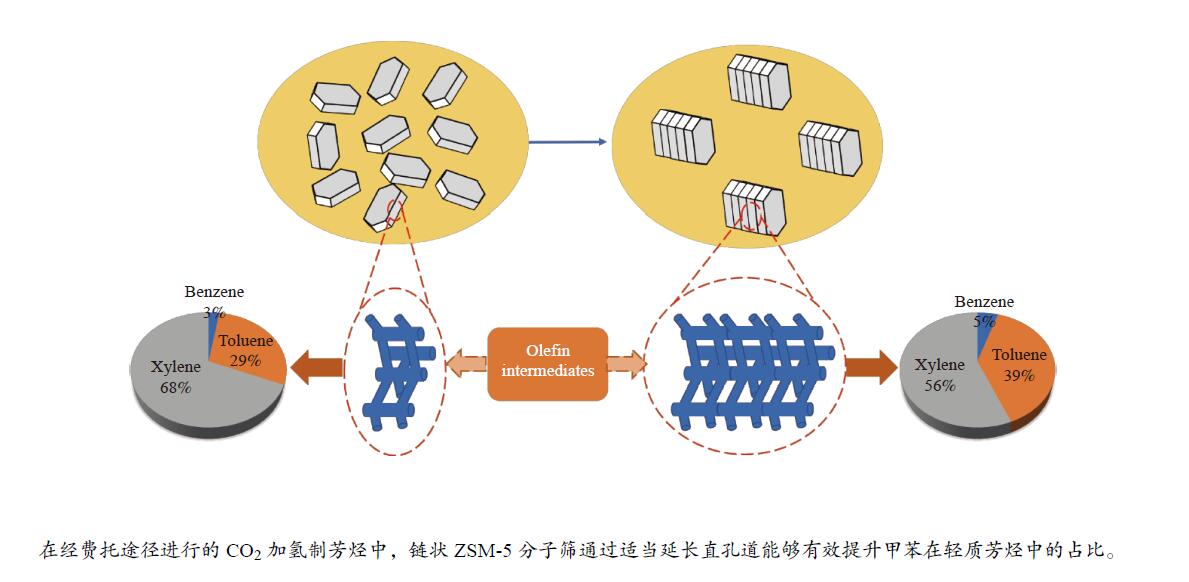
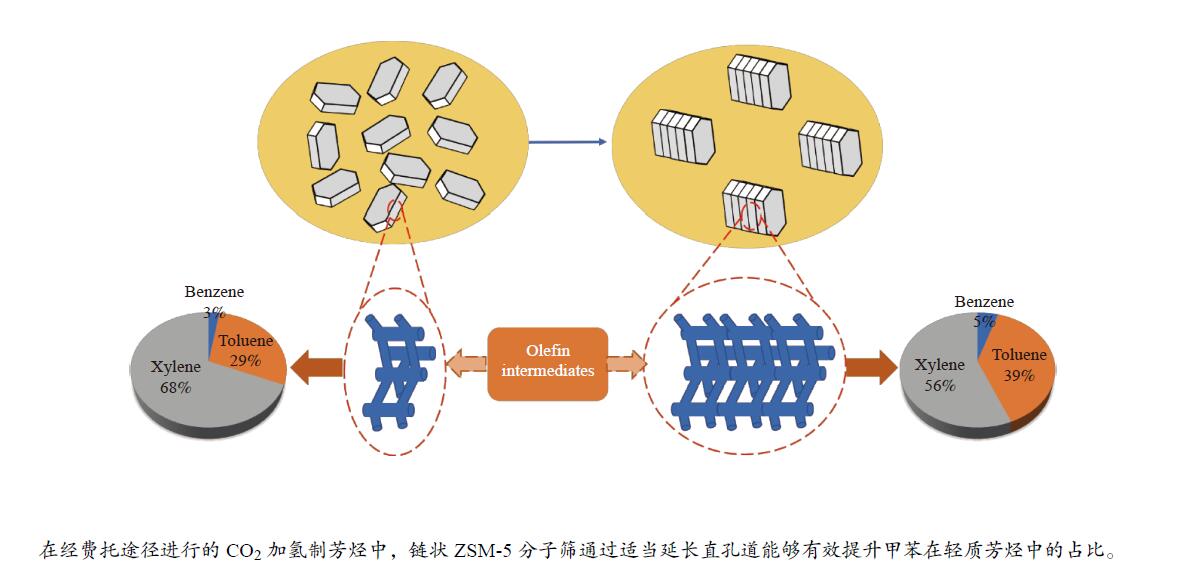
Chain-like ZSM-5 zeolite coupled with Cu-Fe3O4 for CO2 hydrogenation to light aromatics
-
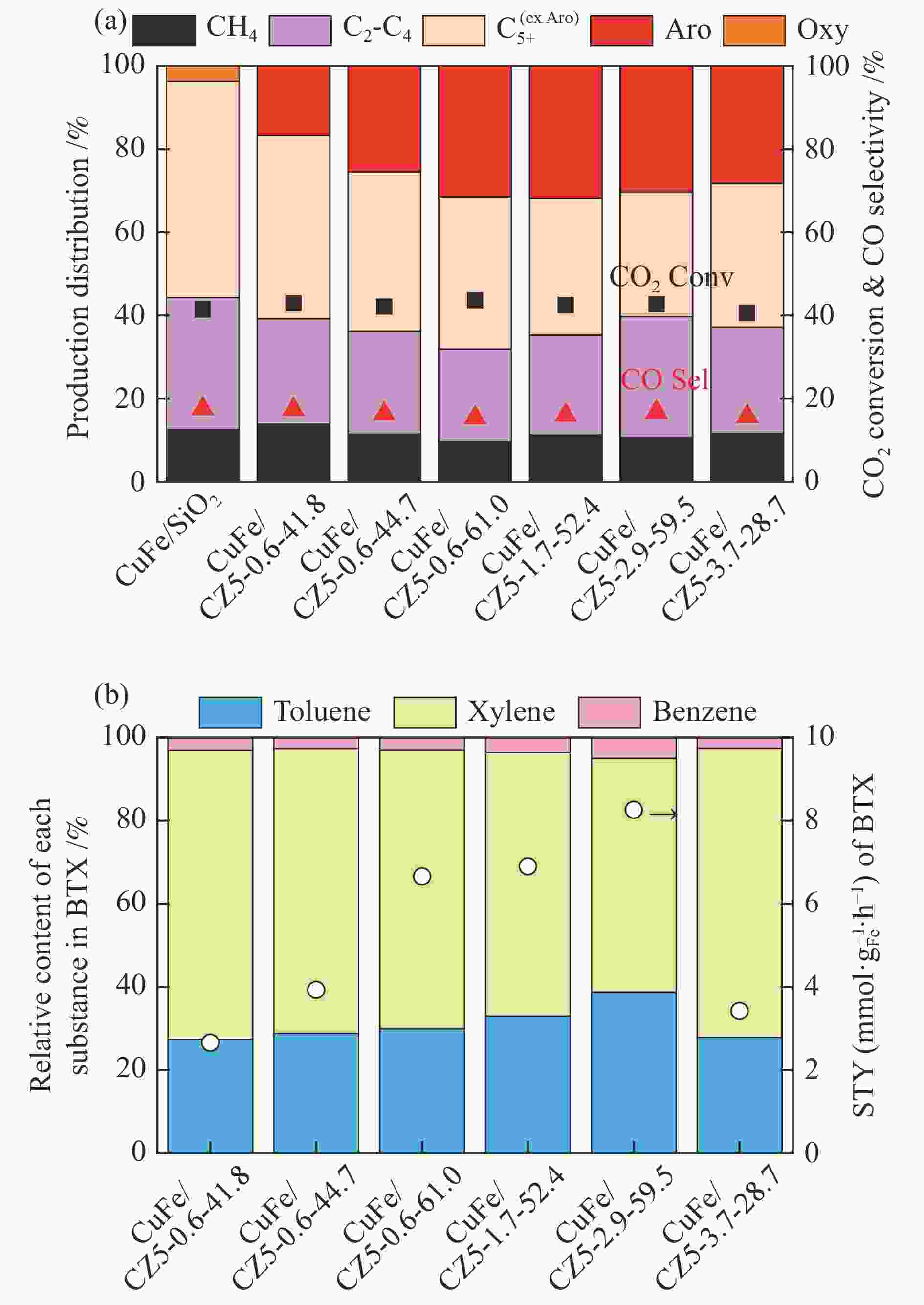
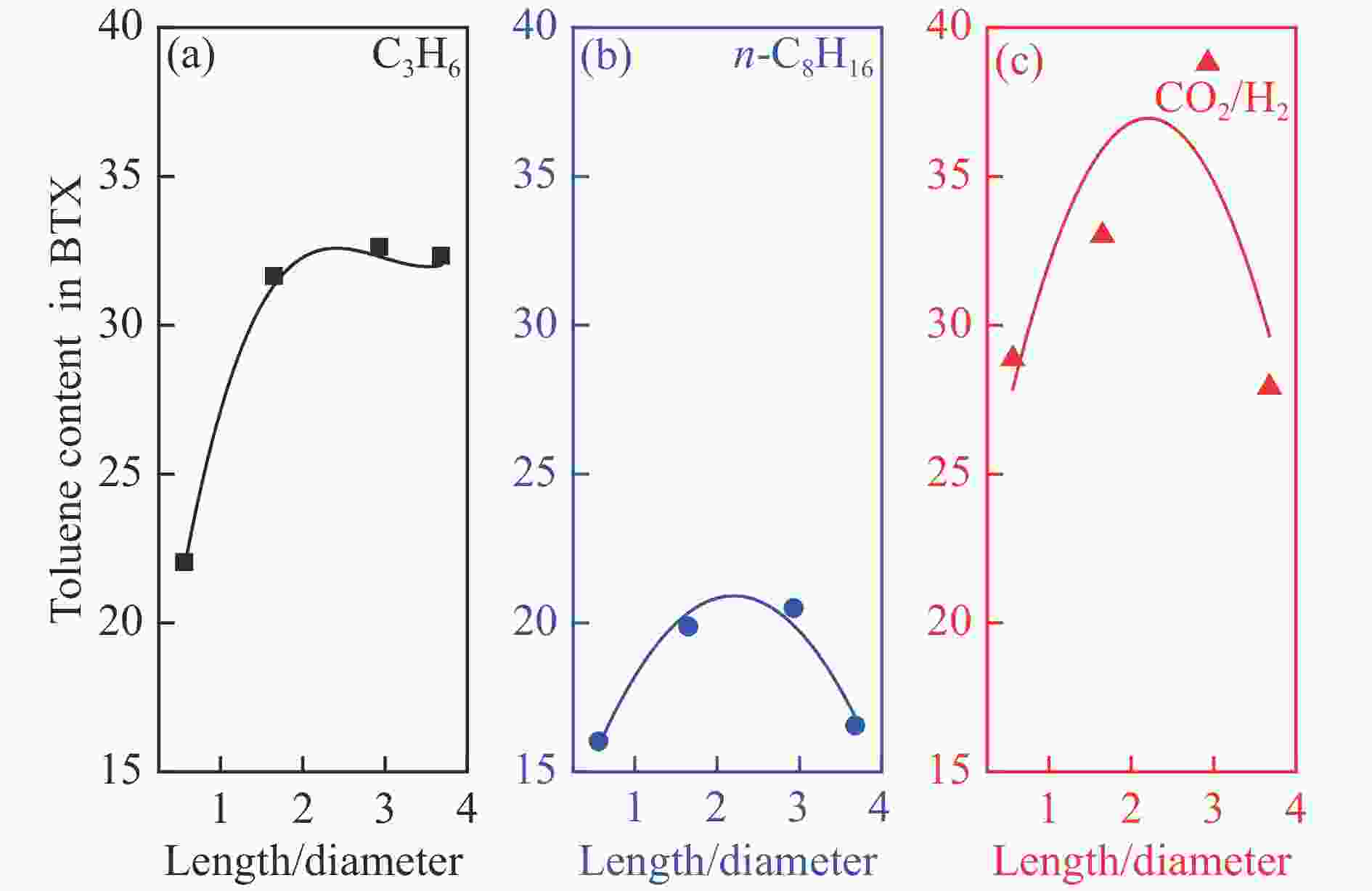
摘要: 近年来,二氧化碳(CO2)的捕获与利用受到广泛关注,尤其CO2加氢直接制轻质芳烃(如苯、甲苯和二甲苯等)已被当作一条具有潜力的化学品生产路线,但有效调控芳烃产物分布及提高目标芳烃选择性仍是一个巨大的挑战。在此,本研究提供了一种由铜改性铁基催化剂耦合链状ZSM-5分子筛组成的双功能催化剂用于CO2加氢一步高效制备轻质芳烃。采用了XRD、SEM、TEM、ICP-AES、Py-FTIR和N2吸附-脱附等表征手段对双功能催化剂组分进行了分析,研究了分子筛酸密度和长径比(即b轴/a轴)对芳烃选择性及分布的影响。结果表明,具有高酸密度及适当长径比的链状ZSM-5分子筛能促进产物的C−C偶联并抑制CH4的生成,从而有效提高芳烃的选择性及甲苯的时空收率(STY)。Abstract: The capture and utilization of carbon dioxide (CO2) have attracted much attention in recent years; in particular, the direct hydrogenation of CO2 to light aromatics has been considered as a potential route to produce high value-added chemicals. However, it is still a big challenge to adjust the aromatic distribution and achieve a high selectivity to the targeted products. In this work, a bifunctional catalyst that combines the Cu-modified Fe3O4 and the chain-like ZSM-5 zeolite is used for the hydrogenation of CO2 to light aromatics. The catalyst components were characterized by XRD, SEM, TEM, ICP-AES, Py-IR and N2 adsorption-desorption; the effect of acid density and length-to-diameter ratio (b-axis/ a-axis) of zeolite moiety on the selectivity and distribution of aromatic products was then investigated. The results indicate that the chain-like ZSM-5 zeolite moiety with high acid density and appropriate length-to-diameter ratio can promote the C–C coupling for CO2 hydrogenation and inhibit the formation of CH4, which can improve the selectivity to aromatics and the space time yield (STY) of toluene.
-
Key words:
- carbon dioxide hydrogenation /
- aromatics /
- ZSM-5 zeolite /
- Cu-Fe3O4 /
- bifunctional catalyst
1) 共同第一作者 -
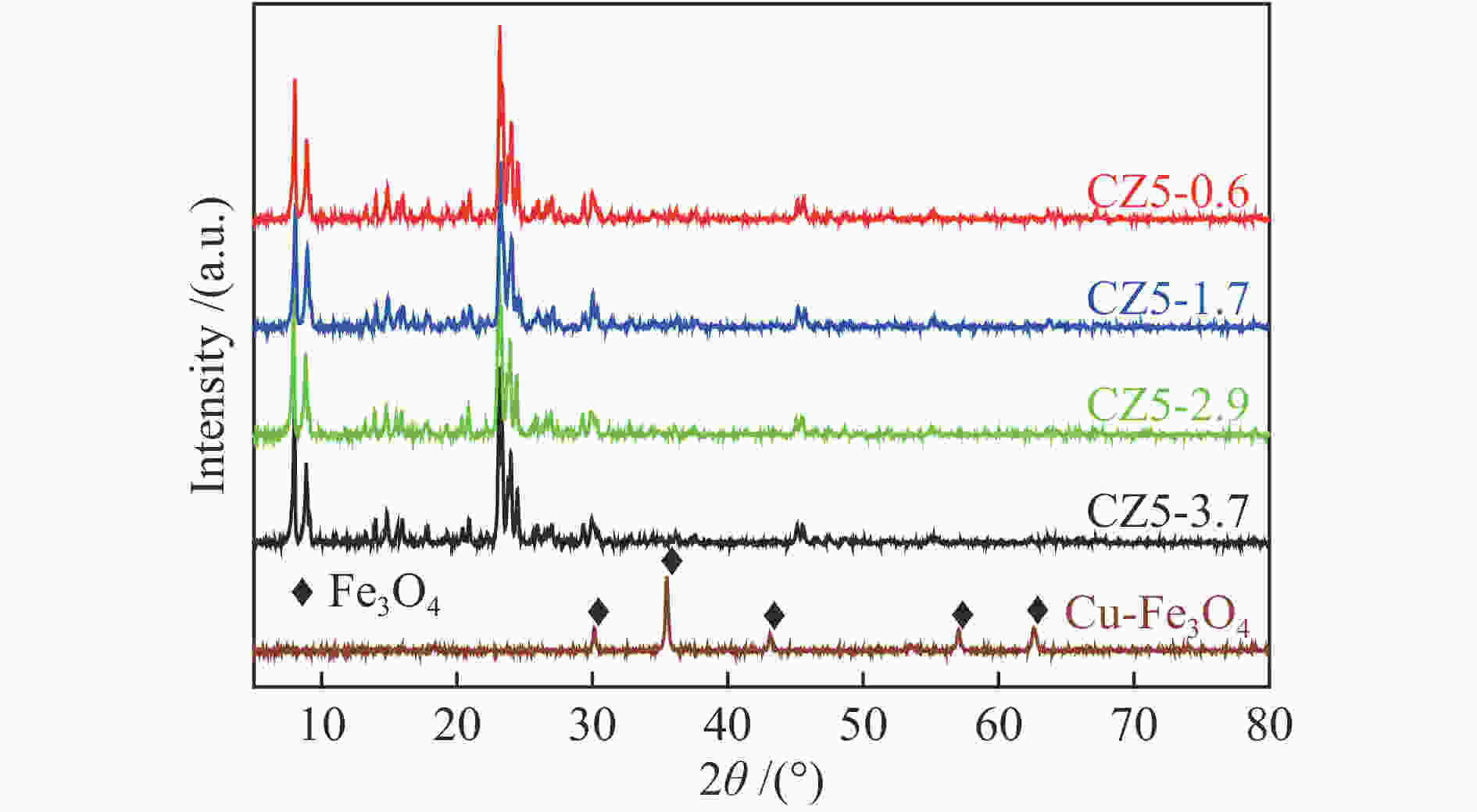
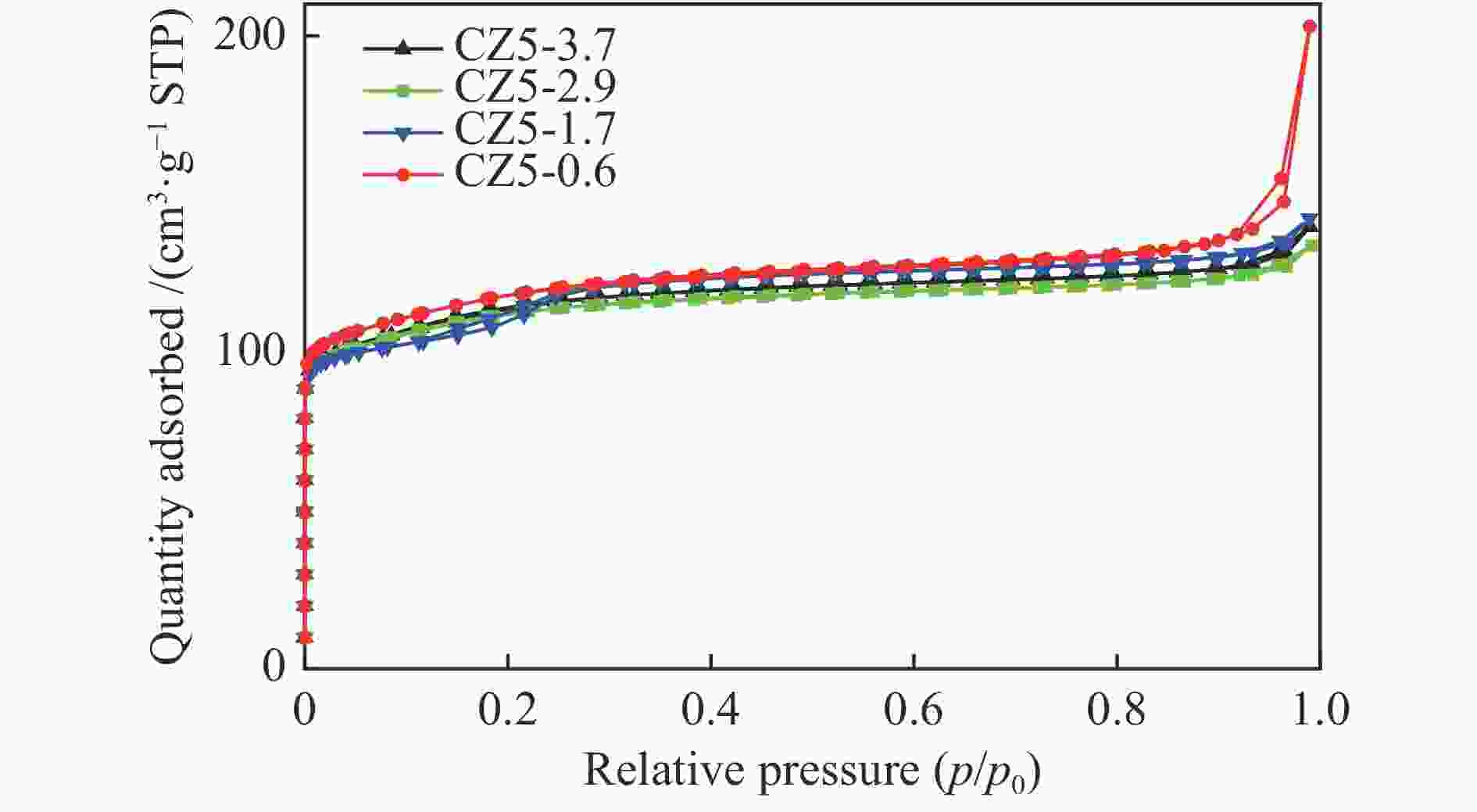
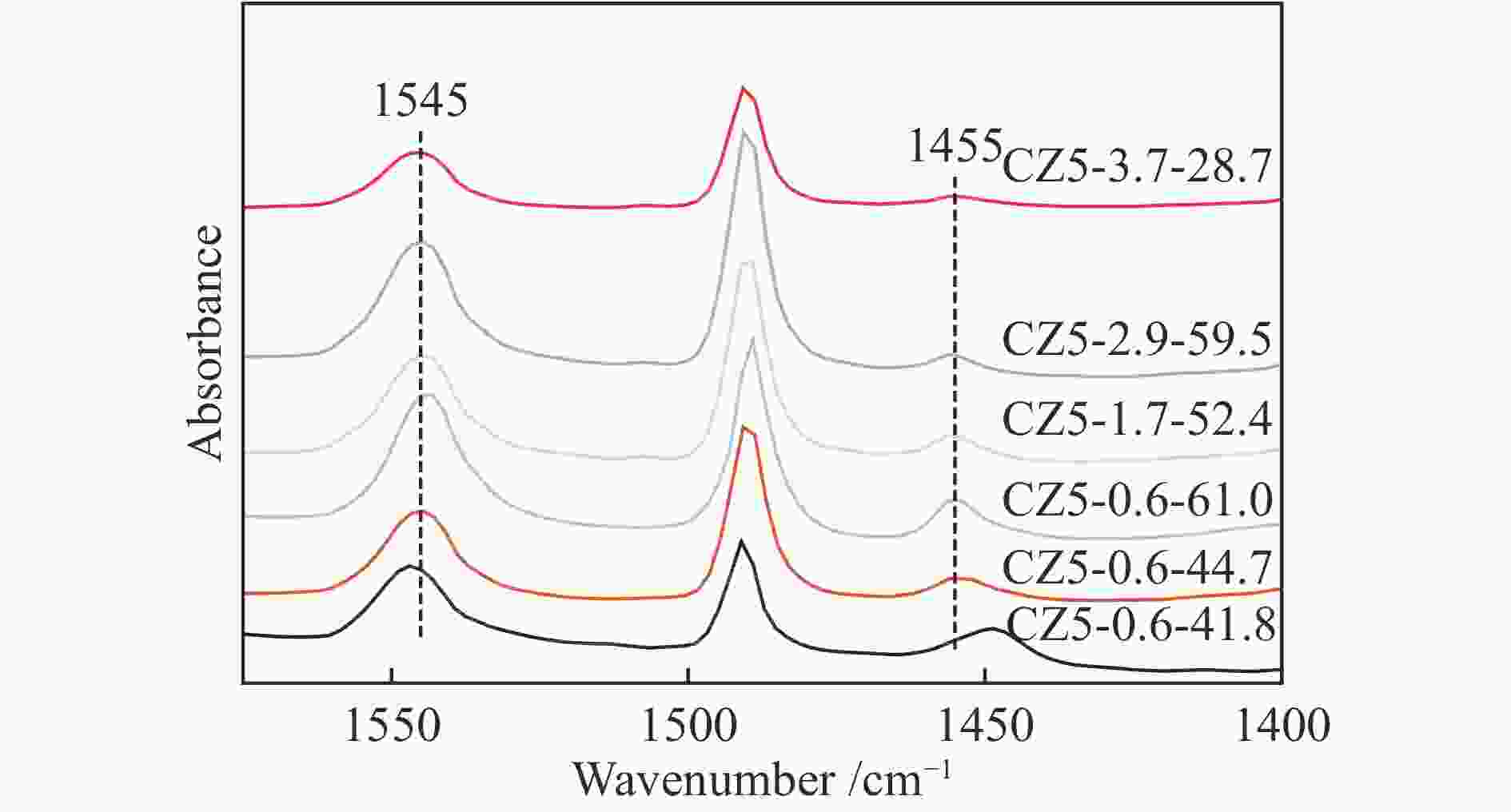
表 1 CZ5样品物理化学性质
Table 1 Physicochemical properties of various CZ5 zeolite samples
Catalyst Si/Al* Lewis acidity/(μmol·g−1) Brønsted acidity/(μmol·g−1) Total acidity/
(μmol·g−1)SBET
/(m2·g−1)CZ5-0.6-41.8 165 9.1 32.7 41.8 − CZ5-0.6-44.7 83 5.2 39.5 44.7 398.8 CZ5-0.6-61.0 61 8.8 52.2 61.0 − CZ5-1.7-52.4 88 4.3 48.1 52.4 377.6 CZ5-2.9-59.5 85 3.1 56.4 59.5 376.3 CZ5-3.7-28.7 90 2.3 26.5 28.7 369.7 *Calculated from ICP-AES -
[1] MAC DOWELL N, FENNELL P S, SHAH N, MAITLAND G C. The role of CO2 capture and utilization in mitigating climate change[J]. Nat Clim Change,2017,7(4):243−249. doi: 10.1038/nclimate3231 [2] RA E C, KIM K Y, KIM E H, LEE H, AN K, LEE J S. Recycling carbon dioxide through catalytic hydrogenation: Recent key developments and perspectives[J]. ACS Catal, 2020, 10(19): 11318−11345. [3] LIM X Z. How to make the most of carbon dioxide[J]. Nature,2015,526(7575):628−630. doi: 10.1038/526628a [4] GAO P, ZHANG L, LI S, ZHOU Z, SUN Y. Novel heterogeneous catalysts for CO2 hydrogenation to liquid fuels[J]. ACS Cent Sci,2020,6(10):1657−1670. doi: 10.1021/acscentsci.0c00976 [5] WEN C, JIANG J, CAI C, TIAN Z, XU X, WU J, WANG C, MA L. Single-step selective conversion of carbon dioxide to aromatics over Na-Fe3O4/hierarchical HZSM-5 zeolite catalyst[J]. Energy Fuels,2020,34(9):11282−11289. [6] WANG Y, GAO W, KAZUMI S, LI H, YANG G, TSUBAKI N. Direct and oriented conversion of CO2 into value-added aromatics[J]. Chemistry,2019,25(20):5149−5153. doi: 10.1002/chem.201806165 [7] GUO L S, CUI Y, ZHANG P P, PENG X B, YONEYAMA Y, YANG G H, TSUBAKI N. Enhanced liquid fuel production from CO2 hydrogenation: Catalytic performance of bimetallic catalysts over a two-stage reactor system[J]. ChemistrySelect,2018,3(48):13705−13711. doi: 10.1002/slct.201803335 [8] AITBEKOVA A, GOODMAN E D, WU L H, BOUBNOV A, HOFFMAN A S, GENC A, CHENG H, CASALENA L, BARE S R, CARGNELLO M. Engineering of ruthenium-iron oxide colloidal heterostructures: Improved yields in CO2 hydrogenation to hydrocarbons[J]. Angew Chem Int Ed,2019,58(48):17451−17457. doi: 10.1002/anie.201910579 [9] WILLIAMSON D L, HERDES C, TORRENTE-MURCIANO L, JONES M D, MATTIA D. N-Doped Fe@CNT for Combined RWGS/FT CO2 Hydrogenation[J]. ACS Sustainable Chem Eng,2019,7(7):7395−7402. doi: 10.1021/acssuschemeng.9b00672 [10] AMOYAL M, VIDRUK-NEHEMYA R, LANDAU M V, HERSKOWITZ M. Effect of potassium on the active phases of Fe catalysts for carbon dioxide conversion to liquid fuels through hydrogenation[J]. J Catal,2017,348:29−39. doi: 10.1016/j.jcat.2017.01.020 [11] ZHANG J F, ZHANG M, CHEN S Y, WANG X X, ZHOU Z L, WU Y Q, ZHANG T, YANG G H, HAN Y Z, TAN Y S. Hydrogenation of CO2 into aromatics over a ZnCrOx-zeolite composite catalyst[J]. Chem Commun,2019,55(7):973−976. doi: 10.1039/C8CC09019J [12] ZHANG X B, ZHANG A F, JIANG X, ZHU J, LIU J H, LI J J, ZHANG G H, SONG C S, GUO X W. Utilization of CO2 for aromatics production over ZnO/ZrO2-ZSM-5 tandem catalyst[J]. J CO2 Util,2019,29:140−145. doi: 10.1016/j.jcou.2018.12.002 [13] NI Y M, CHEN Z Y, FU Y, LIU Y, ZHU W L, LIU Z M. Selective conversion of CO2 and H2 into aromatics[J]. Nat Commun,2018,9(1):3457. [14] ZHOU C, SHI J Q, ZHOU W, CHENG K, ZHANG Q H, KANG J C, WANG Y. Highly active ZnO-ZrO2 aerogels integrated with HZSM-5 for aromatics synthesis from carbon dioxide[J]. ACS Catal,2020,10(1):302−310. doi: 10.1021/acscatal.9b04309 [15] YANG H Y, ZHANG C, GAO P, WANG H, LI X P, ZHONG L S, WEI W, SUN Y H. A review of the catalytic hydrogenation of carbon dioxide into value-added hydrocarbons[J]. Catal Sci Technol,2017,7(20):4580−4598. doi: 10.1039/C7CY01403A [16] DORNER R W, HARDY D R, WILLIAMS F W, WILLAUER H D. Heterogeneous catalytic CO2 conversion to value-added hydrocarbons[J]. Energy Environ Sci,2010,3(7):884−890. doi: 10.1039/c001514h [17] LIU J, ZHANG A, JIANG X, MIN L, SUN Y, SONG C, GUO X. Selective CO2 hydrogenation to hydrocarbons on Cu-promoted Fe-based catalysts: Dependence on Cu-Fe interaction[J]. ACS Sustainable Chem Eng,2018,6:10182−10190. doi: 10.1021/acssuschemeng.8b01491 [18] SONG G, LI M, YAN P, NAWAZ M A, LIU D. High Conversion to aromatics via CO2-FT over a CO-reduced Cu-Fe2O3 catalyst integrated with HZSM-5[J]. ACS Catal,2020,10(19):11268−11279. [19] JAE J, TOMPSETT G A, FOSTER A J, HAMMOND K D, AUERBACH S M, LOBO R F, HUBER G W. Investigation into the shape selectivity of zeolite catalysts for biomass conversion[J]. J Catal,2011,279(2):257−268. doi: 10.1016/j.jcat.2011.01.019 [20] NEZAM I, ZHOU W, GUSMAO G S, REALFF M J, WANG Y, MEDFORD A J, JONES C W. Direct aromatization of CO2 via combined CO2 hydrogenation and zeolite-based acid catalysis[J]. J CO2 Util,2021,45:101405. [21] ZHOU W, CHENG K, KANG J C, ZHOU C, SUBRAMANIAN V, ZHANG Q H, WANG Y. New horizon in C1 chemistry: Breaking the selectivity limitation in transformation of syngas and hydrogenation of CO2 into hydrocarbon chemicals and fuels[J]. Chem Soc Rev,2019,48(12):3193−3228. doi: 10.1039/C8CS00502H [22] OLSON D H, KOKOTAILO G T, LAWTON S L, MEIER W M. Crystal-structure and structure-related properties of ZSM-5[J]. J Phys Chem-Us,1981,85(15):2238−2243. doi: 10.1021/j150615a020 [23] KOKOTAILO G T, LAWTON S L, OLSON D H, OLSON D H, MEIER W M. Structure of synthetic zeolite ZSM-5[J]. Nature,1978,272(5652):437−438. doi: 10.1038/272437a0 [24] WANG N, HOU Y L, SUN W J, CAI D L, CHEN Z H, LIU L M, GE B H, HU L, QIAN W Z, WEI F. Modulation of b-axis thickness within MFI zeolite: Correlation with variation of product diffusion and coke distribution in the methanol-to hydrocarbons conversion[J]. Appl Catal B: Environ,2019,243:721−733. doi: 10.1016/j.apcatb.2018.11.023 [25] WANG T, YANG C, GAO P, ZHOU S, LI S, WANG H, SUN Y. ZnZrOx integrated with chain-like nanocrystal HZSM-5 as efficient catalysts for aromatics synthesis from CO2 hydrogenation[J]. Appl Catal B: Environ,2021,286:119929. [26] ZHANG Y L, MA L L, TU J L, WANG T J, LI X J. One-pot synthesis of promoted porous iron-based microspheres and its Fischer-Tropsch performance[J]. Appl Catal A: Gen,2015,499:139−145. doi: 10.1016/j.apcata.2015.04.017 [27] QUAN Y H, LI S Y, WANG S, LI Z K, DONG M, QIN Z F, CHEN G, WEI Z H, FAN W B, WANG J G. Synthesis of chainlike ZSM-5 zeolites: Determination of synthesis parameters, mechanism of chainlike morphology formation, and their performance in selective adsorption of xylene isomers[J]. ACS Appl Mater Inter,2017,9(17):14899−14910. doi: 10.1021/acsami.7b02738 [28] WANG Y, KAZUMI S, GAO W Z, GAO X H, LI H J, GUO X Y, YONEYAMA Y, YANG G H, TSUBAKI N. Direct conversion of CO2 to aromatics with high yield via a modified Fischer-Tropsch synthesis pathway[J]. Appl Catal B: Environ,2020,269:118792. [29] CUI X, GAO P, LI S G, YANG C G, LIU Z Y, WANG H, ZHONG L S, SUN Y H. Selective production of aromatics directly from carbon dioxide hydrogenation[J]. ACS Catal,2019,9(5):3866−3876. doi: 10.1021/acscatal.9b00640 -




 下载:
下载: