Electrochemical properties of coal pitch-based three-dimensional porous carbon as electrode materials for supercapacitors
-
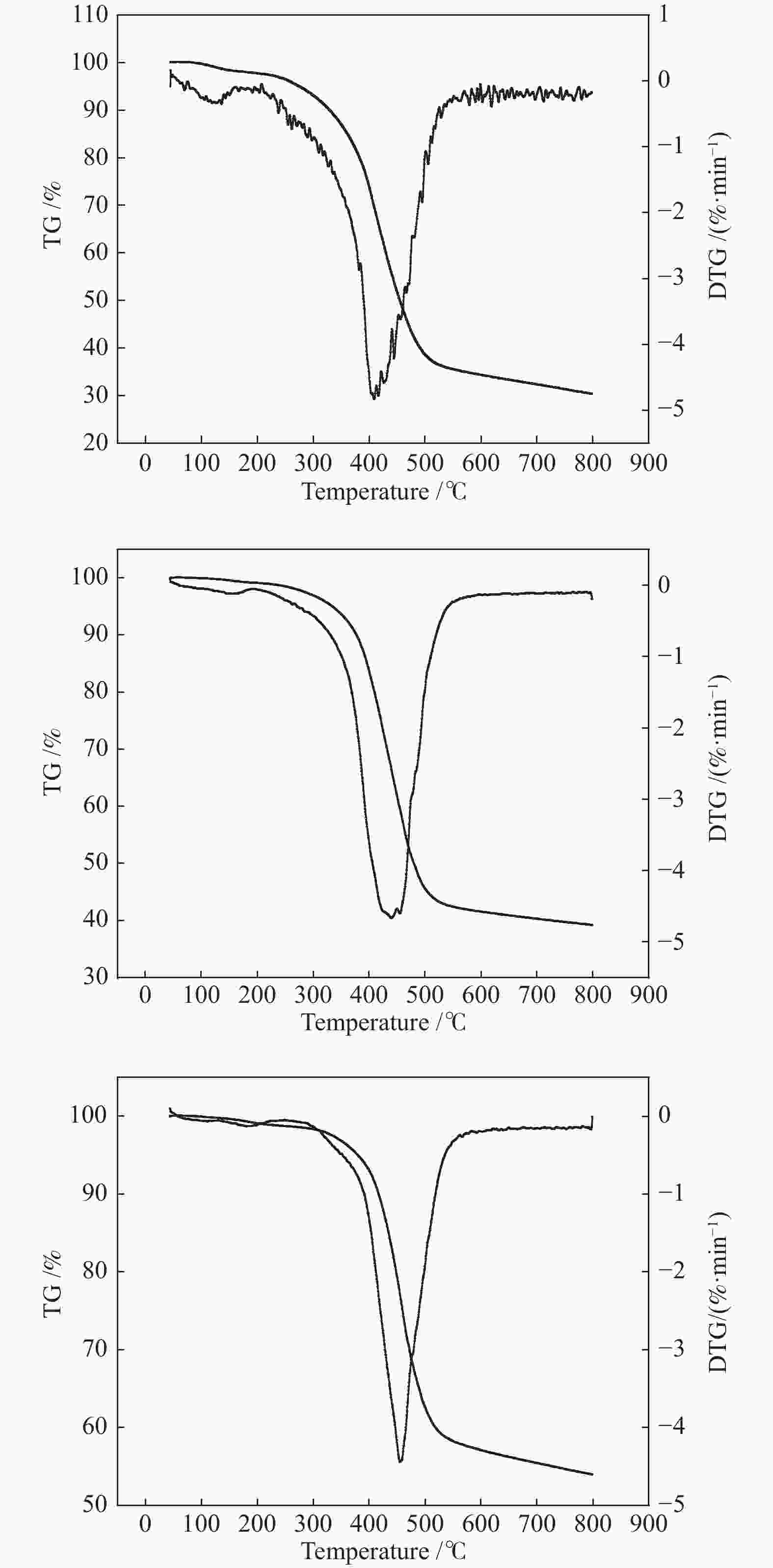
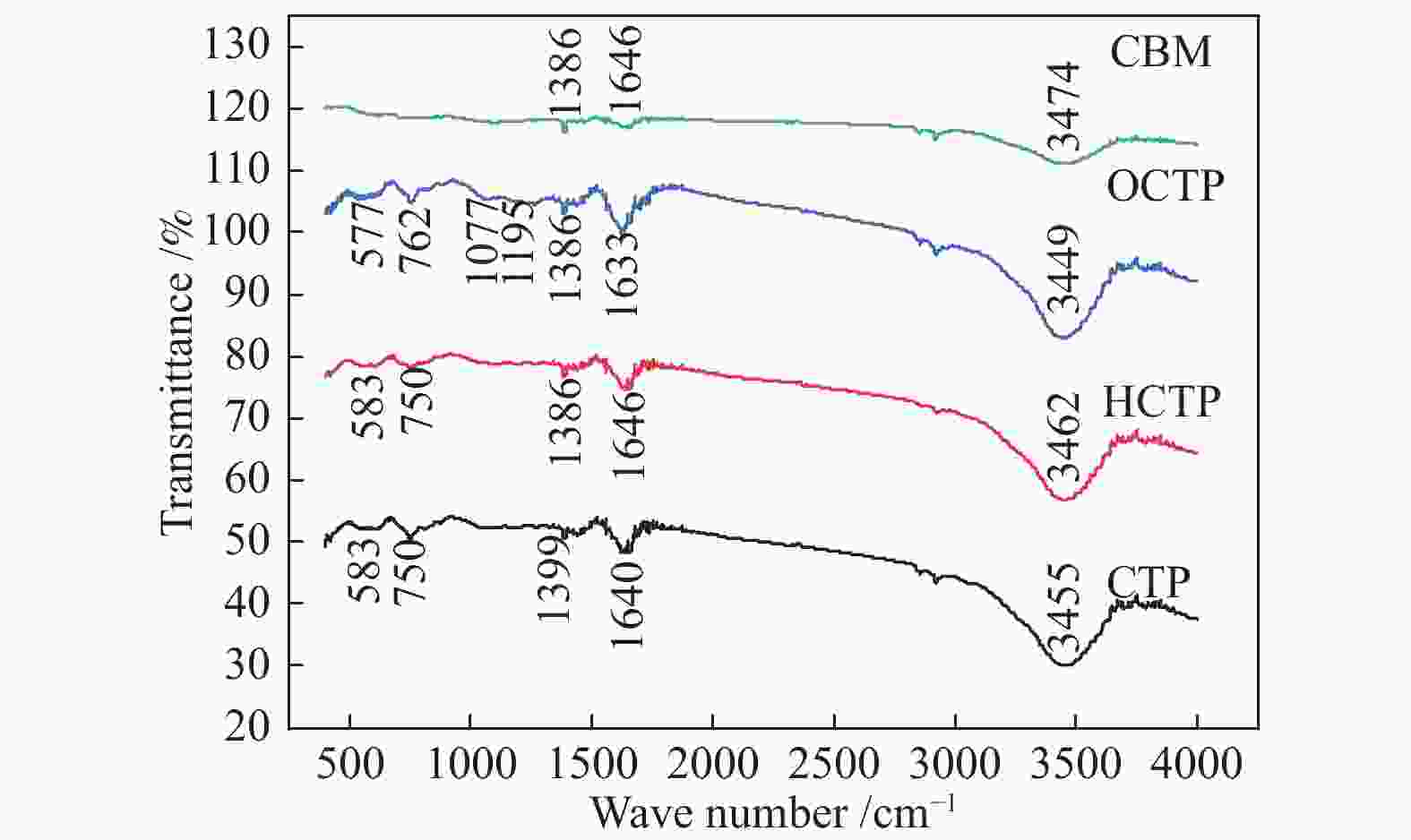
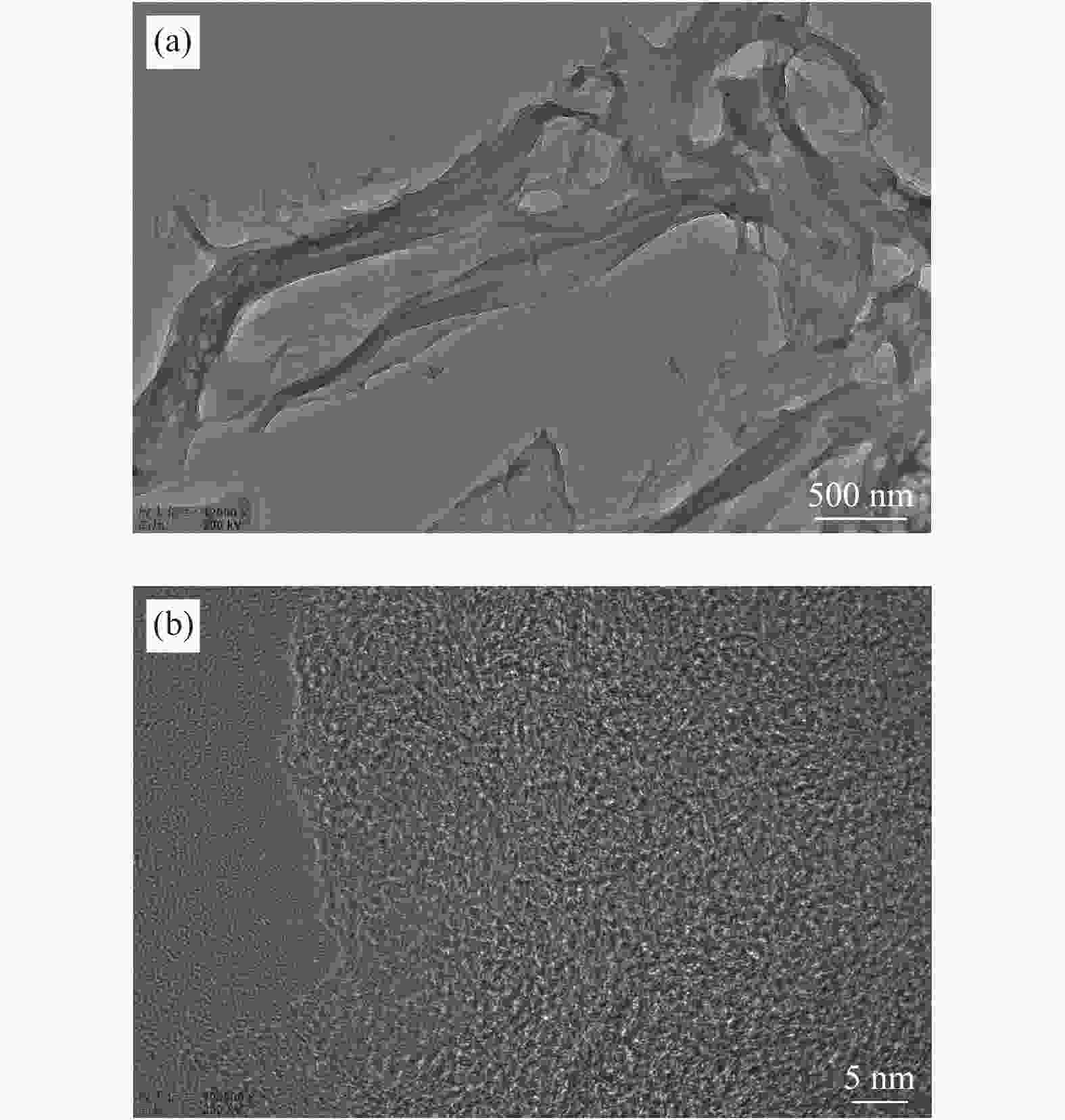
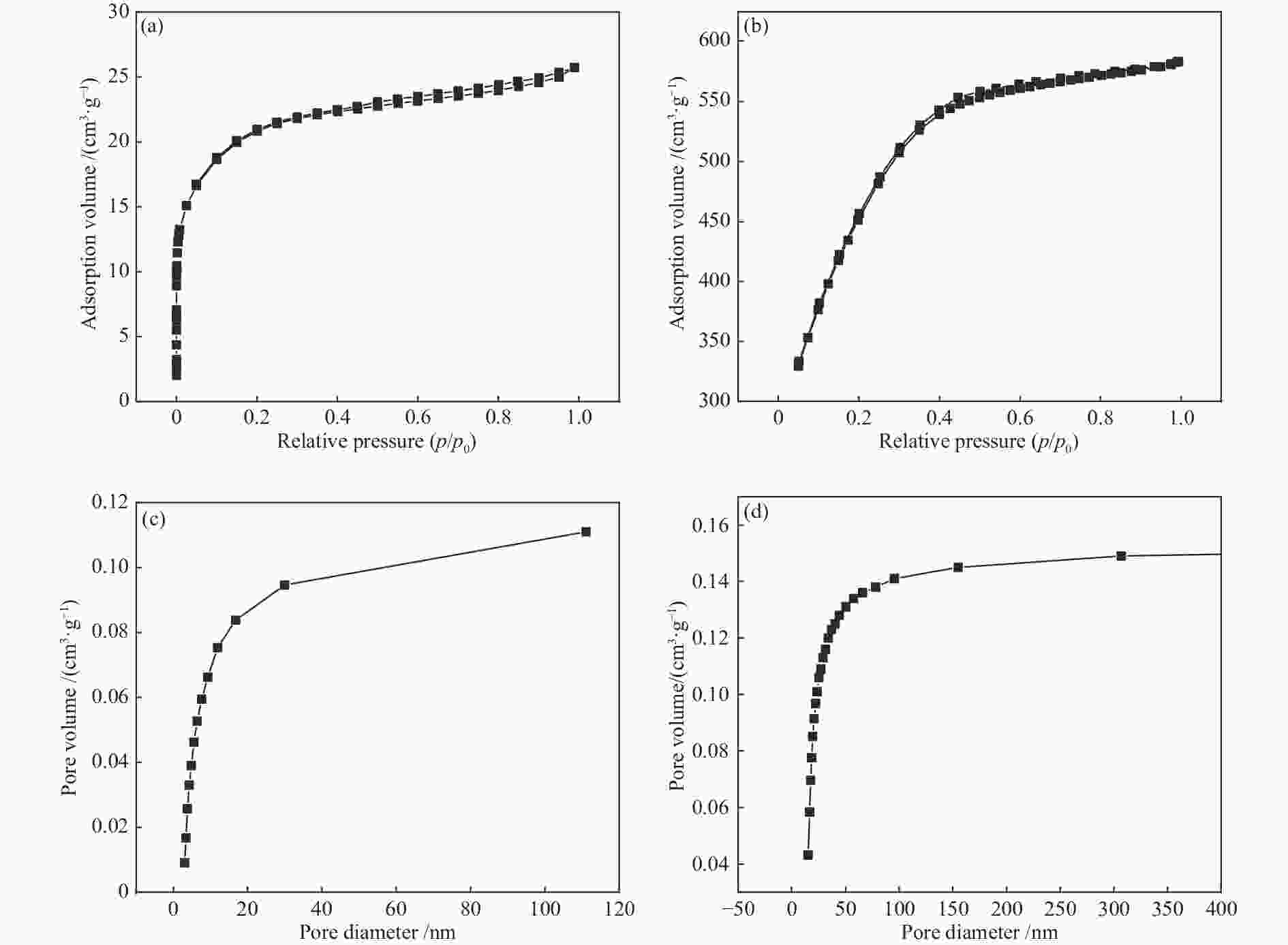
摘要: 以廉价的煤沥青为原料,加入造孔剂,依次经过混合工艺、空气氧化稳定化工艺、炭化工艺得到三维骨架结构的炭材料,再经过水蒸气活化,得到高比表面积的多孔炭材料。通过热重分析和元素分析研究沥青和空气氧化稳定化、炭化后样品的热反应特性和组成结构的变化;利用扫描电子显微镜、比表面积分析仪和电化学工作站等对活化后样品表面形貌、孔结构和电化学性能进行表征。水蒸气活化后的多孔炭比表面积可达到1638 m2/g。以此多孔炭为超级电容器电极材料,制备的水系双电层电容器循环性能良好,1 A/g电流密度下比电容可达252 F/g,经过10000次循环后,比容量仍能保持97.3%。因此,该方法制备的多孔炭材料价格低廉、化学稳定性好,可作为一种理想的超级电容器电极材料。Abstract: Using cheap coal pitch as raw material and adding pore-forming agent, the carbon material with three-dimensional skeleton structure was obtained by mixing process, air oxidation stabilization process and carbonization process successively, and then the porous carbon material with high specific surface area was obtained by water vapor activation. Thermogravimetric and elemental analysis were used to study the thermal reaction characteristics and structural changes of the samples after oxidation stabilization and carbonization. The surface morphology, pore structure and electrochemical properties of the activated samples were characterized by scanning electron microscope, BET method and electrochemical workstation. After water vapor activation, BET of porous carbon can reach 1638 m2/g. The water-based double layer capacitors prepared with this porous carbon as the electrode material of supercapacitors have good cyclic performance, and the specific capacitance can reach 252 F/g at 1 A/g current density. After 10000 cycles, the specific capacity can still maintain 97.3%. Therefore, the porous carbon materials prepared by this method have low cost and good chemical stability, and can be used as an ideal electrode material for supercapacitors.
-
Key words:
- coal pitch /
- porous carbon /
- supercapacitor
-
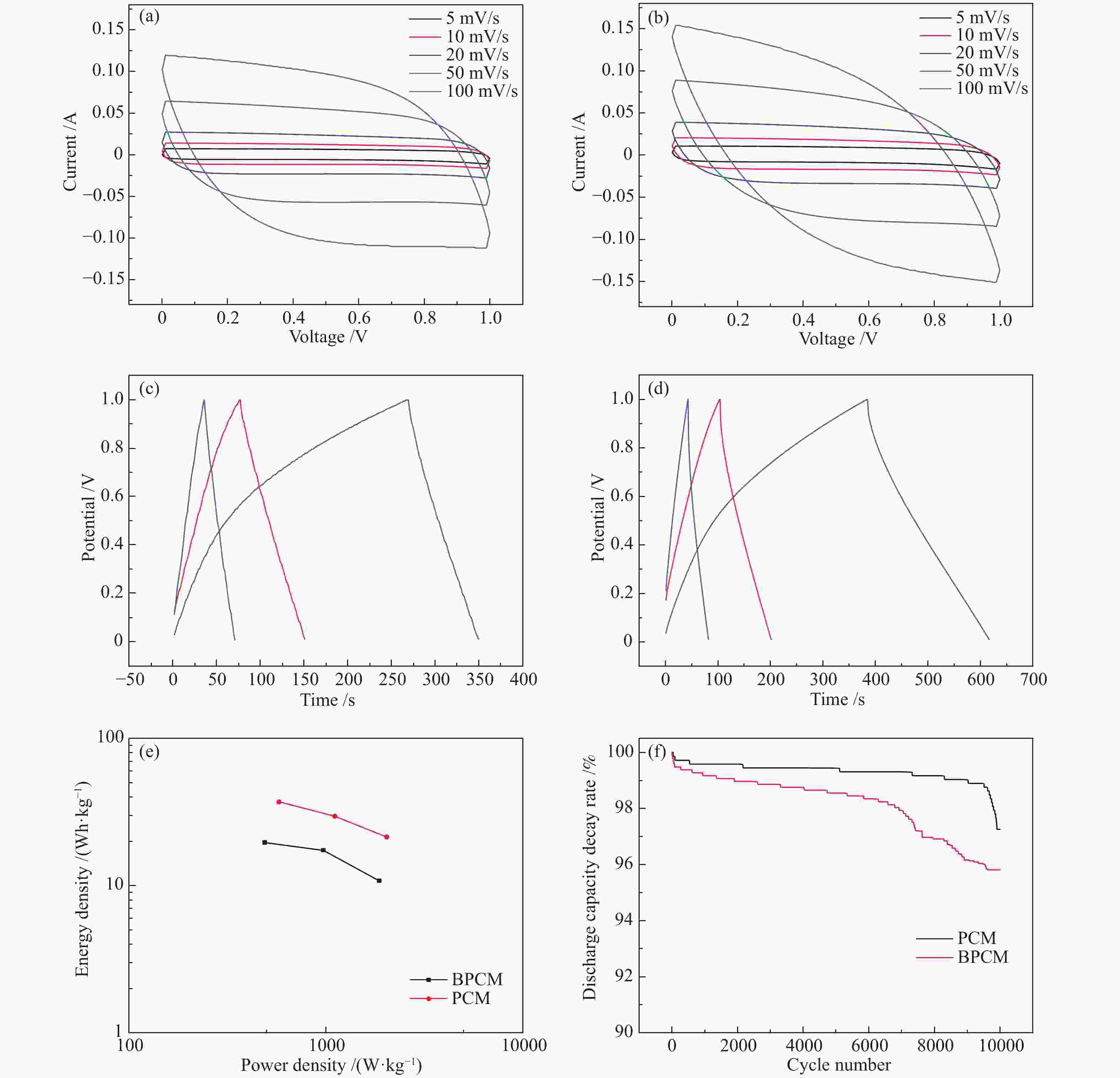
图 6 (a)BPCM和(b)PCM在不同扫描速率下的循环伏安曲线,(c)BPCM和(d)PCM在不同电流密度下的恒流充放电曲线,(e)BPCM和PCM电极的能量/功率密度图,(f)BPCM和PCM电极的循环寿命测试
Figure 6 CV curves of (a) BPCM and (b) PCM electrodes at different scanning rates, charge/discharge curves of (c) BPCM and (d) PCM electrodes at different current densities, (e) ragone plots of BPCM and PCM electrodes,(f) Cycle life tests of BPCM and PCM electrodes
表 1 CTP的基本性质分析
Table 1 Basic analysis of CTP
Sample SP/℃ Content w/% Element composition /% Cv QI TI ash C H N S O* CTP 119.4 58.56 0.16 21.40 trace 91.05 4.37 1.06 0.48 3.04 *: by difference 表 2 氧化反应前后煤沥青性质
Table 2 Analysis of properties of CTP before and after oxidation reaction
Sample SP/℃ Ash/% Element composition /% C H N S O CTP 119.4 trace amount 91.05 4.37 1.06 0.48 3.04 HCTP 260 trace amount 93.64 4.01 1.05 0.48 0.82 OCTP 280 trace amount 87.69 3.27 0.98 0.37 7.69 表 3 不同电流密度下BPCM和PCM的质量比电容
Table 3 Specific capacitances of BPCM and PCM electrodes at different current densities (F/g)
Sample 0.5 A/g 1 A/g 2 A/g BPCM 82.54 152.94 146.06 PCM 290.13 252.64 216.52 -
[1] YANG Z F, TIAN J R, YIN Z F, CUI C J QIAN W Z, WEI F. Carbon nanotube-and graphene-based nanomaterials and applications in high-voltage supercapacitor: A review[J]. Carbon,2019,141:467−480. doi: 10.1016/j.carbon.2018.10.010 [2] YIN J, ZHANG W, ALHEBSHI N A. Synthesis strategies of porous carbon for supercapacitor applications[J]. Small Methods,2020,4(3):1900853. doi: 10.1002/smtd.201900853 [3] CHEN T, DAI L. Carbon nanomaterials for high-performance supercapacitors[J]. Mater Today,2013,16(7-8):272−280. doi: 10.1016/j.mattod.2013.07.002 [4] CHEN X, PAUL R, DAI L. Carbon-based supercapacitors for efficient energy storage[J]. Natl Sci Rev,2017,4(3):453−489. doi: 10.1093/nsr/nwx009 [5] STAAF L G H, LUNDGREN P, ENOKSSON P. Present and future supercapacitor carbon electrode materials for improved energy storage used in intelligent wireless sensor systems[J]. Nano Energy,2014,9:128−141. doi: 10.1016/j.nanoen.2014.06.028 [6] PORTET C, TABERNA P L, SIMON P, FLAHAUT E, LABERTY-ROBERT C. High power density electrodes for carbon supercapacitor applications[J]. Electrochim Acta,2005,50(20):4174−4181. doi: 10.1016/j.electacta.2005.01.038 [7] ZHANG L L, ZHAO X S. Carbon-based materials as supercapacitor electrodes[J]. Chem Soc Rev,2009,38(9):2520−2531. doi: 10.1039/b813846j [8] WANG G, ZHANG L, ZHANG J. A review of electrode materials for electrochemical supercapacitors[J]. Chem Soc Rev,2012,41(2):797−828. doi: 10.1039/C1CS15060J [9] SAIKIA B K, BENOY S M, BORA M, TAMULY J, PANDEY M, BHATTACHARYA D. A brief review on supercapacitor energy storage devices and utilization of natural carbon resources as their electrode materials[J]. Fuel,2020,282(15):118796. [10] BORCHARDT L, OSCHATZ M, KASKEL S. Tailoring porosity in carbon materials for supercapacitor applications[J]. Mater Horizons,2014,1(2):157−168. doi: 10.1039/C3MH00112A [11] XIE K, QIN X T, WANG X Z, WANG Y N, TAO H S, WU Q, YANG L J HU Z. Carbon nanocages as supercapacitor electrode materials[J]. Adv Mater,2012,24(3):347−352. doi: 10.1002/adma.201103872 [12] XIE L, SUN G, SU F. Hierarchical porous carbon microtubes derived from willow catkins for supercapacitor applications[J]. J Mater Chem A,2016,4(5):1637−1646. doi: 10.1039/C5TA09043A [13] JIANG H, MA J, LI C Z. Mesoporous carbon incorporated metal oxide nanomaterials as supercapacitor electrodes[J]. Adv Mater,2012,24(30):4197−4202. doi: 10.1002/adma.201104942 [14] LI H Q, LUO J Y, ZHOU X F. An ordered mesoporous carbon with short pore length and its electrochemical performances in supercapacitor applications[J]. J Electrochem Soc,2007,154(8):A731. doi: 10.1149/1.2741198 [15] 刘海晶. 电化学超级电容器多孔碳电极材料的研究[D]. 上海: 复旦大学, 2011.LIU Hai-jing. Study on porous carbon electrode materials for electrochemical supercapacitors[D]. Shanghai: Fudan University, 2011. [16] 席云龙. 超级电容器用新型聚苯胺/二氧化锰/多孔碳电极材料的制备与研究[D]. 吉林: 吉林大学, 2017.XI Yun-long. Preparation and study of novel polyaniline / manganese dioxide / porous carbon electrode materials for supercapacitors[D]. Jilin: Jilin University, 2017. [17] LE VAN K, THI T T L. Activated carbon derived from rice husk by NaOH activation and its application in supercapacitor[J]. Prog Nat Sci-Mater,2014,24(3):191−198. doi: 10.1016/j.pnsc.2014.05.012 [18] BABEL K, JUREWICZ K. KOH activated carbon fabrics as supercapacitor material[J]. J Phys Chem Solids,2004,65(2/3):275−280. doi: 10.1016/j.jpcs.2003.08.023 [19] JÄNES A, KURIG H, LUST E. Characterisation of activated nanoporous carbon for supercapacitor electrode materials[J]. Carbon,2007,45(6):1226−1233. doi: 10.1016/j.carbon.2007.01.024 -





 下载:
下载: