Study on the mechanism of oxidation of nitrogen-containing char by CO2 based on density functional theory
-
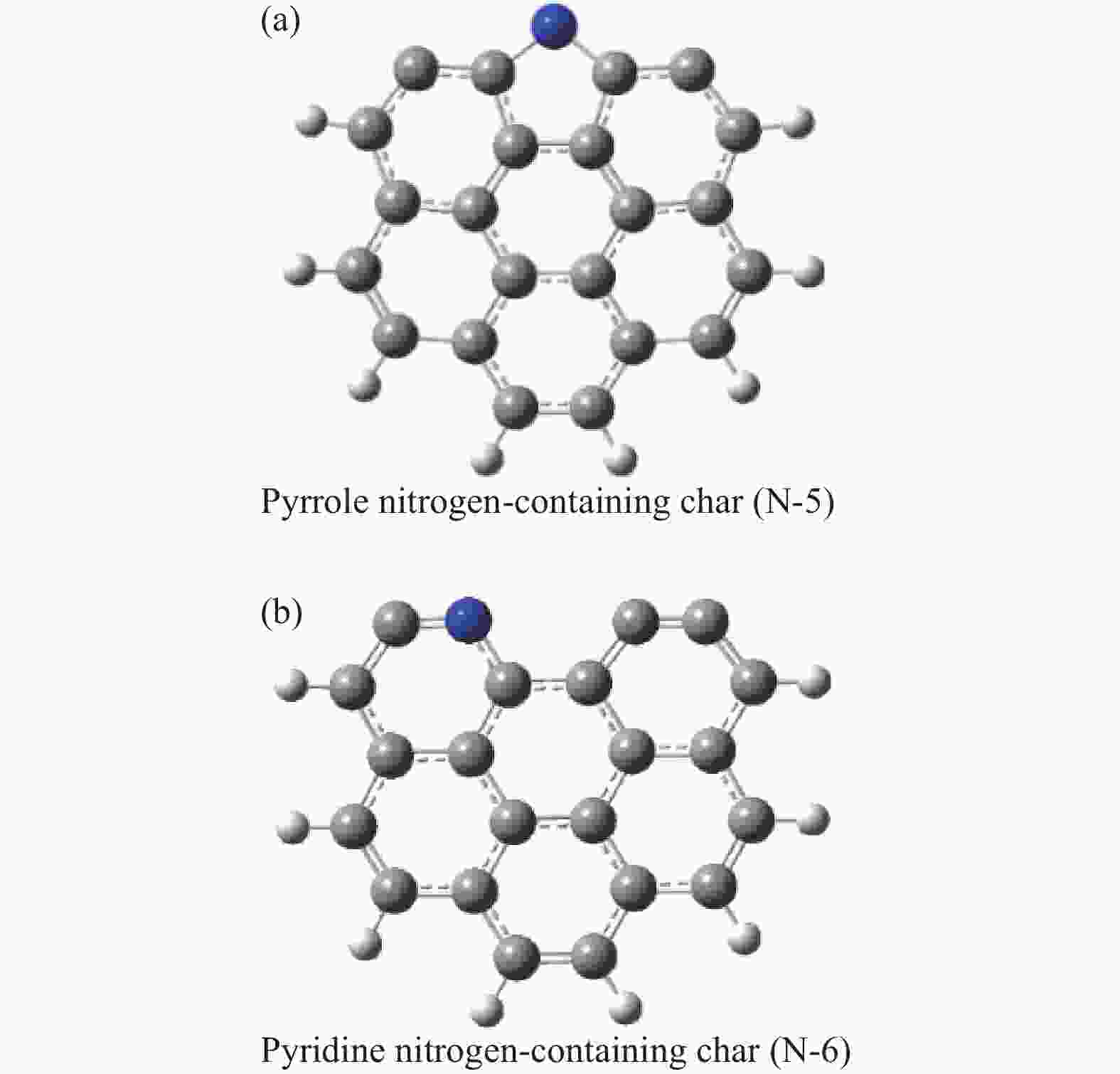
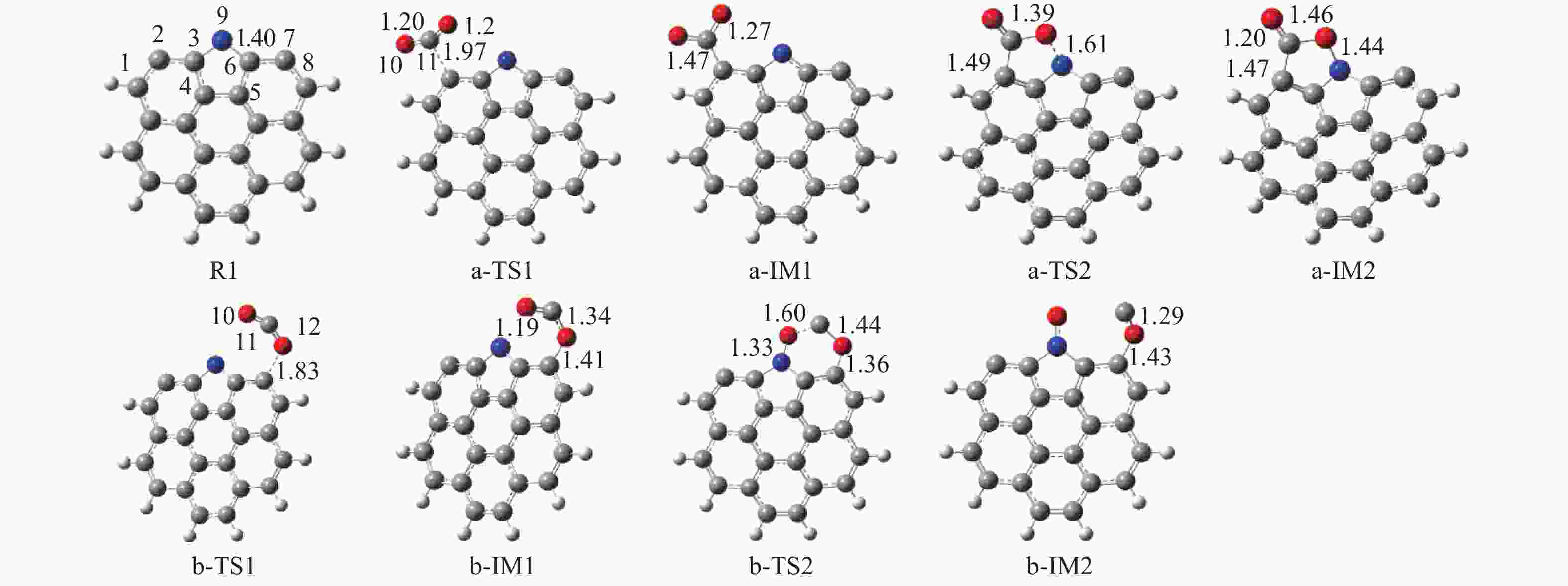
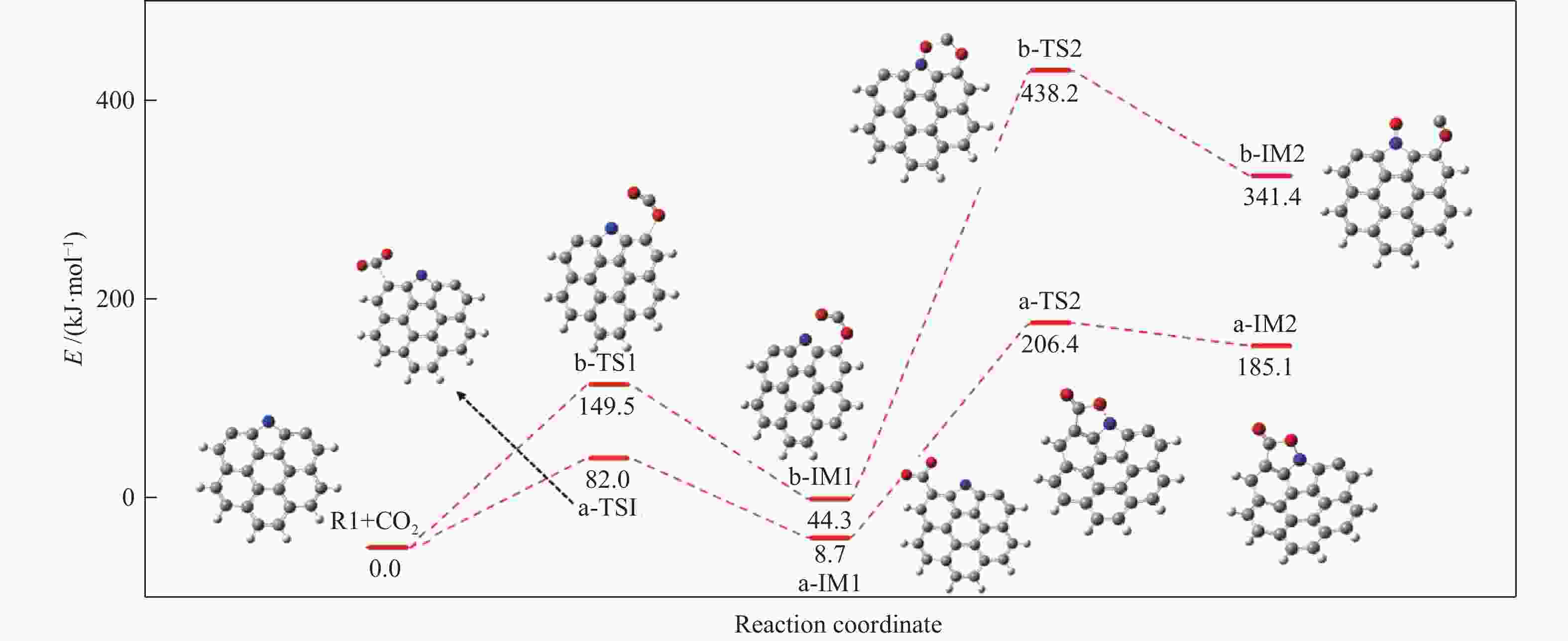
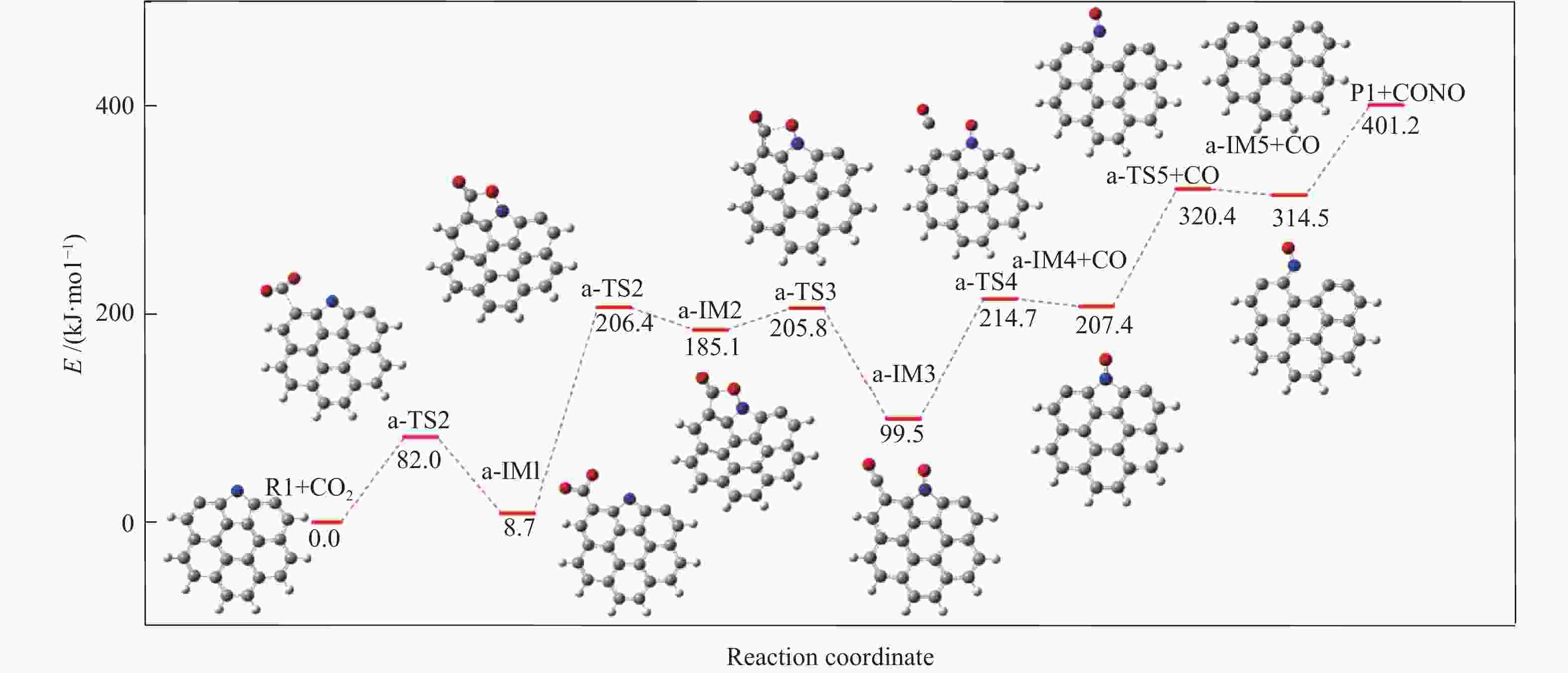
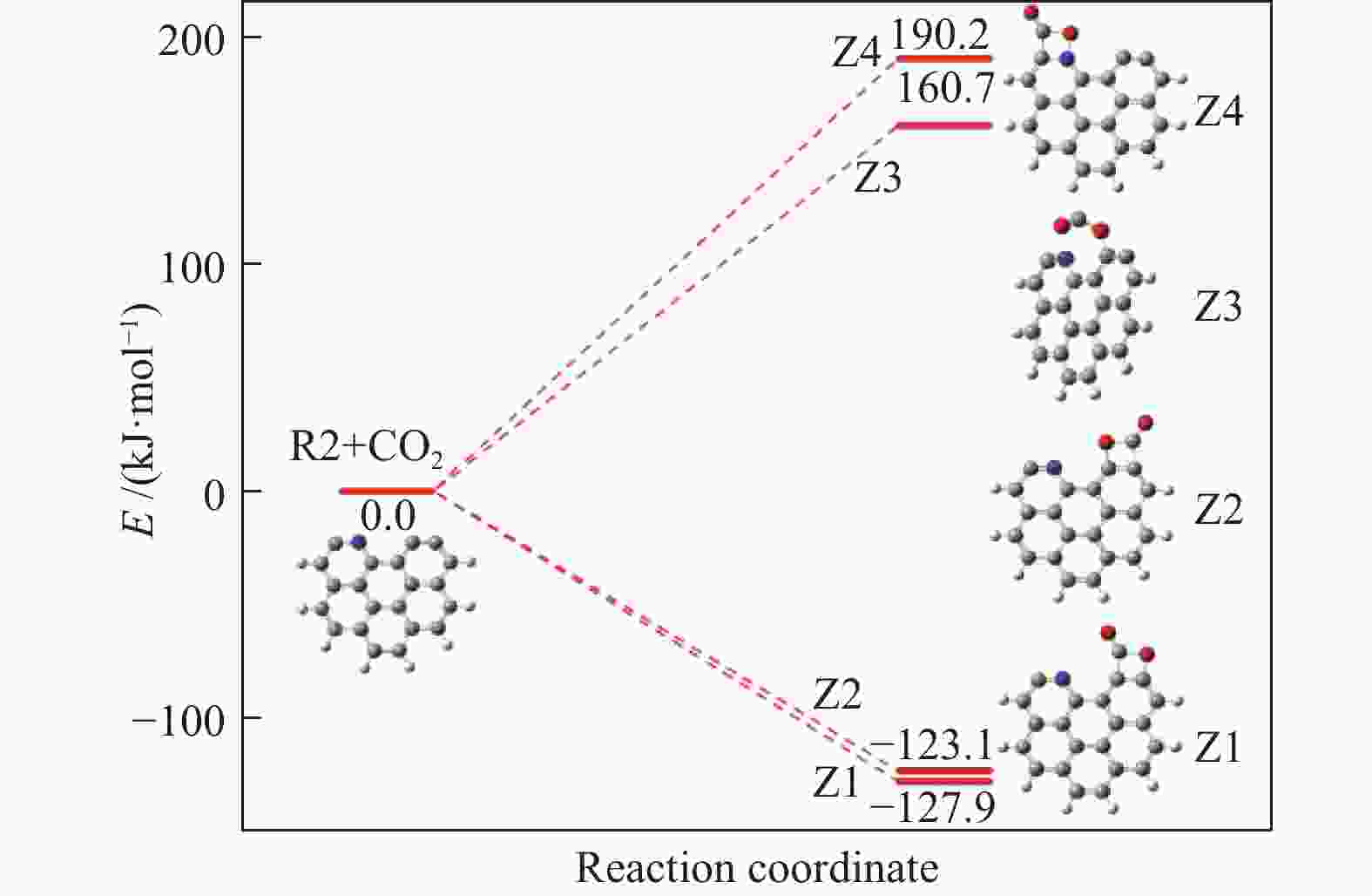
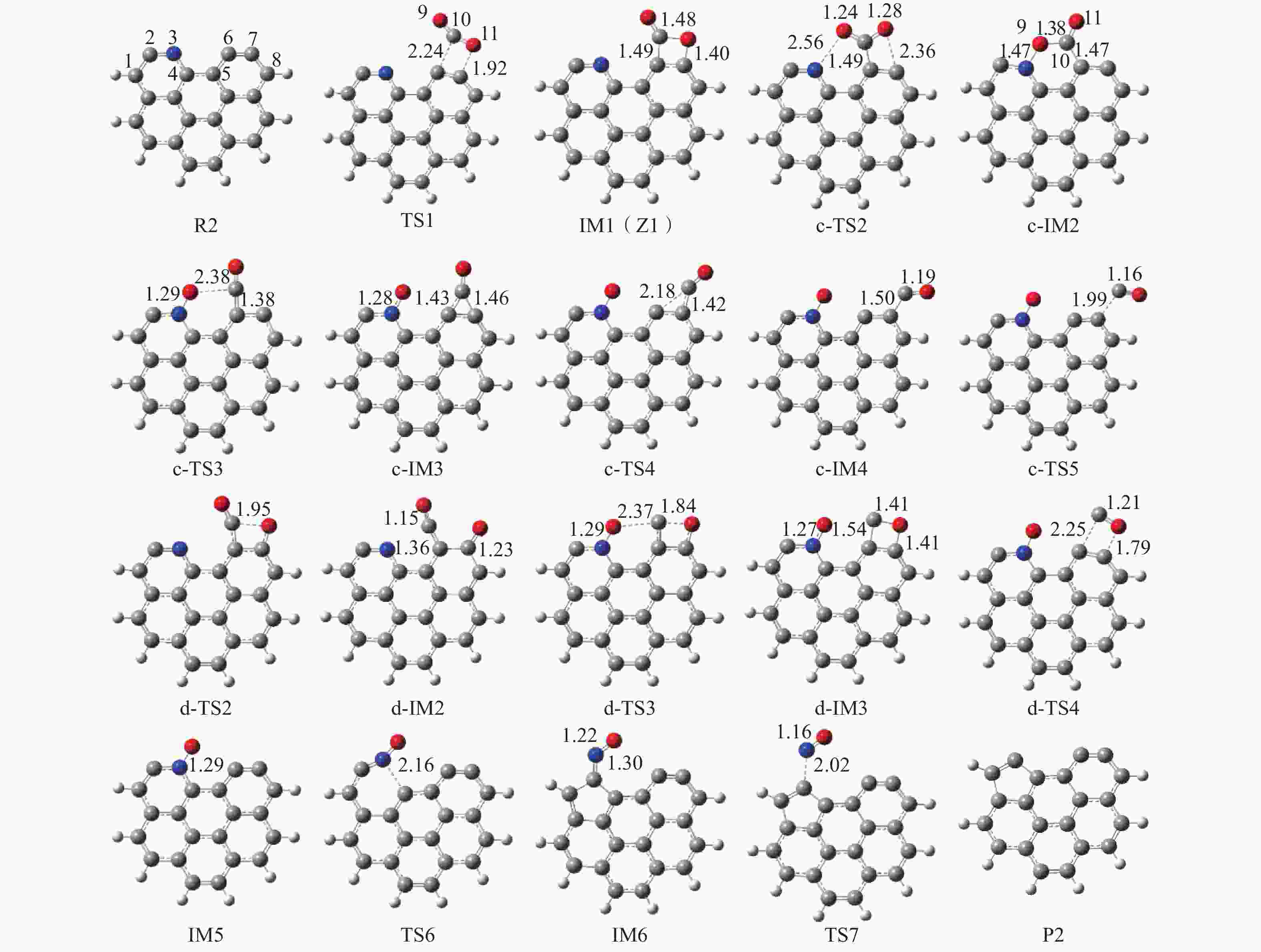
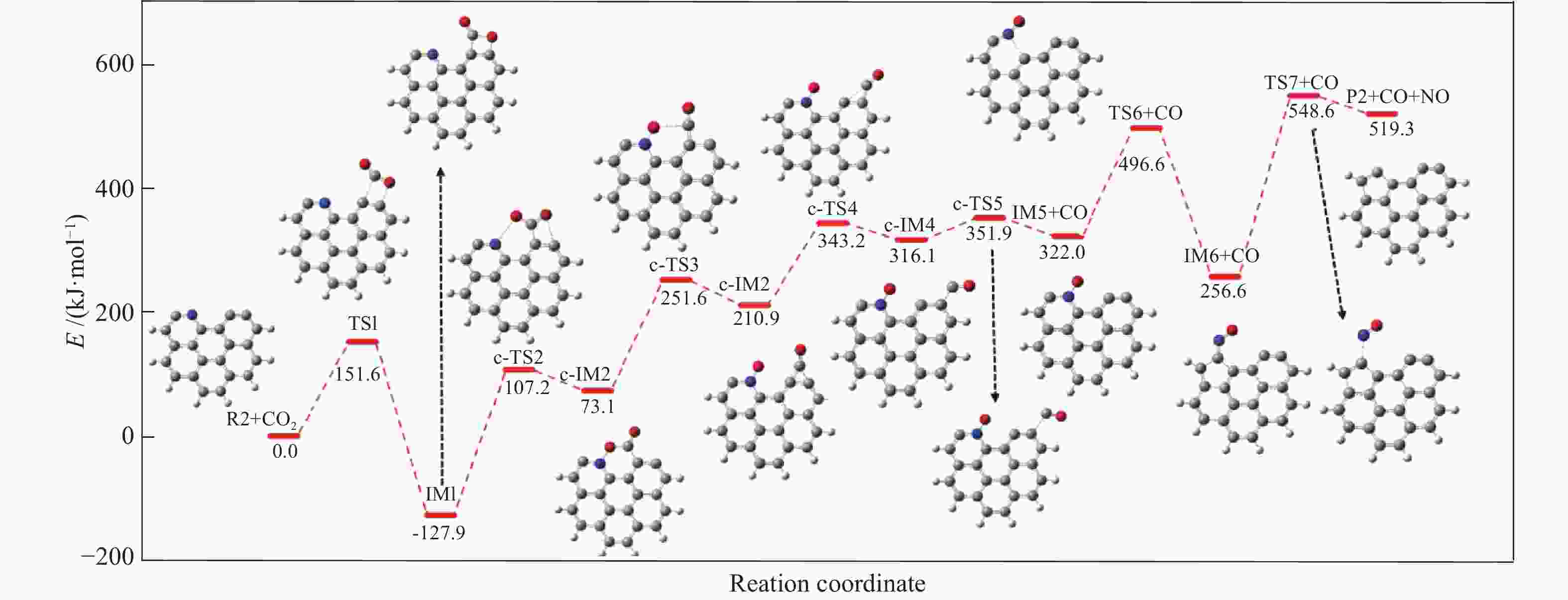
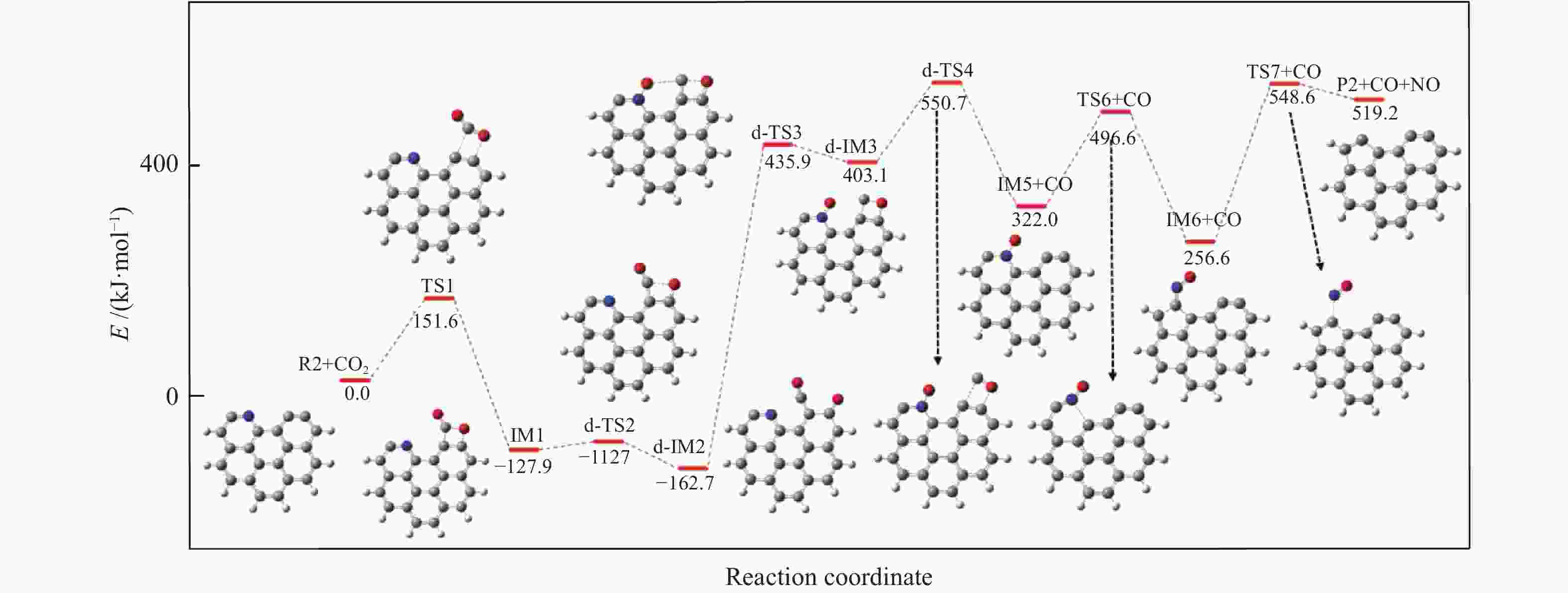
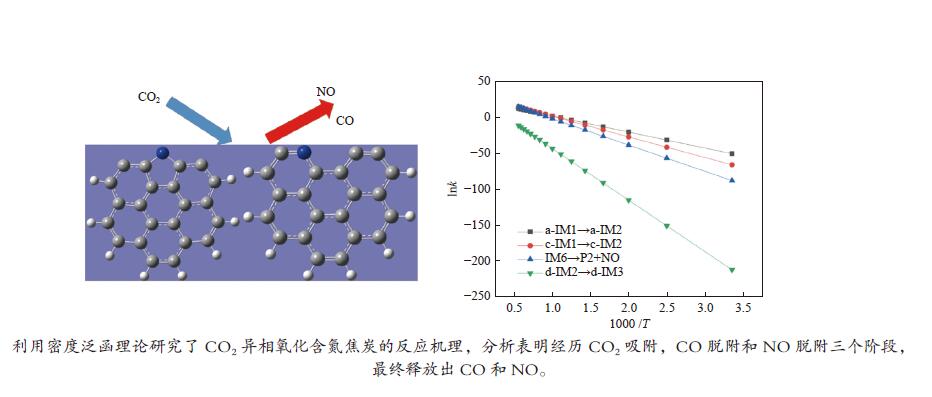
摘要: 本研究基于密度泛函理论,选取简化的含吡咯氮(N-5)或吡啶氮(N-6)焦炭模型,在分子水平上对CO2氧化含氮焦炭的异相反应机理进行研究。结构优化采用B3LYP-D3/6-31G(d)方法,单点能计算采用B3LYP-D3/def2-TZVP方法。计算结果表明,CO2氧化含氮焦炭过程分为CO2吸附、CO脱附和NO脱附三个阶段。CO2异相氧化含吡咯氮焦炭的反应中,CO2分子倾向于以C−O−down模式(N−O结合、C−C结合)吸附形成含氮和氧的五元杂环结构。然后五元环中原CO2分子的C−O键断裂形成表面羰基和表面氮氧结构,分别解吸附出CO和NO。该反应吸热401.2 kJ/mol,决速步能垒为197.6 kJ/mol。CO2异相氧化含吡啶氮焦炭的反应中,CO2分子以C−O−down和C−C结合、C−O结合模式吸附后倾向于先形成含氮和氧的六元杂环,再发生CO和NO分子的脱附。该反应吸收598.6 kJ/mol的热量,决速步能垒为292.0 kJ/mol。Abstract: In order to obtain the NO formation mechanism during the coal combustion, the heterogeneous oxidation of nitrogen-containing char by CO2 were investigated based on density functional theory. Simplified char models containing pyrrole nitrogen or pyridine nitrogen were selected as the carbonaceous surfaces. Geometric optimizations were carried out at the B3LYP-D3/6-31G(d) level. Energies of optimized geometries were calculated at the B3LYP-D3/def2-TZVP level. The results show that CO2 oxidation of nitrogen-containing char is composed of three stages: namely CO2 adsorption, CO desorption and NO desorption. In the reaction of CO2 heterogeneous oxidation of pyrrole nitrogen-containing char, CO2 molecules tend to absorb in the C−O−down mode (C−C bonding, N−O bonding) to form a five-membered heterocyclic structure containing nitrogen and oxygen atoms. Then, the surface carbonyl groups and N(O) are formation as the C−O bonds of the original CO2 molecules in the five-membered ring broken to desorb CO and NO, respectively. The reaction is 401.2 kJ/mol endothermic, and the highest energy barrier is 197.6 kJ/mol. In the reaction of CO2 heterogeneous oxidation of pyridine nitrogen-containing char, CO2 molecules tend to form six-membered heterocyclic ring containing nitrogen and oxygen atoms after adsorption in the C−O−down and C−C bonding and C−O bonding mode. And then CO and NO molecules are desorbed. The reaction is 598.6 kJ/mol endothermic, and the energy barrier of rate-determining step is 292.0 kJ/mol.
-
Key words:
- CO2 /
- nitrogen-containing char /
- heterogeneous oxidation /
- CO /
- NO /
- density functional theory
-
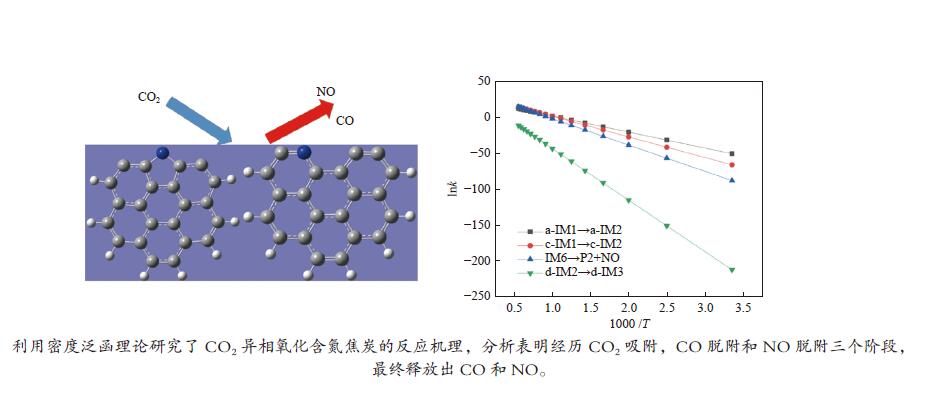
表 1 拟合所得动力学反应参数
Table 1 Fitted kinetic parameters of Arrhenius expressions
Reaction A /s−1 Ea /(kJ·mol−1) a-IM1→a-IM2 7.82 × 1010 186.0 c-IM1→c-IM2 4.57 × 1013 240.2 IM6→P2+NO 3.59 × 1015 305.7 d-IM2→d-IM3 5.49 × 1012 598.2 -
[1] 国家统计局. 中国统计年鉴2020[J]. 北京: 中国统计出版社, 2020.National Bureau of Statistics of China. China Statistical Yearbook[J]. Beijing: China Statistics Press, 2020. [2] 毛洪钧, 李悦宁, 林应超, 王婷, 李维尊, 鞠美庭, 朱复东. 生物质锅炉氮氧化物排放控制技术研究进展[J]. 工程科学学报,2019,41(1):4−14.MAO Hong-jun, LI Yue-ning, LIN Ying-chao, WANG Ting, LI Wei-zun, JU Mei-ting, ZHU Fu-dong. Overview of advances in emission control technologies for nitric oxides from biomass boilers[J]. Chin J Eng,2019,41(1):4−14. [3] DE SOETE G G, CROISET E, RICHARD J R. Heterogeneous formation of nitrous oxide from char-bound nitrogen[J]. Combust Flame,1999,117(1):140−154. [4] MILLER J A, BOWMAN C T. Mechanism and modeling of nitrogen chemistry in combustion[J]. Prog Energy Combust Sci,1989,15(4):287−338. doi: 10.1016/0360-1285(89)90017-8 [5] WINTER F, WARTHA C, LÖFFLER G, HOFBAUER H. The NO and N2O formation mechanism during devolatilization and char combustion under fluidized-bed conditions[J]. Symp (Int) Combust,1996,26(2):3325−3334. doi: 10.1016/S0082-0784(96)80180-9 [6] 车得福. 煤氮热变迁与氮氧化物生成[M]. 西安: 西安交通大学出版社, 2013.CHE De-fu. Thermal Coal-N Transformation and Nitrogen oxide Generation[M]. Xi'an: Xi'an Jiaotong University Press, 2013. [7] GLARBORG P, JENSEN A D, JOHNSSON J E. Fuel nitrogen conversion in solid fuel fired systems[J]. Prog Energy Combust Sci,2003,29(2):89−113. doi: 10.1016/S0360-1285(02)00031-X [8] 张秀霞, 周志军, 周俊虎, 刘建忠, 岑可法. O2氧化含氮焦炭释放CO和NO的量子化学研究[J]. 煤炭学报,2011,36(1):129−134.ZHANG Xiu-xia, ZHOU Zhi-jun, ZHOU Jun-hu, LIU Jian-zhong, CEN Ke-fa. A quantum chemistry study of CO and NO desorption from oxidation of nitrogen-containing char by oxygen[J]. J China Coal Soc,2011,36(1):129−134. [9] 张秀霞. 焦炭燃烧过程中氮转化机理与低NOx燃烧技术的开发[D]. 杭州: 浙江大学, 2012.ZHANG Xiu-xia. Nitrogen conversion mechanism during char combustion and develepment of low NOx technology[D]. Hangzhou: Zhejiang University, 2012. [10] WANG X B, HU Z F, DENG S H, XIONG Y Y, TAN H Z. Effect of biomass/coal co-firing and air staging on NOx emission and combustion efficiency in a drop tube furnace[J]. Energy Procedia,2014,61:2331−2334. doi: 10.1016/j.egypro.2014.11.1196 [11] SALZMANN R, NUSSBAUMER T. Fuel staging for NOx reduction in biomass combustion: experiments and modeling[J]. Energy Fuels, 15(3): 575–582. [12] 张泰, 柳朝晖, 黄晓宏, 陈松涛, 王勇, 皮里刚, 郑楚光. 3 MWth富氧燃烧气体污染物生成与排放特性研究[J]. 工程热物理学报,2014,35(8):1652−1655.ZHANG Tai, LIU Zhao-hui, HUANG Xiao-hong, CHEN Song-tao, WANG Yong, PI Li-gang, ZHENG Chu-guang. Experimental study of gaseous pollutant formation and emission on 3 MWth oxy-fuel pilot test facility[J]. J Eng Therm,2014,35(8):1652−1655. [13] OHTSUKA Y, WU Z. Nitrogen release during fixed-bed gasification of several coals with CO2: Factors controlling formation of N2[J]. Fuel,1999,78(5):521−527. doi: 10.1016/S0016-2361(98)00187-2 [14] PARK D–C, DAY S J, NELSON P F. Nitrogen release during reaction of coal char with O2, CO2, and H2O[J]. Proc Combust Ins,2005,30(2):2169−2175. doi: 10.1016/j.proci.2004.08.051 [15] ZHANG H, JIANG X M, LIU J X. Updated effect of carbon monoxide on the interaction between NO and char bound nitrogen: A combined thermodynamic and kinetic study[J]. Combust Flame,2020,220:107−118. doi: 10.1016/j.combustflame.2020.06.032 [16] ZHANG H, LIU J X, LIU J G, LUO L, JIANG X M. DFT study on the alternative NH3 formation path and its functional group effect[J]. Fuel,2018,214(FEB):108−114. [17] ZHANG H, LIU J X, WANG X Y, LUO L, JIANG X M. DFT study on the C(N)-NO reaction with isolated and contiguous active sites[J]. Fuel,2017,203(SEP):715−724. [18] 刘艳华, 车得福, 李荫堂, 惠世恩, 徐通模. X射线光电子能谱确定铜川煤及其焦中氮的形态[J]. 西安交通大学学报,2001,35(7):661−665. doi: 10.3321/j.issn:0253-987X.2001.07.001LIU Yan-hua, CHE De-fu, LI Yin-tang, HUI Shi-en, XU Tong-mo. X-Ray photoelectron spectroscopy determination of the forms of nitrogen in Tongchuan coal and its chars[J]. J Xi'an Jiaotong Univ,2001,35(7):661−665. doi: 10.3321/j.issn:0253-987X.2001.07.001 [19] ZHAO S H, SUN R Y, BI X L, PAN X J, SU Y. Density functional theory study of the heterogenous interaction between char-bound nitrogen and CO2 during oxy-fuel coal combustion[J]. Combust Flame,2020,216:136−145. doi: 10.1016/j.combustflame.2020.02.026 [20] CHEN N, YANG R T. Ab initio molecular orbital calculation on graphite Selection of molecular system and model chemistry[J]. Carbon,1998,36(7−8):1061−1070. doi: 10.1016/S0008-6223(98)00078-5 [21] 刘烨鸣. 高浓度CO2燃烧条件下NOx转化机理的研究[D]. 扬州: 扬州大学, 2018.LIU Ye-ming. Study on NOx conversion mechanism under O2/CO2 combustion with high concentration of CO2[D]. Yangzhou: Yangzhou University, 2018. [22] STEPHENS P J, DEVLIN F J, CHABALOWSKI C F, FRISCH M J. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields[J]. J Phys Chem,1994,98(45):11623−11627. doi: 10.1021/j100096a001 [23] SENDT K, HAYNES B S. Density functional study of the chemisorption of O2 on the zigzag surface of graphite[J]. Combust Flame,2005,143(4):629−643. doi: 10.1016/j.combustflame.2005.08.026 [24] ZHU Z H, FINNERTY J, LU G Q, YANG R T. A comparative study of carbon gasification with O2 and CO2 by density functional theory calculations[J]. Energy Fuels,2002,16(6):1359−1368. doi: 10.1021/ef0200020 [25] ZHANG H, JIANG X M, LIU J X, SHEN J. Application of density functional theory to the nitric oxide heterogeneous reduction mechanism in the presence of hydroxyl and carbonyl groups[J]. Energy Convers Manage,2014,83(JUL):167−176. [26] MONTOYA A, TRUONG T N, SAROFIM A F. Application of density functional theory to the study of the reaction of NO with char–bound nitrogen during combustion[J]. J Phys Chem A,2000,104(36):8409−8417. doi: 10.1021/jp001045p [27] STEFAN G. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu[J]. J Chem Phys,2010,15(132):1−19. [28] 钟俊, 高正阳, 丁艺, 余岳溪, 杨维结. Zigzag煤焦表面异相还原N2O反应[J]. 煤炭学报,2017,42(11):3028−3034.ZHONG Jun, GAO Zheng-yang, DING Yi, YU Yue-xi, YANG Wei-jie. Heterogeneous reduction reaction of N2O by char based on zigzag carbonaceous model[J]. J China Coal Soc,2017,42(11):3028−3034. [29] GONZALEZ C, SCHLEGEL H B. Reaction path following in mass-weighted internal coordinates[J]. J Phys Chem,1990,94(14):5523−5527. doi: 10.1021/j100377a021 [30] FRISCH M J, TRUCKS G W, SCHLEGEL H B, SCUSERIA G E. Gaussian 09 Rev. D. 01[M]. Wallingford, CT. 2009. [31] 傅献彩. 物理化学[M]. 5版. 北京: 高等教育出版社, 2005.FU Xian-cai. Physical Chemistry[M]. 5th ed. Beijing: Higher Education Press, 2005. [32] CHEN P, GU M Y, CHEN G, LIU F S, LIN Y Y. DFT study on the reaction mechanism of N2O reduction with CO catalyzed by char[J]. Fuel,2019,192(9):1682−706. [33] 田向红. 焦炭氧化的密度泛函理论研究[D]. 郑州: 郑州大学, 2019.TIAN Xiang-hong. Study on coke oxidation with density functional theory[D]. Zhengzhou: Zhengzhou University, 2019. -




 下载:
下载: