Catalytic performance of Cu/Hβ catalysts for selective catalytic reduction of NO with NH3
-
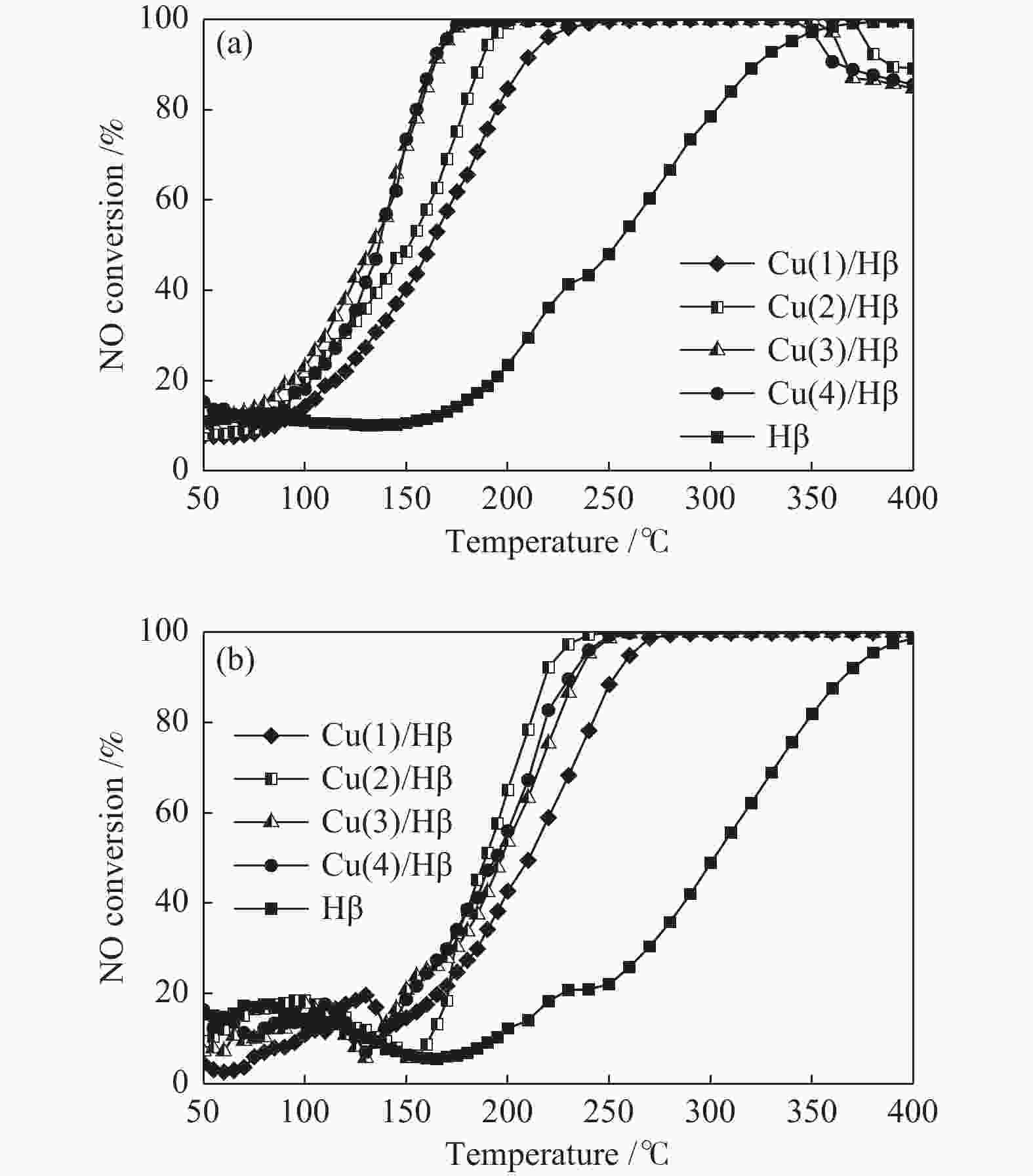
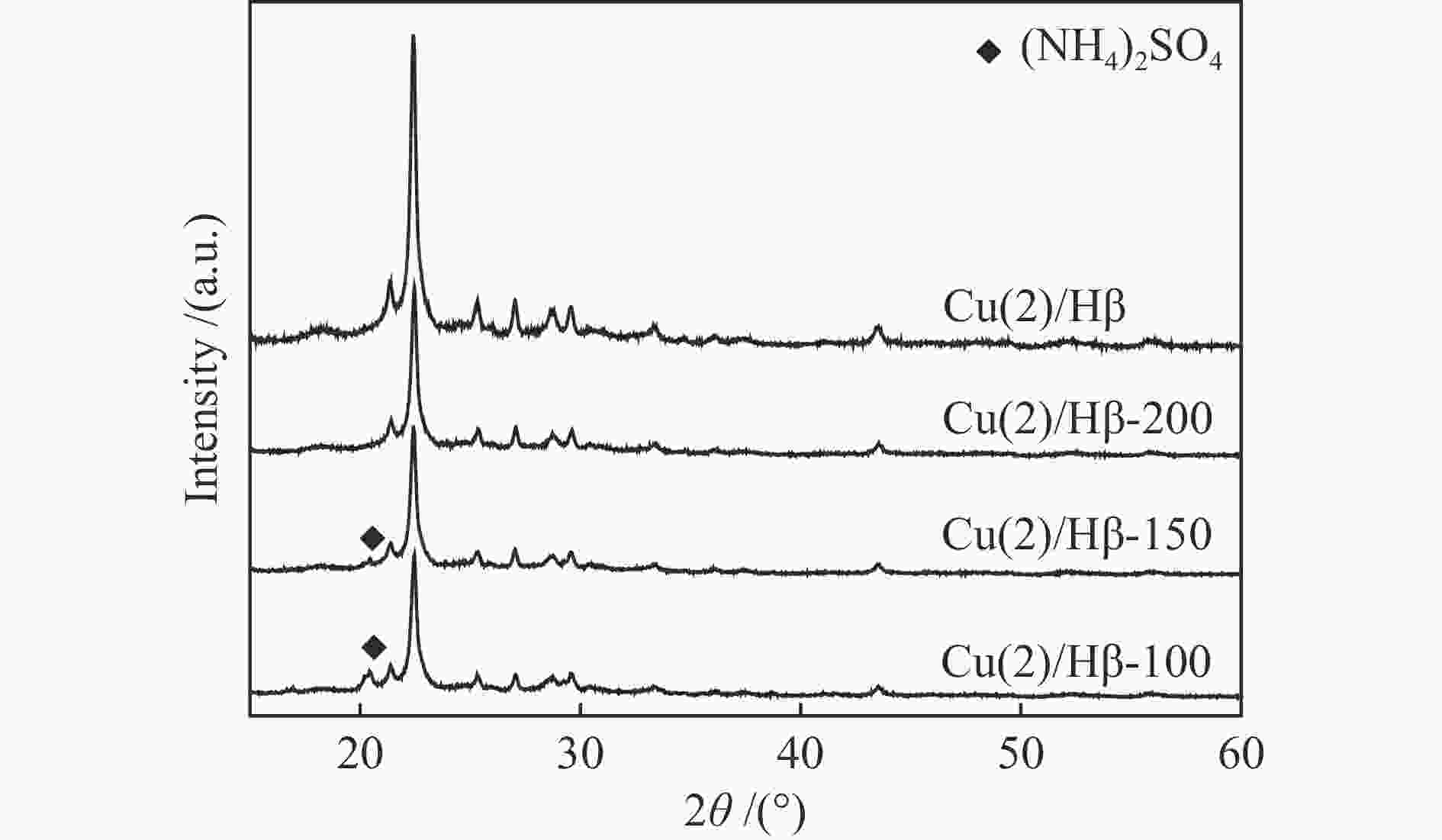
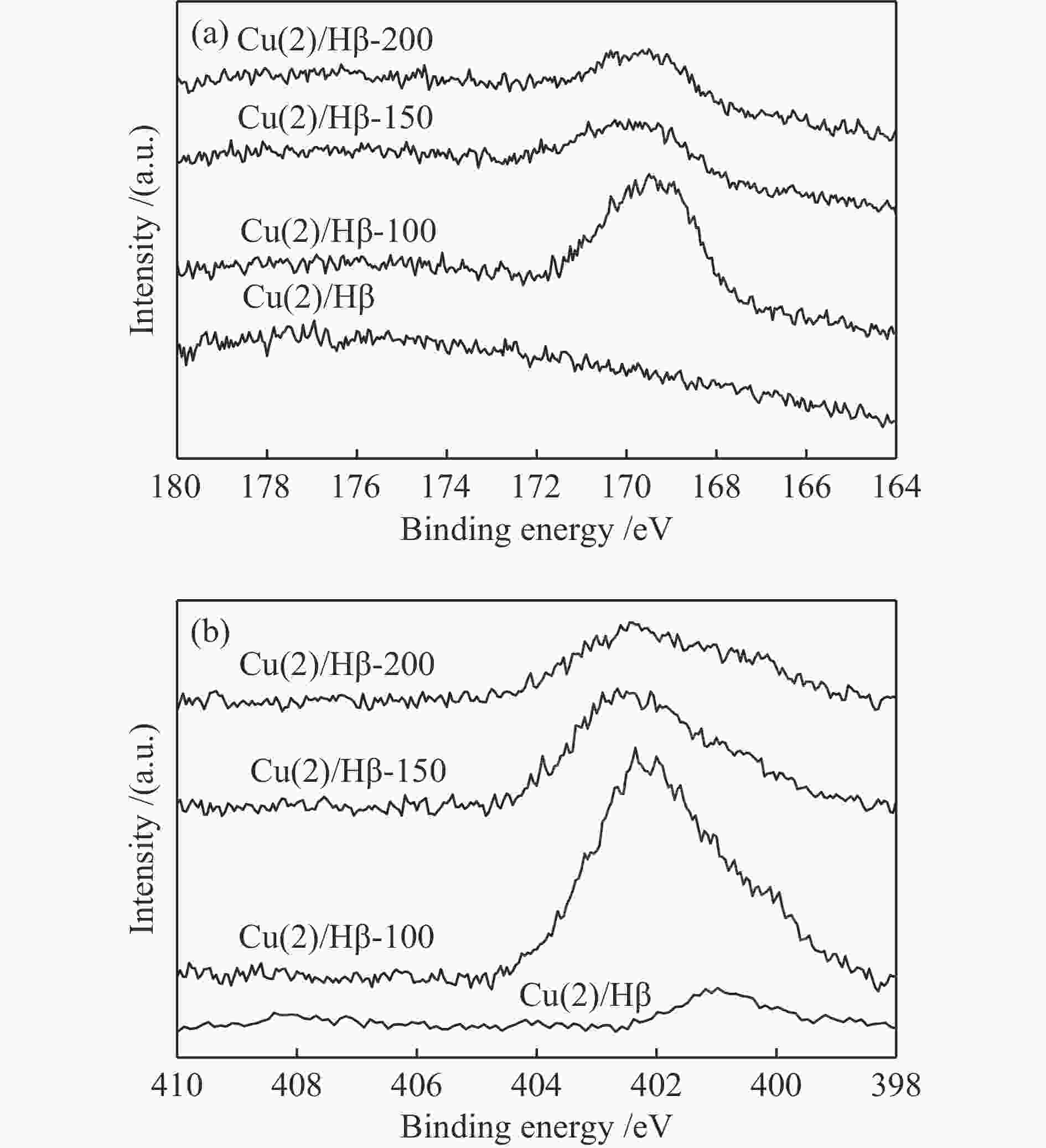
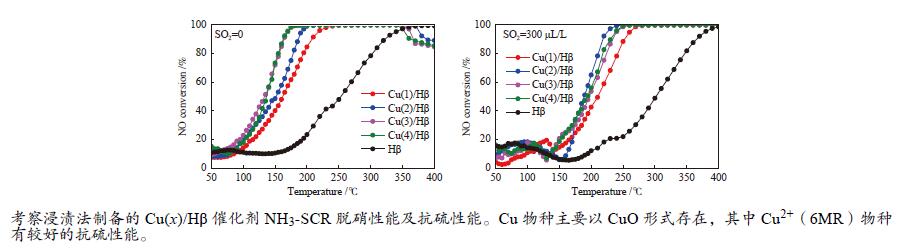
摘要: 采用浸渍法制备了以Hβ分子筛为载体的负载氧化铜催化剂,考察了Cu负载量对催化剂NH3选择性催化还原NO反应(NH3-SCR)性能的影响,通过XRD、N2吸附-脱附、NH3-TPD、NO-TPD、H2-TPR、EDS和XPS等表征技术研究了催化剂的物理化学性质和SO2存在条件下催化剂活性降低的原因。结果表明,反应气体不含SO2,Cu负载量为3%,即Cu(3)/Hβ催化剂有较高的反应活性,t95为169 ℃;反应气体含SO2,Cu负载量为2%时,即Cu(2)/Hβ催化剂的反应活性较好,t95为225 ℃。反应前后催化剂的分析结果表明,SO2存在条件下催化剂活性降低的主要原因是在低温条件下,SO2与NH3反应生成的硫铵盐覆盖了催化剂活性中心。Abstract: A series of Cu(x)/Hβ catalysts were prepared by the impregnation method and the effect on the performance of the catalysts for the selective catalytic reduction of NO with NH3 (NH3-SCR) was investigated. Characterization techniques such as XRD, N2 adsorption-desorption, NH3-TPD, NO-TPD, H2-TPR, EDS and XPS were used to investigate the physical and chemical properties of the catalysts and the reason of the decrease of catalyst activity in the presence of SO2. It was shown that the catalyst Cu(3)/Hβ, the Cu loading was 3% and Cu(2)/Hβ, the Cu loading was 2%, exhibited the better catalytic activity when the initial reaction material contained no SO2 and SO2, and t95 was 169 and 225 ℃, respectively. The analysis results of the catalyst before and after the reaction showed that the main reason for the decrease of catalyst activity in the presence of SO2 was that the ammonium sulfur salt which was formed by the reaction of SO2 and NH3 in the low reaction temperature covers the catalyst active center.
-
Key words:
- Hβ /
- copper /
- selective catalytic reduction /
- NH3 /
- NO
-
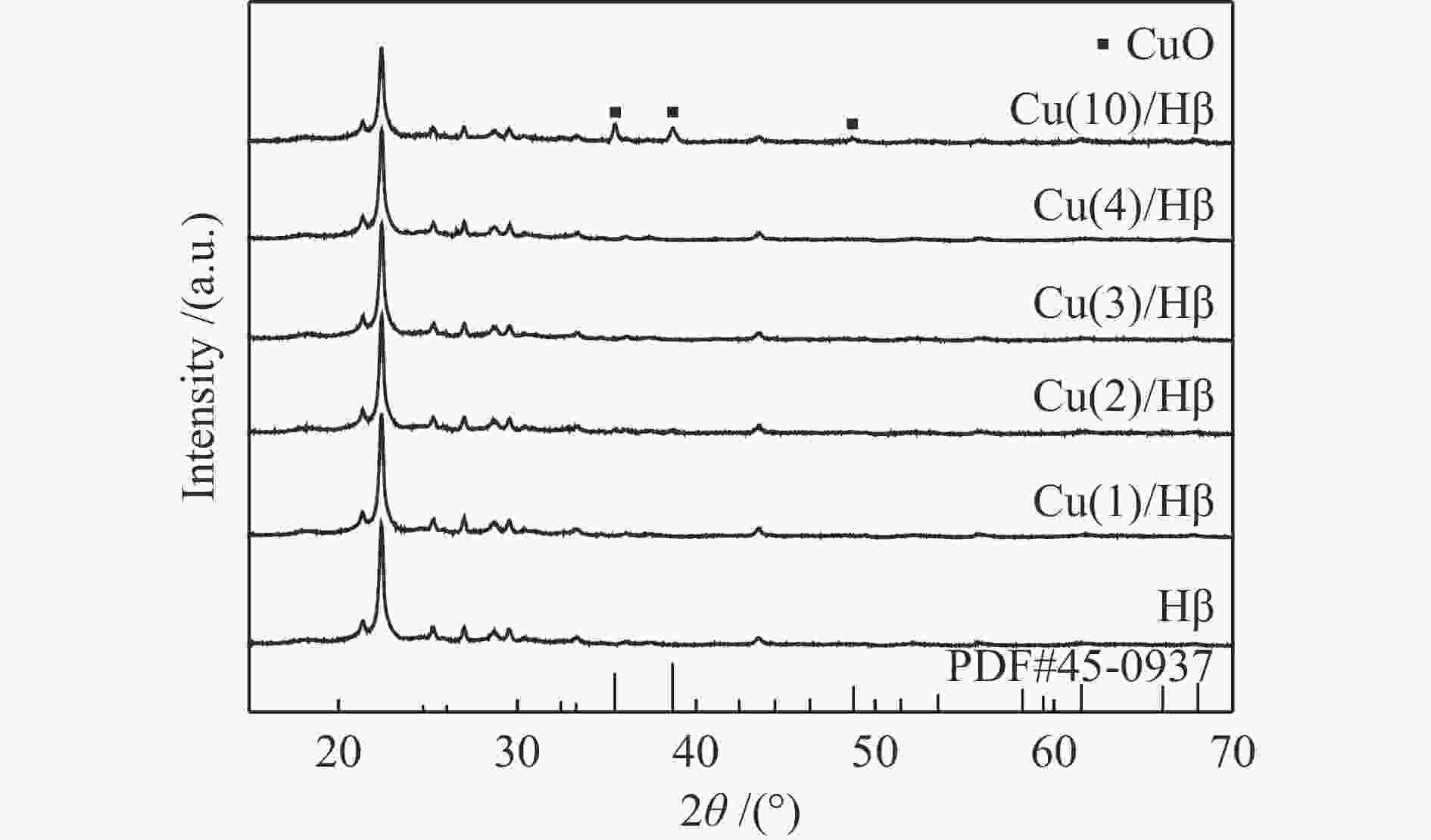
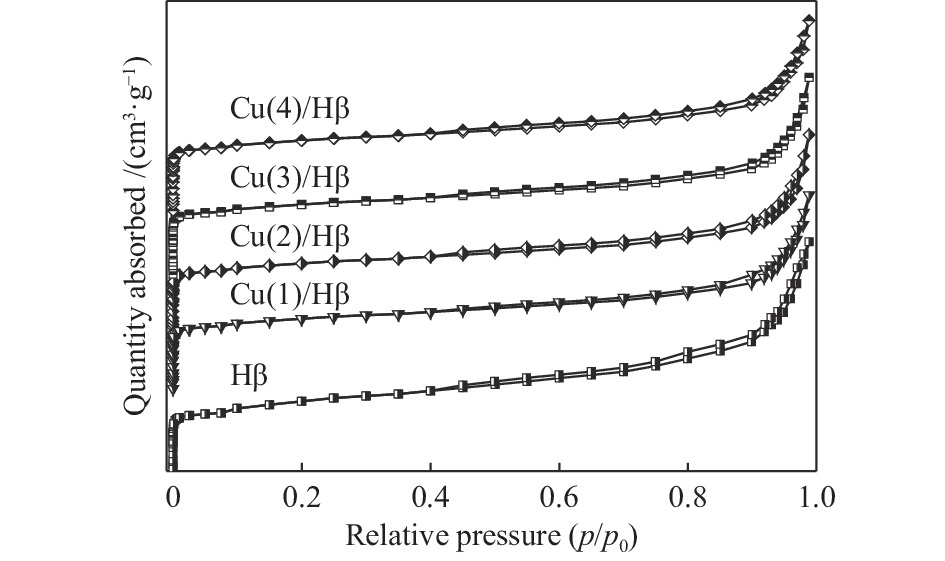
表 1 Cu(x)/Hβ催化剂上N2脱附峰面积和温度
Table 1 Texture property data of Cu( x )/Hβ catalysts
Sample SBET /
(m2·g−1)Smicro /
(m2·g−1)Sext /
(m2·g−1)vtotal/
(cm3·g−1)vmicro /
(cm3·g−1)vext /
(cm3·g−1)Hβ 537 416 121 0.42 0.19 0.23 Cu(1)/Hβ 511 402 109 0.40 0.18 0.22 Cu(2)/Hβ 476 381 95 0.37 0.17 0.20 Cu(3)/Hβ 474 376 98 0.37 0.17 0.20 Cu(4)/Hβ 425 338 87 0.34 0.15 0.19 表 2 Cu(x)/Hβ催化剂上NH3脱附峰面积和温度
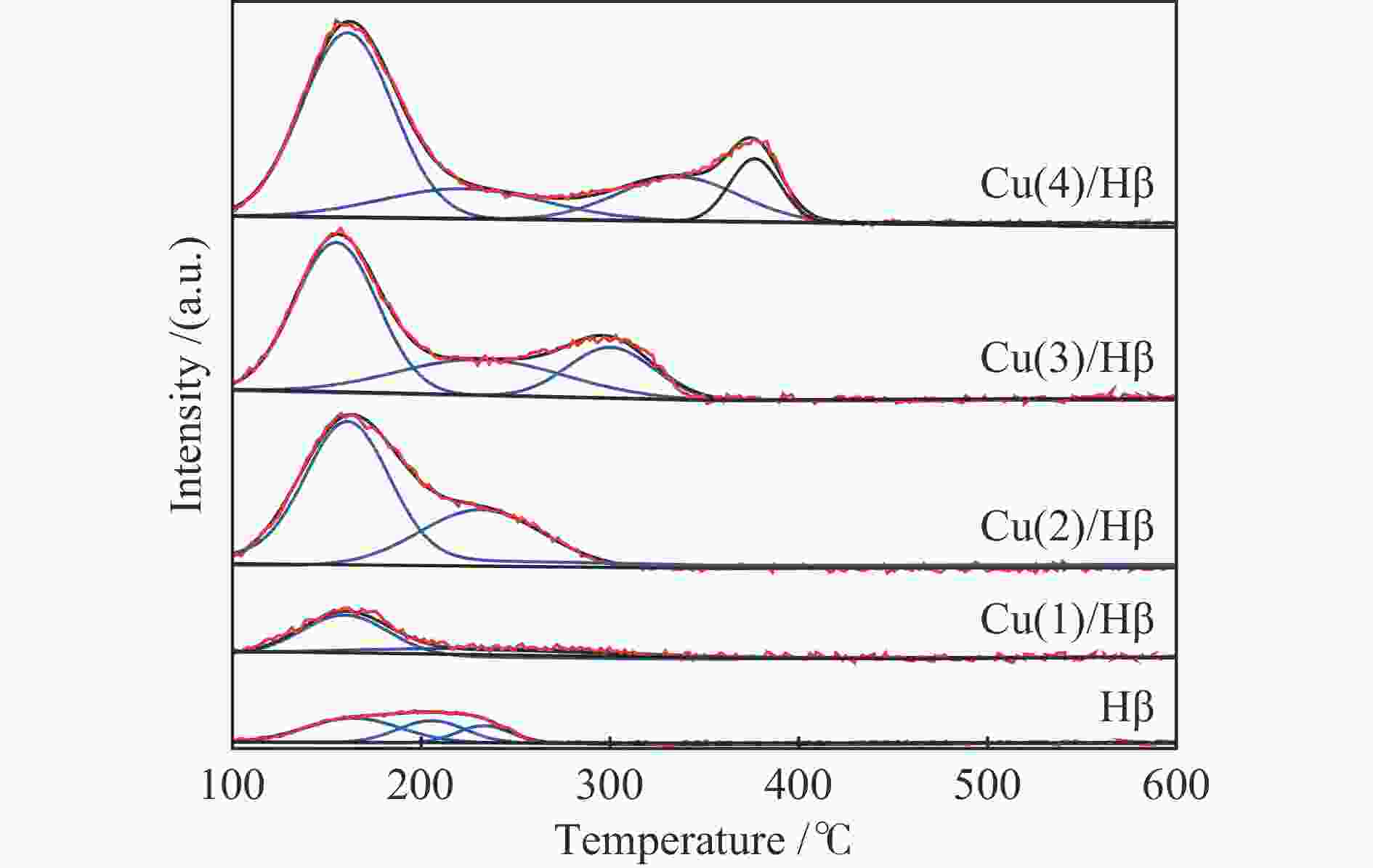
Table 2 Area and temperature of NH3 desorption peak over the Cu(x)/Hβ catalysts
Sample Area of acid sites /(a.u.) (Temperature /℃) Total weak acid medium strong acid strong acid Hβ 3.02(173) 4.26(231) 3.68(369) 10.96 Cu(1)/Hβ 2.05(169) 2.42(214) 2.16(325) 6.63 Cu(2)/Hβ 2.72(169) 2.87(212) 2.34(296) 7.93 Cu(3)/Hβ 2.74(167) 2.90(206) 2.83(295) 8.47 Cu(4)/Hβ 2.30(175) 2.84(225) 2.47(351) 7.61 表 3 Cu(x)/Hβ催化剂上NO脱附峰面积和温度
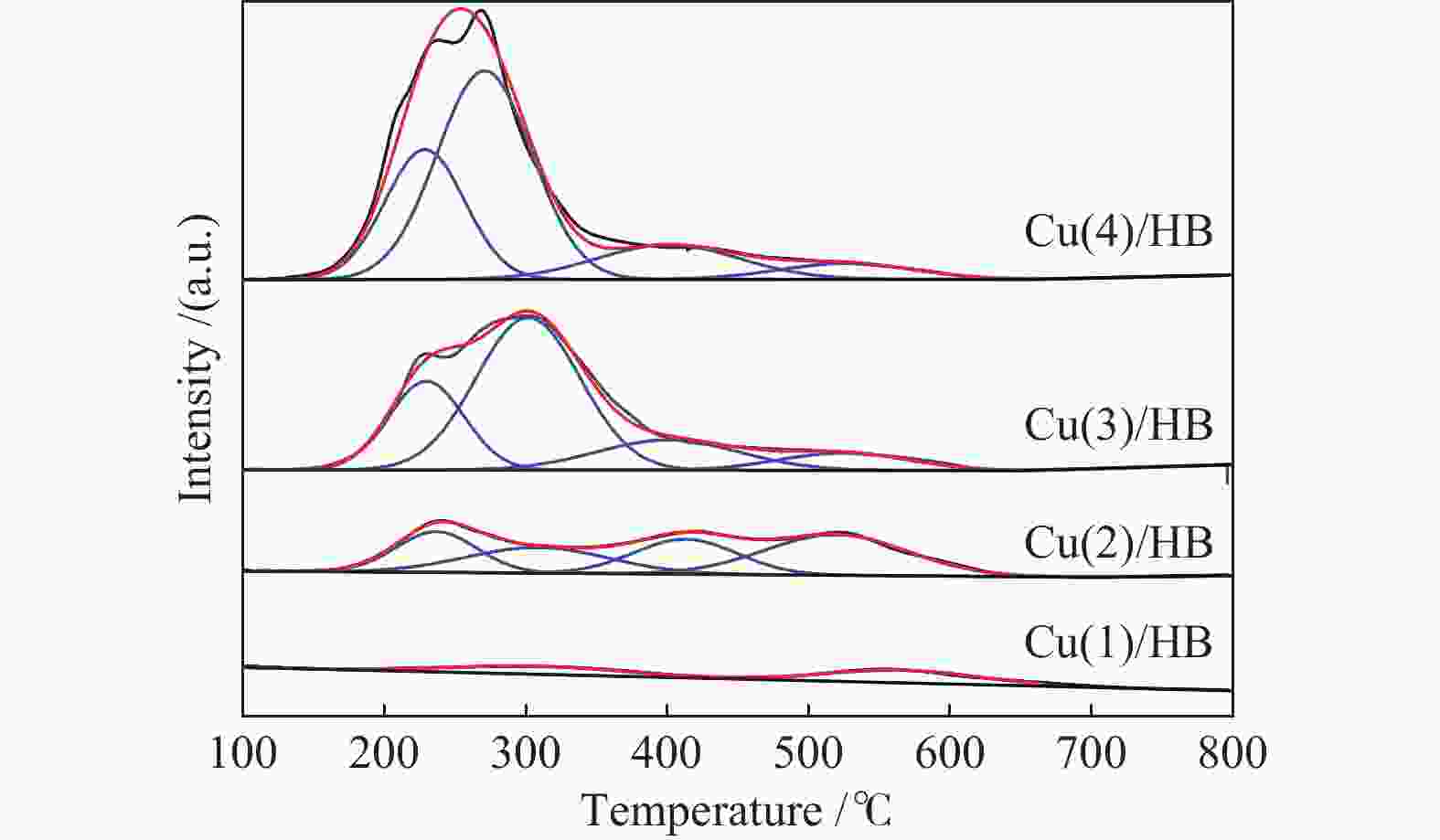
Table 3 Area and temperature of NO desorption peak over the Cu(x)/Hβ catalysts
Peak Area /a.u. (Temperature /℃) Hβ Cu(1)/Hβ Cu(2)/Hβ Cu(3)/Hβ Cu(4)/Hβ 1 0.59(164) 0.92 (160) 3.64 (161) 3.16 (155) 4.06(161) 2 0.38(205) 0.72(236) 1.80 (231) 1.55 (232) 1.09(224) 3 0.24(233) – – 1.11 (300) 1.28(334) 4 – – – – 0.76(375) Total 1.21 1.64 5.44 5.82 7.19 表 4 Cu(x)/Hβ催化剂上H2-TPR表征
Table 4 H2-TPR results of the Cu(x)/Hβ catalysts
Peak Cu(1)/Hβ Cu(2)/Hβ Cu(3)/Hβ Cu(4)/Hβ t/℃ area/(a.u) (pct./%) t/℃ area/(a.u) (pct./%) t/℃ area/(a.u) (pct./%) t/℃ area/(a.u) (pct./%) A – – 237 4.64 (20.0) 230 9.50 (23.0) 229 14.67 (27.8) B 308 2.03(42.3) 308 5.17 (22.3) 302 22.53 (54.5) 271 28.17 (53.2) C – – 413 5.13 (22.1) 400 6.11 (14.8) 401 7.01 (13.3) D 567 2.76(56.7) 519 8.23 (35.5) 525 3.18 (7.7) 527 3.01 (5.7) Total 4.79 23.17 41.32 52.85 表 5 Cu(2)/Hβ催化剂反应前后的电子能谱
Table 5 EDS analysis results of the catalyst at different reaction temperatures
Sample Cu(2)/Hβ Cu(2)/Hβ-100 Cu(2)/Hβ-150 Cu(2)/Hβ-200 Sulfur /% 0 3.1 1.9 0.6 -
[1] 张文博, 陈佳玲, 郭立, 郑伟, 王光华, 郑申棵, 吴晓琴. 金属负载型分子筛催化剂的NH3-SCR机理研究进展[J]. 燃料化学学报,2021,49(9):1294−1315. doi: 10.1016/S1872-5813(21)60080-4ZHANG Wen-bo, CHEN Jia-ling, GUO Li, ZHENG Wei, WANG Guang-hua, ZHEN Sheng-ke, WU Xiao-qin. Research progress on NH3-SCR mechanism of metal-supported zeolite catalysts[J]. J Fuel Chem Technol,2021,49(9):1294−1315. doi: 10.1016/S1872-5813(21)60080-4 [2] 张先龙, 胡晓芮, 刘仕雯, 杨祥瑾, 吴雪平, 王钧伟, 肖客松. 锰基累托石低温NH3-SCR催化剂的制备方法[J]. 环境化学,2022,41(3):1−9.ZHANG Xian-long, HU Xiao-rui, LIU Shi-wen, YANG Xiang-jing, WU Xue-ping, WANG Jun-wei, XIAO Ke-song. The preparation method of manganese-based rectorite low-temperature NH3-SCR catalyst[J]. Environ Chem,2022,41(3):1−9. [3] 汤常金, 孙敬方, 董林. 超低温(<150 ℃)SCR脱硝技术研究进展[J]. 化工学报,2020,71(11):4873−4884, 5362.TANG Chang-jin, SUN Jing-fang, DONG Lin. Recent progress on elimination of NOx from flue gas via SCR technology under ultra-low temperatures (< 150 ℃)[J]. CIESC J,2020,71(11):4873−4884, 5362. [4] 卞若愚, 安忠义, 李启超, 朱纯, 孙镇坤, 段伦博. O3-NH3协同活性焦脱硫脱硝的均相预反应特性研究[J]. 中国环境科学,2021,41(10):4476−4483. doi: 10.3969/j.issn.1000-6923.2021.10.002BIAN Ruo-yu, AN Zhong-yi, LI Qi-chao, ZHU Chun, SUN Zhen-kun, DUAN Lun-bo. Characteristics of simultaneous removal of NOxand SO2 by O3-NH3 synergy[J]. China Environ Sci,2021,41(10):4476−4483. doi: 10.3969/j.issn.1000-6923.2021.10.002 [5] MA L, CHENG Y, CAVATAIO G, MCCABE R W, FU L, LI J. Characterization of commercial Cu-SSZ-13 and Cu-SAPO-34 catalysts with hydrothermal treatment for NH3-SCR of NOx in diesel exhaust[J]. Chem Eng J,2013,225:323−330. doi: 10.1016/j.cej.2013.03.078 [6] 付金艳, 王振峰, 白心蕊, 崔梦壳, 武文斐. γ-Al2O3酸性修饰稀土尾矿NH3-SCR脱硝性能[J]. 中国环境科学,2020,40(9):3741−3747. doi: 10.3969/j.issn.1000-6923.2020.09.004FU Jin-yan, WANG Zhen-feng, BAI Xin-rui, CUI Men-kai, WU Wen-fei. Denitration performance of NH3-SCR from γ-Al2O3 acid modified rare earth tailings[J]. China Environ Sci,2020,40(9):3741−3747. doi: 10.3969/j.issn.1000-6923.2020.09.004 [7] ZHAN S, ZHANG H, ZHANG Y, SHI Q, LI Y, LI X. Efficient NH3-SCR removal of NOx with highly ordered mesoporous WO3(CHI)-CeO2 at low temperatures[J]. Appl Catal B: Environ,2017,203:199−209. doi: 10.1016/j.apcatb.2016.10.010 [8] LEE S M, PARK K H, Hong S C. MnOx/CeO2-TiO2 mixed oxide catalysts for the selective catalytic reduction of NO with NH3 at low temperature[J]. Chem Eng J,2012,195−196:323−331. [9] LI Y, LI Y, WANG P, HU W, ZHANG S, SHI Q, ZHAN S. Low-temperature selective catalytic reduction of NOx with NH3 over MnFeOx nanorods[J]. Chem Eng J,2017,330:213−222. doi: 10.1016/j.cej.2017.07.018 [10] ZHAO Z, YU R, ZHAO R, SHI C, GIES H, XIAO F-S, DE VOS D, YOKOI T, BAO X, KOLB U, FEYEN M, MCGUIRE R, MAURER S, MOINI A, MüLLER U, ZHANG W. Cu-exchanged Al-rich SSZ-13 zeolite from organotemplate-free synthesis as NH3-SCR catalyst: Effects of Na+ ions on the activity and hydrothermal stability[J]. Appl Catal B: Environ,2017,217:421−428. doi: 10.1016/j.apcatb.2017.06.013 [11] ZHAO H, ZHAO Y, MA Y, YONG X, WEI M, CHEN H, ZHANG C, LI Y. Enhanced hydrothermal stability of a Cu-SSZ-13 catalyst for the selective reduction of NOx by NH3 synthesized with SAPO-34 micro-crystallite as seed[J]. J Catal,2019,377:218−223. doi: 10.1016/j.jcat.2019.07.023 [12] YUE Y, LIU B, QIN P, LV N, WANG T, BI X, ZHU H, YUAN P, BAI Z, CUI Q, BAO X. One-pot synthesis of FeCu-SSZ-13 zeolite with superior performance in selective catalytic reduction of NO by NH3 from natural aluminosilicates[J]. Chem Eng J,2020,398:125515. doi: 10.1016/j.cej.2020.125515 [13] 马子然, 王宝冬, 路光杰, 肖雨亭, 杨建辉, 陆金丰, 李歌, 周佳丽, 王红妍, 赵春林. 粉煤灰基SAPO-34分子筛脱硝催化剂的合成及其脱硝性能[J]. 化工进展,2020,39(10):4051−4060. doi: 10.16085/j.issn.1000-6613.2020-0011MA Zi-ran, WANG Bao-dong, LU Guang-jie, XIAO Yu-ting, YANG Jian-hui, LU Jin-feng, LI Ge, ZHOU Jia-li, WANG Hong-yan, ZHAO Chun-lin. Preparation and performance of SAPO-34 based SCR catalyst derived from fly ash[J]. Chem Ind Eng Prog,2020,39(10):4051−4060. doi: 10.16085/j.issn.1000-6613.2020-0011 [14] LI Q J, PEIMM, YAOP, XU S, XU S H, LIUS, XU HD, DAN Y, CHEN Y Q. Determining hydrothermal deacyivation mechanisms on Cu/SAPO-34 NH3-SCR catalysts at low- and hing-reaction regions: establishing roles of different reaction sites[J]. Rare Metals,2022,41(6):1899−1910. doi: 10.1007/s12598-021-01933-8 [15] 其其格吉日嘎拉, 李晨曦, 叶青, 程锦, 程水源, 康天放. La-Cu/ZSM-5催化剂NH3选择性催化还原NO的性能[J]. 环境化学,2020,39(9):2567−2575. doi: 10.7524/j.issn.0254-6108.2019062503MUNKHJARGAL TSETSEGJARGAL, LI Chen-xi, YE Qing, CHENG Jin, CHENG Shui-yuan, KANG Tian-fang. La-Cu/ZSM-5 catalysts electively reduced NO by NH3-SCR[J]. Environ Chem,2020,39(9):2567−2575. doi: 10.7524/j.issn.0254-6108.2019062503 [16] 邱爽, 肖永厚, 刘建辉, 贺高红. 一步法制备高活性NH3-SCR催化剂Cu-SAPO-34: Si含量的影响[J]. 化工学报,2021,72(5):2578−2585.QIU Shuang, XIAO Yong-hou, LIU Jian-hui, HE Gao-hong. Enhanced NH3-SCR performance over Cu-SAPO-34: Si prepared by one-step synthesis: Effect of Si contents[J]. CIESC J,2021,72(5):2578−2585. [17] IMAI H, HAYASHIDA N, YOKOI T, TATSUMI T. Direct crystallization of CHA-type zeolite from amorphous aluminosilicate gel by seed-assisted method in the absence of organic-structure-directing agents[J]. Microporous Mesoporous Mater,2014,196:341−348. doi: 10.1016/j.micromeso.2014.05.043 [18] XIE B, ZHANG H, YANG C, LIU S, REN L, ZHANG L, MENG X, YILMAZ B, MULLER U, XIAO F S. Seed-directed synthesis of zeolites with enhanced performance in the absence of organic templates[J]. Chem Commun,2011,47(13):3945−3947. doi: 10.1039/c0cc05414c [19] LIU Q, LIU Z, WU W. Effect of V2O5 additive on simultaneous SO2 and NO removal from flue gas over a monolithic cordierite-based CuO/Al2O3 catalyst[J]. Catal Today,2009,147:S285−S289. doi: 10.1016/j.cattod.2009.07.013 [20] GAO F, WALTER E D, KARP E M, LUO J, TONKYN R G, KWAK J H, SZANYI J, PEDEN C H F. Structure-activity relationships in NH3-SCR over Cu-SSZ-13 as probed by reaction kinetics and EPR studies[J]. J Catal,2013,300:20−29. doi: 10.1016/j.jcat.2012.12.020 [21] 任翠涛, 胡颖智, 魏浩宇, 李滨, 王虹, 丁福臣, 李翠清, 宋永吉. SO2存在条件下M/REY催化剂NH3选择性还原NO性能研究[J]. 燃料化学学报,2013,41(10):1241−1247.REN Cui-tao, HU Ying-zhi, WEI Hao-yu, LI Bin, WANG Hong, DING Fu-chen, LI Cui-qing, SONG Yong-ji. NH3 selective catalytic reduction of NO over M/REY catalysts in presence of SO2[J]. J Fuel Chem Technol,2013,41(10):1241−1247. [22] SHEVLIN S. Looking deeper into zeolites[J]. Nat Mater,2020,19(10):1038−1039. doi: 10.1038/s41563-020-0787-4 [23] HINCAPIE B O, GARCES L J, ZHANG Q, SACCO A, SUIB S L. Synthesis of mordenite nanocrystals[J]. Microporous Mesoporous Mater,2004,67(1):19−26. doi: 10.1016/j.micromeso.2003.09.026 [24] RAVI M, SUSHKEVICH V L, VAN B J A. Towards a better understanding of Lewis acidic aluminium in zeolites[J]. Nat Mater,2020,19(10):1047−1056. doi: 10.1038/s41563-020-0751-3 [25] SU W, LI Z, PENG Y, LI J. Correlation of the changes in the framework and active Cu sites for typical Cu/CHA zeolites (SSZ-13 and SAPO-34) during hydrothermal aging[J]. Phys Chem Chem Phys,2015,17(43):29142−29149. doi: 10.1039/C5CP05128B [26] XU M, WANG J, YU T, WANG J, SHEN M. New insight into Cu/SAPO-34 preparation procedure: Impact of NH4-SAPO-34 on the structure and Cu distribution in Cu-SAPO-34 NH3-SCR catalysts[J]. Appl Catal B: Environ,2018,220:161−170. doi: 10.1016/j.apcatb.2017.08.031 [27] LIU B, LV N G, WANG C, ZHANG H W, YUE Y Y, JINGDONG XU J D, BI X T, BAO X J. Redistributing Cu species in Cu-SSZ-13 zeolite as NH3-SCR catalyst via a simple ion-exchange[J]. Chin J Chem Eng,2022,41:329−341. doi: 10.1016/j.cjche.2021.10.027 [28] WANG H M, LI W, XU S Y, LIU M, HAO J M, NING P, QIULIN ZHANG Q L. Insights into the impact of lanthanum on hydrothermal-induced migration and transformation of copper species in Cu/SAPO-34 catalyst for NH3-SCR[J]. Mol Catal,2021,515:111914. doi: 10.1016/j.mcat.2021.111914 [29] MING S J, PANG L, CHEN Z, GUO Y B, GUO L, LIU Q, LIU P, DONG Y H, ZHANG S T, LI T. Insight into SO2 poisoning over Cu-SAPO-18 used for NH3-SCR[J]. Microporous Mesoporous Mater,2020,303:110294. doi: 10.1016/j.micromeso.2020.110294 [30] 白书立, 张晓玉, 薛瑶佳, 李换英, 郏建波. 碳化硅负载氧化铜催化剂低温NH3选择性催化还原NOx的性能[J]. 燃料化学学报,2020,48(6):723−727. doi: 10.3969/j.issn.0253-2409.2020.06.011BAI Shu-li L, ZHANG Xiao-yu, XUE Yao-jia, LI Huan-ying, JIA Jian-bo. Silicon carbon-supported copper oxide catalysts for the selective catalytic reduction of NOx with NH3 at low temperature[J]. J Fuel Chem Technol,2020,48(6):723−727. doi: 10.3969/j.issn.0253-2409.2020.06.011 [31] YE D, REN X, QU R, LIU S, ZHENG C, GAO X. Designing SO2-resistant cerium-based catalyst by modifying with Fe2O3 for the selective catalytic reduction of NO with NH3[J]. Mol Catal,2019,462:10−18. doi: 10.1016/j.mcat.2018.10.007 [32] 焦坤灵, 赵阳国, 武文斐, 王振峰, 龚志军. SO2对稀土精矿催化剂NH3-SCR脱硝催化性能的影响[J]. 化工学报,2019,70(12):4645−4653.JIAO Kun-ling, ZHAO Yang-guo, WU Wen-fei, WANG Zhen-feng, GONG Zhi-jun. Effect of SO2 on catalytic performance of rare earth concentrate catalyst for NH3-SCR denitrification[J]. CIESC J,2019,70(12):4645−4653. [33] 魏永林, 陈红萍, 侯欣辛, 杨旭, 李泽清. Fe-Mn/TiO2低温NH3-SCR脱硝催化剂的SO2中毒机理[J]. 功能材料,2021,52(4):4132−4139, 4146. doi: 10.3969/j.issn.1001-9731.2021.04.020WEI Yong-lin, CHEN Hong-ping, HOU Xin-xin, YANG Xu, LI Ze-qing. SO2 poisoning mechanism of Fe-Mn/TiO2 catalyst for low-temperature NH3-SCR deniteation[J]. J Funct Mater,2021,52(4):4132−4139, 4146. doi: 10.3969/j.issn.1001-9731.2021.04.020 [34] 陈潇雪, 宋敏, 孟凡跃, 卫月星. FexMnCe1-AC低温SCR催化剂SO2中毒机理研究[J]. 化工学报,2019,70(8):3000−3010.CHEN Xiao-xue, SONG Min, MENG Fan-yue, WEI Yue-xing. Mechanism study on SO2 poisoning of FexMnCe1-AC catalyst for low-temperature SCR[J]. CIESC J,2019,70(8):3000−3010. [35] 肖雨亭, 吴鹏, 王玲, 张亚平. Ce改性Fe-Mn/TiO2低温SCR脱硝催化剂硫中毒机理[J]. 化工环保,2019,39(4):431−436. doi: 10.3969/j.issn.1006-1878.2019.04.011XIAO Yu-ting, WU Peng, WANG Ling, ZHANG Ya-ping. Mechanism of sulfur poisoning on low-temperature SCR denitration catalyst Ce-modified Fe-Mn/TiO2[J]. Environ Prot Chem Ind,2019,39(4):431−436. doi: 10.3969/j.issn.1006-1878.2019.04.011 [36] ROMANO E J, SCHULZ K H. A XPS investigation of SO2 adsorption on ceria-zirconia mixed-metal oxides[J]. Appl Surf Sci,2005,246(1/3):262−270. -




 下载:
下载: