Effect of hydrolysis of tetraethyl orthosilicate (TEOS) on titanium distribution of TS-1 zeolite
-
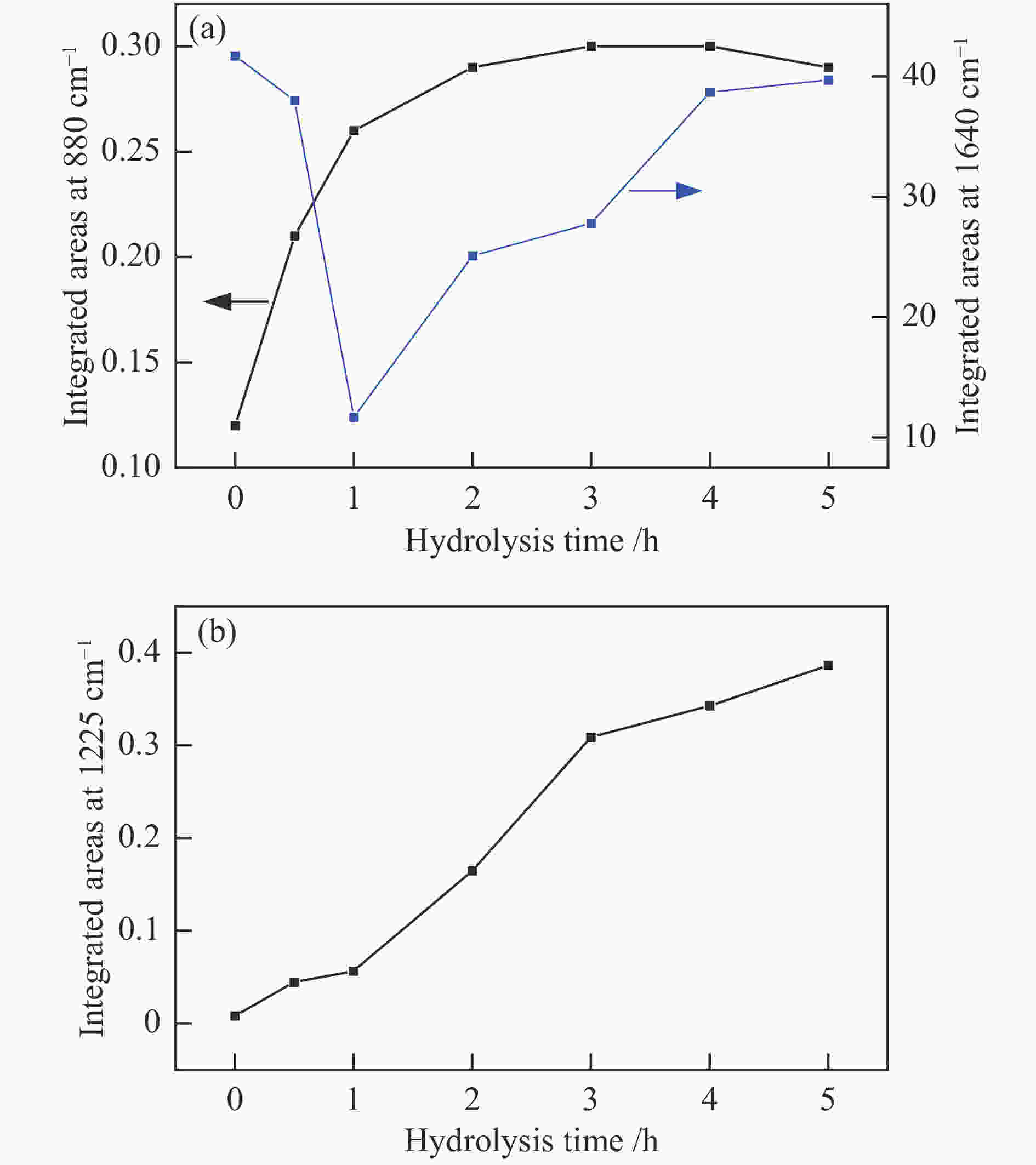
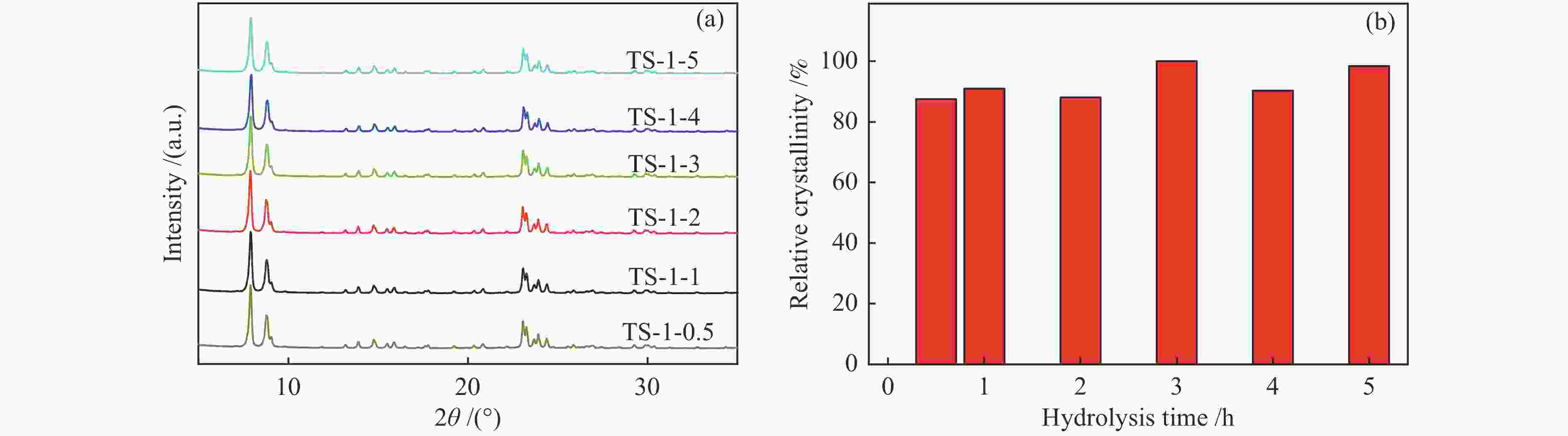
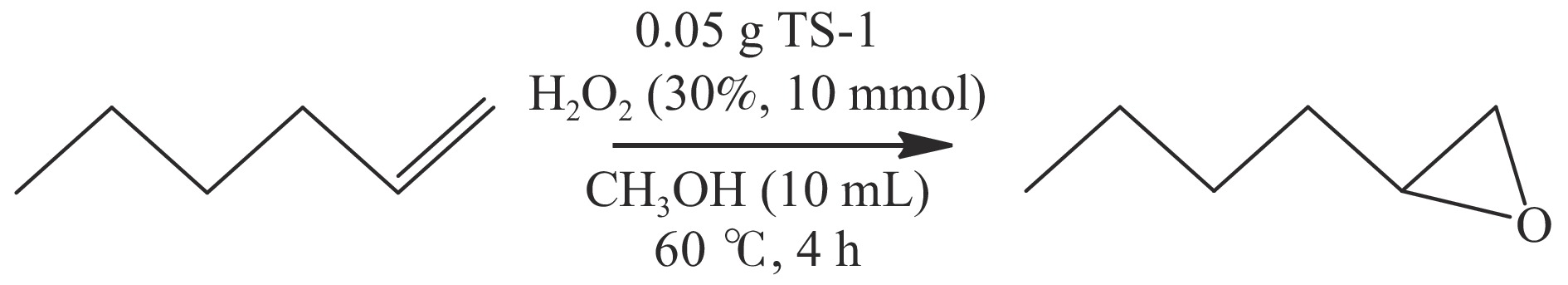
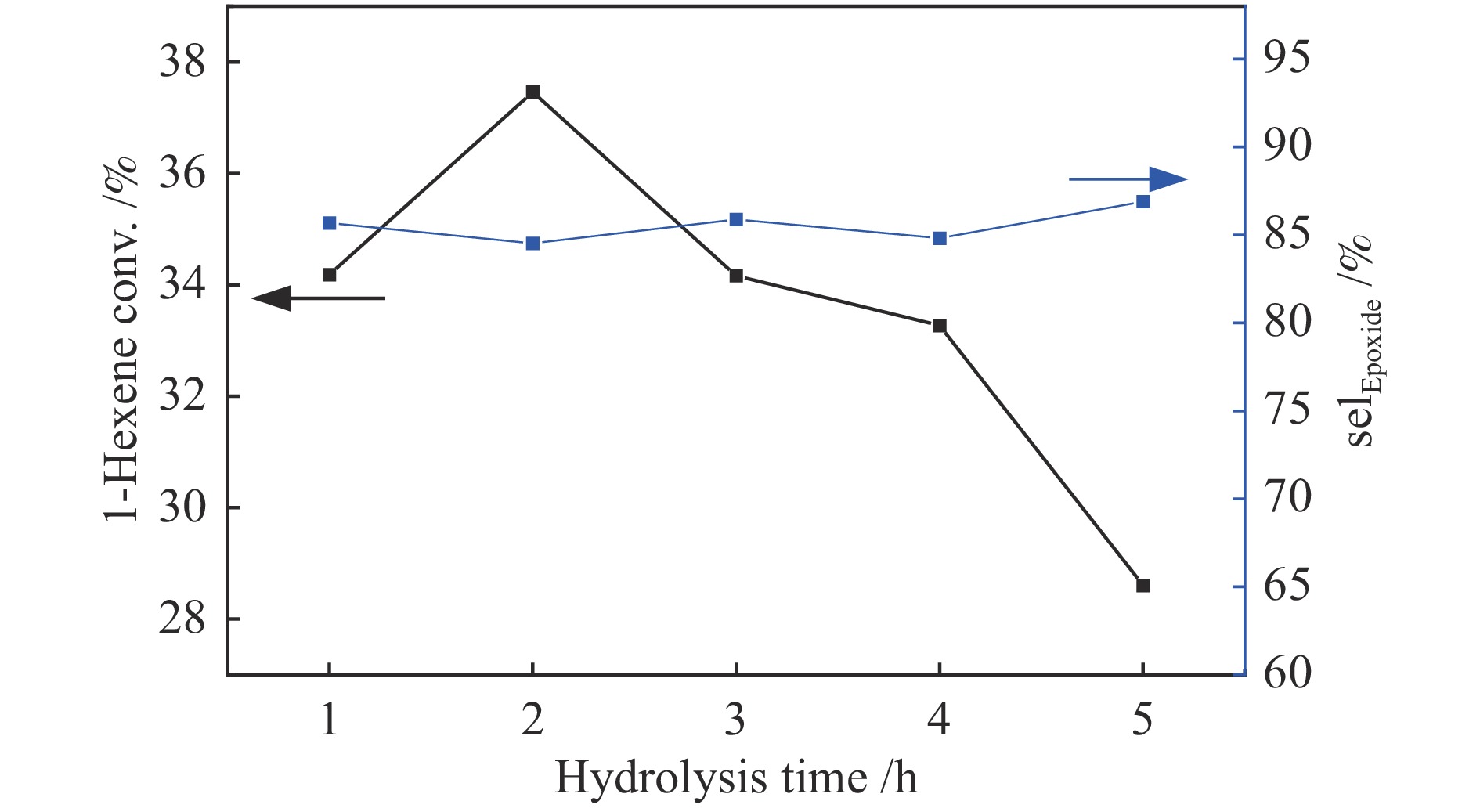
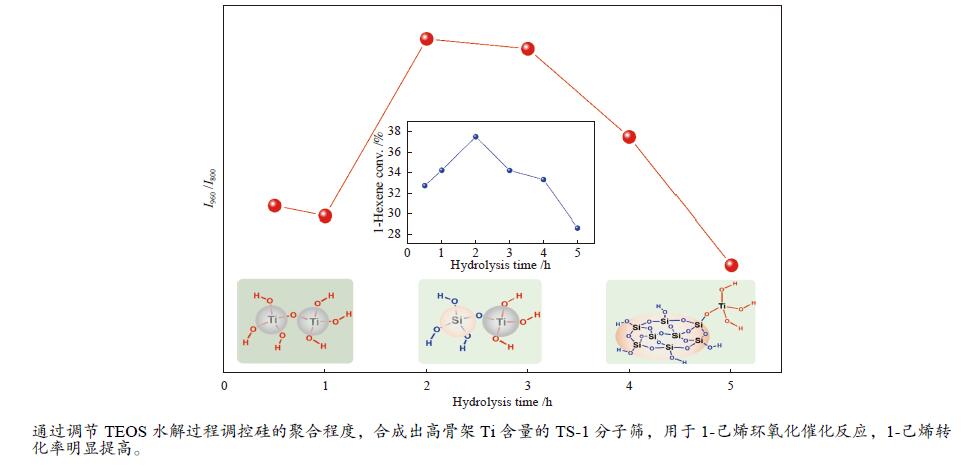
摘要: 采用FT-IR技术跟踪正硅酸四乙酯 (TEOS) 水解过程并结合XRF、UV-vis等表征探讨了水解时间对TS-1分子筛中钛物种分布的影响。以1-己烯环氧化为模型反应对TS-1分子筛的催化氧化性能进行了评价。结果表明,TEOS适度水解能有效抑制非骨架钛形成,提高骨架钛含量,而水解时间较短或过长均不利于钛进入骨架,并会明显降低TS-1分子筛的催化性能。TEOS水解2 h 后制得的TS-1分子筛具有最佳的1-己烯环氧化催化性能,在常压60 ℃反应条件下1-己烯转化率可达到37.5%,产物1,2-环氧己烷选择性可维持在85%左右,这一结果对费托烯烃环氧化催化剂的开发具有指导作用。Abstract: The hydrolysis process of tetraethyl orthosilicate (TEOS) was tracked by FT-IR analysis, and the effect of hydrolysis time on the distribution of titanium species in TS-1 zeolites was discussed through XRF and UV-Vis. The catalytic oxidation performance of TS-1 zeolite was tested by 1-hexene epoxidation as a model reaction. The results show that moderate hydrolysis of tetraethyl orthosilicate can effectively inhibit the formation of extra-framework Ti and increase the content of framework Ti. However, shorter or longer hydrolysis time is not conducive to the entry of titanium into the skeleton, and reduce the catalytic performance of TS-1 zeolites. TS-1 prepared with 2 h TEOS hydrolysis time has the best catalytic performance for epoxidation of 1-hexene. Under the reaction conditions of atmospheric pressure and 60 ℃, the conversion of 1-hexene can reach 37.5% and the selectivity of 1,2-epoxy hexane can maintain at about 85%, and the result has a guiding role for the development of olefin epoxidation catalyst.
-
Key words:
- TS-1 zeolites /
- tetraethyl orthosilicate /
- hydrolysis /
- 1-hexene epoxidation
-
表 1 不同TS-1分子筛样品的Si/Ti值和固收率
Table 1 Si/Ti values and solid yield of different TS-1 samples
Sample Solid yielda/% Si/Tib Ti/(Si+Ti)c/% Average particle sized/nm TS-1-0 0 − − − TS-1-0.5 40.1 45.3 0.049 352 TS-1-1 89.9 43.6 0.053 358 TS-1-2 90.5 48.5 0.043 366 TS-1-3 91.9 55.7 0.032 275 TS-1-4 91.8 58.0 0.030 224 TS-1-5 91.5 64.4 0.024 187 a: ${\rm{Solid}}\;{\rm{yield}} =\dfrac{{m}_{2}\times {M}_{1}}{{m}_{1}\times {M}_{2}}\times 100\%\; $, m1 is the amount of TEOS added during feeding; M1 is the relative molecular weight of TEOS, and its value is 208.33 g/mol; M2 is the relative molecular weight of SiO2, and its value is 60.08 g/mol; M2 is the mass of SiO2 actually obtained;
b, c: Si/Ti and Ti/(Si+Ti) in solid is determined by XRF;
d: Average particle size is obtained by SEM image表 2 不同TS-1分子筛样品的UV-vis分峰
Table 2 Diffuse reflectance UV-visible result of different TS-1 samples
Sample Deconvoluted bands and assignments: λmax, nm (relative intensity/%) Ti(OSi)4 (~210 nm) anatase TiO2 (~330 nm) TS-1-0.5 91.9 8.1 TS-1-1 93.5 6.5 TS-1-2 100.0 0 TS-1-3 100.0 0 TS-1-4 96.9 3.1 TS-1-5 97.3 2.7 -
[1] YIN J, ZHANG T Q, SCHULMAN E, LIU D X, MENG J Q. Hierarchical porous metallized poly-melamine formaldehyde (PMF) as low-cost and high-efficiency catalyst for cyclic carbonate synthesis from CO2 and epoxides[J]. J Mater Chem A,2018,6(18):8441−8448. doi: 10.1039/C8TA00625C [2] OGAWA H, MIYAMOTO Y, FUJIGAKI T, CHIHARA T. Ring-opening of 1, 2-epoxyalkane with alcohols over H-ZSM-5 in liquid phase[J]. Catal Lett,1996,40(3/4):253−255. [3] DING J, XU L, YU Y, WU H H, HUANG S J, YANG Y L, WU J, WU P. Clean synthesis of acetaldehyde oxime through ammoximation on titanosilicate catalysts[J]. Catal Sci Technol,2013,3(10):2587−2595. doi: 10.1039/c3cy00471f [4] WU G Q, XIAO J H, ZHANG L, WANG W J, HONG Y P, HUANG H J, JIANG Y, LI L, WANG C R. Copper-modified TS-1 catalyzed hydroxylation of phenol with hydrogen peroxide as the oxidant[J]. RSC Adv,2016,6(103):101071−101078. doi: 10.1039/C6RA20980G [5] DU Q, GUO Y P, DUAN H N, LI H, CHEN Y J, LIU H Z. Synthesis of hierarchical TS-1 zeolite via a novel three-step crystallization method and its excellent catalytic performance in oxidative desulfurization[J]. Fuel,2017,188:232−238. doi: 10.1016/j.fuel.2016.10.045 [6] 刘斌, 李钢, 王祥生. TS-1/H2O2催化模拟汽油中噻吩的选择氧化研究[J]. 燃料化学学报,2006,34(5):629−632. doi: 10.3969/j.issn.0253-2409.2006.05.023LIU Bin, LI Gang, WANG Xiang-sheng. Selective oxidation of thiophene from model gasoline over TS-1/H2O2[J]. J Fuel Chem Technol,2006,34(5):629−632. doi: 10.3969/j.issn.0253-2409.2006.05.023 [7] CLERICI M G, INGALLINA P. Epoxidation of lower olefins with hydrogen-peroxide and titanium silicalite[J]. J Catal,1993,140(1):71−83. doi: 10.1006/jcat.1993.1069 [8] 马书启, 李钢, 王祥生, 金长子, 刘民, 郭新闻. 钛硅分子筛催化1-丁烯环氧化研究[J]. 燃料化学学报,2005,33(4):509−512. doi: 10.3969/j.issn.0253-2409.2005.04.027MA Shu-qi, LI Gang, WANG Xiang-sheng, JIN Chang-zi, LIU Min, GUO Xin-wen. 1-Butylene epoxidation over various titanosilicate molecular sieves[J]. J Fuel Chem Technol,2005,33(4):509−512. doi: 10.3969/j.issn.0253-2409.2005.04.027 [9] BORDIGA S, COLUCCIA S, LAMBERTI C, MARCHESE L, ZECCHINA A. XAFS study of Ti-Silicalite: structure of framework Ti(IV) in the presence and absence of reactive molecules (H2O, NH3) and comparison with ultraviolet-visible and IR results[J]. J Phys Chem,1994,98(15):4125−4132. doi: 10.1021/j100066a036 [10] ALBA-RUBIO A C, FIERRO J L G, LEÓN-REINA L, MARISCAL J A, DUMESIC M, GRANADOS L. Oxidation of furfural in aqueous H2O2 catalysed by titanium silicalite: deactivation processes and role of extraframework Ti oxides[J]. Appl Catal B: Environ,2017,202:269−280. doi: 10.1016/j.apcatb.2016.09.025 [11] MILLINI R, MASSARA E P, PEREGO G, BELLUSSI G. Framework composition of titanium silicalite-1[J]. J Catal,1992,137(2):497−503. doi: 10.1016/0021-9517(92)90176-I [12] THANGARAJ A, KUMAR R, MIRAJKAR S, RATNASAMY P. Catalytic properties of crystalline titanium silicalites I: synthesis and characterization of titanium-rich zeolites with MFI structure[J]. J Catal,1991,130(1):1−8. doi: 10.1016/0021-9517(91)90086-J [13] SASIDHARAN M, WU P, TAKASHI T. Direct formation of pinacols from olefins over various titano-silicates[J]. J Catal,2002,209:260−265. doi: 10.1006/jcat.2002.3595 [14] SERRANO D P, SANZ R, PIZARRO P, PERAL A, MORENO I. Improvement of the hierarchical TS-1 properties by silanization of protozeolitic units in presence of alcohols[J]. Microporous Mesoporous Mater,2013,166:59−66. doi: 10.1016/j.micromeso.2012.04.050 [15] WRÓBLEWSKA A, TOŁPA J, KŁOSIN D, MIĄDLICKI P, KOREN Z C, MICHALKIEWICZ B. The application of TS-1 materials with different titanium contents as catalysts for the autoxidation of α-pinene[J]. Microporous Mesoporous Mater,2020,305:110384. doi: 10.1016/j.micromeso.2020.110384 [16] ZHANG M, REN S Y, GUO Q X, SHEN B J. Synthesis of hierarchically porous zeolite TS-1 with small crystal size and its performance of 1-hexene epoxidation reaction[J]. Microporous Mesoporous Mater,2021,326:111395. doi: 10.1016/j.micromeso.2021.111395 [17] XUE Y, XIE Y L, WEI H J, WEN Y Q, WANG X Y, LI B J. Improving the performance of TS-1 catalysts for continuous cyclohexanone ammoximation through controlment of active species distribution[J]. New J Chem,2014,38(9):4229−4234. doi: 10.1039/C4NJ00455H [18] 林民, 舒兴田, 汪燮卿, 赵琦, 韩秀文, 林励吾, 包信和. TS-1分子筛的合成 Ⅰ. 29Si和 1H NMR研究正硅酸乙酯的水解[J]. 催化学报,1999,20(1):29−34. doi: 10.3321/j.issn:0253-9837.1999.01.009LIN Min, SHU Xing-tian, WANG Xie-qin, ZHAO Qi, HAN Xiu-wen, LIN Li-wu, BAO Xin-he. Synthesis of TS-1 zeolite I. Hydrolysis of ethyl orthosilicate by 29Si and 1H NMR[J]. Chin J Catal,1999,20(1):29−34. doi: 10.3321/j.issn:0253-9837.1999.01.009 [19] 范峰滔, 冯兆池, 李灿. 正硅酸乙酯水解过程的原位紫外拉曼光谱研究[C]//第十四届全国光散射学术会议论文摘要集. 北京, 2007: 179–179.FAN Feng-tao, FENG Zhao-chi, LI Can. Study on hydrolysis of ethyl orthosilicate by in situ UV Raman spectroscopy[C]//Abstracts of the 14th National Conference on Light Scattering. Beijing, 2007: 179–179. [20] RUBIO F, RUBIO J, OTEO J L. A FT-IR study of the hydrolysis of Tetraethylorthosilicate (TEOS)[J]. Spectrosc Lett,1998,31(1):199−219. doi: 10.1080/00387019808006772 [21] 隋学叶, 刘世权, 程新. 正硅酸乙酯的水解缩聚反应及多孔SiO2粉体的制备[J]. 中国粉体技术,2006,3:35−39. doi: 10.3969/j.issn.1008-5548.2006.05.010SUI Xue-ye, LIU Shi-quan, CHENG Xin. Hydrolytic polycondensation of ethyl orthosilicate and preparation of porous SiO2 powder[J]. China Powder Sci Technol,2006,3:35−39. doi: 10.3969/j.issn.1008-5548.2006.05.010 [22] XUE T, LIU H, WANG Y, WU H H, WU P, HE M Y. Seed-induced synthesis of small-crystal TS-1 using ammonia as alkali source[J]. Chin J Catal,2015,36(11):1928−1935. doi: 10.1016/S1872-2067(15)60955-X [23] WANG J G, WANG Y B, TATSUMI T, ZHAO Y L. Anionic polymer as a quasi-neutral medium for low-cost synthesis of titanosilicate molecular sieves in the presence of high-concentration alkali metal ions[J]. J Catal,2016,338:321−328. doi: 10.1016/j.jcat.2016.03.027 [24] WANG W H, LI G, LI W G, LIU L P. Synthesis of hierarchical TS-1 by caramel templating[J]. Chem Commun,2011,47(12):3529−3531. [25] SHAKERI M, DEHGHANPOUR S B. Preparation of efficient TS-1 with small particle size and enhanced framework Ti[J]. ChemistrySelect,2019,4(16):4771−4774. doi: 10.1002/slct.201900457 [26] KLAAC J, KULAWIK K, SCHULZ-EKLOFF G, JAEGER N. Comparative spectroscopic study of TS-1 and zeolite-hosted extraframework Titanium oxide dispersions[J]. Stud Surf Sci Catal,1994,84:2261−2268. [27] LV Q, LI G, SUN H. Synthesis of hierarchical TS-1 with convenient separation and the application for the oxidative desulfurization of bulky and small reactants[J]. Fuel,2014,130:70−75. doi: 10.1016/j.fuel.2014.04.042 [28] 刘瑶, 苏雪筠. TiO2溶胶的制备及其性能研究[J]. 佛山陶瓷,2016,9(117):1−5.LIU Yao, SU Xue-jun. Preparation and properties of TiO2 sol[J]. Foshan Ceramics,2016,9(117):1−5. -




 下载:
下载: