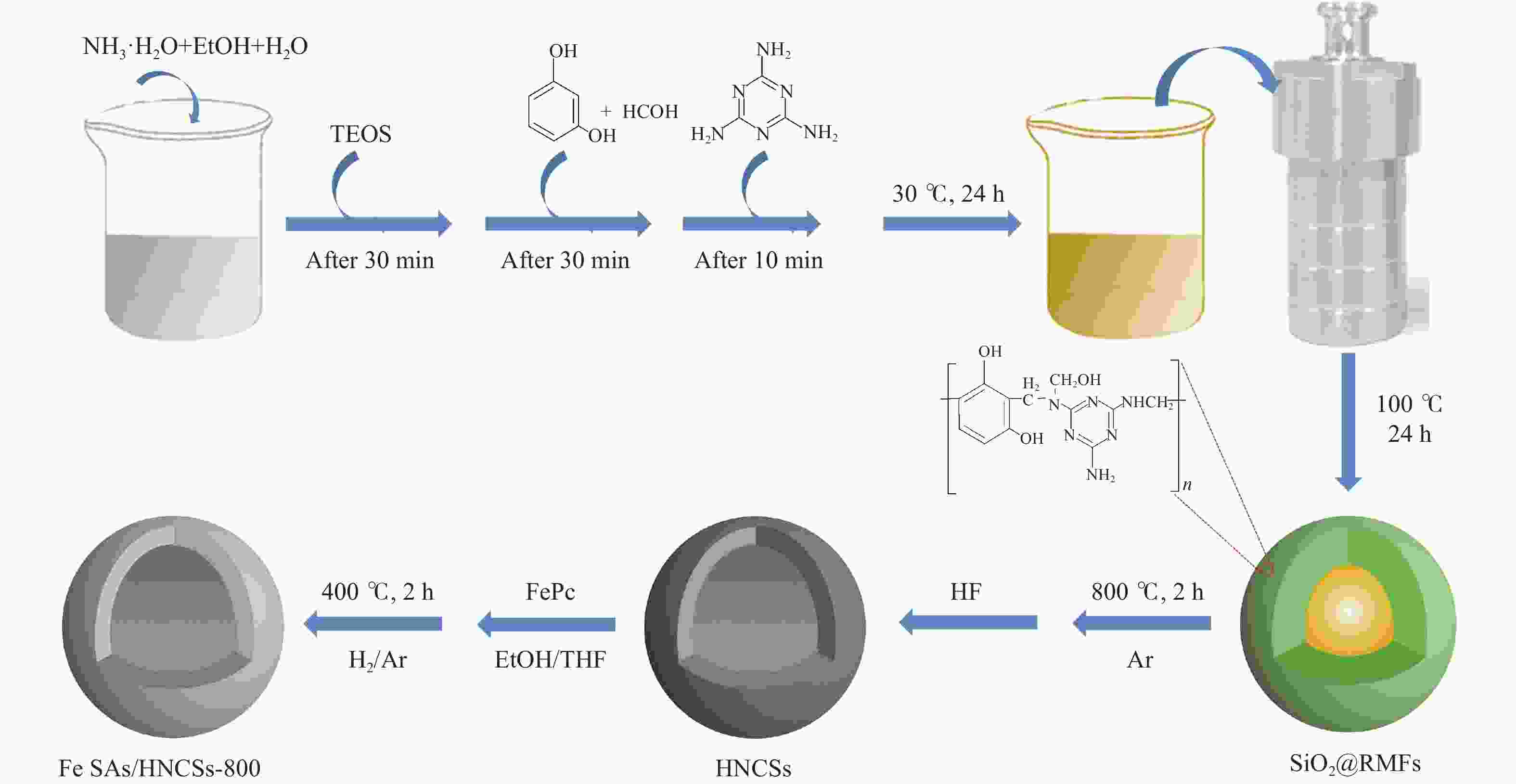
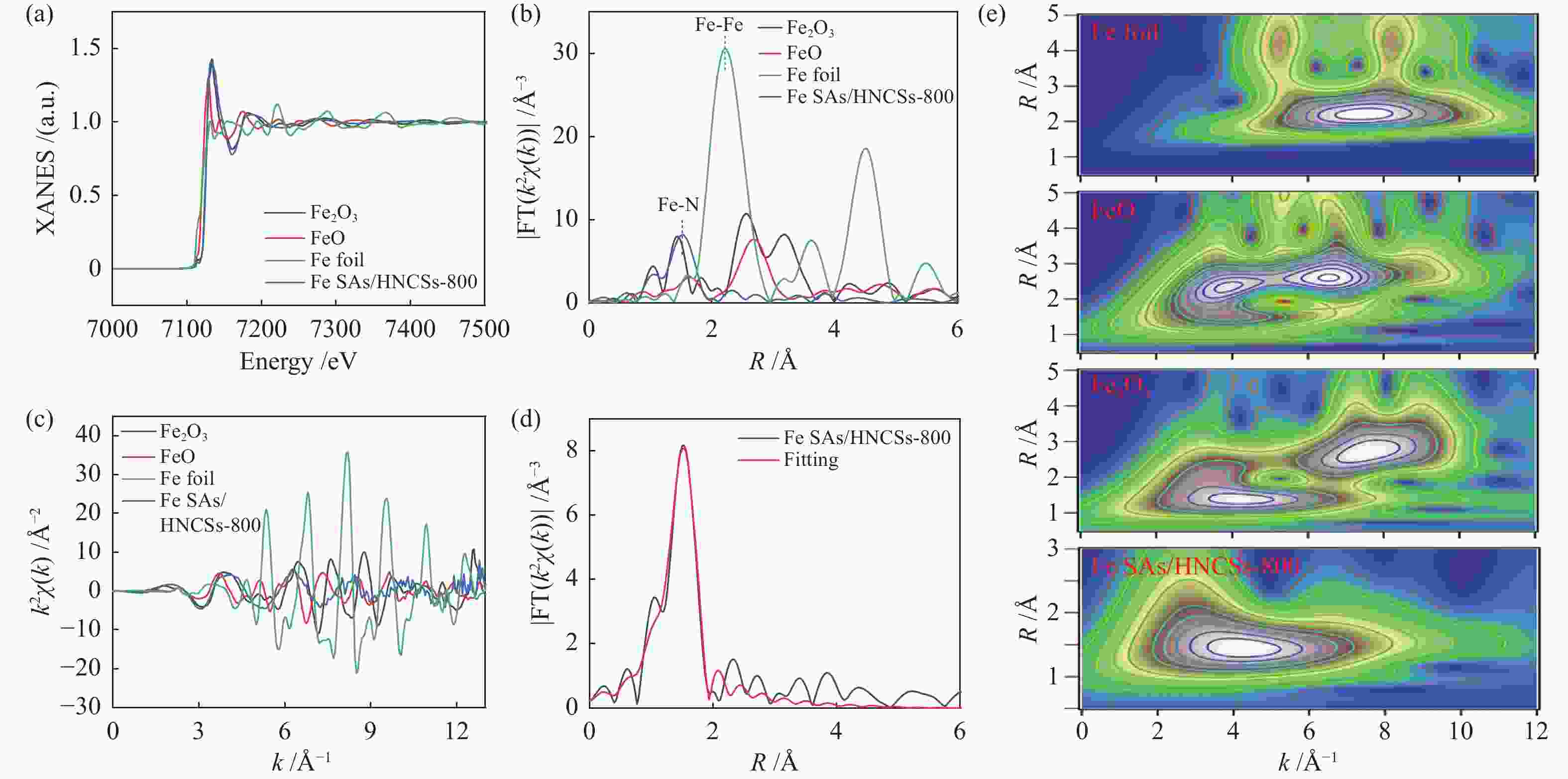
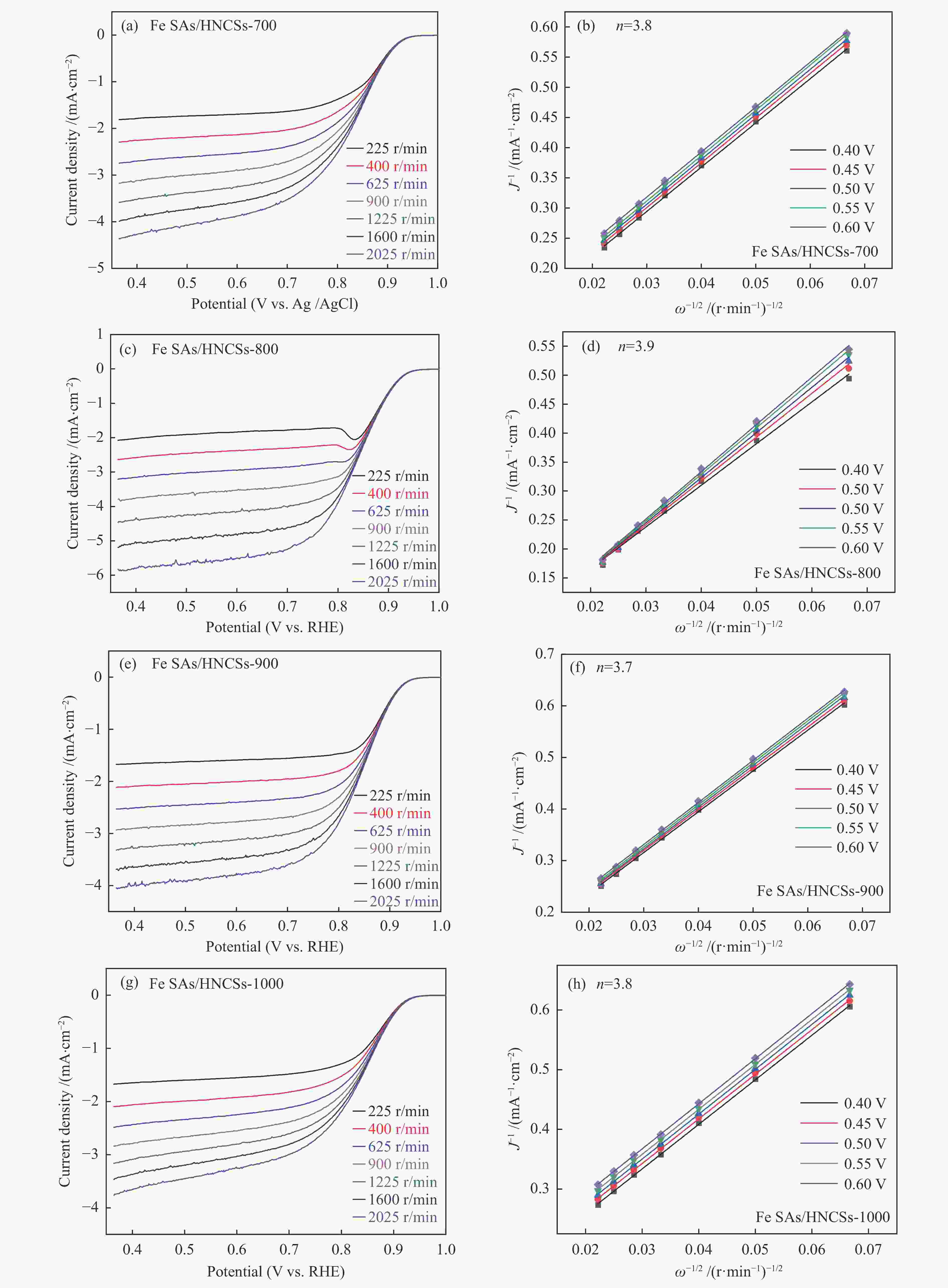
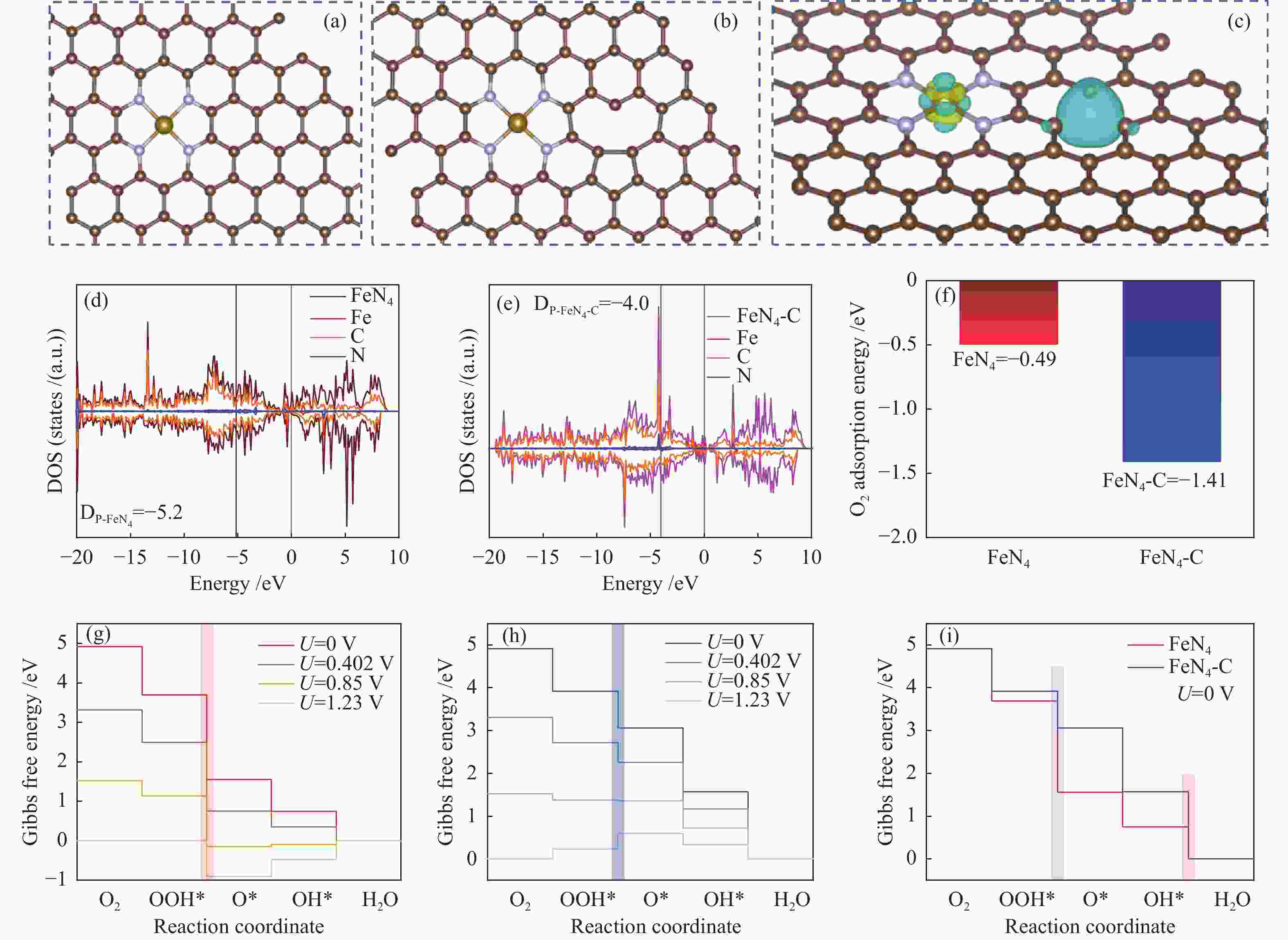
Hollow N-doped carbon spheres with anchored single-atom Fe sites for efficient electrocatalytic oxygen reduction
-
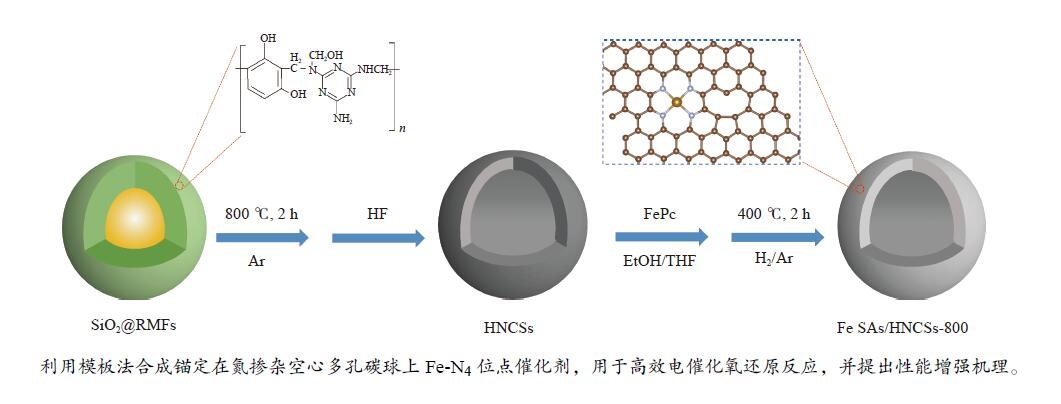
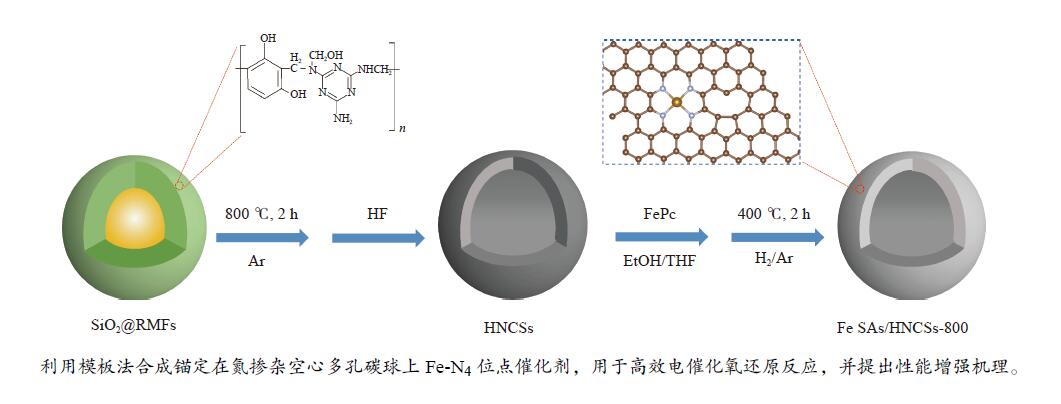
摘要: 本研究将单原子分散的Fe-N4位点锚定在氮掺杂空心多孔碳球上用于电催化氧还原反应,研究表明,所制备的Fe SAs/HNCSs-800催化剂表现出优异的电催化氧还原性能,其起始电位为0.925 V,半波电位为0.867 V。球差电镜和同步辐射X射线吸收光谱证实了催化剂中存在高度分散的Fe-N4单原子位点。通过密度泛函理论计算证明单原子Fe-N4位点是氧还原反应有效的活性位点,其相邻的C缺陷可以有效调控单原子Fe的电子结构,进而提高电催化氧还原性能。Abstract: We anchored atomically dispersed Fe-N4 sites on hollow N-doped carbon spheres (Fe SAs/HNCSs-800) for electrocatalytic ORR; the obtained material exhibited electrocatalytic activity and stability comparable to that of commercial Pt/C, with an onset potential of 0.925 V and a half-wave potential of 0.867 V. Aberration-corrected high-angle annular dark-field scanning transmission electron microscopy and X-ray absorption spectroscopy results confirmed the presence of highly dispersed Fe single atoms in Fe SAs/HNCSs-800. The results of experiments and theoretical calculations show that the single-atom dispersed Fe-N4 serve as the ORR active sites, and the adjacent C defects can effectively regulate the electronic structure of Fe atoms and improve the electrocatalytic ORR activity.
-
Figure 4 (a) LSV curves, (b) Tafel plots, (c) cyclic voltammetry (CV) curves, and (d) kinetic current density (jk) curves of Fe SAs/HNCSs; (e) jk at 0.85 V and E1/2 values; (f) LSV curves before and after 3000-cycle durability test, with the inset showing the i-t curves of Fe SAs/HNCSs-800 and 20% Pt/C
-
[1] WANG M, ZHANG H, LIU Y, PAN Y. Research progress of precise structural regulation of single atom catalyst for accelerating electrocatalytic oxygen reduction reaction[J]. J Energy Chem,2022,72:56−72. doi: 10.1016/j.jechem.2022.05.007 [2] ZHANG J, YUAN Y, GAO L, ZENG G, LI M, HUANG H. Stabilizing Pt-based electrocatalysts for oxygen reduction reaction, Fundamental understanding and design strategies[J]. Adv Mater,2021,33(20):2006494. doi: 10.1002/adma.202006494 [3] WANG Y, CHU F, ZENG J, WANG Q, NAREN T, LI Y, CHENG Y, LEI Y, WU F. Single atom catalysts for fuel cells and rechargeable batteries: Principles, advances, and opportunities[J]. ACS Nano,2021,15(1):210−239. doi: 10.1021/acsnano.0c08652 [4] CUI L, FAN K, ZONG L, LU F, ZHOU M, LI B, ZHANG L, FENG L, LI X, CHEN Y, WANG L. Sol-gel pore-sealing strategy imparts tailored electronic structure to the atomically dispersed Ru sites for efficient oxygen reduction reaction[J]. Energy Storage Mater,2022,44:469−476. doi: 10.1016/j.ensm.2021.11.007 [5] LI J, BANIS M N, REN Z, ADAIR K R, DOYLE DAVIS K, MEIRA D M, FINFROCK Y Z, ZHANG L, KONG F, SHAM T K, LI R, LUO J, SUN X. Unveiling the nature of Pt single-atom catalyst during electrocatalytic hydrogen evolution and oxygen reduction reactions[J]. Small,2021,17(11):2007245. doi: 10.1002/smll.202007245 [6] YANG S, KIM J, TAK Y J, SOON A, LEE H. Single-atom catalyst of platinum supported on titanium nitride for selective electrochemical reactions[J]. Angew Chem Int Ed,2016,55(6):2058−2062. doi: 10.1002/anie.201509241 [7] LIU Q, LI Y, ZHENG L, SHANG J, LIU X, YU R, SHUI J. Sequential synthesis and active-site coordination principle of precious metal single-atom catalysts for oxygen reduction reaction and PEM fuel cells[J]. Adv Energy Mater,2020,10(20):2000689. doi: 10.1002/aenm.202000689 [8] LIU J, BAK J, ROH J, LEE K, CHO A, HAN J W, CHO E. Reconstructing the coordination environment of platinum single-atom active sites for boosting oxygen reduction reaction[J]. ACS Catal,2021,11(1):466−475. doi: 10.1021/acscatal.0c03330 [9] WANG M, ZHENG X, QIN D, LI M, SUN K, LIU C, CHEONG W C, LIU Z, CHEN Y, LIU S, WANG B, LI Y, LIU Y, LIU C, YANG X, FENG X, YANG C, CHEN C, PAN Y. Atomically dispersed CoN3C1-TeN1C3 diatomic sites anchored in N-doped carbon as efficient bifunctional catalyst for synergistic electrocatalytic hydrogen evolution and oxygen reduction[J]. Small,2022,18(29):2201974. doi: 10.1002/smll.202201974 [10] PAN Y, LIU S, SUN K, CHEN X, WANG B, WU K, CAO X. A bimetallic Zn/Fe polyphthalocyanine derived single-atom Fe-N4 catalytic site, A superior trifunctional catalyst for overall water splitting and Zn-air batteries.[J]. Angew Chem Int Ed,2018,57(28):8614−8618. doi: 10.1002/anie.201804349 [11] QI W, HUANG W, NIU J, ZHANG B, ZHANG Z, LI W. The role of S in the Co-N-S-C catalysis system towards the ORR for proton exchange membrane fuel cells[J]. Appl Surf Sci,2021,540:148325. doi: 10.1016/j.apsusc.2020.148325 [12] YIN P, YAO T, WU Y, ZHENG L, LIN Y, LIU W, JU H, ZHU J, HONG X, DENG Z, ZHOU G, WEI S, LI Y. Single cobalt atoms with precise N-coordination as superior oxygen reduction reaction catalysts[J]. Angew Chem Int Ed,2016,55(36):10800−10805. doi: 10.1002/anie.201604802 [13] LU F, FAN K, CUI L, YANG Y, WANG W, ZHANG G, WANG C, ZHANG Q, LI B, ZONG L, WANG L. Cu-N4 single atoms derived from metal-organic frameworks with trapped nitrogen-rich molecules and their use as efficient electrocatalysts for oxygen reduction reaction[J]. Chem Eng J,2022,431:133242. doi: 10.1016/j.cej.2021.133242 [14] ZONG L, LU F, ZHANG W, FAN K, CHEN X, JOHANNESSEN B, QI D, BEDFORD N M, WARREN M, SEGRE C U, LIU P, WANG L, ZHAO H. Atomically-dispersed Mn-(N-C2)2(O-C2)2 sites on carbon for efficient oxygen reduction reaction[J]. Energy Storage Mater,2022,49:209−218. doi: 10.1016/j.ensm.2022.04.016 [15] CHEN G, LIU P, LIAO Z, SUN F, HE Y, ZHONG H, ZHANG T, ZSCHECH E, CHEN M, WU G, ZHANG J, FENG X. Zinc-mediated template synthesis of Fe-N-C electrocatalysts with densely accessible Fe-Nx active sites for efficient oxygen reduction[J]. Adv Mater,2020,32(8):1907399. doi: 10.1002/adma.201907399 [16] MUN Y, LEE S, KIM K, KIM S, LEE S, HAN J W, LEE J. Versatile strategy for tuning ORR activity of a single Fe-N4 site by controlling electron-withdrawing/donating properties of a carbon plane[J]. J Am Chem Soc,2019,141(15):6254−6262. doi: 10.1021/jacs.8b13543 [17] YANG L, CHENG D, XU H, ZENG X, WAN X, SHUI J, XIANG Z, CAO D. Unveiling the high-activity origin of single-atom iron catalysts for oxygen reduction reaction[J]. Proc Natl Acad Sci,2018,115(26):6626−6631. doi: 10.1073/pnas.1800771115 [18] WANG M, LI M, ZHAO Y, SHI N, ZHANG H, ZHAO Y, ZHANG Y, ZHANG H, WANG W, SUN K, PAN Y, LIU S, ZHU H, GUO W, LI Y, LIU Y, LIU C. Construction of N-doped carbon frames anchored with Co single atoms and Co nanoparticles as robust electrocatalyst for hydrogen evolution in the entire pH range[J]. J Energy Chem,2022,67:147−156. doi: 10.1016/j.jechem.2021.09.037 [19] ZONG L, FAN K, WU W, CUI L, ZHANG L, JOHANNESSEN B, QI D, YIN H, WANG Y, LIU P, WANG L, ZHAO H. Anchoring single copper atoms to microporous carbon spheres as high-performance electrocatalyst for oxygen reduction reaction[J]. Adv Funct Mater,2021,31(41):2104864. doi: 10.1002/adfm.202104864 [20] LU F, FAN K, CUI L, LI B, YANG Y, ZONG L, WANG L. Engineering FeN4 active sites onto nitrogen-rich carbon with tubular channels for enhanced oxygen reduction reaction performance[J]. Appl Catal B: Environ,2022,313:121464. doi: 10.1016/j.apcatb.2022.121464 [21] PAN Y, CHEN Y, WU K, CHEN Z, LIU S, CAO X, CHEONG W, MENG T, LUO J, ZHENG L, LIU C, WANG D, PENG Q, LI J, CHEN C. Regulating the coordination structure of single-atom Fe-NxCy catalytic sites for benzene oxidation[J]. Nat Commun,2019,10(1):4290. doi: 10.1038/s41467-019-12362-8 -




 下载:
下载: