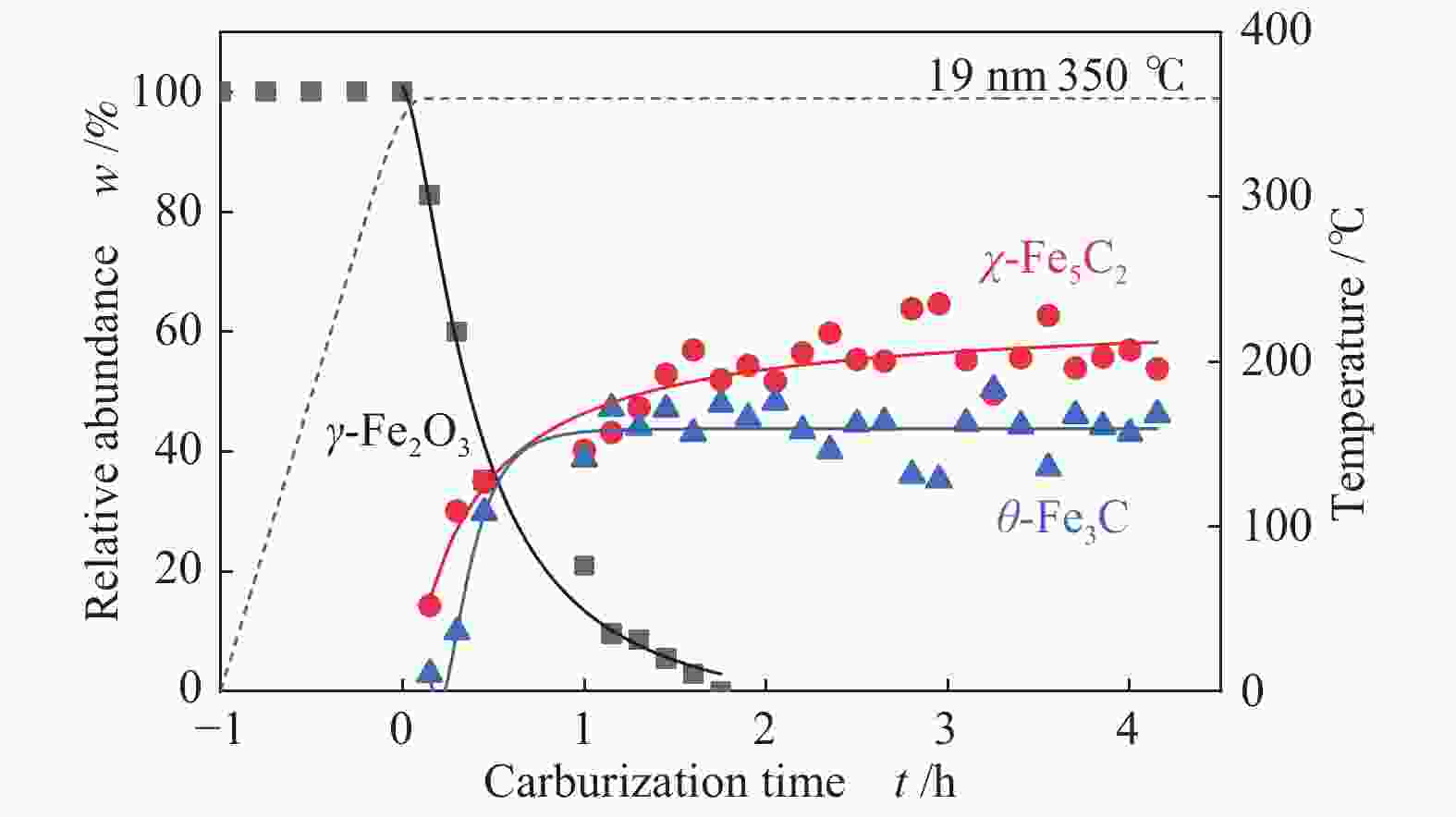
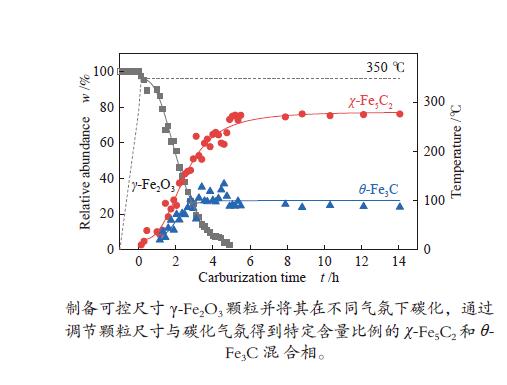
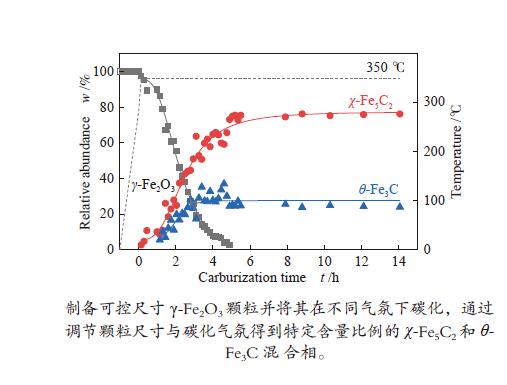
摘要: 采用油酸铁热分解法制备出不同尺寸(4−19 nm)的γ-Fe2O3纳米颗粒,在350 ℃下,于5%CO/He、5%CO/10%H2/He和5%CO/20%H2/He的三种气氛中,使用原位XRD反应装置研究了γ-Fe2O3纳米颗粒的碳化过程与物相变化规律,同时结合Raman、CO-TPR和TEM等手段对样品进行了表征。结果表明,γ-Fe2O3纳米颗粒完全碳化后会形成稳定比例的χ-Fe5C2和θ-Fe3C的混合相;在相同碳化气氛下,随γ-Fe2O3颗粒尺寸增大完全碳化所需时间缩短,尺寸较小的γ-Fe2O3颗粒表面残留炭较多,会抑制碳化反应进程,碳化相中θ-Fe3C相对含量随γ-Fe2O3纳米颗粒尺寸增大而增高;相同尺寸的γ-Fe2O3颗粒在不同气氛下碳化,完全碳化所需时间随H2分压增大先缩短后略有延长,碳化相中θ-Fe3C相对含量随H2分压增大而增高。通过调节γ-Fe2O3颗粒尺寸和碳化气氛可定向制得合适比例的χ-Fe5C2和θ-Fe3C混合相,这一结果有益于费托合成铁基催化剂中的铁碳化物活性相结构的优化。
-
关键词:
- γ-Fe2O3纳米颗粒 /
- 原位XRD /
- 碳化 /
- 物相变化
Abstract: Different sizes of γ-Fe2O3 nanoparticles (4−19 nm) were prepared by thermal decomposition of iron oleate and carburized in three different gas atmosphere of 5%CO/He, 5%CO/10%H2/He and 5%CO/20%H2/He at 350 ℃. The carburization process and phase transformation of γ-Fe2O3 nanoparticles were investigated by in situ XRD, Raman spectroscopy, CO-TPR and TEM. The results showed that χ-Fe5C2 and θ-Fe3C phases with a stable ratio were formed after carburization. The time to complete carburization was shortened for increasing sizes of γ-Fe2O3 particles under the same carburization atmosphere. While the smaller γ-Fe2O3 particles showed more residual carbon on the surface, which could inhibit the carburization process. The relative content of θ-Fe3C increased with the increase of the size of γ-Fe2O3 nanoparticles. For γ-Fe2O3 nanoparticles with the same sizes, the time to complete carburization in different atmospheres was firstly shortened and then slightly lengthened with the increase of H2 partial pressure, while the relative content of θ-Fe3C increased with the increase of H2 partial pressure. By adjusting the particle size of γ-Fe2O3 and the carburization atmosphere, the mixed phases of χ-Fe5C2 and θ-Fe3C can be optimized.-
Key words:
- γ-Fe2O3 nanoparticle /
- in situ XRD /
- carburization /
- phase transformation
-
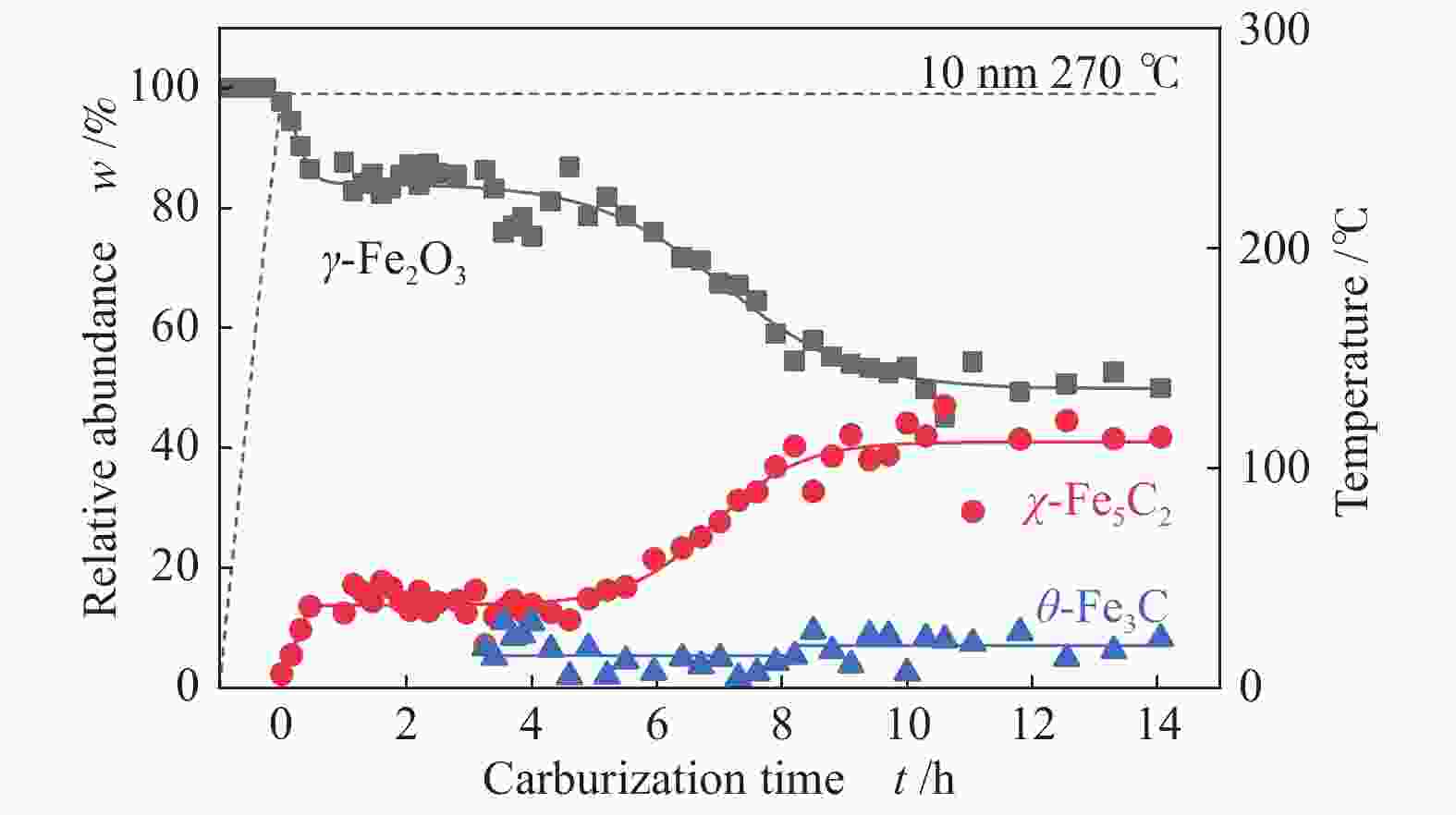
表 1 350 ℃下不同尺寸γ-Fe2O3在不同气氛下完全碳化所需时间及碳化产物分布
Table 1 Time required for complete carburization of different sizes of γ-Fe2O3 in different atmospheres and the distribution of carburization products at 350 ℃
Sample 5%CO/He w/% 5%CO/10%H2/He w/% 5%CO/20%H2/He w/% 4 nm 15 h 4 h 3.5 h Fe5C2 (0) Fe5C2 (85%) Fe5C2 (65%) Fe3C (0) Fe3C (15%) Fe3C (35%) 10 nm 5 h 1 h 0.5 h Fe5C2 (80%) Fe5C2 (60%) Fe5C2 (50%) Fe3C (20%) Fe3C (40%) Fe3C (50%) 19 nm 3 h 1.3 h 1.3 h Fe5C2 (20%) Fe5C2 (60%) Fe5C2 (55%) Fe3C (80%) Fe3C (40%) Fe3C (45%) -
[1] VAN STEEN E, CLAEYS M. Fischer-Tropsch catalysts for the biomass to liquid process[J]. Chem Eng Technol,2008,31(5):655−666. doi: 10.1002/ceat.200800067 [2] YANG Y, XU J, LIU Z Y, GUO Q, YE M, WANG G, GAO J, WANG J, ZHU Z, GE W, LIU Z, WANG F, LI Y. Progress in coal chemical technologies of China[J]. Rev Chem Eng,2020,36(1):21−66. [3] 温晓东, 杨勇, 相宏伟, 焦海军, 李永旺. 费托合成铁基催化剂的设计基础: 从理论走向实践[J]. 中国科学:化学,2017,47(11):1298−1311. doi: 10.1360/N032017-00111WEN Xiao-dong, YANG Yong, XIANG Hong-wei, JIAO Hai-jun, LI Yong-wang. The design principle of iron-based catalysts for fischer-tropsch synthesis: from theory to practice[J]. Sci Sin Chim,2017,47(11):1298−1311. doi: 10.1360/N032017-00111 [4] DICTOR R A, BELL A T. Fischer-Tropsch synthesis over reduced and unreduced iron-oxide catalysts[J]. J Catal,1986,97(1):121−136. doi: 10.1016/0021-9517(86)90043-6 [5] TORRES GALVIS H M, BITTER J H, KHARE C B, RUITENBEEK M, DUGULAN A L, DE JONG K P. Supported iron nanoparticles as catalysts for sustainable production of lower olefins[J]. Science,2012,335(6070):835−838. doi: 10.1126/science.1215614 [6] 定明月, 杨勇, 相宏伟, 李永旺. 费托合成Fe基催化剂中铁物相与活性的关系[J]. 催化学报,2010,31(9):1145−1150.DING Ming-yue, YANG Yong, XIANG Hong-wei, LI Yong-wang. Relationship between iron phase and activity of iron-based Fischer-Tropsch synthesis catalyst[J]. Chin J Catal,2010,31(9):1145−1150. [7] BIAN G Z, OONUKI A, KOIZUMI N, NOMOTO H, YAMADA M. Studies with a precipitated iron Fischer-Tropsch catalyst reduced by H2 or CO[J]. J Mol Catal A: Chem,2002,186(1/2):203−213. doi: 10.1016/S1381-1169(02)00186-3 [8] YANG C, ZHAO H, HOU Y, MA D. Fe5C2 nanoparticles: A facile bromide-induced synthesis and as an active phase for Fischer-Tropsch synthesis[J]. J Am Chem Soc,2012,134(38):15814−15821. doi: 10.1021/ja305048p [9] MA C, ZHANG W, CHANG Q, WANG X, WANG H, CHEN H, WEI Y, ZHANG C, XIANG H, YANG Y, LI Y. θ-Fe3C dominated Fe@C core-shell catalysts for Fischer-Tropsch synthesis: roles of θ-Fe3C and carbon shell[J]. J Catal,2021,393:238−246. doi: 10.1016/j.jcat.2020.11.033 [10] LO J M H, ZIEGLER T. Density functional theory and kinetic studies of methanation on iron surface[J]. J Phys Chem C,2007,111(29):11012−11025. doi: 10.1021/jp0722206 [11] PARK J, AN K J, HWANG Y S, PARK J G, NOH H J, KIM J Y, PARK J H, HWANG N M, HYEON T W. Ultra-large-scale syntheses of monodisperse nanocrystals[J]. Nat Mater,2004,3(12):891−895. doi: 10.1038/nmat1251 [12] YU W W, FALKNER J C, YAVUZ C T, COLVIN V L. Synthesis of monodisperse iron oxide nanocrystals by thermal decomposition of iron carboxylate salts[J]. Chem Commun,2004,20:2306−2307. [13] KIM B H, LEE N, KIM H, AN K, PARK Y I, CHOI Y, SHIN K, LEE Y, KWON S G, NA H B, PARK J G, AHN T Y, KIM Y W, MOON W K, CHOI S H, HYEON T. Large-scale synthesis of uniform and extremely small-sized iron oxide nanoparticles for high-resolution T1 magnetic resonance imaging contrast agents[J]. J Am Chem Soc,2011,133(32):12624−12631. doi: 10.1021/ja203340u [14] 郭天雨, 刘粟侥, 青明, 冯景丽, 吕振刚, 王洪, 杨勇. 原位XRD反应装置下H2O对Fe5C2的物相及F-T反应性能影响的研究[J]. 燃料化学学报,2020,48(1):75−82. doi: 10.3969/j.issn.0253-2409.2020.01.009GUO Tian-yu, LIU Su-yao, QING Ming, FENG Jing-li, LV Zhen-gang, WANG Hong, YANG Yong. In situ XRD study of the effect of H2O on Fe5C2 phase and Fischer-Tropsch performance[J]. J Fuel Chem Technol,2020,48(1):75−82. doi: 10.3969/j.issn.0253-2409.2020.01.009 [15] LI C, STAIR P C. An advance in Raman studies of catalysts: Ultraviolet resonance Raman spectroscopy[J]. Stud Surf Sci Catal,1996,101:881−890. [16] BUTOVSKY E, PERELSHTEIN I, GEDANKEN A. Air stable core-shell multilayer metallic nanoparticles synthesized by RAPET: Fabrication, characterization and suggested applications[J]. J Mater Chem,2012,22(30):15025−15030. doi: 10.1039/c2jm32528d [17] 孙峰. Rietveld方法精修及定量分析研究[D]. 青岛: 中国海洋大学, 2009.SUN Feng. Research of Rietveld method in refinement of crystal structure and quantitative phase analysis[D]. Qingdao: Ocean University of China, 2009. [18] ZHAO S, LIU X, HUO C, LI Y, WANG J, JIAO H. Surface morphology of Hagg iron carbide (χ-Fe5C2) from ab initio atomistic thermodynamics[J]. J Catal,2012,294:47−53. doi: 10.1016/j.jcat.2012.07.003 [19] DE S E, CINQUINI F, BEALE A M, SAFONOVA O V, BEEK W V, SAUTET P, WECKHUYSEN B M. Stability and reactivity of ε-χ-θ iron carbide catalyst phases in Fischer-Tropsch synthesis: controlling μc[J]. J Am Chem Soc,2010,132(42):14928−14941. doi: 10.1021/ja105853q [20] 刘兴武. 铁碳化合物的制备、物相转变以及费托反应性能研究[D]. 北京: 中国科学院大学, 2016.LIU Xing-wu. Preparation, phase transition and properties of iron carbides during Fischer-Tropsch synthesis[D]. Beijing: University of Chinese Academy of Sciences, 2016. [21] COHN E M, HPFER L J E. Some thermal reactions of the higher iron carbides[J]. J Chem Phys,1953,21(2):354−359. doi: 10.1063/1.1698884 -




 下载:
下载: