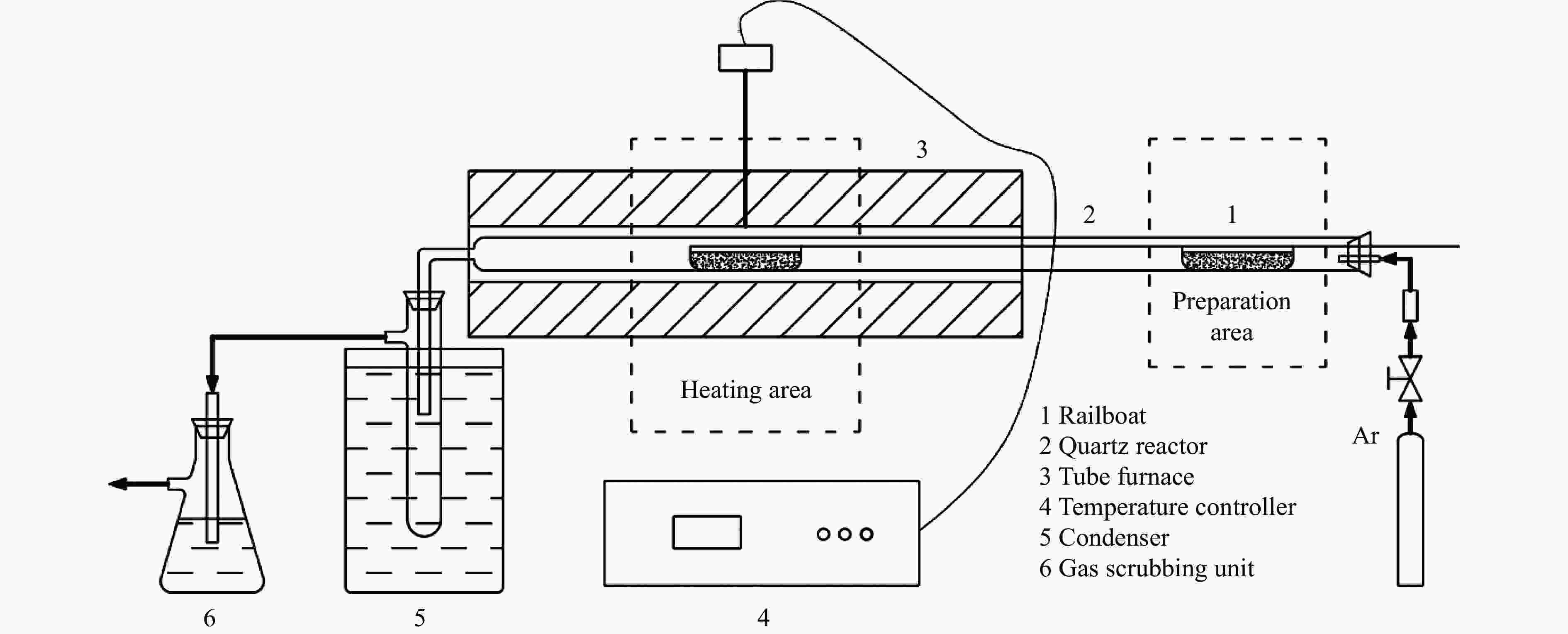
Effect of molten salt on re-pyrolysis behaviors of heavy bio-oil
-
摘要: 研究了KCl-ZnCl2熔融盐在400、500、600 ℃下对重质生物油再热解特性及产物分布的影响。结果表明,熔融盐提高了重质生物油热解的固体产率,同时使气体产率下降;对苯酚、甲基苯酚、乙基苯酚、对丙基苯酚等部分化合物具有较好的富集效果,尤其在400 ℃下甲基苯酚的相对含量从8.82%提升到了20.85%,而苯酚在600 ℃下相对含量从2.18%提升到了8.62%;在炭形成过程中,熔融盐使C元素含量降低,O元素含量提高,增大了孔隙的BET比表面积和总孔容积,促进了固体产物孔隙结构的形成,增大了孔隙的平均孔径。Abstract: The effects of KCl-ZnCl2 molten salt on the pyrolysis characteristics and pyrolysis products of heavy bio-oil at 400, 500 and 600℃ were studied. The results showed that molten salt increased the solid yield of heavy bio-oil pyrolysis and decreased the gas yield. Some compounds such as phenol, cresol, ethylphenol and 4-propylphenol had good enrichment effect, especially the relative concentration of cresol increased from 8.82% to 20.85% at 400℃, while the relative concentration of phenol increased from 2.18% to 8.62% at 600℃. During formation of char, molten salt reduced the content of carbon and increased the content of oxygen, increased the BET surface area and total pore volume of pores. Molten salt promoted formation of pore structure of the solid product and increased its average pore diameter.
-
Key words:
- heavy bio-oil /
- molten salt /
- pyrolysis /
- component analysis
-
表 1 重质生物油理化性质
Table 1 Properties and elemental compositions of heavy bio-oil
Moisture w/% Ultimate analysisa w/% C H Ob N 5.17 66.36 7.22 25.67 0.75 a: dry and ash-free basis, b: by difference 表 2 重质生物油主要有机组分相对峰面积
Table 2 Relative peak area of main organic components in heavy bio-oil
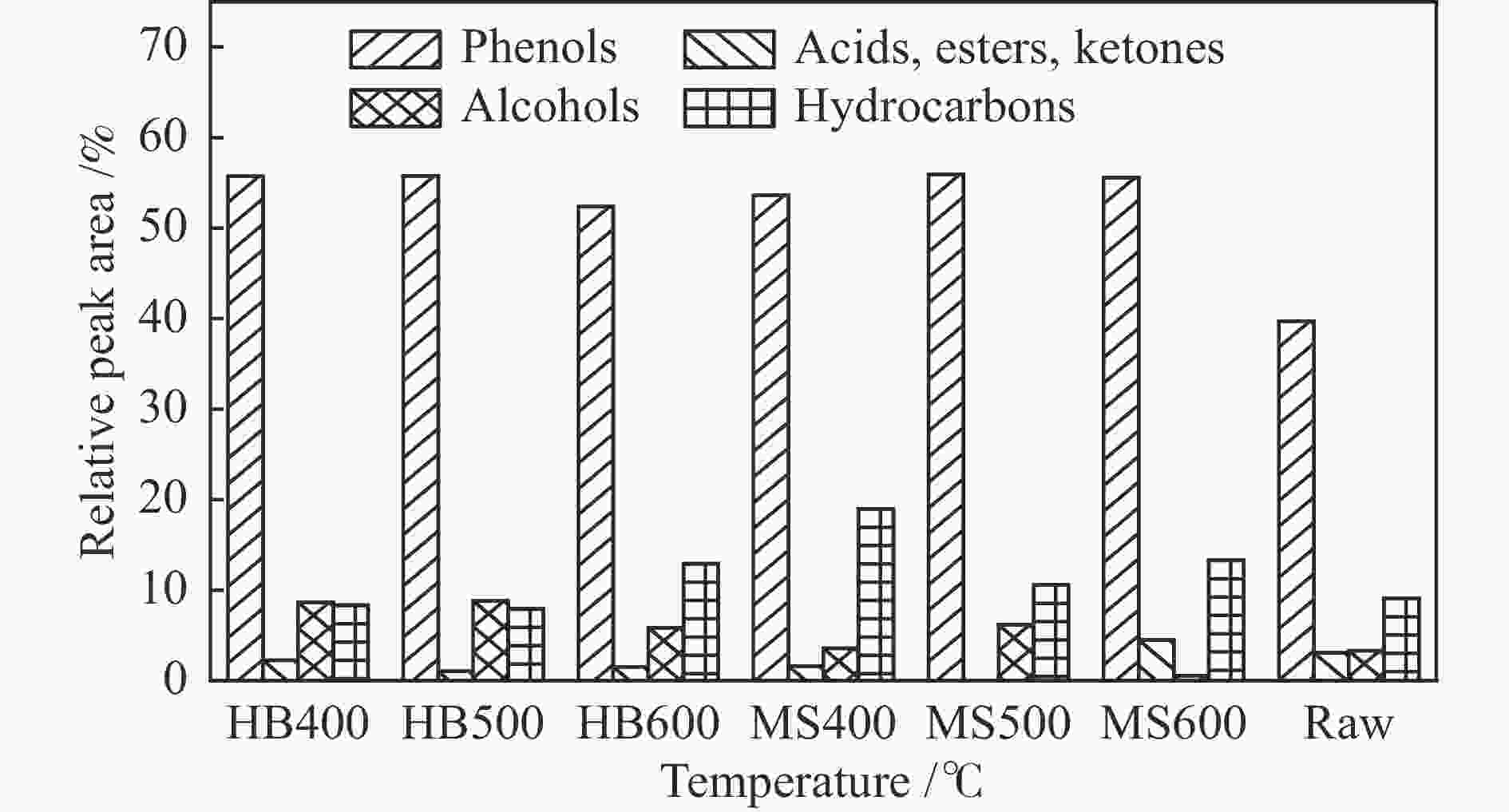
Component Phenols Acids, Esters, Ketones Alcohols Hydrocarbons Relative peak area /% 39.71 3.08 3.31 9.11 表 3 原油及再热解液体产物各组分的相对峰面积
Table 3 Relative peak area of each component of crude oil and re-pyrolysis liquid products
Component Relative peak area /% HB400 HB500 HB600 MS400 MS500 MS600 raw Acetic acid − − − − − − 1.01 Methyl formate 1.01 1.05 1.01 0.94 − − − Ethyl acetate − − − − − 0.53 − n-Ethyl propanoate − − − − − 1.26 − Hexyl butyrate − − − − − 0.72 − Toluene 0.54 0.5 0.81 0.6 − − − 3-cyclohexyl-1-propanol − 1.21 1.28 − 1.19 − − Cyclohexaneacetic acid 1.24 − − − − − − Styrene − − 0.54 − − − − 2,4-dimethylfuran − − 0.51 0.6 0.5 − − 2-methylcyclopentenone − − − − − 0.96 − Phenol 4.43 5.24 0.54 7.38 6.01 8.62 2.18 Indene 0.7 0.74 0.8 0.55 0.62 − 0.81 Methyl cyclopentenolone 0.55 − − 0.58 − − − Cresol 13.95 13.34 13.67 20.85 15.55 19.97 8.82 Guaiacol − − − − 2.13 − 1.44 7-methylbenzofuran − − − − − 0.59 − 3-methyl-1H-indene 0.56 0.52 0.76 − 0.71 − − Xylenol 5.94 5.7 5.53 6.03 6.53 9.41 4.09 Ethylphenol − − 0.98 4.94 2.54 3.01 − Naphthalene 2.75 2.9 3.22 3.47 4.16 4.37 2.98 2-methoxy-4-methylphenol 4.63 4.07 2.08 3.93 3.44 1.72 4.28 Trimethylphenol 1.71 2.05 2.53 0.55 2.76 1.04 0.53 Catechol 3.15 3.78 4.65 − 1.98 0.99 − 1,5,5-trimethyl-6-methylidenecyclohexene − − 1.68 1.88 − 1.82 0.83 6-ethyl-m-cresol − − − 1.86 − 2.48 1.13 4-Propylphenol 0.96 0.88 − 1.71 1.65 1.88 − 4-ethyl-2-methoxyphenol 4.34 3.66 2.34 3.76 3.45 1.72 2.86 1-(2-methoxyphenyl)ethanol − − − − 0.67 − 0.52 1-methylnaphthalene 1.4 1.53 2.27 1.99 2.4 2.6 2.47 1-(4-hydroxy-3-methylphenyl)ethanone − − − 0.67 − 1.02 − 4-methylcatechol 3.21 3.46 3.91 1.49 2.08 1.59 − 2-methoxy-5-prop-2-enyl-phenol 2.79 2.69 3.63 − 2.04 0.56 9.78 4-n-propylanisole − − − 1.22 1.26 1.2 − 1-(4-methoxyphenyl)propan-1-ol − − − − − − 1.17 Vanillyl alcohol 3.5 3.02 1.68 − 1.78 − − Dihydroeugenol − − − 1.57 0.87 0.99 − Diphyl − − − − − 0.54 − 1-ethylnaphthalene − − − − − 0.6 − trans-isoeugenol 4.26 4.46 2.16 2.05 0.71 − − 4-ethylcatechol 2.64 3.03 3.31 2.38 2.96 1.01 − 5,6-dimethoxy-3-methyl-2,3-dihydroinden-1-one − − − − − − 2.07 2,6-dimethoxyphenol − − − 1.34 − − 1.62 Coniferyl alcohol 1.8 2.07 2.04 0.54 − − − 4,4'-dimethoxybenzhydrol 2.82 2.54 2.29 2.47 2.53 0.55 − Vanillin − − 0.74 − − − − 1,4-dimethoxy-2,3-dimethylbenzene 0.99 1.15 0.82 − 0.72 − − 5-tert-butylpyrogallol 2.03 1.72 1.04 1.18 1.23 − 1.77 4-allyl-2,6-dimethoxyphenol 1.68 1.69 1.31 − − − 2.83 2-tert-butyl-4-methoxyphenol − − 0.56 − − − 0.71 Phenalene − − − 0.54 0.5 0.6 0.78 1-phenylpropylbenzene 0.73 − − 0.53 − − − Anthracene 0.68 0.6 0.74 0.83 0.85 0.99 1.24 note:− relative peak area < 0.5% 表 4 随受热温度变化的固体产物的元素分析
Table 4 Ultimate analysis of solid products varying with heating temperature
Sample Ultimate analysisa w/% C H Ob N HB400 80.65 3.75 13.35 2.25 HB500 83.45 3.38 11.09 2.08 HB600 86.91 2.76 8.18 2.15 MS400 64.79 3.98 29.30 1.93 MS500 62.46 3.35 32.65 1.54 MS600 63.35 2.90 32.28 1.47 a: dry and ash-free basis; b: by difference 表 5 随受热温度变化的固体产物的孔隙特性
Table 5 Pore characteristics of solid products varying with heating temperature
Sample BET surface area /(m2·g−1) Total pore volume /(cm3·g−1) Average pore size /nm HB400 − − − HB500 4.5983 0.002320 2.0179 HB600 14.5326 0.003612 0.9242 MS400 2.3882 0.004051 6.7856 MS500 7.4815 0.007779 4.1592 MS600 58.4711 0.030234 2.0683 note:HB400 few pores cannot be measured -
[1] HU H S, WU Y L, YANG M D. Fractionation of bio-oil produced from hydrothermal liquefaction of microalgae by liquid-liquid extraction[J]. Biomass Bioenerg,2018,108:487−500. doi: 10.1016/j.biombioe.2017.10.033 [2] 马亚凯, 袁鑫华, 罗泽军, 朱锡锋. 精馏系统内真空度对生物油模型化合物蒸馏特性的影响[J]. 燃料化学学报,2022,50(2):160−165. doi: 10.1016/S1872-5813(21)60140-8MA Ya-kai, YUAN Xin-hua, LUO Ze-jun, ZHU Xi-feng. Influence of vacuum degrees in rectification system on distillation characteristics of bio-oil model compounds[J]. J Fuel Chem Technol,2022,50(2):160−165. doi: 10.1016/S1872-5813(21)60140-8 [3] 闫彩辉, 赵炜, 盛晨, 吴晓娜. 柱层析法在生物油分离方面应用的研究进展[J]. 林产化学与工业,2011,31(4):123−126.YAN Cai-hui, ZHAO Wei, SHENG Chen, WU Xiao-na. Research progress for separation of bio-oil by column chromatography[J]. Chem Ind Forest Prod,2011,31(4):123−126. [4] 王储, 朱锡锋. 分级冷凝生物油组分富集与组分稳定性研究[J]. 燃料化学学报,2018,46(11):1315−1322. doi: 10.3969/j.issn.0253-2409.2018.11.005WANG Chu, ZHU Xi-feng. Study on component enrichment and storage stability of bio-oils obtained from fractional condensation[J]. J Fuel Chem Technol,2018,46(11):1315−1322. doi: 10.3969/j.issn.0253-2409.2018.11.005 [5] LI W, PAN C Y, SHENG L, LIU Z, CHEN P, LOU H, ZHENG X M. Upgrading of high-boiling fraction of bio-oil in supercritical methanol[J]. Bioresour Technol,2011,102(19):9223−8. doi: 10.1016/j.biortech.2011.07.071 [6] ZHANG X H, TANG W W, ZHANG Q, LI Y P, CHEN L G, XU Y, WANG C G, MA L L. Production of hydrocarbon fuels from heavy fraction of bio-oil through hydrodeoxygenative upgrading with Ru-based catalyst[J]. Fuel,2018,215:825−34. doi: 10.1016/j.fuel.2017.11.111 [7] JIANG H T, AI N, WANG M, JI D X, JI J B. Experimental study on thermal pyrolysis of biomass in molten salt media[J]. Electrochem,2009,77(8):730−5. doi: 10.5796/electrochemistry.77.730 [8] ZENG K, YANG X Y, XIE Y P, YANG H P, LI J, ZHONG D, ZUO H Y, NZIHOU A, ZHU Y J, CHEN H P. Molten salt pyrolysis of biomass: The evaluation of molten salt[J]. Fuel,2021,302:121103. [9] XIE Y P, ZENG K, FLAMANT G, YANG H P, LIU N, HE X, YANG X Y, NZIHOU A, CHEN H P. Solar pyrolysis of cotton stalk in molten salt for bio-fuel production[J]. Energy,2019,179:1124−32. doi: 10.1016/j.energy.2019.05.055 [10] KONG W X, ZHAO F, GUAN H J, ZHAO Y F, ZHANG H S, ZHANG B. Highly adsorptive mesoporous carbon from biomass using molten-salt route[J]. J Mater Sci,2016,51(14):6793−800. doi: 10.1007/s10853-016-9966-8 [11] NYGÅRD H S, OLSEN E. Effect of salt composition and temperature on the thermal behavior of beech wood in molten salt pyrolysis[J]. Energy Procedia,2014,58:221−8. doi: 10.1016/j.egypro.2014.10.432 [12] KUDSY M, KUMAZAWA H, SADA E. Pyrolysis of kraft lignin in molten ZnCl2-KCl media with tetralin vapor addition[J]. Can J Chem Eng,1995,73(3):411−5. doi: 10.1002/cjce.5450730319 [13] AI N, ZENG G N, ZHOU H Y, HE Y T. Co-production of activated carbon and bio-oil from agricultural residues by molten salt pyrolysis[J]. Bioresour,2013,8(2):1551−62. [14] 谢迎谱. 生物质熔融碳酸盐热解与气化过程实验研究[D]. 武汉: 华中科技大学, 2020.XIE Yin-pu. Experimental study on biomass pyrolysis and gasification with molten carbonates[D]. Wuhan: Huazhong University of Science and Technology, 2020. [15] WANG W L, REN X Y, CHANG J M, CAI L P, SHI S Q. Characterization of bio-oils and bio-chars obtained from the catalytic pyrolysis of alkali lignin with metal chlorides[J]. Fuel Process Technol,2015,138:605−11. doi: 10.1016/j.fuproc.2015.06.048 [16] 杨耀钧, 刁瑞, 王储, 朱锡锋. 不同金属氧化物对重质生物油再裂解的比较性研究[J]. 化工学报,2021,72(11):5820−5830. doi: 10.11949/0438-1157.20210895YANG Yao-jun, DIAO Rui, WANG Chu, ZHU Xi-feng. Catalytic effect of different metal oxides on pyrolysis behaviors of heavy bio-oil: A comparative study[J]. CIESC J,2021,72(11):5820−5830. doi: 10.11949/0438-1157.20210895 [17] LUO Z J, ZHU X F, WANG C, WANG Y S, ZHU X F. Comparative study on the evolution of physicochemical properties of tar obtained from heavy fraction of bio-oil at different heating rates[J]. J Anal Appl Pyrolysis,2020,150:104854. [18] SEVILLA M, MOKAYA R. Energy storage applications of activated carbons: Supercapacitors and hydrogen storage[J]. Energy Environ Sci,2014,7(4):1250−1280. doi: 10.1039/C3EE43525C [19] SHANG H S, LU Y J, ZHAO F, CHAO C, ZHANG B, ZHANG H S. Preparing high surface area porous carbon from biomass by carbonization in a molten salt medium[J]. RSC Adv,2015,5(92):75728−75734. doi: 10.1039/C5RA12406A [20] MOLINA S M, RODRÍGUEZ R F. Role of chemical activation in the development of carbon porosity[J]. Colloid Surface A,2004,241(1/3):15−25. doi: 10.1016/j.colsurfa.2004.04.007 [21] PORADA S, SCHIPPER F, ASLAN M, ANTONIETTI M, PRESSER V, FELLINGER T P. Capacitive deionization using biomass-based microporous salt-templated heteroatom-doped carbons[J]. ChemSusChem,2015,8(11):1867−1874. doi: 10.1002/cssc.201500166 [22] DÍEZ N, FUERTES A B, SEVILLA M. Molten salt strategies towards carbon materials for energy storage and conversion[J]. Energy Storage Mater,2021,38:50−69. doi: 10.1016/j.ensm.2021.02.048 -





 下载:
下载: