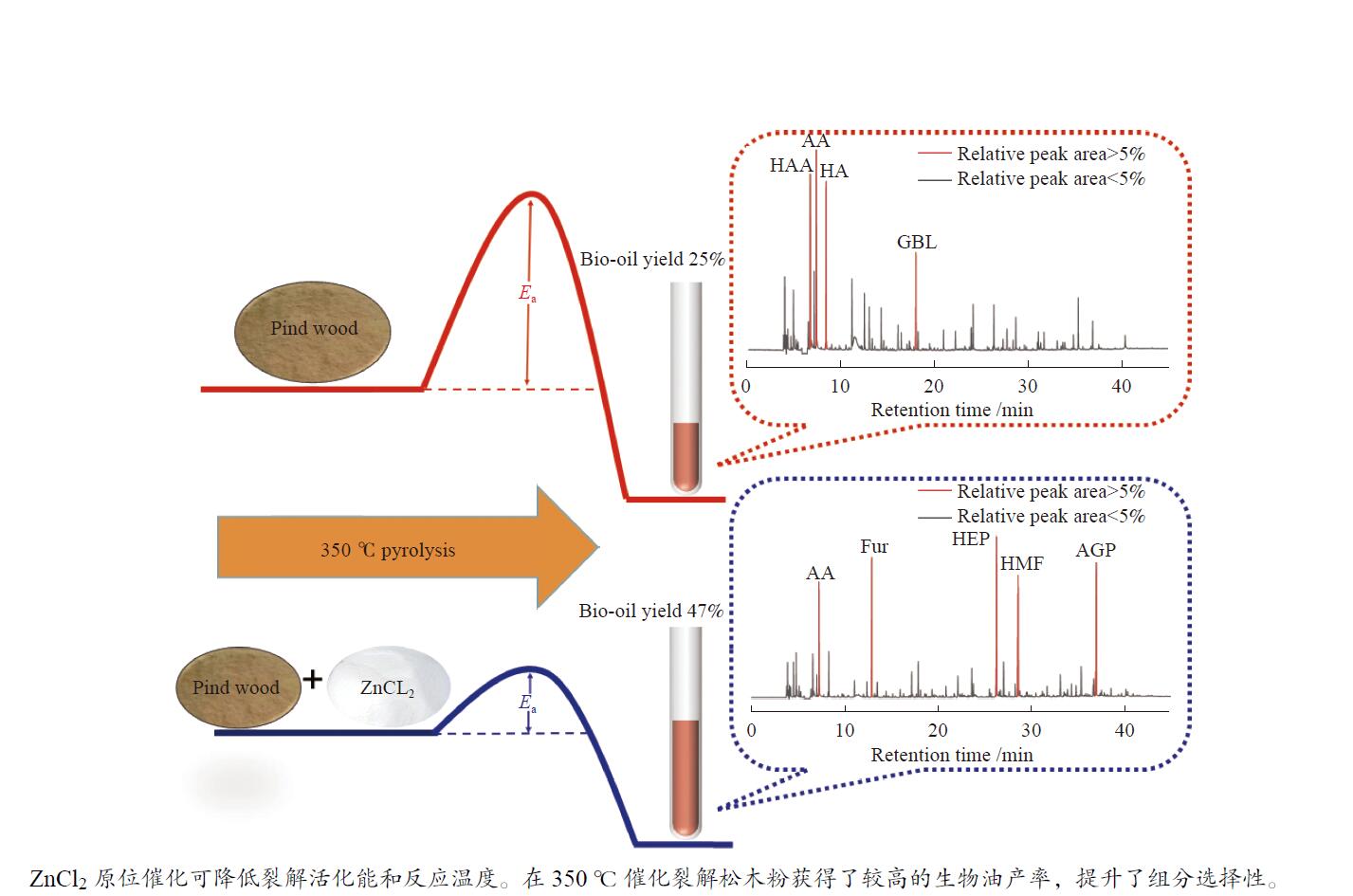
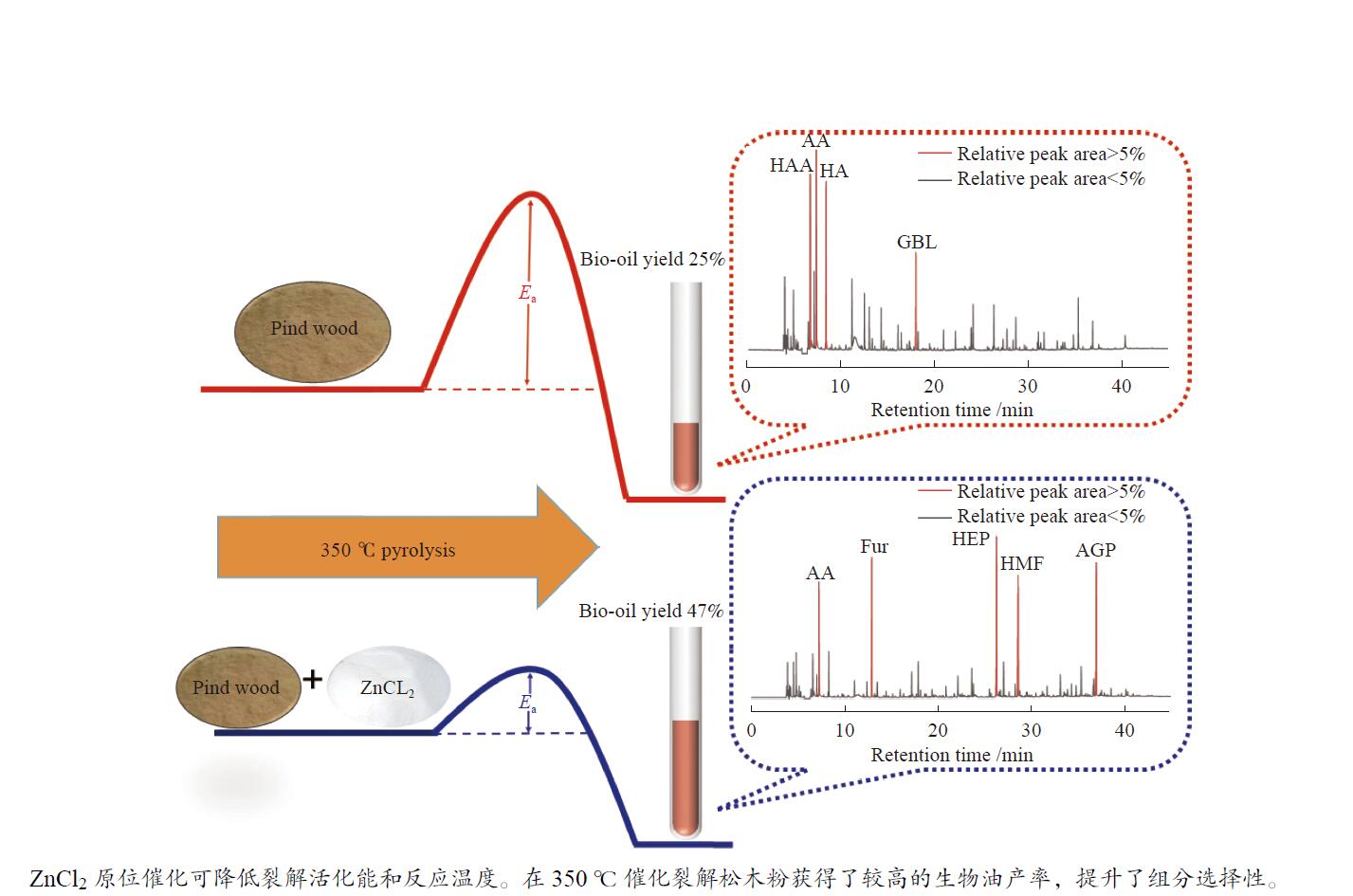
In-situ catalytic pyrolysis of pine powder by ZnCl2 to bio-oil under mild conditions and application of biochar
-
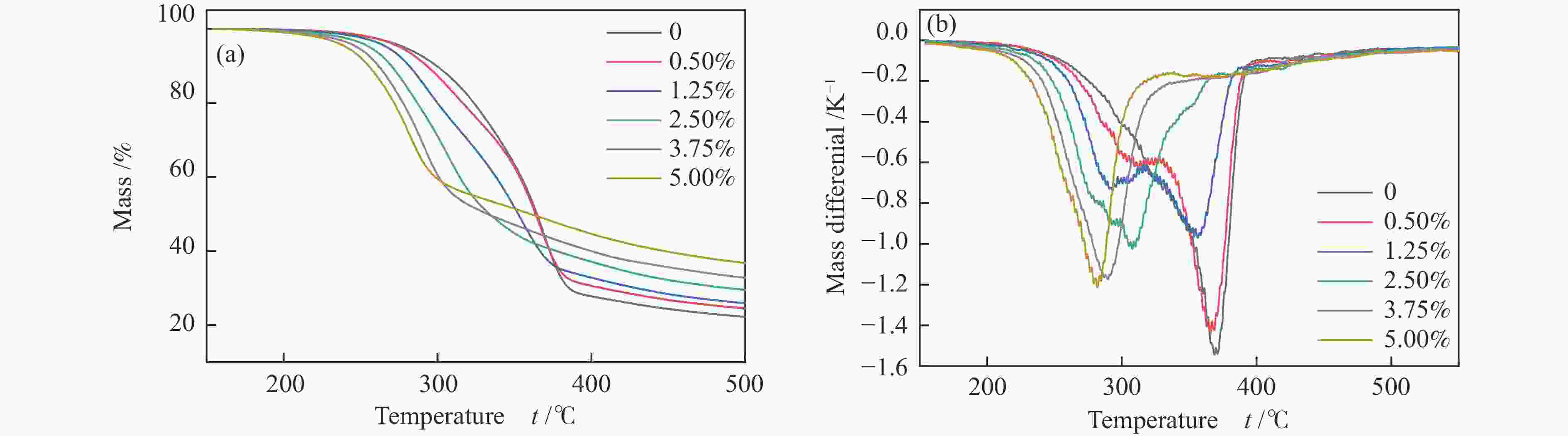
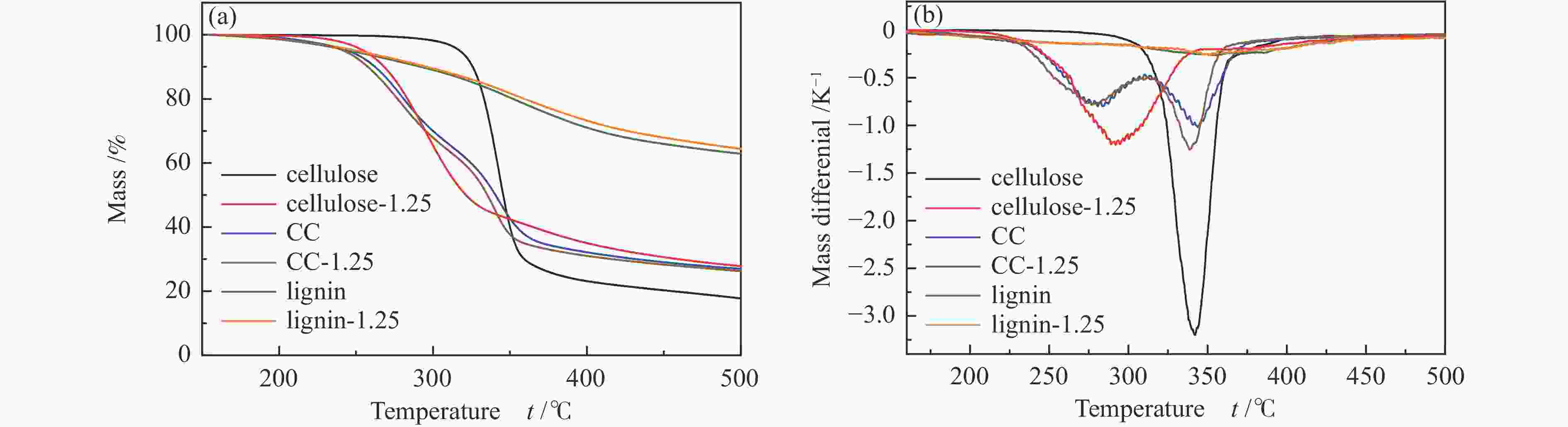
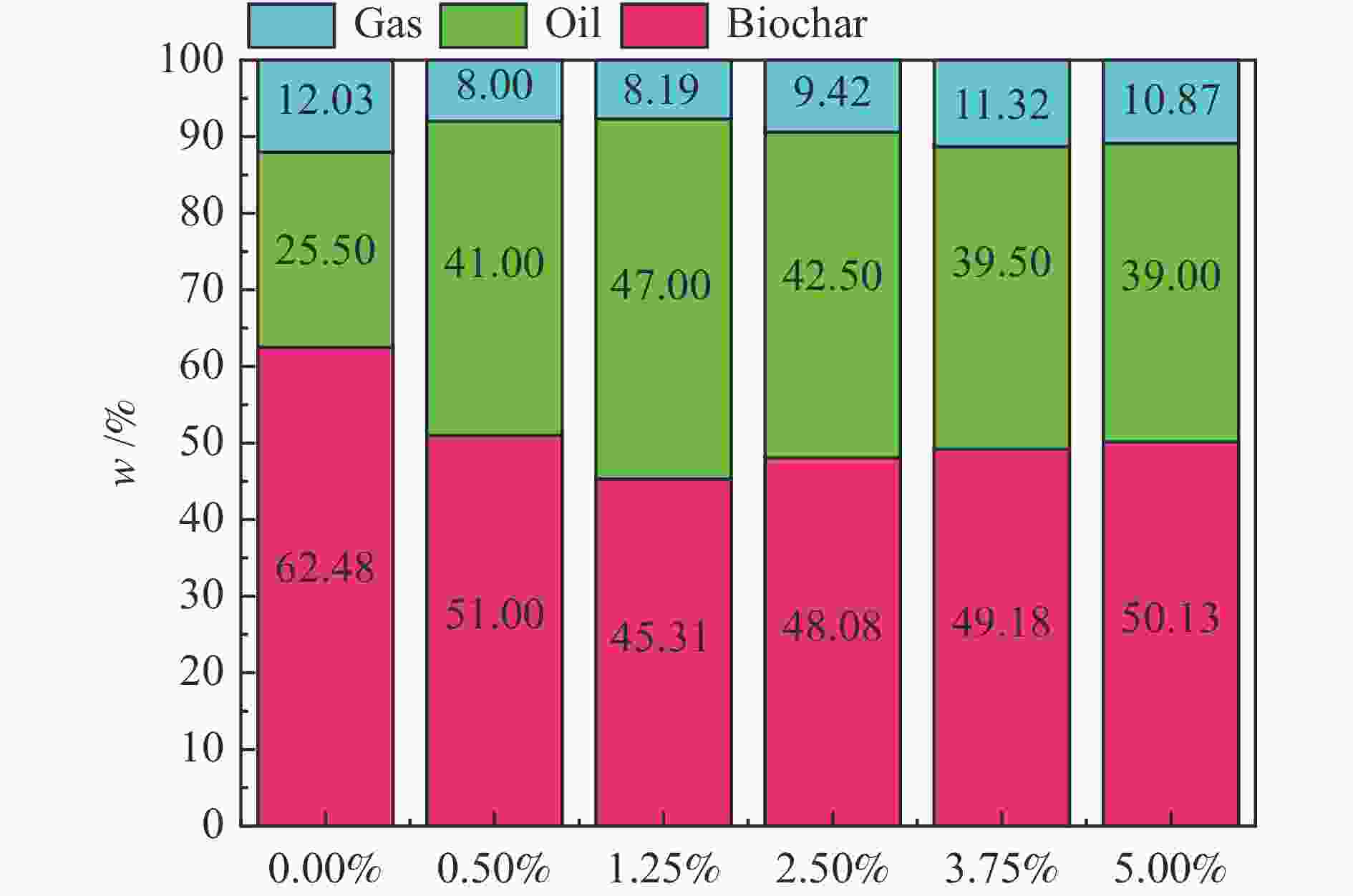
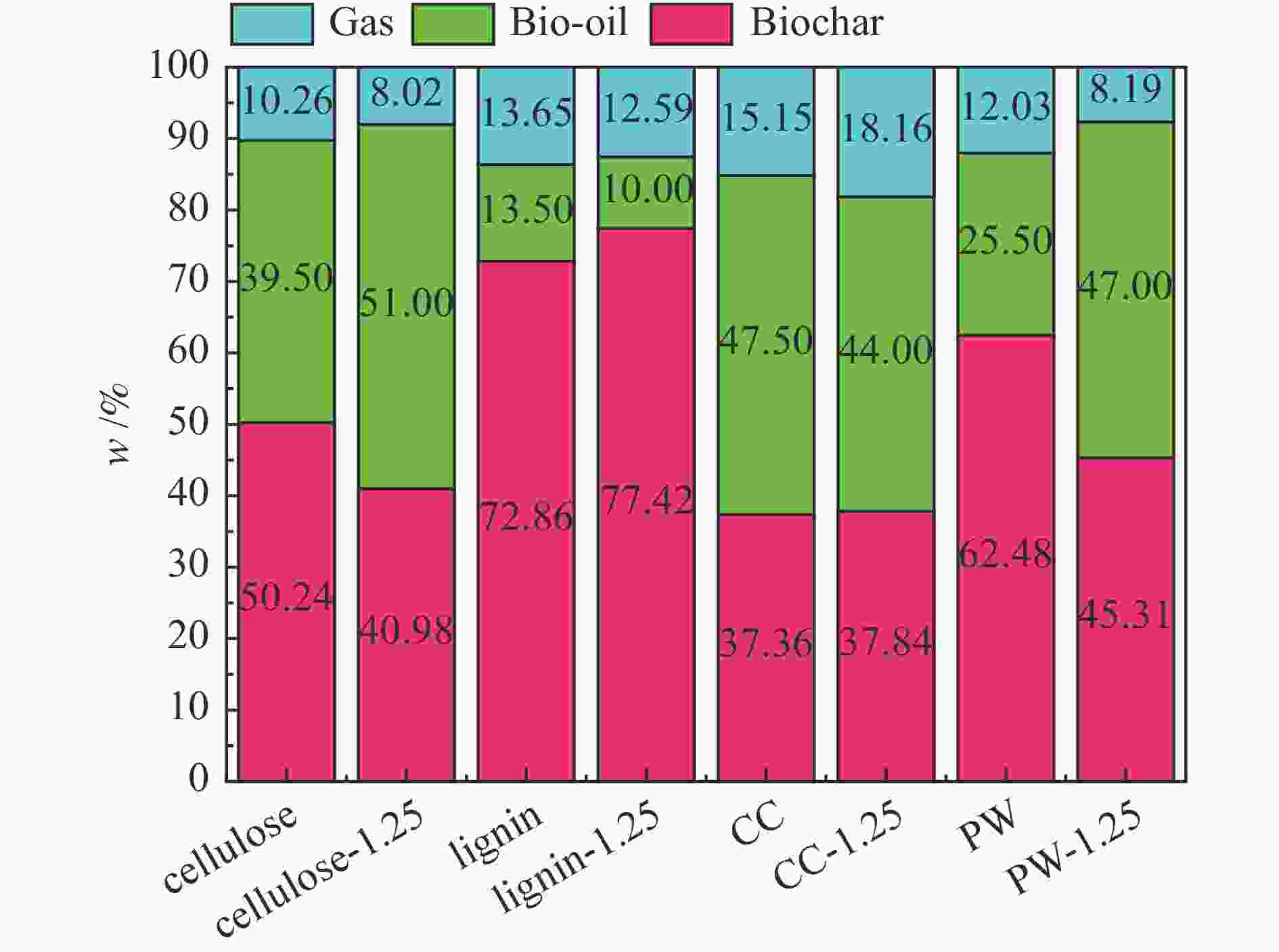
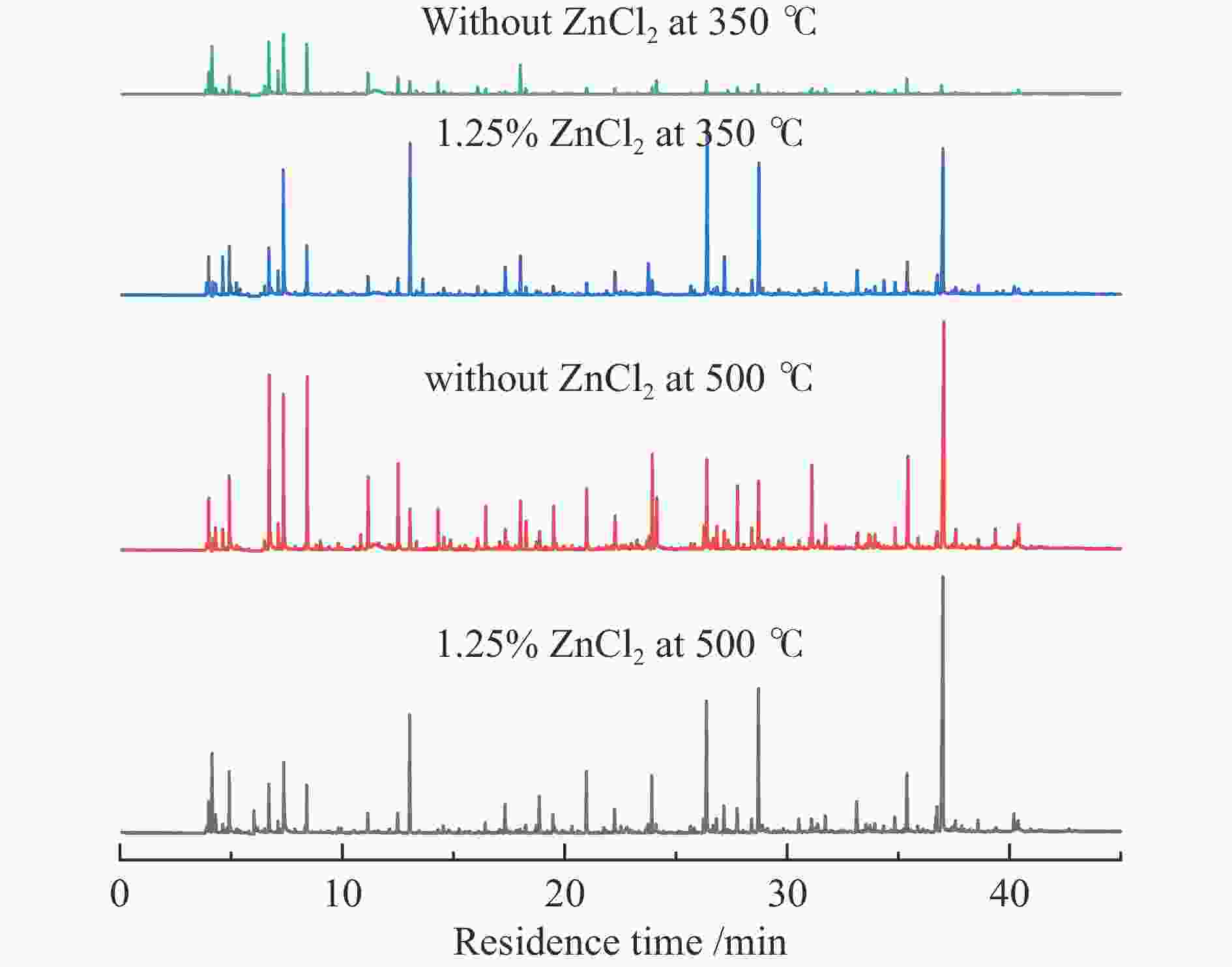
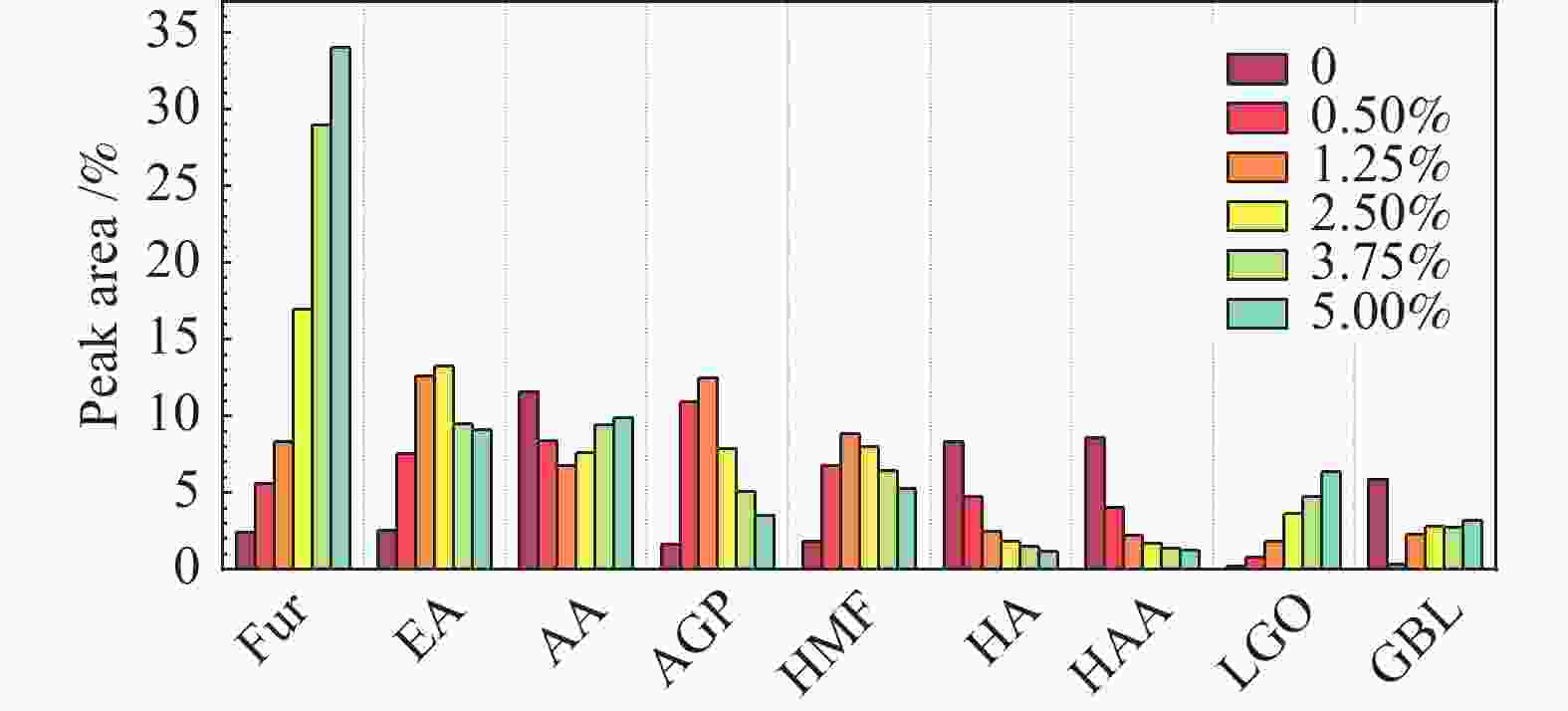
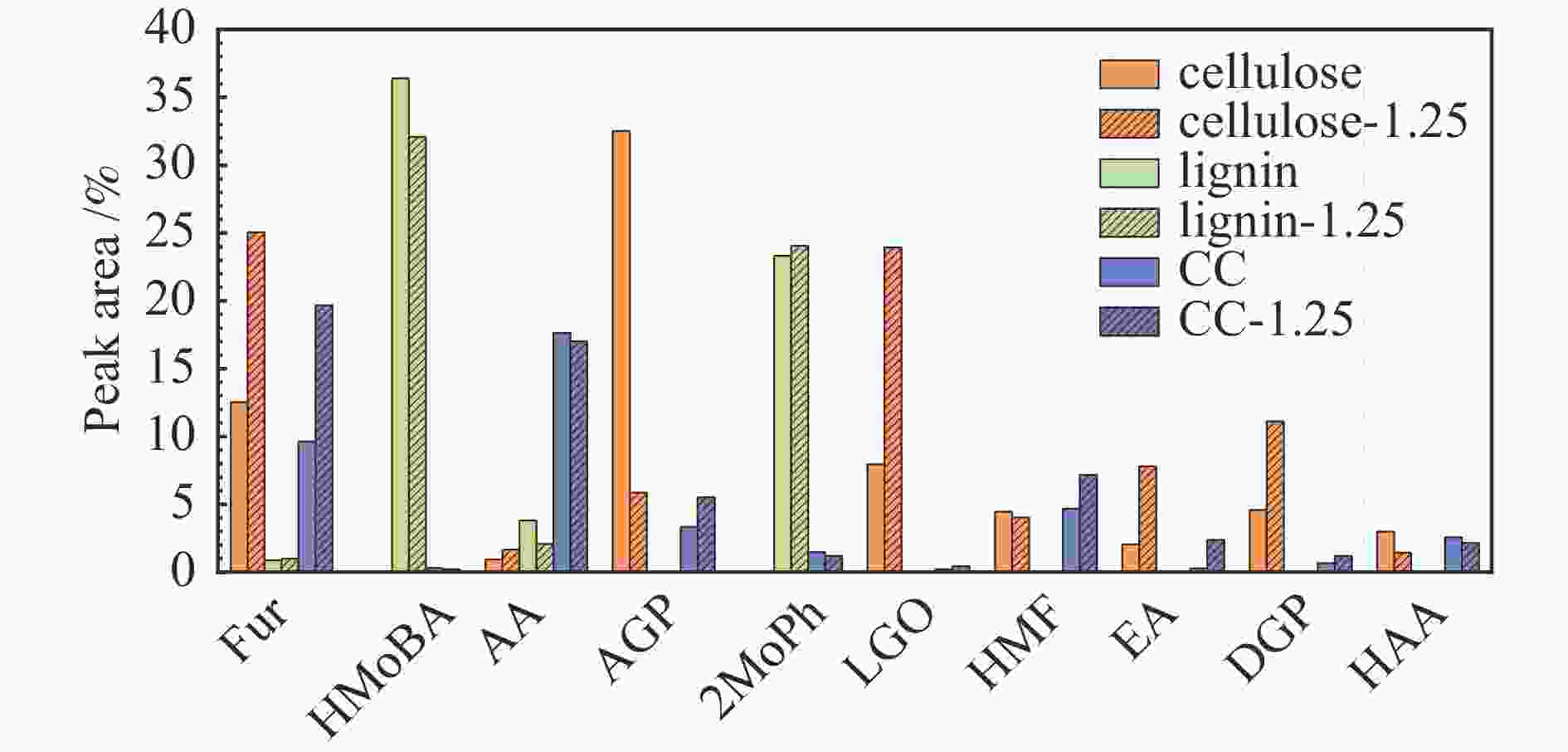
摘要: 生物质快速热裂解是生物质转化利用的有效途径,但常因是非催化过程,裂解温度高导致生物油成分复杂难控。本实验以ZnCl2为催化剂,研究了木质素、纤维素、玉米芯和松木粉的热解过程,旨在探索原位催化对快速热裂解的强化作用。本实验通过热重曲线拟合,获得了热裂解的活化能;通过快速热裂解实验,研究了催化作用下热解油组成变化。结果表明,ZnCl2催化可显著降低生物质裂解温度,简化生物油组成。在350 ℃快速热裂解松木粉获得了47%生物油产率,主要成分是纤维素和半纤维素的衍生物。ZnCl2可显著降低纤维素裂解的活化能(由304.78 kJ/mol降低至112.46 kJ/mol),而对木质素的裂解影响不大。裂解后的碳渣在600 ℃二次碳化可获得性能良好的活性炭,苯酚吸附容量可达165 mg/g。Abstract: Fast pyrolysis of biomass is an effective way for biomass conversion and utilization. However, the pyrolysis temperature is usually high because it is a non-catalytic process, resulting in the complicated composition of bio-oil and difficulty to control. Aiming to explore in-situ catalysis in this paper, the fast pyrolysis of lignin, cellulose, corncob and pine wood powder was studied using ZnCl2 as the catalyst. The activation energies of non-catalytic pyrolysis and catalytic pyrolysis were obtained based on kinetic fitting of their thermal gravimetric curves. The variation in pyrolysis oil composition was analyzed. It was found that ZnCl2 in-situ catalysis could not only significantly reduce the pyrolysis temperature, but also simplify the resultant bio-oil composition. Even under pyrolysis temperature as low as 350 ℃, fast pyrolysis of pine wood powder could achieve a yield of 47% of bio-oil, which was predominantly composed of the derivatives of cellulose and hemicellulose. ZnCl2 in-situ catalysis could significantly decrease the activation energy of cellulose cracking from 304.78 to 112.46 kJ/mol, but has little effect on that of lignin. The carbon residue from ZnCl2-catalyzed pyrolysis was further carbonized at 600 ℃, affording activated carbon with adsorption capacity of phenol up to 165 mg/g. The research work provides guidance and reference for the development of in-situ catalytic pyrolysis technology with high efficiency.
-
Key words:
- biomass /
- fast pyrolysis /
- in-situ catalysis /
- zinc chloride /
- pine wood
-
表 1 不同生物质的工业分析和元素分析
Table 1 Ultimate and proximate analyses of biomass
Material Proximate analysis wad/% Ultimate analysis wad/% A M V FC C H O N S Lignin 8.49 14.65 52.99 23.87 52.52 4.64 15.22 0.19 4.28 Cellulose 5.92 0.01 87.90 6.17 43.17 6.4 44.41 0.09 0 CC 1.80 1.60 82.95 13.65 45.73 6.05 44.41 0.41 0 PW 2.17 1.43 81.52 14.88 47.91 6.21 42.10 0.18 0 ad: air dried basis; A: ash; M: moisture; V: volatile matter; FC: fixed carbon 表 2 不同样品的热解动力学参数
Table 2 Kinetic parameters of different samples
Sample Stage Ⅰ Stage Ⅱ E /(kJ·mol−1) A /s−1 R2 E /(kJ·mol−1) A /s−1 R2 Cellulose 304.78 9.77 × 1023 0.998 Cellulose-1.25 112.46 1.22 × 108 0.997 Lignin 66.71 1.01 × 103 0.997 Lignin-1.25 62.89 4.48 × 102 0.998 CC 105.81 6.60 × 107 0.999 248.29 1.74 × 1019 0.999 CC-1.25 112.91 3.10 × 108 0.999 208.99 4.97 × 1015 0.999 PW 106.45 1.19 × 107 0.996 317.06 7.56 × 1023 0.997 PW-0.5 129.34 4.65 × 109 0.998 222.24 1.55 × 1016 0.999 PW-1.25 143.38 1.38 × 1011 0.999 169.94 1.19 × 1012 0.998 PW-2.5 161.91 3.25 × 1013 0.994 92.72 1.16 × 106 0.996 PW-3.75 124.81 2.97 × 109 0.999 PW-5 126.23 6.91 × 109 0.999 表 3 生物炭微观结构数据信息
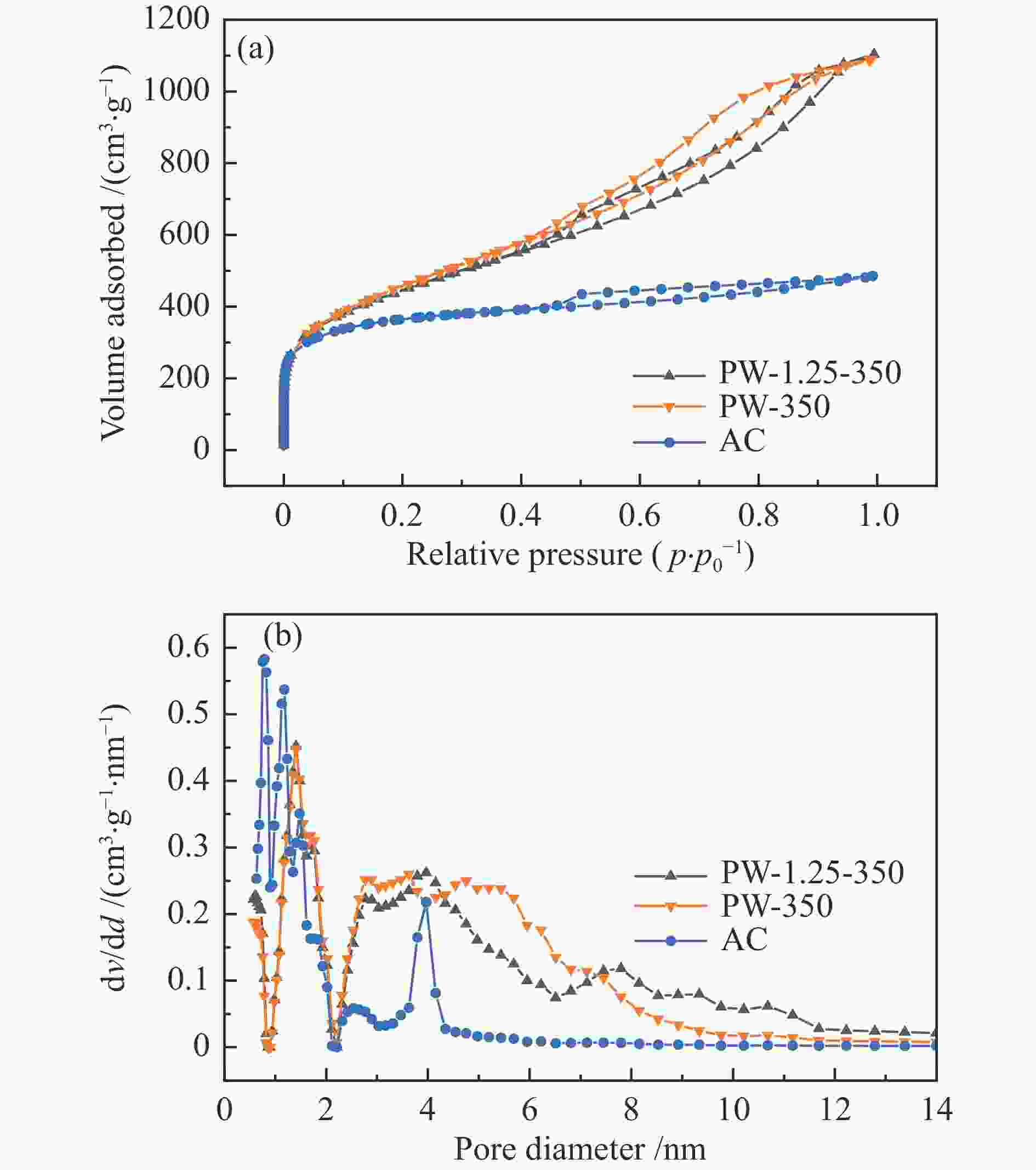
Table 3 Microscopic structure information of the biochar
Sample Specific surface area /(m2·g−1) Pore volume / (cm3·g−1) Average pore size /nm total micro- meso- total micro- meso- PW-1.25-350 1151 671 480 1.57 0.37 1.20 4.37 PW-350 1150 628 522 1.57 0.36 1.21 4.16 AC 1025 919 106 0.67 0.46 0.21 2.59 表 4 活性炭吸附能力比较
Table 4 Comparison of the biochar with the activated carbon reported in the literature
Biomass Activation agent Temperature /℃ Adsorption capacity /(mg·g−1) Ref. Bamboo Urea + KHCO3 700 169 [33] Eucalyptus KOH 800 222 [34] Pine shell – 550 25 [35] Poplar* Urea 900 160.5 [36] Bamboo** KC2O4 700 169.5 [37] Corn straw ZnCl2 + FeCl3 900 183.43 [38] Pine wood ZnCl2 600 165 this work *: with CO2-assisted activation; **: with NH3-assisted activation -
[1] LIANG J, SHAN G C, SUN Y F. Catalytic fast pyrolysis of lignocellulosic biomass: Critical role of zeolite catalysts[J]. Renewable Sustainable Energy Rev,2021,139:110707. doi: 10.1016/j.rser.2021.110707 [2] 李承宇, 张军, 袁浩然, 王树荣, 陈勇. 纤维素热解转化研究进展[J]. 燃料化学学报,2021,49(12):1733−1851.LI Cheng-yu, ZHANG Jun, YUAN Hao-ran, WANG Shu-rong, CHEN Yong. Research progress of cellulose pyrolysis transformation[J]. J Fuel Chem Technol,2021,49(12):1733−1851. [3] WANG S R, DAI G X, YANG H P, LUO Z Y. Lignocellulosic biomass pyrolysis mechanism: A state-of-the-art review[J]. Prog Energy Combust Sci,2017,62:33−86. doi: 10.1016/j.pecs.2017.05.004 [4] BHOI P R, OUEDRAOGO A S, SOLOIU V, QUIRINO R. Recent advances on catalysts for improving hydrocarbon compounds in bio-oil of biomass catalytic pyrolysis[J]. Renewable Sustainable Energy Rev,2020,121:109676. doi: 10.1016/j.rser.2019.109676 [5] RAHMAN M M, LIU R, CAI J. Catalytic fast pyrolysis of biomass over zeolites for high quality bio-oil – A review[J]. Fuel Process Technol,2018,180:32−46. doi: 10.1016/j.fuproc.2018.08.002 [6] ZHANG C T, ZHANG L J, LI Q Y, WANG Y, LIU Q, WEI T, DONG D H, SALAVATI S, GHOLIZADEH M, HU X. Catalytic pyrolysis of poplar wood over transition metal oxides: Correlation of catalytic behaviors with physiochemical properties of the oxides[J]. Biomass Bioenergy,2019,124:125−141. doi: 10.1016/j.biombioe.2019.03.017 [7] DAI G X, WANG S R, HUANG S Q, ZOU Q. Enhancement of aromatics production from catalytic pyrolysis of biomass over HZSM-5 modified by chemical liquid deposition[J]. J Anal Appl Pyrolysis,2018,134:439−445. doi: 10.1016/j.jaap.2018.07.010 [8] HASSAN N S, JALIL A A, HITAM C N C, VO D V N, NABGAN W. Biofuels and renewable chemicals production by catalytic pyrolysis of cellulose: a review[J]. Environ Chem Lett,2020,18(5):1625−1648. doi: 10.1007/s10311-020-01040-7 [9] LIU C J, WANG H M, KARIM A M, SUN J M, WANG Y. Catalytic fast pyrolysis of lignocellulosic biomass[J]. Chem Soc Rev,2014,43(22):7594−7623. doi: 10.1039/C3CS60414D [10] 尹海云, 李小华, 张蓉仙, 樊永胜, 俞宁, 蔡忆昔. HZSM-5在线提质生物油及催化剂失活机理分析[J]. 燃料化学学报,2014,42(9):1077−1086.YIN Hai-yun, LI Xiao hua, ZHANG Rong-xian, FAN Yong-sheng, YU Ning, CAI Yi-xi. HZSM-5 online enhancement of bio-oil and analysis of catalyst deactivation mechanism[J]. J Fuel Chem Technol,2014,42(9):1077−1086. [11] CHENG S L, ZHANG Z M, ZHANG D M, DENG Y. Microwave irradiation pyrolysis of rice straw in ionic liquid ([Emim]Br)[J]. Bioresources,2013,8(3):3994−4003. [12] ZHOU X W, MAYES H B, BROADBELT L J, NOLTE M W, SHANKS B H. Fast pyrolysis of glucose‐based carbohydrates with added NaCl part 1: Experiments and development of a mechanistic model[J]. AlChE J,2015,62(3):766−777. [13] LENG E W, WANG Y, GONG X, ZHANG B, ZHANG Y, XU M H. Effect of KCl and CaCl2 loading on the formation of reaction intermediates during cellulose fast pyrolysis[J]. Proc Combust Inst,2017,36(2):2263−2270. doi: 10.1016/j.proci.2016.06.167 [14] OH S-J, CHOI G-G, KIM J-S. Fast pyrolysis of corn stover using ZnCl2: Effect of washing treatment on the furfural yield and solvent extraction of furfural[J]. Energy,2015,88:697−702. doi: 10.1016/j.energy.2015.05.101 [15] LU Q, DONG C Q, ZHANG X M, TIAN H Y, YANG Y P, ZHU X F. Selective fast pyrolysis of biomass impregnated with ZnCl2 to produce furfural: Analytical Py-GC/MS study[J]. J Anal Appl Pyrolysis,2011,90(2):204−212. doi: 10.1016/j.jaap.2010.12.007 [16] ASTM E871-82(2019), Standard Test Method for Moisture Analysis of Particulate Wood Fuels[S]. [17] ASTM E1755-01(2007), Standard Test Method for Ash in Biomass[S]. [18] ASTM E872-82(2006), Standard Test Method for Volatile Matter in the Analysis of Particulate Wood Fuels[S]. [19] NY/T 3498—2019, 农业生物质原料成分测定--元素分析仪法[S].NY/T 3498—2019, Determination of the composition of agricultural biomass raw materials by Elemental analyzer method[S]. [20] ZHANG Y, CUI H Y, YI W M, SONG F, ZHAO P P, WANG L H, CUI J Y. Highly effective decarboxylation of the carboxylic acids in fast pyrolysis oil of rice husk towards ketones using CaCO3 as a recyclable agent[J]. Biomass Bioenergy,2017,102:13−22. doi: 10.1016/j.biombioe.2017.04.004 [21] RAZA M, ABU-JDAYIL B, AL-MARZOUQI A H, INAYAT A. Kinetic and thermodynamic analyses of date palm surface fibers pyrolysis using Coats-Redfern method[J]. Renew Energy,2022,183:67−77. doi: 10.1016/j.renene.2021.10.065 [22] HU J, SHEN D K, WU S L, XIAO R. Insight into the effect of ZnCl2 on analytical pyrolysis behavior of cellulolytic enzyme corn stover lignin[J]. J Anal Appl Pyrolysis,2017,127:444−450. doi: 10.1016/j.jaap.2017.07.005 [23] CHEN D Y, CEN K H, ZHUANG X Z, GAN Z Y, ZHOU J B, ZHANG Y M, ZHANG H. Insight into biomass pyrolysis mechanism based on cellulose, hemicellulose, and lignin: Evolution of volatiles and kinetics, elucidation of reaction pathways, and characterization of gas, biochar and bio‐oil[J]. Combust Flame,2022,242:112142. doi: 10.1016/j.combustflame.2022.112142 [24] CHAKAR F S, RAGAUSKAS A J. Review of current and future softwood kraft lignin process chemistry[J]. Ind Crops Prod,2004,20(2):131−141. doi: 10.1016/j.indcrop.2004.04.016 [25] KUMAR P, BARRETT D M, DELWICHE M J, STROEVE P. Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production[J]. Ind Eng Chem Res,2009,48(8):3713−3729. doi: 10.1021/ie801542g [26] MA S W, ZHANG L Q, ZHU L, ZHU X F. Preparation of multipurpose bio-oil from rice husk by pyrolysis and fractional condensation[J]. J Anal Appl Pyrolysis,2018,131:113−119. doi: 10.1016/j.jaap.2018.02.017 [27] LIU W J, JIANG H, YU H Q. Development of biochar-based functional materials: Toward a sustainable platform carbon material[J]. Chem Rev,2015,115(22):12251−12285. doi: 10.1021/acs.chemrev.5b00195 [28] ZHANG H Y, LIU X J, LU M Z, HU X Y, LU L G, TIAN X N, JI J B. Role of Bronsted acid in selective production of furfural in biomass pyrolysis[J]. Bioresour Technol,2014,169:800−803. doi: 10.1016/j.biortech.2014.07.053 [29] LI Y, HU B, NAQVI S R, ZHANG Z X, LI K, LU Q. Selective preparation of 5-hydroxymethylfurfural by catalytic fast pyrolysis of cellulose over zirconium-tin mixed metal oxides[J]. J Anal Appl Pyrolysis,2021,155:105103. doi: 10.1016/j.jaap.2021.105103 [30] QU G F, HE W W, CAI Y Y, HUANG X, NING P. Catalytic pyrolysis of cellulose in ionic liquid [bmim]OTf[J]. Carbohydr Polym,2016,148:390−396. doi: 10.1016/j.carbpol.2016.04.052 [31] OH S-J, CHOI G-G, KIM J-S. Production of acetic acid-rich bio-oils from the fast pyrolysis of biomass and synthesis of calcium magnesium acetate deicer[J]. J Anal Appl Pyrolysis,2017,124:122−129. doi: 10.1016/j.jaap.2017.01.032 [32] ESSANDOH M, KUNWAR B, PITTMAN C U, MOHAN D, MLSNA T. Sorptive removal of salicylic acid and ibuprofen from aqueous solutions using pine wood fast pyrolysis biochar[J]. Chem Eng J,2015,265:219−227. doi: 10.1016/j.cej.2014.12.006 [33] LI Z H, XING B, DING Y, LI Y C, WANG S R. A high-performance biochar produced from bamboo pyrolysis with in-situ nitrogen doping and activation for adsorption of phenol and methylene blue[J]. Chin J Chem Eng,2020,28(11):2872−2880. doi: 10.1016/j.cjche.2020.03.031 [34] SINGH R, DUTTA R K, NAIK D V, RAY A, KANAUJIA P K. High surface area Eucalyptus wood biochar for the removal of phenol from petroleum refinery wastewater[J]. Environm Challenges,2021,5:100353. doi: 10.1016/j.envc.2021.100353 [35] MOHAMMED N A S, ABU-ZURAYK R A, HAMADNEH I, AL-DUJAILI A H. Phenol adsorption on biochar prepared from the pine fruit shells: Equilibrium, kinetic and thermodynamics studies[J]. J Environ Manage,2018,226:377−385. doi: 10.1016/j.jenvman.2018.08.033 [36] GAO W R, LIN Z X, CHEN H R, YAN S S, ZHU H N, ZHANG H, SUN H Q, ZHANG S, ZHANG S J, WU Y L. Roles of graphitization degree and surface functional groups of N-doped activated biochar for phenol adsorption[J]. J Anal Appl Pyrolysis,2022,167:105700. doi: 10.1016/j.jaap.2022.105700 [37] LI Y C, LI Z H, XING B, LI H M, MA Z Q, ZHANG W B, REUBROYCHAROEN P, WANG S R. Green conversion of bamboo chips into high-performance phenol adsorbent and supercapacitor electrodes by simultaneous activation and nitrogen doping[J]. J Anal Appl Pyrolysis,2021,155:105072. doi: 10.1016/j.jaap.2021.105072 [38] FENG D D, GUO D W, ZHANG Y, SUN S Z, ZHAO Y J, SHANG Q, SUN H L, WU J Q, TAN H P. Functionalized construction of biochar with hierarchical pore structures and surface O-/N-containing groups for phenol adsorption[J]. Chem Eng J,2021,410:127707. doi: 10.1016/j.cej.2020.127707 -




 下载:
下载: