Morphology study of nickel and vanadium in asphaltenes via hydropyrolysis
-
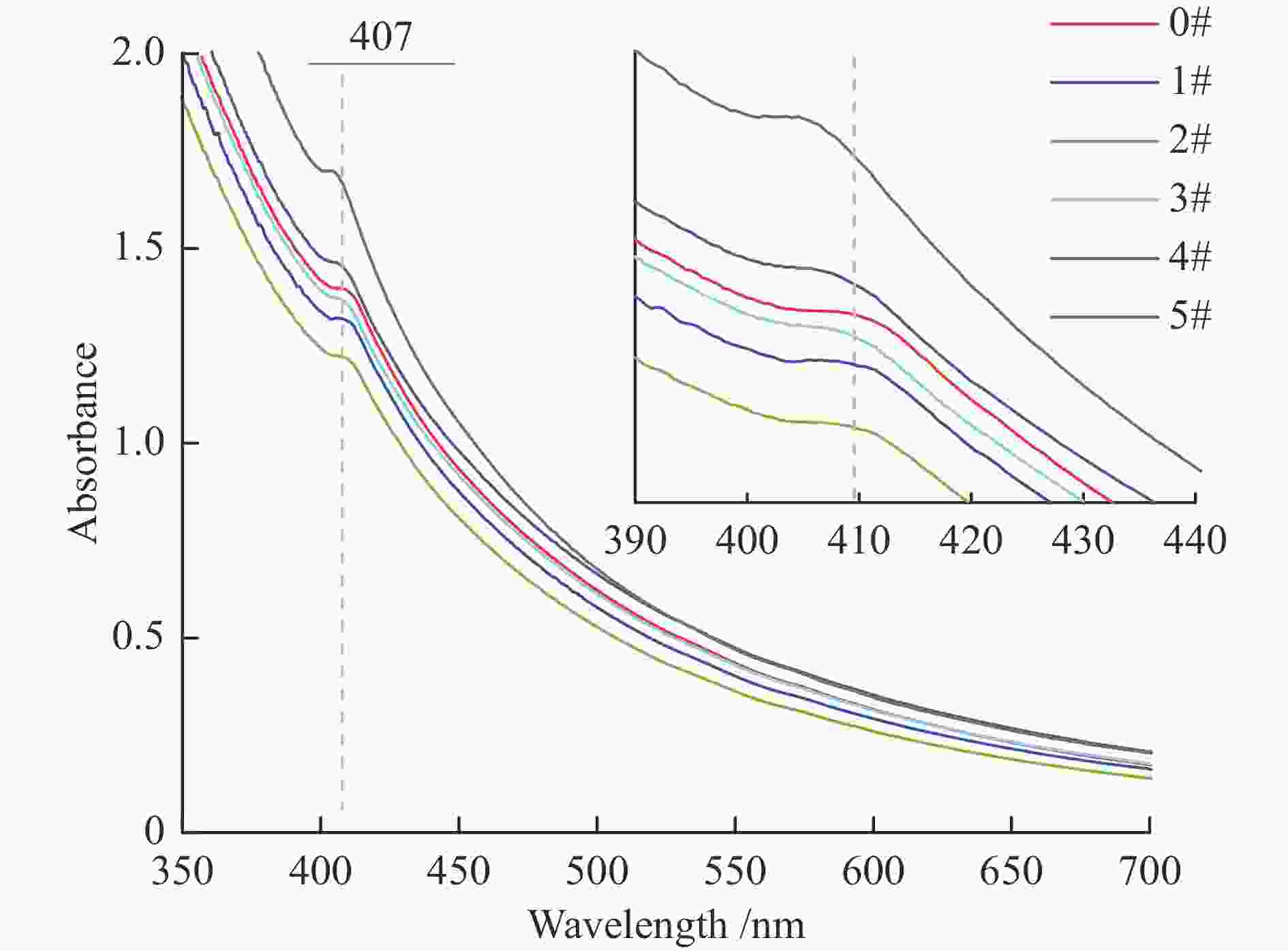
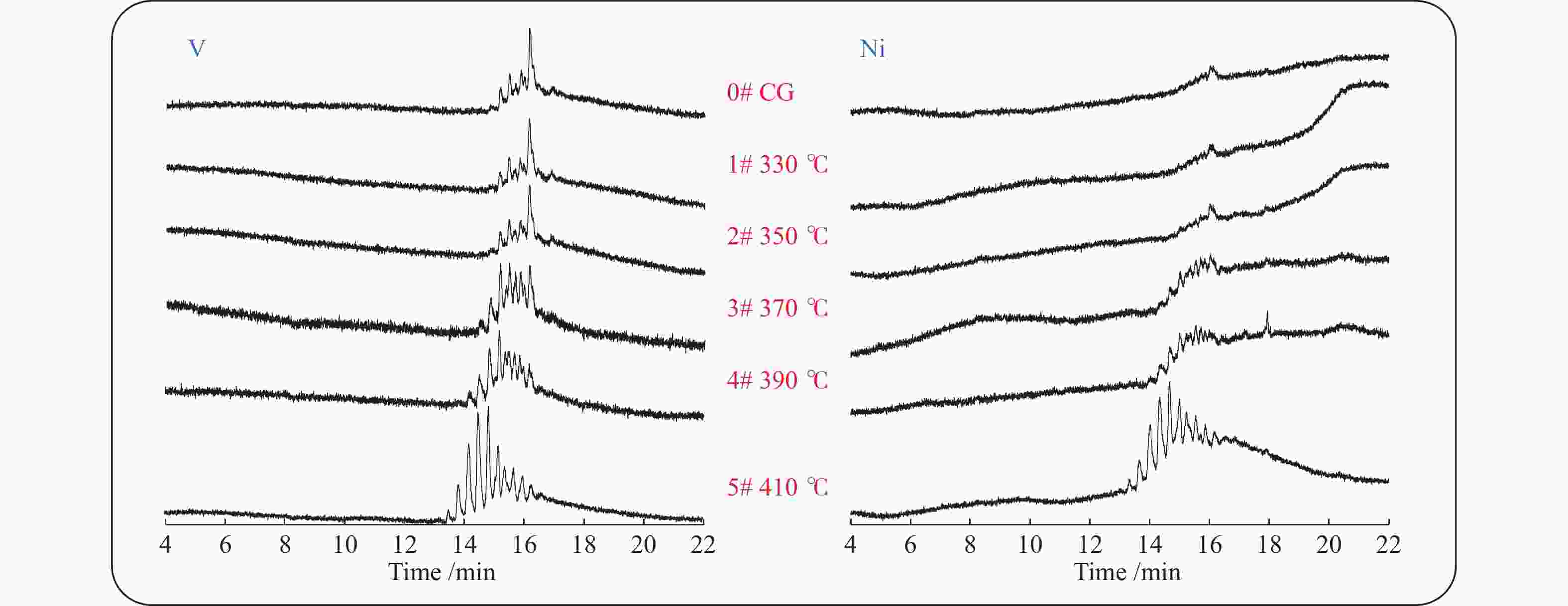
摘要: 本研究通过临氢热解方法处理委内瑞拉减压渣油沥青质,通过电感偶合等离子体质谱仪(ICP MS)、紫外-可见光光谱仪(UV-vis)、高温气相色谱-原子发射检测器联用仪(HT GC-AED)和傅里叶变换离子回旋共振质谱(FT-ICR MS)等手段分析表征反应产物,探究沥青质的分子组成与结构,以及镍和钒化合物的存在形态。实验结果表明,随着临氢热解反应温度从330 ℃升高至410 ℃,反应产物的甲苯可溶物收率由64%下降至19%,可被GC-AED检测到的镍、钒化合物的含量大幅度升高,镍和钒卟啉的分子组成分布也随反应温度的升高呈现出规律性的变化。Abstract: The morphology of nickel and vanadium compounds in the asphaltenes were investigated via hydropyrolysis with the help of inductively coupled plasma mass spectrometer (ICP-MS), ultraviolet-visible (UV-Vis), high-temperature gas chromatography atomic emission detection (HT GC-AED), and positive-ion electrospray ionization Fourier transform ion cyclotron resonance mass spectrometry (+ESI FT-ICR MS). The results showed that the toluene soluble yields of products decreased from 64% to 19% as the hydropyrolysis temperature increased from 330 to 410 ℃, while the abundance of nickel and vanadium compounds detected by GC-AED increased significantly. The molecular composition distribution of nickel and vanadyl porphyrins showed rhythmic changes with different temperatures in the hydropyrolysis of asphaltenes.
-
Key words:
- hydropyrolysis /
- asphaltenes /
- nickel /
- vanadium /
- porphyrin
-
图 3 沥青质临氢热解产物中N4VO类化合物的DBE与碳数分布(图中给出了C30H31N4O1V1 和 C36H31N4O1V1钒卟啉的可能结构)
Figure 3 Relative ion abundance plots of DBE versus carbon number of N4VO class species assigned from positive-ion ESI FT-ICR mass spectra of the hydropyrolysis treated asphaltenes The proposed structure of C30H31N4O1V1 and C36H31N4O1V1 are shown based on the molecular composition
表 1 委内瑞拉重油减渣组分C7沥青质临氢热解反应后甲苯可溶物收率
Table 1 Toluene soluble yields of hydropyrolysis products derived from the asphaltenes of Venezuelan VR
Sample 0#
CG1#
330 ℃2#
350 ℃3#
370 ℃4#
390 ℃5#
410 ℃Yield w/% 98 64 60 51 45 19 表 2 沥青质临氢热解反应后甲苯可溶物中镍和钒含量
Table 2 Nickel and vanadium contents of hydropyrolysis products from ICP MS
Sample 0#
CG1#
330 ℃2#
350 ℃3#
370 ℃4#
390 ℃5#
410 ℃V /(μg·g−1) 1896 1640 1400 1473 1609 1305 Ni /(μg·g−1) 439 400 412 404 441 310 表 3 委内瑞拉减渣C7沥青质临氢热解后各样品中镍、钒元素的GC-AED信号丰度与对照组的比率
Table 3 Signal peak area ratio of nickel and vanadium in reaction group and control group detect by GC-AED
Sample 0#
CG1#
330 ℃2#
350 ℃3#
370 ℃4#
390 ℃5#
410 ℃V(ratio) 1.0 0.9 0.9 1.0 1.1 3.6 Ni(ratio) 1.0 1.0 1.3 1.9 3.8 14.4 -
[1] CHILINGARIAN G V, YEN T F. Bitumens, Asphalts, and Tar Sands[M]. Amsterdam: Elsevier, 1978. [2] E H, WATKINSON P. A study of asphaltene solubility and precipitation[J]. Fuel,2004,83(14):1881−1887. [3] ENAYAT S, RAJAN BABU N, KUANG J, REZAEE S, LU H, TAVAKKOLI M, WANG J, VARGAS F M. On the development of experimental methods to determine the rates of asphaltene precipitation, aggregation, and deposition[J]. Fuel,2020,260:116250. doi: 10.1016/j.fuel.2019.116250 [4] SPEIGHT J G. Petroleum asphaltenes - Part 1: Asphaltenes, resins and the structure of petroleum[J]. Oil Gas Sci Technol,2004,59(5):467−477. [5] RIBEIRO F S, SOUZA MENDES P R, BRAGA S L. Obstruction of pipelines due to paraffin deposition during the flow of crude oils[J]. Int J Heat Mass Transfer,1997,40(18):4319−4328. doi: 10.1016/S0017-9310(97)00082-3 [6] MULLINS O C, SHEU E Y, HAMMAMI A, MARSHALL A G. Asphaltenes, Heavy Oils, and Petroleomics[M]. Berlin: Springer Science & Business Media, 2007. [7] YEN T F. Role of Trace Metals in Petroleum[M]. United States: Ann Arbor Science Publishers, Ann Arbor, MI, 1975. [8] ELLIOT J D. Delayed coker design and operation: Recent trends and innovations[J]. Foster Wheeler USA Corporation, 1996. [9] DECHAINE G P, GRAY M R. Chemistry and association of vanadium compounds in heavy oil and bitumen, and implications for their selective removal†[J]. Energy Fuels,2010,24(5):2795−2808. [10] TREIBS A. Chlorophyll- und Häminderivate in bituminösen Gesteinen, Erdölen, Erdwachsen und Asphalten. Ein Beitrag zur Entstehung des Erdöls[J]. Justus Liebigs Ann Chem,1934,510(1):42−62. doi: 10.1002/jlac.19345100103 [11] TREIBS A. Chlorophyll‐ und Häminderivate in organischen Mineralstoffen[J]. Angew Chem,1936,49(38):682−686. doi: 10.1002/ange.19360493803 [12] YEN T F, CHILINGARIAN G V. Asphaltenes and asphalts, 2[M]. Amsterdam: Elsevier, 2000. [13] CAUMETTE G, LIENEMANN C P, MERDRIGNAC I, BOUYSSIERE B, LOBINSKI R. Fractionation and speciation of nickel and vanadium in crude oils by size exclusion chromatography-ICP MS and normal phase HPLC-ICP MS[J]. J Anal At Spectrom,2010,25(7):1123−1129. doi: 10.1039/c003455j [14] MCKENNA A M, PURCELL J M, RODGERS R P, MARSHALL A G. Identification of vanadyl porphyrins, in a heavy crude oil and raw asphaltene by atmospheric pressure photoionization Fourier Transform Ion Cyclotron Resonance (FT-ICR) mass spectrometry[J]. Energy Fuels,2009,23(3-4):2122−2128. [15] QIAN K, EDWARDS K E, MENNITO A S, WALTERS C C, KUSHNERICK J D. Enrichment, resolution, and identification of nickel porphyrins in petroleum asphaltene by cyclograph separation and atmospheric pressure photoionization Fourier transform ion cyclotron resonance mass spectrometry[J]. Anal Chem,2010,82(1):413−419. doi: 10.1021/ac902367n [16] XU Z, YU L, XU C, YAN Y, ZHANG Y, ZHANG Q, ZHAO S, CHUNG K, GRAY M R, QUAN S. Separation and characterization of vanadyl porphyrins in Venezuela Orinoco heavy crude oil[J]. Energy Fuels,2013,27(6):2874−2882. [17] ZHENG F, HSU C S, ZHANG Y, SUN Y, WU Y, LU H, SUN X, SHI Q. Simultaneous detection of vanadyl, nickel, iron, and gallium porphyrins in marine shales from the eagle ford formation, south texas[J]. Energy Fuels,2018,32(10):10382−10390. [18] FAN S, LIU H, WANG J, CHEN H, BAI R, GUO A, CHEN K, HUANG J, WANG Z. Microwave-assisted petroporphyrin release from asphaltene aggregates in polar solvents[J]. Energy Fuels,2020,34(3):2683−2692. [19] ZHANG L, XU Z, QUAN S, SUN X, NA Z, ZHANG Y, CHUNG K H, XU C, ZHAO S. Molecular characterization of polar heteroatom species in venezuela orinoco petroleum vacuum residue and its supercritical fluid extraction subfractions[J]. Energy Fuels,2012,26(9):5795−5803. [20] FREEMAN D H, SAINT MARTIN D C, BOREHAM C J. Identification of metalloporphyrins by third-derivative UV/VIS diode array spectroscopy[J]. Energy Fuels,1993,7(2):194−199. [21] FOSTER N S, DAY J W, FILBY R H, ALFORD A, ROGERS D. The role of Na-montmorillonite in the evolution of copper, nickel, and vanadyl geoporphyrins during diagenesis[J]. Org Geochem,2002,33(8):907−919. doi: 10.1016/S0146-6380(02)00065-7 [22] MOULIAN R, ZHENG F, SALVATO VALLVERDU G, BARRERE-MANGOTE C, SHI Q, GIUSTI P, BOUYSSIERE B. Understanding the vanadium-asphaltene nanoaggregate link with silver triflate complexation and GPC ICP-MS analysis[J]. Energy Fuels,2020,34(11):13759−13766. [23] ZHENG F, MOULIAN R, CHACÓN-PATIÑO M L, RODGERS R P, BARRÈRE-MANGOTE C, GRAY M R, GIUSTI P, SHI Q, BOUYSSIERE B. Tracking changes in asphaltene nanoaggregate size distributions as a function of silver complexation via gel permeation chromatography inductively coupled plasma mass spectrometry[J]. Energy Fuels,2021,35(22):18125−18134. [24] ZHAO X, SHI Q, GRAY M R, XU C. New vanadium compounds in venezuela heavy crude oil detected by positive-ion electrospray ionization Fourier transform ion cyclotron resonance mass spectrometry[J]. Sci Rep,2014,4(1):5373. [25] QIAN K, FREDRIKSEN T R, MENNITO A S, ZHANG Y, HARPER M R, MERCHANT S, KUSHNERICK J D, RYTTING B M, KILPATRICK P K. Evidence of naturally-occurring vanadyl porphyrins containing multiple S and O atoms[J]. Fuel,2019,239:1258−1264. doi: 10.1016/j.fuel.2018.09.115 -





 下载:
下载: