Effect of support factors on the selective catalytic oxidation of benzyl alcohol over Au/BN catalyst
-
摘要: 本研究以三种不同结构特点的氮化硼(BN)充当载体,负载Au纳米颗粒进行苯甲醇选择性氧化反应,发现载体的结晶性、比表面积对活性相Au的尺寸具有显著影响。Au/BN500的比表面积是晶化程度高的Au/BN600、Au/BN700催化剂的四倍以上。相较于Au/BN700而言,Au/BN500催化剂Au纳米颗粒具有更好的分散性以及更小的粒径(13 vs. 3.2 nm),且Au/BN500的活性是其他催化剂的两倍,但其催化活性在5 h以内有30%左右的流失。Abstract: The selective oxidation of primary alcohols represents a premier route for the synthesis of aldehydes as intermediates of multiple commercial fine chemicals such as drugs and perfumes. In particular, catalytically selective oxidation of primary alcohol in use of oxygen is of great interest, owning to its high efficiency, solvent-free, and easy separation. As such, choosing this route to pursue desired atomic economy has been an essential topic of common concern in both academic and industrial circles in recent years. Boron nitride with graphite like structure is a new catalyst developed in recent years, which has the characteristics of stability and good thermal conductivity. In this contribution, three kinds of boron nitride (BN) with different structural characteristics were used as carriers to support Au nano metal for selective oxidation of benzyl alcohol. It is found that the crystallinity and specific surface area of the carriers have a great influence on the size of active phase Au. The specific surface area of Au/BN500 is four times higher than those of the Au/BN600 and Au/BN700. Compared with Au/BN700, Au/BN500 catalyst has better dispersion and smaller particle size (13 vs. 3.2 nm). The catalytic activity of Au/BN500 is about twice as much as those of the other two, and about 30% activity is lost within 5 h. The results in this paper provide enriched experimental and theoretical references for rational design and development of novel high-performance boron nitride-based oxidative dehydrogenation catalysts.
-
Key words:
- boron nitride /
- selective oxidation /
- gold /
- primary alcohol /
- heterogeneous catalysis
-
图 2 三种催化剂的形态结构
Figure 2 Transmission electron microscopy characterizations of three catalysts
(a), (b): Au/BN500, (c), (d): Au/BN600 (e), (f): Au/BN700 low-magnification and high-magnification electron spectroscopies are shown in left and right column, respectively,the illustration of Figures (b), (d) and (f) show the statistical distribution of particle size
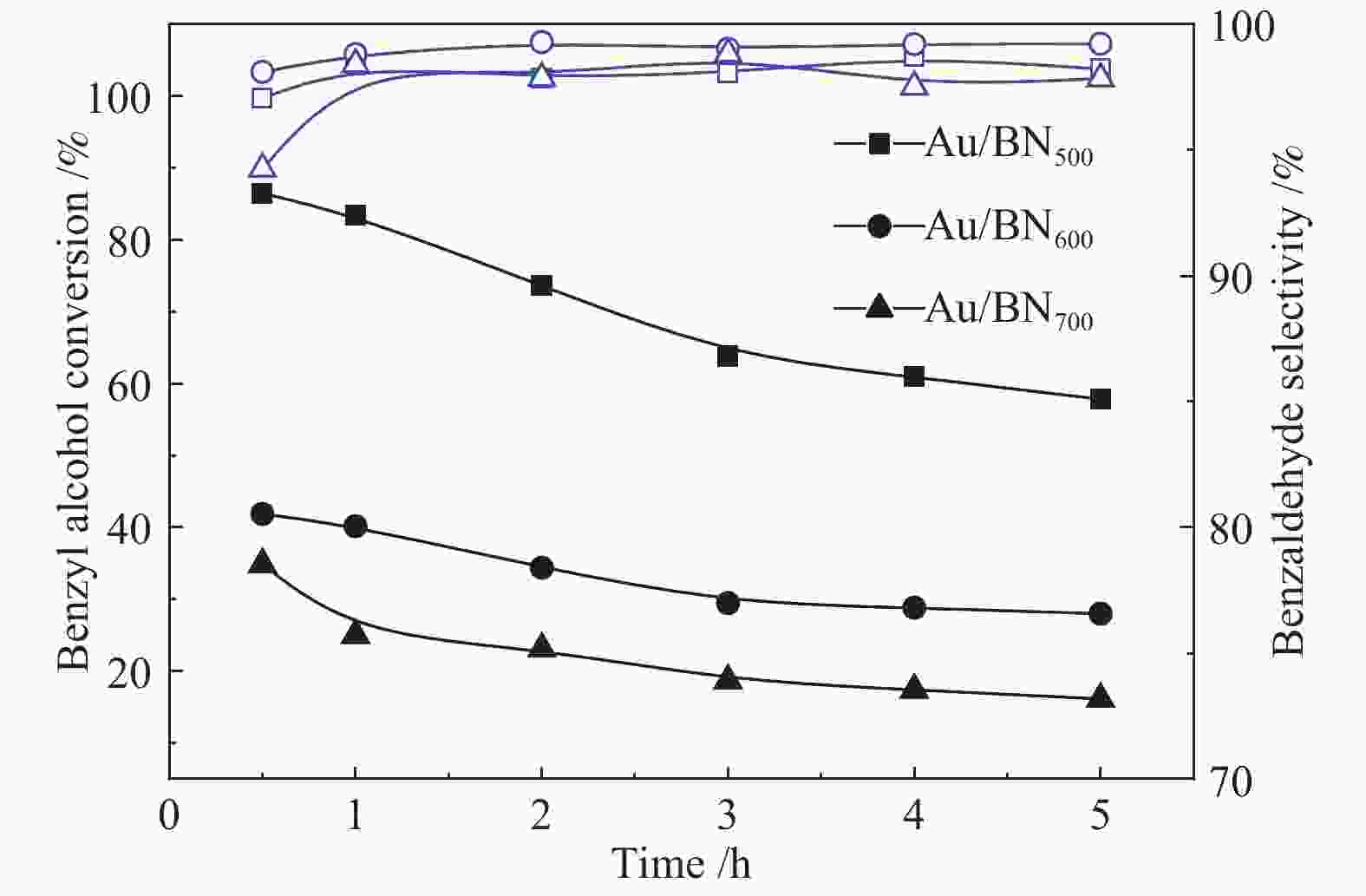
图 4 三种Au/BN催化剂以模拟空气为氧化剂,选择性催化氧化苯甲醇制备苯甲醛
Figure 4 Catalytic performance of three Au/BN catalysts in selective oxidization of benzyl alcohol to synthesize benzaldehyde
from top to bottom (■): Au/BN500, (●): Au/BN600, (▲): Au/BN700, where the black dotted line represents conversion rate and blue dotted line denotes selectivity as function of time
-
[1] DELLA PINA C, FALLETTA E, PRATI L, ROSSI M. Selective oxidation using gold[J]. Chem Soc Rev,2008,37(9):2077−2095. doi: 10.1039/b707319b [2] CHEN J, ZHANG Q H, WANG Y, WAN H L. Size-dependent catalytic activity of supported palladium nanoparticles for aerobic oxidation of alcohols[J]. Adv Synth Catal,2008,350(3):453−464. doi: 10.1002/adsc.200700350 [3] RODRIGUEZ-GOMEZ A, HOLGADO J P, CABALLERO A. cobalt carbide identified as catalytic site for the dehydrogenation of ethanol to acetaldehyde[J]. ACS Catal,2017,7(8):5243−5247. doi: 10.1021/acscatal.7b01348 [4] ADNAN R H, GOLOVKO V B. Benzyl alcohol oxidation using gold catalysts derived from Au- clusters on TiO2[J]. Catal Lett,2019,149(2):449−455. doi: 10.1007/s10562-018-2625-8 [5] DIMITRATOS N, LOPEZ-SANCHEZ J A, MORGAN D, CARLEY A, PRATI L, HUTCHING G J. Solvent free liquid phase oxidation of benzyl alcohol using Au supported catalysts prepared using a sol immobilization technique[J]. Catal Today,2007,122(3/4):317−324. [6] CHEN Y, WANG H, LIU C J, ZENG Z Y, ZHANG H, ZHOU C M, JIA X L, YANG Y H. Formation of monometallic Au and Pd and bimetallic Au-Pd nanoparticles confined in mesopores via Ar glow-discharge plasma reduction and their catalytic applications in aerobic oxidation of benzyl alcohol[J]. J Catal,2012,289:105−117. doi: 10.1016/j.jcat.2012.01.020 [7] ZHOU Q Y, ZHOU C Y, ZHOU Y H, HONG W, ZOU S H, GONG X Q, LIU J J, XIAO L P, FAN J. More than oxygen vacancies: A collective crystal- plane effect of CeO2 in gas- phase selective oxidation of benzyl alcohol[J]. Catal Sci Technol,2019,9(11):2960−2967. [8] GAO Y, ZHANG L, VAN HOOF A J F, FRIEDRICH H, HENSEN E J M. A robust Au/ZnCr2O4 catalyst with highly dispersed gold nanoparticles for gas-phase selective oxidation of cyclohexanol to cyclohexanone[J]. ACS Catal,2019,9(12):11104−11115. doi: 10.1021/acscatal.9b02821 [9] LIU P, HENSEN E J M. Highly efficient and robust Au/MgCuCr2O4 catalyst for gas-phase oxidation of ethanol to acetaldehyde[J]. J Am Chem Soc,2013,135(38):14032−14035. doi: 10.1021/ja406820f [10] HOU W B, DEHM N A, SCOTT R W J. Alcohol oxidations in aqueous solutions using Au, Pd, and bimetallic AuPd nanoparticle catalysts[J]. J Catal,2008,253(1):22−27. doi: 10.1016/j.jcat.2007.10.025 [11] DAI Y, YAN X, TANG Y, LIU X, XIAO L, FAN J. Low-temperature gas-phase oxidation of benzyl alcohol on mesoporous K-Cu-TiO2through oxidative dehydrogenation[J]. ChemCatChem,2012,4(10):1603−1610. [12] ZHAO G, LI Y, ZHANG Q, DENG M, CAO F, LU Y. Galvanic deposition of silver on 80-μm-Cu-fiber for gas-phase oxidation of alcohols[J]. AlChE J,2014,60(3):1045−1053. doi: 10.1002/aic.14295 [13] MAO J, DENG M, CHEN L, LIU Y, LU Y. Novel microfibrous-structured silver catalyst for high efficiency gas-phase oxidation of alcohols[J]. AlChE J,2010,56(6):1545−1556. doi: 10.1002/aic.12088 [14] JACOBSEN C, BORON NITRIDE. A novel support for ruthenium-based ammonia synthesis catalysts[J]. J Catal,2001,200(1):1−3. doi: 10.1006/jcat.2001.3200 [15] OHASHI T, WANG Y, SHINADA S. Preparation and high catalytic performance of hollow BN spheres-supported Ni for hydrogen production from methanol[J]. J Mater Chem,2010,20(24):5129−5135. doi: 10.1039/c0jm00412j [16] POSTOLE G, GERVASINI A, GUIMON C, AUROUX A, BONNETOT B. Influence of the preparation method on the surface characteristics and activity of boron-nitride-supported noble metal catalysts[J]. J Phys Chem B,2006,110(25):12572−12580. doi: 10.1021/jp060183x [17] LIN C A, WU J C S, PAN J W, YEH C T. Characterization of boron-nitride-supported Pt catalysts for the deer oxidation of benzene[J]. J Catal,2002,210(1):39−45. doi: 10.1006/jcat.2002.3638 [18] UOSAKI K, ELUMALAI G, NOGUCHI H, MASUDA T, LYALIN A, NAKAYAMA A, TAKETSUGU T. Boron nitride nanosheet on gold as an electrocatalyst for oxygen reduction reaction: theoretical suggestion and experimental proof[J]. J Am Chem Soc,2014,136(18):6542−6545. doi: 10.1021/ja500393g [19] WANG L, HANG R, XU Y, GUO C, QIAN Y. From ultrathin nanosheets, triangular plates to nanocrystals with exposed (102) facets, a morphology and phase transformation of sp2 hybrid BN nanomaterials[J]. RSC Adv,2014,4(27):14233. doi: 10.1039/c3ra47005a [20] CHUBAROV M, PEDERSEN H, HÖGBERG H, CZIGÄNY Z, GARBRECHT M, HENRY A. Polytype pure sp2-BN thin films as dictated by the substrate crystal structure[J]. Chem Mater,2015,27(5):1640−1645. doi: 10.1021/cm5043815 [21] SUZUKI K, YAMAGUCHI T, MATSUSHITA K, IITSUKA C, MIURA J, AKAOGI T, ISHIDA H. Aerobic oxidative esterification of aldehydes with alcohols by gold-nickel oxide nanoparticle catalysts with a core-shell structure[J]. ACS Catal,2013,3(8):1845−1849. doi: 10.1021/cs4004084 [22] YAP Y K. B−C−N Nanotubes and Related Nanostructures[M].Berlin: Springer, 2009. [23] DELLA PINA C, FALLETTA E, ROSSI M. Highly selective oxidation of benzyl alcohol to benzaldehyde catalyzed by bimetallic gold-copper catalyst[J]. J Catal,2008,260(2):384−386. doi: 10.1016/j.jcat.2008.10.003 [24] HAO C, ZHENZHEN Y, ZIHAO Z, ZITAO C, MIAOFANG C, SONG W, JIE F, SHENG D. Construction of a nanoporous highly crystalline hexagonal boron nitride from an amorphous precursor for catalytic dehydrogenation[J]. Angew Chem Int Ed,2019,58:1−6. doi: 10.1002/anie.201813481 [25] LEI W W, PORTEHAULT D, LIU D, QIN S, CHEN Y. Porous boron nitride nanosheets for effective water cleaning[J]. Nat Commun,2013,4:1777. [26] LIU M, TAN L, ZHOU B, LI L, MI Z, LI C J. Group-III nitrides catalyzed transformations of organic molecules[J]. Chem,2021,7(1):64−92. doi: 10.1016/j.chempr.2020.09.014 [27] XIANGZHAN M, ZENGXI L, YONGQIANG Z, RUIYI Y, HUI W. Deactivation behavior and aggregation mechanism of supported Au T nanoparticles in the oxidation of monoethanolamine to glycine[J]. Catal Commun,2020,(146):106127. [28] ZHONG L, YU F, AN Y, ZHAO Y, SUN Y, LI Z, LIN T, LIN Y, QI X, DAI Y, GU L, HU J, JIN S, SHEN Q, WANG H. Cobalt carbide nanoprisms for direct production of lower olefins from syngas[J]. Nature,2016,538(7623):84−87. doi: 10.1038/nature19786 -





 下载:
下载: