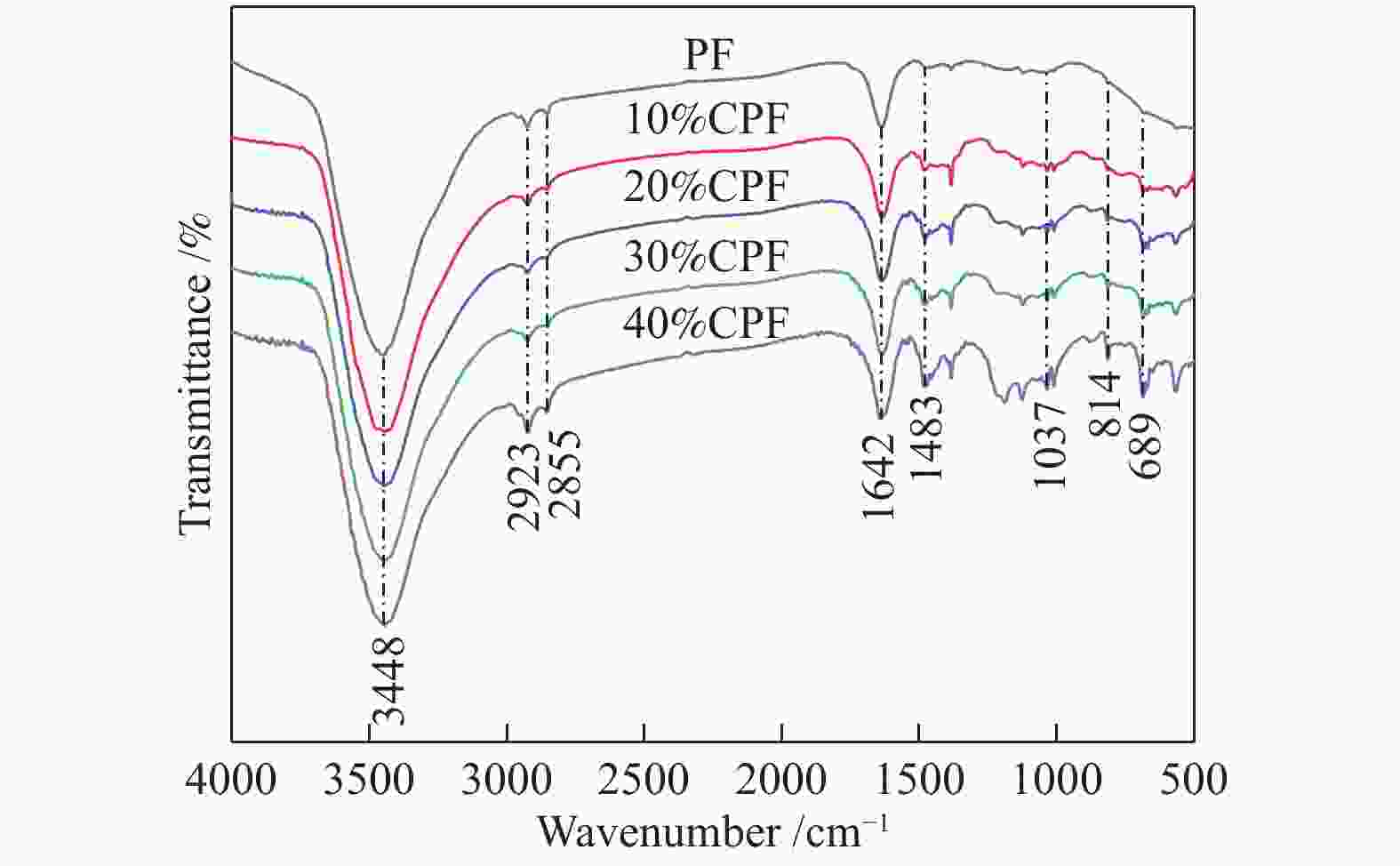
Preparation and characterization of low-temperature coal tar toughened phenolic foams
-
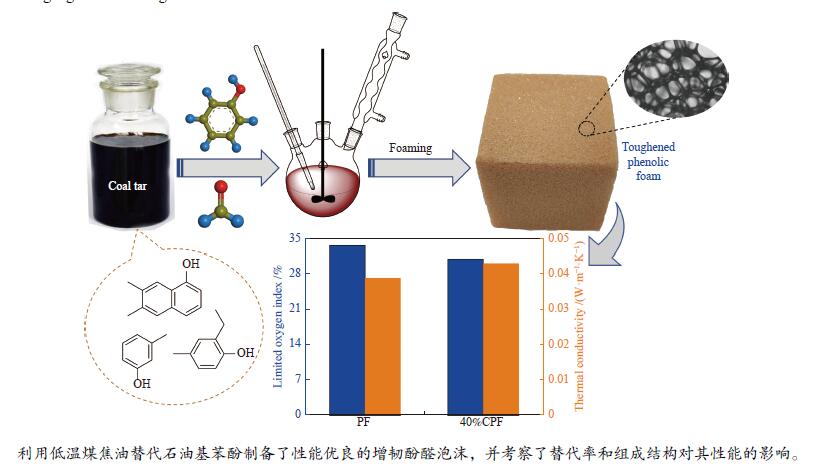
摘要: 本研究以低温煤焦油为原料,部分替代石油基苯酚制备煤焦油基酚醛泡沫(CPF),对CPFs的化学结构、表观形貌、压缩强度、粉化率、热稳定性、阻燃性能和隔热性能进行了表征。结果表明,CPFs与常规酚醛泡沫的化学结构相似。与常规酚醛泡沫相比,30%CPF和40%CPF的压缩强度分别增加了18.3%和55.9%;且由于脂肪结构如烷基酚的引入,使得泡沫的韧性显著提高,其粉化率分别下降了22.9%和50.8%。此外,CPFs在低温下的热稳定性增加。尽管CPFs的极限氧指数下降,导热系数增加,但依然保持较好的阻燃和隔热性能。这说明低温煤焦油能够高比例地替代苯酚制备出性能优良的酚醛泡沫,为低温煤焦油的高值化利用提供了新的思路。Abstract: In this study, coal tar-based phenolic foam (CPF) was prepared using low-temperature coal tar as raw material to partially replace phenol. The chemical structure, apparent morphology, compressive strength, thermal stability, flame retardancy and thermal insulation properties of CPFs were characterized. The results show that CPFs have similar chemical structures to conventional phenolic foam. Comparing with conventional phenolic foam, the compressive strength of 30%CPF and 40%CPF increases by 18.3% and 55.9%, and the pulverization rate decreases by 22.9% and 50.8%, respectively. The results indicated that toughness was significantly strengthened due to the incorporation of aliphatic structures such as alkylphenols. In addition, the thermal stability of CPFs in the low temperature stage also improves. Although the limited oxygen index of CPFs decreases and thermal conductivity of CPFs increases, they still maintain good flame retardancy and thermal insulation properties. The obtained results prove that low-temperature coal tar can significantly replace phenol to prepare phenolic foam with good performance, which provides a new idea for the high-value utilization of low-temperature coal tar.
-
Key words:
- low-temperature coal tar /
- phenolic foams /
- toughness /
- characterization
-
Table 1 Formulations of PR and CPRs with different substitution rates
Substitution rate /% Low-temp. coal tar /g Phenol /g 37% aqueous formaldehyde /g 15% aqueous NaOH /mL 0 0 50 77.54 15 10 5 45 71.34 15 20 10 40 65.13 15 30 15 35 58.93 15 40 20 30 52.73 15 Table 2 Basic properties of PR and CPRs
Resin PR 10%CPR 20%CPR 30%CPR 40%CPR Viscosity /(Pa·s) 4.1 6.9 10.4 20.7 48.7 Solid content /% 79.2 77.7 76.6 75.4 74.3 Table 3 Arenes with RC > 0.2% detected in low-temperature coal tar by GC/MS
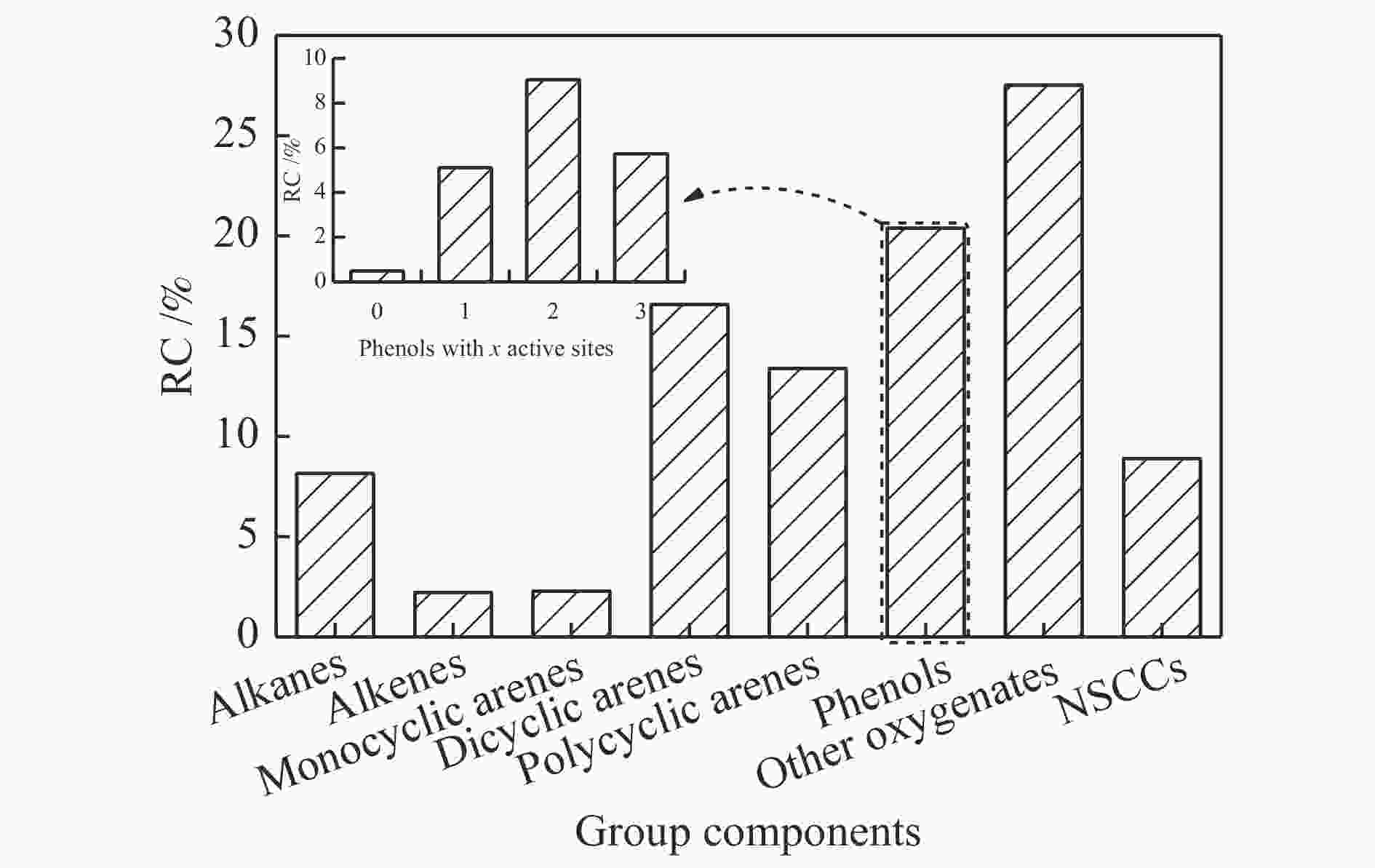
Arene RC Arene RC Naphthalene 0.32 2-methyl-9H-fluorene 0.63 5H-benzo[7]annulene 0.60 2,6-dimethylbiphenyl 0.49 2-methylnaphthalene 0.36 2,4,6-trimethylbiphenyl 0.67 1,2,4-trimethyl-5-(prop-1-en-2-yl)benzene 0.21 3,3',4,4'-tetramethylbiphenyl 0.22 2,6-dimethylnaphthalene 0.31 3,4-diethylbiphenyl 0.70 1,7-dimethylnaphthalene 0.75 1a,9b-dihydro-1H-cyclopropa[l]phenanthrene 1.16 3-ethyl-1,2,4,5-tetramethylbenzene 0.43 2,3-dimethylphenanthrene 0.75 1,1,4,5,6-pentamethylindane 0.29 pyrene 0.26 4,6,8-trimethylazulene 0.84 2,3,5-trimethylphenanthrene 1.16 1,6,7-trimethylnaphthalene 0.85 1-methylpyrene 0.46 Phenalene 0.29 2-isopropyl-10-methylphenanthrene 1.22 1-allylnaphthalene 0.28 1-methyl-4-((4-propylphenyl)ethynyl)benzene 0.21 4-methylbiphenyl 0.21 4,8-dimethyl-6-phenylazulene 0.30 3,4'-dimethylbiphenyl 0.42 1,3-dimethyl-8-isopropyl-phenanthrene 0.29 6-isopropyl-1,4-dimethylnaphthalene 0.25 tetracene 0.22 RC: relative content Table 4 Other oxygenates with RC > 0.2% detected in low-temperature coal tar by GC/MS
Other oxygenate RC Other oxygenate RC 2-acetyl-2-carene 0.21 1-heneicosyl formate 0.24 2-methylindan-1-ol 0.33 3-hydroxy-estra-1,3,5(10),9(11)-tetraen-17-one 0.40 2,3,4,5-tetramethylbenzaldehyde 0.20 1-heneicosanol 0.52 2-methyl-4-(2,6,6-trimethylcyclohex-1-en-1-yl)but-3-enal 0.31 methyl 3-(3-methylbut-2-en-1-yl)-1,4-dioxo-1,4-dihydronaphthalene-2-carboxylate 0.31 1,2a,3,4,5,7,8,9-octahydro-2H-benzo[cd]pyren-5-ol 0.30 11-methyl-10,11-dihydrotetraphene-10,11-diol 0.29 2,3-diphenylcycloprop-2-en-1-one 0.25 1,2-dimethylnaphtho[2,1-b]furan 0.26 4,6,8-trimethyl-1-azulenecarbaldehyde 0.45 Table 5 Phenols detected in low-temperature coal tar by GC/MS
Phenols RC Phenol RC With 0 active site 2,3,5-trimethylphenol 0.14 6,6'-methylenebis(2-(tert-butyl)-4-methylphenol) 0.50 2,6-dimethyl-1,4-benzenediol 0.35 With 1 active sites 2,5-dimethyl-1,3-benzenediol 0.04 2,4-xylenol 1.48 2-(2-methylallyl)phenol 0.98 2,3,6-trimethylphenol 0.16 4,5-dimethyl-1,3-benzenediol 0.08 2-allyl-6-cresol 0.01 6-methyl-4-indanol 0.27 2-ethyl-6-cresol 0.34 5,8-dihydronaphthalen-1-ol 0.16 3-methylcatechol 0.30 [1,1'-biphenyl]-2-ol 0.03 2-ethyl-4-cresol 1.26 naphthalen-1-ol 0.61 2,3,6-trimethylphenol 0.23 9H-fluoren-2-ol 0.80 2-ethyl-4,5-xyenol 0.23 5,7-dimethylnaphthalen-1-ol 0.08 6-propyl-2-cresol 0.10 6,7-dimethylnaphthalen-1-ol 1.74 4-methyl-2-(pent-3-en-2-yl)phenol 0.17 2-styrylphenol 0.11 2-methylnaphthalen-1-ol 0.18 4-(cyclohepta-2,4,6-trien-1-yl)phenol 0.17 3,6-dimethyl-2-(2-methylbut-3-en-2-yl)phenol 0.07 4-benzylphenol 0.04 2,5,8-trimethylnaphthalen-1-ol 0.28 dibenzo[b,d]furan-2-ol 0.19 1,2,3,4-tetrahydrophenanthren-9-ol 0.28 4-styrylphenol 0.21 with 2 active sites phenanthren-2-ol 0.22 2-ethylphenol 0.22 1-phenyl-1H-inden-4-ol 0.54 2,5-xyenol 0.44 with 3 active sites 3,4-xyenol 0.39 phenol 0.78 Catechol 0.20 3-cresol 2.84 2-ethyl-5-cresol 0.21 3-ethylphenol 1.52 3,4,5-trimethylphenol 0.41 orcinol 0.09 4-methylcatechol 0.31 3,5-diethylphenol 0.50 Thymol 0.09 Table 6 Basic characteristics of PF and CPFs
Foam Density /
(kg·m−3)Compressive strength /
MPaPulverization rate /
%Thermal conductivity /
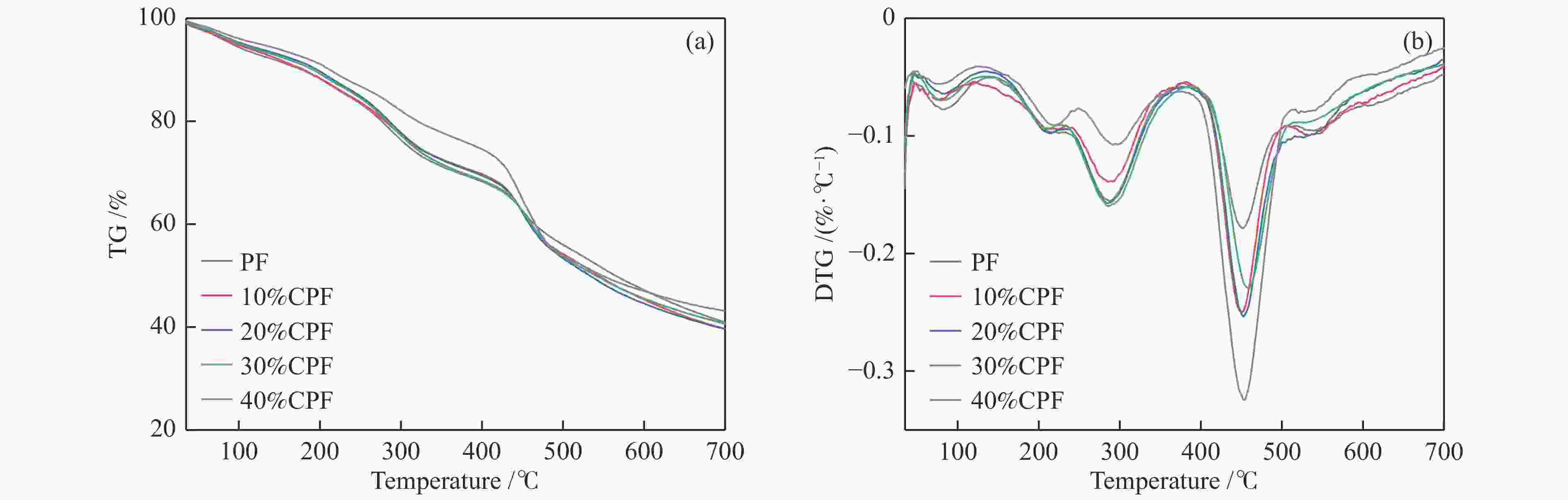
(W·m−1·K−1)PF 50.8 0.172 11.8 0.0391 10%CPF 50.2 0.107 11.6 0.0395 20%CPF 62.4 0.166 9.1 0.0414 30%CPF 93.0 0.268 5.8 0.0443 40%CPF 77.3 0.203 9.1 0.0432 Table 7 TG and DTG analysis of PF and CPFs
Sample t-5% /°C tmax /°C Residual
mass /%step II step III PF 90.9 288.1 451.5 40.9 10%CPF 96.4 285.2 450.4 39.7 20%CPF 106.2 284.8 451.1 39.6 30%CPF 103.0 286.2 458.3 40.6 40%CPF 123.6 291.4 453.2 43.1 -
[1] HUANG Y, FENG J, LIANG C H, HUANG P, ZHANG X W, XIE Q, LI W Y. Co-production of naphthenic oil and phenolic compounds from medium- and low-temperature coal tar[J]. Ind Eng Chem Res,2021,60(16):5890−5902. doi: 10.1021/acs.iecr.1c00390 [2] YAO Q, LI Y, TANG X, GAO J, WANG R, ZHANG Y, SUN M, MA X. Separation of petroleum ether extracted residue of low temperature coal tar by chromatography column and structural feature of fractions by TG-FTIR and PY-GC/MS[J]. Fuel,2019,245:122−130. doi: 10.1016/j.fuel.2019.02.074 [3] XIE K. Reviews of clean coal conversion technology in China: Situations & challenges[J]. Chin J Chem Eng,2021,35:62−69. doi: 10.1016/j.cjche.2021.04.004 [4] LIU J, AHMAD F, ZHANG Q, LIANG L, HUANG W, PENG Z, YUAN Q, XIANG X. Interactive tools to assist convenient group-type identification and comparison of low-temperature coal tar using GC × GC-MS[J]. Fuel,2020,278:118314. doi: 10.1016/j.fuel.2020.118314 [5] GUO X H, WEI X Y, HU L, LIU X J, CHEN M X, XU M L, ZONG Z M. Molecular characterization of a middle/low-temperature coal tar by multiple mass spectrometries[J]. Fuel,2021,306:121435. doi: 10.1016/j.fuel.2021.121435 [6] GANG Y, PAN L, NIU M, ZHANG X, LI D, LI W. Catalytic hydrogenation of Low temperature coal tar into jet fuel by using two-reactors system[J]. J Anal Appl Pyrolysis,2018,134:202−208. doi: 10.1016/j.jaap.2018.06.009 [7] ZHANG X Q, KANG Y H, GAO J, XIONG L, GAO Y, CHEN T, LIU G H, WANG A M, WEI X Y, ZONG Z M, BAI H C. Effective hydroconversion of heteroatom-containing organic species from the extraction of low-temperature coal tar to cycloalkanes over a Y/Beta composite zeolite supported nickel nanoparticles[J]. Fuel,2022,321:124062. doi: 10.1016/j.fuel.2022.124062 [8] GAI H, QIAO L, ZHONG C, ZHANG X, XIAO M, SONG H. Designing ionic liquids with dual Lewis basic sites to efficiently separate phenolic compounds from low-temperature coal tar[J]. ACS Sustainable Chem Eng,2018,6(8):10841−10850. doi: 10.1021/acssuschemeng.8b02119 [9] GAI H, QIAO L, ZHONG C, ZHANG X, XIAO M, SONG H. A solvent based separation method for phenolic compounds from low-temperature coal tar[J]. J Clean Prod,2019,223:1−11. doi: 10.1016/j.jclepro.2019.03.102 [10] LI Y, LUO H A, AI Q, YOU K, ZHAO F, XIAO W. Efficient separation of phenols from coal tar with aqueous solution of amines by liquid-liquid extraction[J]. Chin J Chem Eng,2021,35:180−188. doi: 10.1016/j.cjche.2021.01.008 [11] ZHANG Z, LOU B, ZHAO N, YU E, WANG Z, DU H, CHEN Z, LIU D. Co-carbonization behavior of the blended heavy oil and low temperature coal tar for the preparation of needle coke[J]. Fuel,2021,302:121139. doi: 10.1016/j.fuel.2021.121139 [12] TIAN Y, HUANG Y, YU X, GAO F, GAO S, WANG F, LI D, XU X, CUI L, FAN X, DONG H, LIU J. Co-carbonization of medium- and low-temperature coal tar pitch and coal-based hydrogenated diesel oil prepare mesophase pitch for needle coke precursor[J]. Adv Eng Mater,2021,23(10):2001523. doi: 10.1002/adem.202001523 [13] PYSHYEV S, DEMCHUK Y, POLIUZHYN I, KOCHUBEI V. Obtaining and use adhesive promoters to bitumen from the phenolic fraction of coal tar[J]. Inter Adhes Adhes,2022,118:103191. doi: 10.1016/j.ijadhadh.2022.103191 [14] HUANG R, YUAN X, YAN L, HAN L, BAO W, CHANG L, LIU J, WANG J, OK Y S. Carbon precursors in coal tar: Extraction and preparation of carbon materials[J]. Sci Total Environ,2021,788:147697. doi: 10.1016/j.scitotenv.2021.147697 [15] SANDHYA P K, SREEKALA M S, SABU T. Phenolic Based Foams[M]. Berlin: Springer, 2022: 1−3. [16] MOUGEL C, GARNIER T, CASSAGNAU P, SINTES-ZYDOWICZ N. Phenolic foams: A review of mechanical properties, fire resistance and new trends in phenol substitution[J]. Polymer,2019,164:86−117. doi: 10.1016/j.polymer.2018.12.050 [17] XU Y, GUO L, ZHANG H, ZHAI H, REN H. Research status, industrial application demand and prospects of phenolic resin[J]. RSC Adv,2019,9(50):28924−28935. doi: 10.1039/C9RA06487G [18] ZHOU M, SHI H, LI C, SHENG X, SUN Y, HOU M, NIU M, PAN X. Depolymerization and activation of alkali lignin by solid acid-catalyzed phenolation for preparation of lignin-based phenolic foams[J]. Ind Eng Chem Res,2020,59(32):14296−14305. doi: 10.1021/acs.iecr.0c01753 [19] LI B, YUAN Z, SCHMIDT J, XU C. New foaming formulations for production of bio-phenol formaldehyde foams using raw kraft lignin[J]. Eur Polym J,2019,111:1−10. doi: 10.1016/j.eurpolymj.2018.12.011 [20] GAO C, LI M, ZHU C, HU Y, SHEN T, LI M, JI X, LYU G, ZHUANG W. One-pot depolymerization, demethylation and phenolation of lignin catalyzed by HBr under microwave irradiation for phenolic foam preparation[J]. Compos Part B: Eng,2021,205:108530. doi: 10.1016/j.compositesb.2020.108530 [21] GAO Z, LANG X, CHEN S, ZHAO C. Mini-review on the synthesis of lignin-based phenolic resin[J]. Energy Fuels,2021,35(22):18385−18395. doi: 10.1021/acs.energyfuels.1c03177 [22] ISSAOUI H, DE HOYOS-MARTINEZ P L, PELLERIN V, DOURGES M-A, DELEUZE H, BOURBIGO S, CHARRIER-EL BOUHTOURY F. Effect of catalysts and curing temperature on the properties of biosourced phenolic foams[J]. ACS Sustainable Chem Eng,2021,9(18):6209−6223. doi: 10.1021/acssuschemeng.0c08234 [23] CHEN S, XIN Y, ZHAO C. Multispectroscopic analysis in the synthesis of lignin-based biophenolic resins[J]. ACS Sustainable Chem Eng,2021,9(46):15653−15660. doi: 10.1021/acssuschemeng.1c06135 [24] CHENG J Y, LI Z K, YAN H L, LEI Z P, YAN J C, REN S B, WANG Z C, KANG S G, SHUI H F. Preparation and performance of high-temperature coal tar toughened phenolic foams[J]. J Fuel Chem Technol,2022,50(5):530−537. [25] YU Y, WANG Y, XU P, CHANG J. Preparation and characterization of phenolic foam modified with bio-oil[J]. Materials (Basel),2018,11(11):2228. doi: 10.3390/ma11112228 [26] LI B, WANG Y, MAHMOOD N, YUAN Z, SCHMIDT J, XU C. Preparation of bio-based phenol formaldehyde foams using depolymerized hydrolysis lignin[J]. Ind Crop Prod,2017,97:409−416. doi: 10.1016/j.indcrop.2016.12.063 [27] LI Z K, WANG H T, YAN H L, YAN J C, LEI Z P, REN S B, WANG Z C, KANG S G, SHUI H F. Catalytic ethanolysis of Xilinguole lignite over layered and mesoporous metal oxide composites to platform chemicals[J]. Fuel,2021,287:119560. doi: 10.1016/j.fuel.2020.119560 [28] SUN M, WANG L, ZHONG J, YAO Q, CHEN H, JIAO L, HAO Q, MA X. Chemical modification with aldehydes on the reduction of toxic PAHs derived from low temperature coal tar pitch[J]. J Anal Appl Pyrolysis,2020,148:104822. doi: 10.1016/j.jaap.2020.104822 [29] CUI Y, HOU X, WANG W, CHANG J. Synthesis and characterization of bio-oil phenol formaldehyde resin used to fabricate phenolic based materials[J]. Materials (Basel),2017,10(6):668. [30] WANG G, LIU X, ZHANG J, SUI W, JANG J, SI C. One-pot lignin depolymerization and activation by solid acid catalytic phenolation for lightweight phenolic foam preparation[J]. Ind Crop Prod,2018,124:216−225. doi: 10.1016/j.indcrop.2018.07.080 [31] SONG F, JIA P, BO C, REN X, HU L, ZHOU Y. The mechanical and flame retardant characteristics of lignin-based phenolic foams reinforced with MWCNTs by in-situ polymerization[J]. J Disper Sci Technol,2020,42(7):1042−1051. [32] LI Q, CHEN L, LI X, ZHANG J, ZHENG K, ZHANG X, TIAN X. Effect of nano-titanium nitride on thermal insulating and flame-retardant performances of phenolic foam[J]. J Appl Poly Sci,2016,133(32):43765. -




 下载:
下载: