Effect of manganese modification on the low-temperature formaldehyde oxidation performance of ZIF-67 derived Co3O4
-
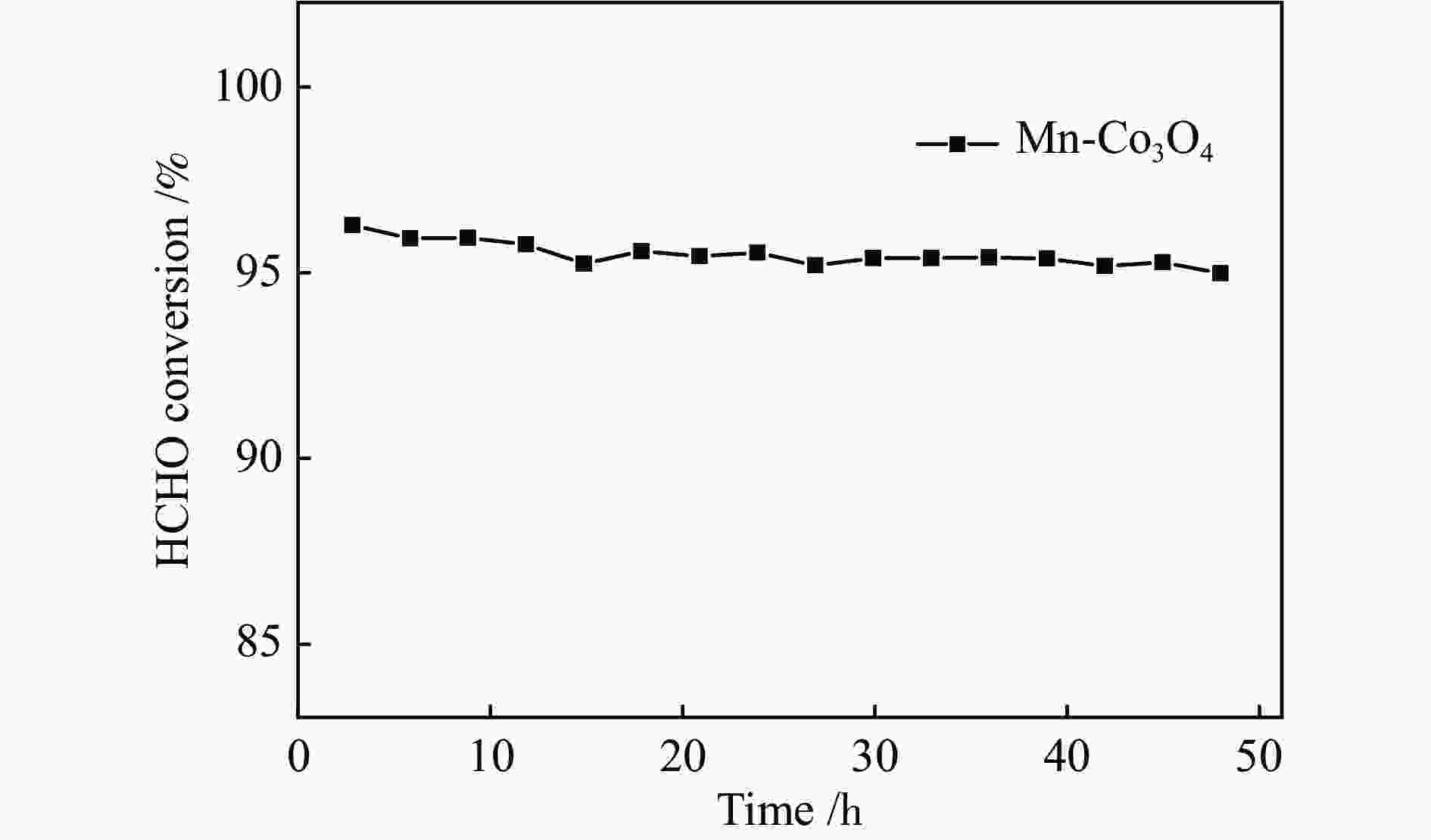
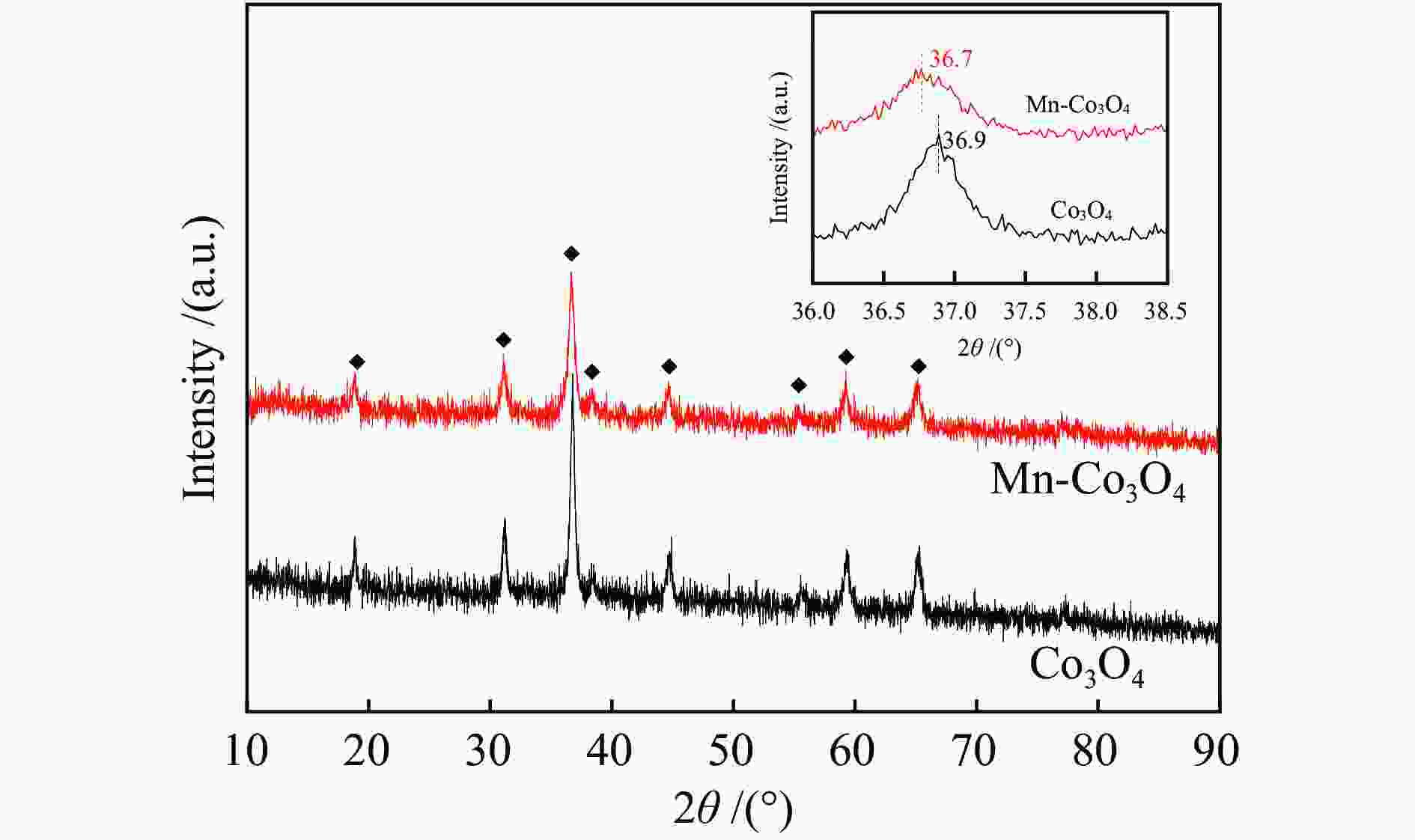
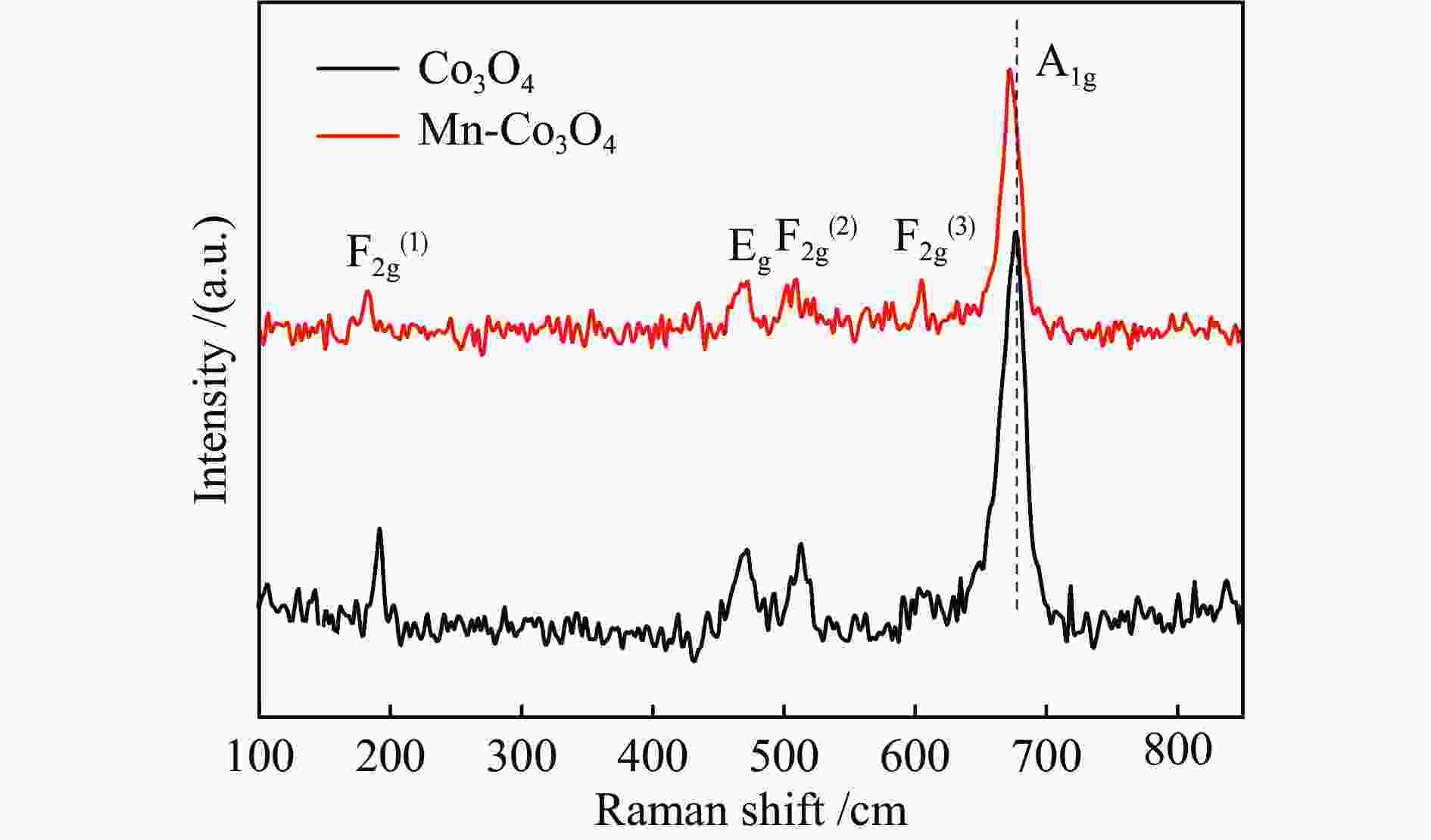
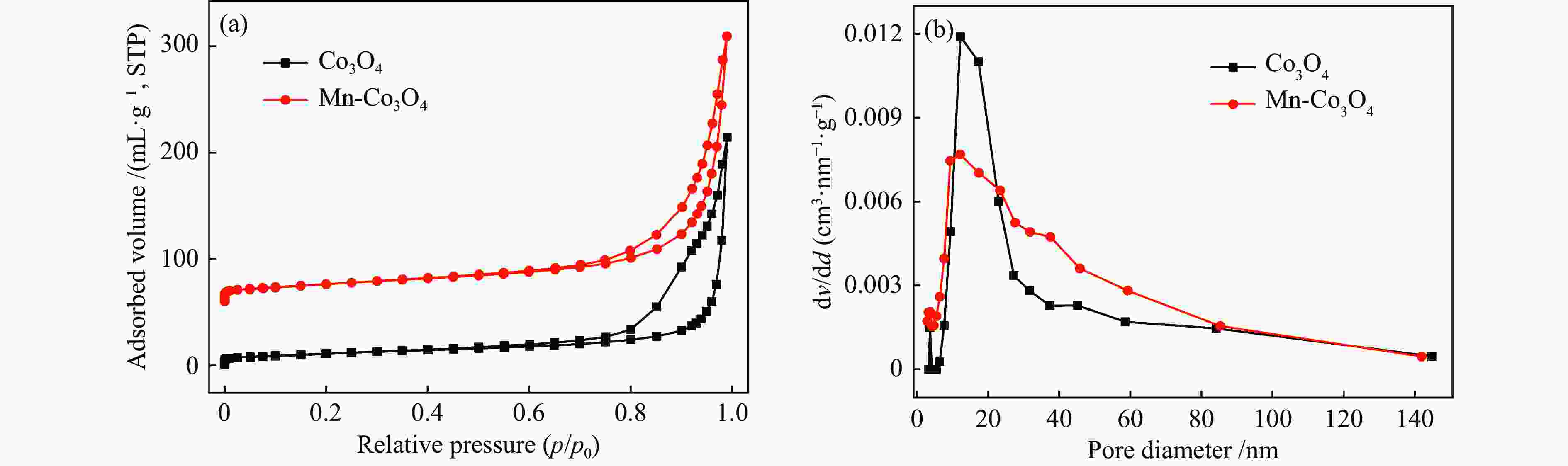
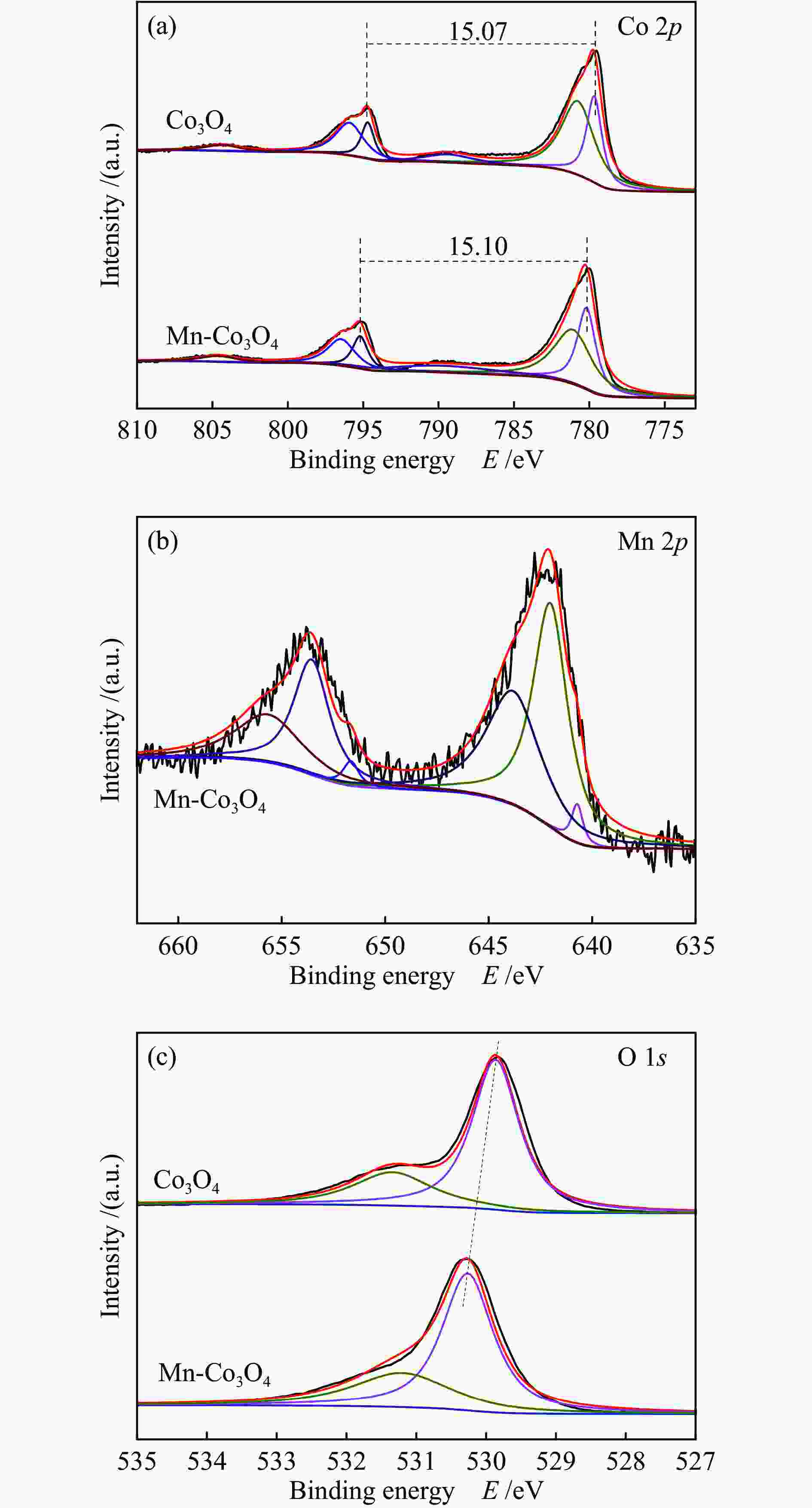
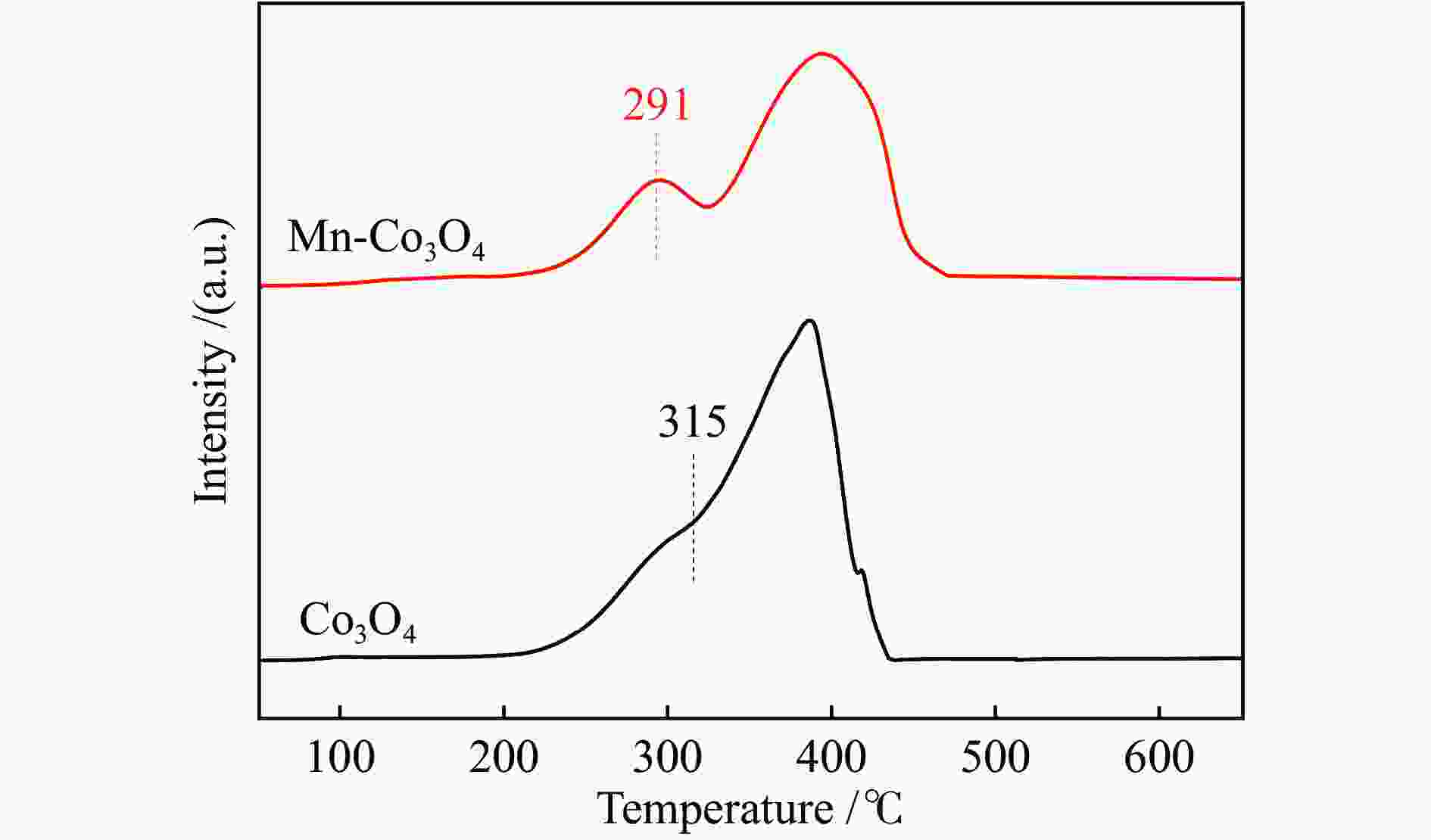
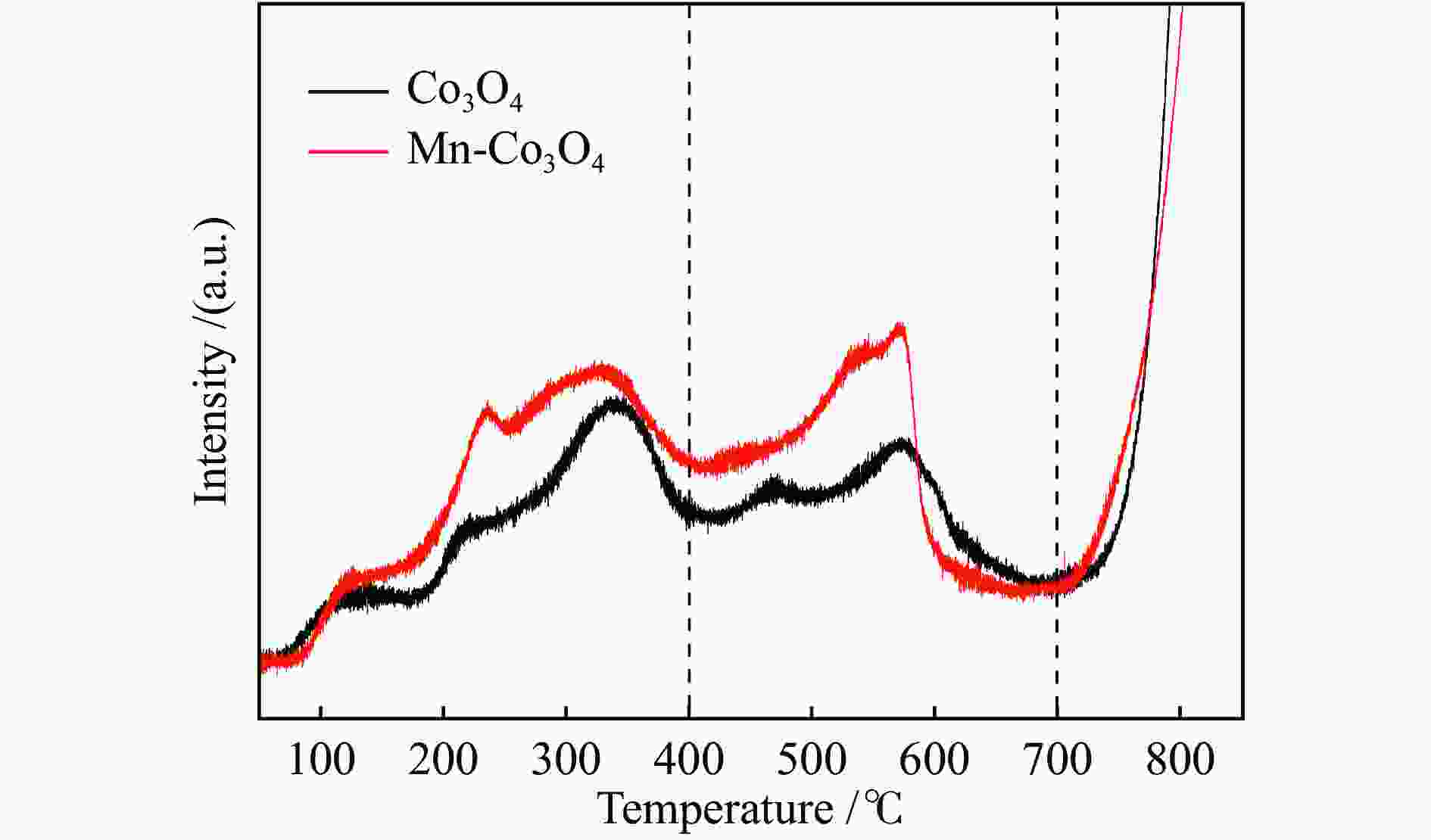
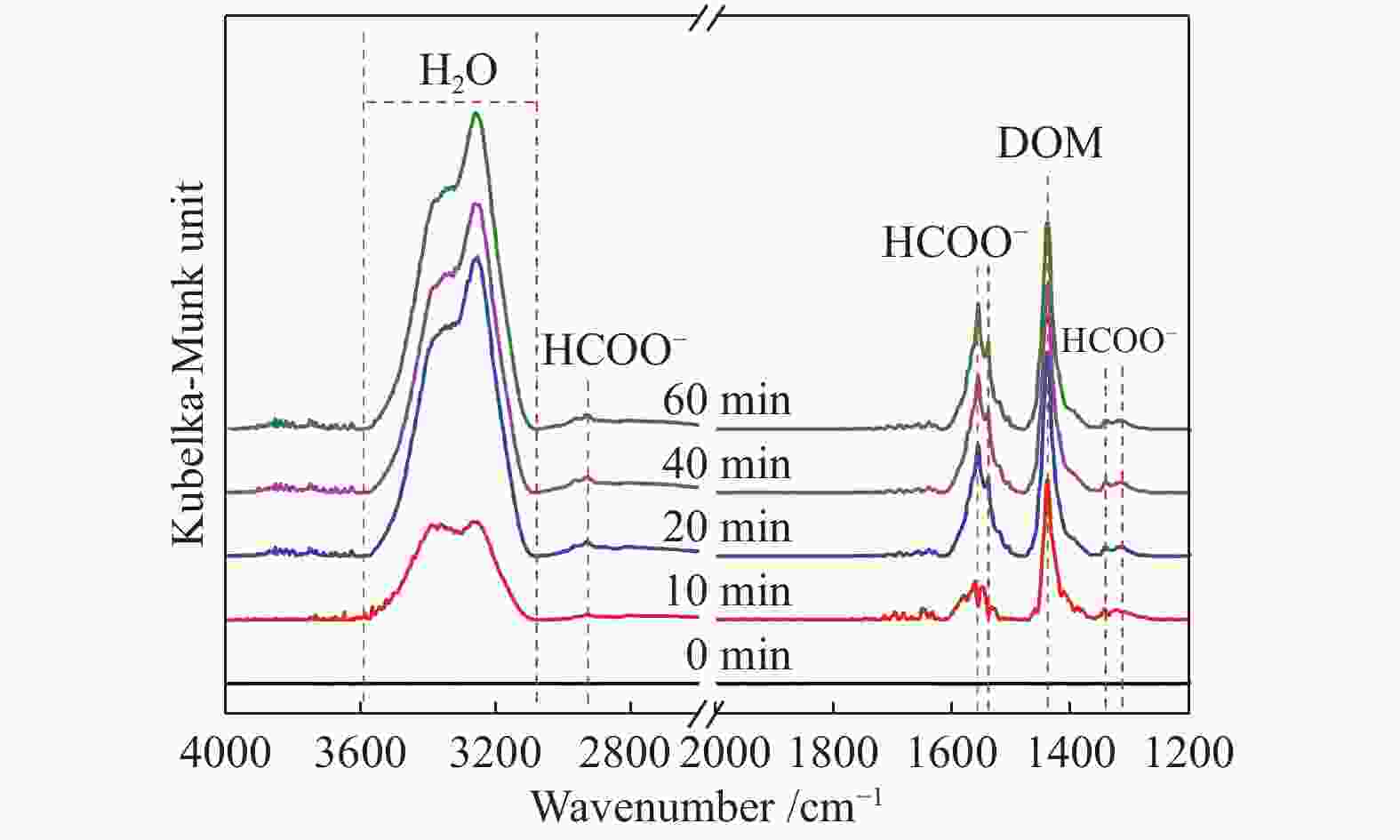
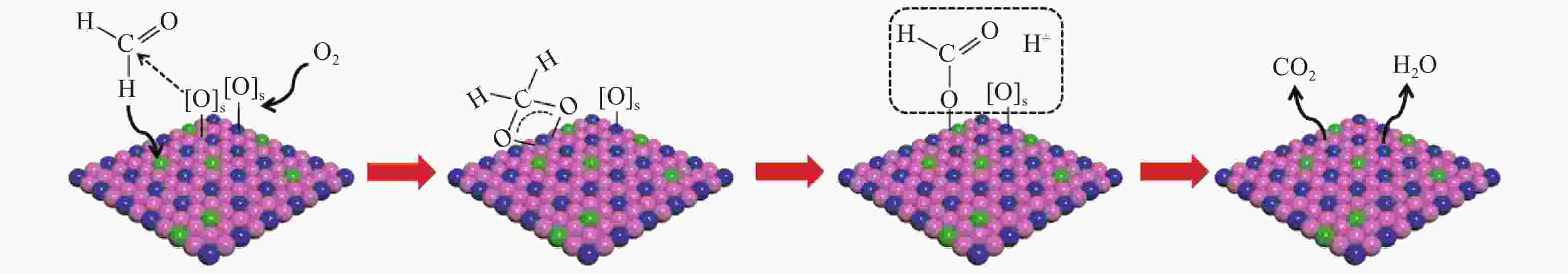
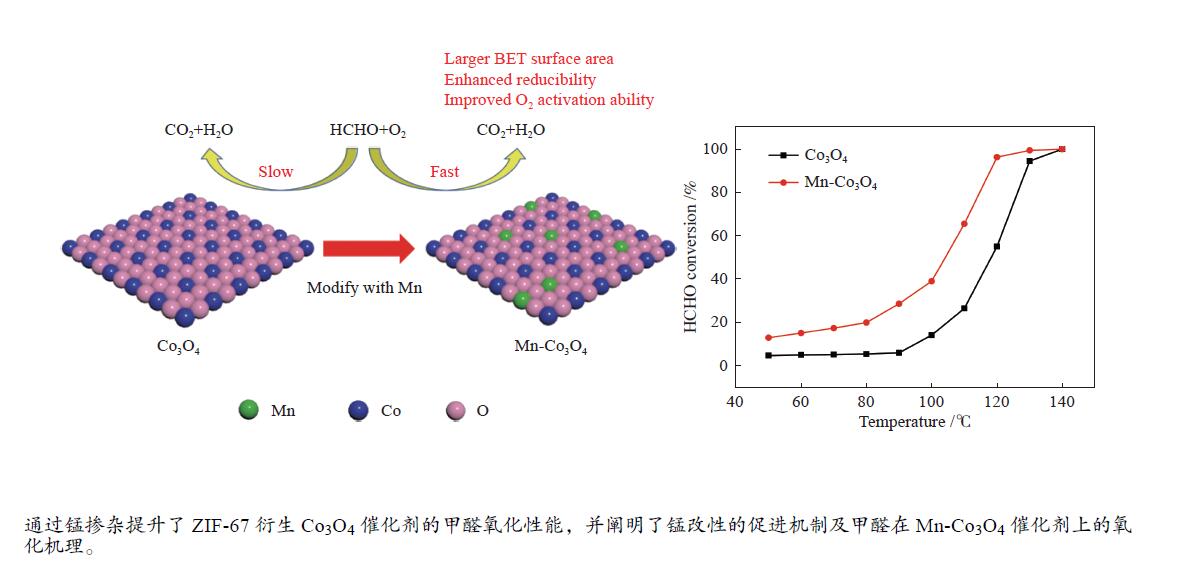
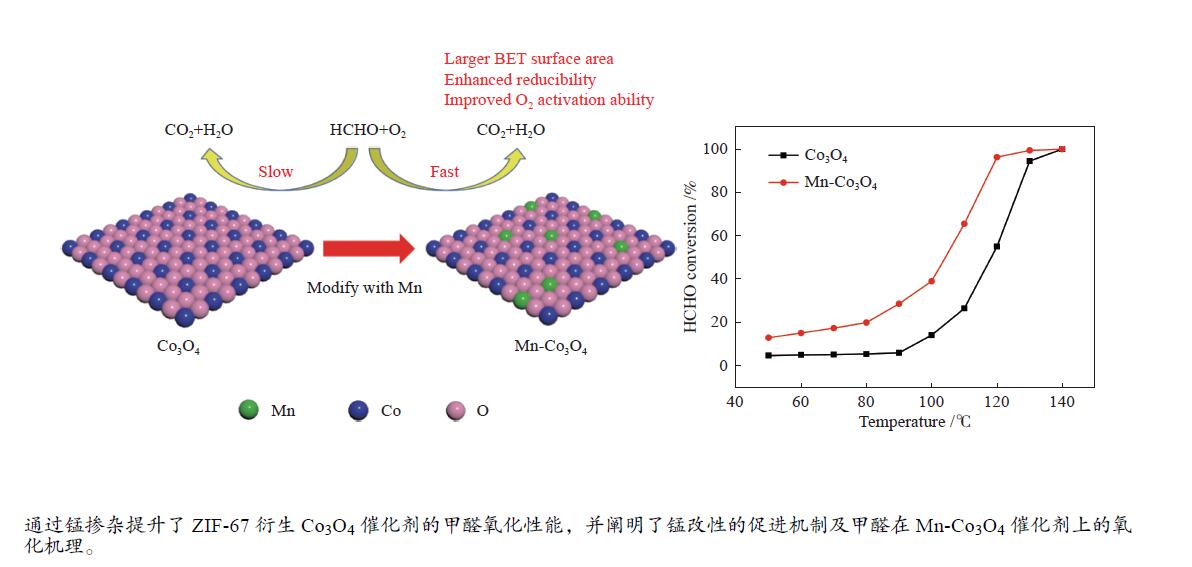
摘要: 针对ZIF-67衍生Co3O4催化剂低温甲醛氧化性能不佳的问题,采用锰(Mn)对Co3O4催化剂进行改性以提升其低温甲醛氧化性能。活性评价结果表明,相比于未改性的Co3O4催化剂,Mn改性后的Mn-Co3O4催化剂甲醛氧化活性显著提升,在118 ℃下即可实现90%的甲醛转化率(进口甲醛浓度为98.16 mg/m3,空速为60000 mL /(gcat·h))。XRD、Raman和BET结果显示,Mn改性后催化剂的结晶度降低,缺陷增加,比表面积增大,这有利于反应物分子的吸附和活性位点的暴露。XPS、H2-TPR和O2-TPD表征结果表明,Mn-Co间存在的强相互作用显著改善了Mn-Co3O4催化剂的低温氧化还原性能和氧活化能力,使其具有更加丰富的Co3+和表面吸附氧物种。最终,这些因素共同促进了Mn-Co3O4催化剂对甲醛的降解。此外,in-situ DRIFTS结果表明,亚甲二氧基和甲酸盐物种是甲醛在Mn-Co3O4催化剂上催化氧化的主要中间物种。Abstract: In consideration of the inferior performance of ZIF-67 derived Co3O4 catalyst in the low-temperature formaldehyde oxidation, manganese was utilized to modify Co3O4 catalyst. The results showed that the Mn-Co3O4 catalyst exhibited the superior HCHO oxidation activity and achieved 90% HCHO conversion at a WHSV of 60000 mL/(gcat·h) and inlet HCHO concentration of 98.16 mg/m3 at 118 ℃. XRD, Raman and BET results demonstrated that the Mn-Co3O4 catalyst possessed lower crystallinity, more defects and specific surface area, which was conducive to the adsorption of reactants and exposure of more active sites. XPS, H2-TPR and O2-TPD results indicated that the strong interaction between Mn and Co species prominently improved the low temperature reducibility and O2 activation performance of Mn-Co3O4 catalyst, which endowed it with more abundant Co3+ and surface-adsorbed oxygen species. Therefore, the Mn-Co3O4 catalyst exhibited superior HCHO oxidation performance. Based on in-situ DRIFTS results, dioxymethylene and formate species were recognized as the main reaction intermediates of HCHO oxidation over the Mn-Co3O4 catalyst.
-
Key words:
- formaldehyde /
- catalytic oxidation /
- ZIF-67 /
- Mn-Co oxide
-
表 1 Co3O4和Mn-Co3O4催化剂的结构参数
Table 1 Properties of Co3O4 and Mn-Co3O4 catalysts
Sample Crystallite size /nm Lattice parameter /Å SBET /(m2·g−1) Pore volume /(cm3·g−1) Average pore diameter /nm Co3O4 21.5 8.073 39.75 0.33 33.38 Mn-Co3O4 16.4 8.085 59.38 0.39 26.00 表 2 Co3O4和Mn-Co3O4催化剂的XPS表征和表面吸附氧脱附量
Table 2 XPS results and desorption amounts of surface-adsorbed oxygen of Co3O4 and Mn-Co3O4 catalysts
Sample Co3+/Co2+ Mn4+/Mn Oα/Oβ Desorption amounts
of surface-adsorbed
oxygen /(μmol·g−1)Co3O4 0.55 − 0.36 140.23 Mn-Co3O4 0.74 0.43 0.48 168.80 -
[1] 张长斌. 室内空气污染物催化氧化研究[J]. 环境化学,2015,34(5):817−823. doi: 10.7524/j.issn.0254-6108.2015.05.2015010509ZHANG Chang-bin. Study of catalytic oxidation of indoor air pollutants[J]. Environ Chem,2015,34(5):817−823. doi: 10.7524/j.issn.0254-6108.2015.05.2015010509 [2] SALTHAMMER T, MENTESE S, MARUTZKY R. Formaldehyde in the indoor environment[J]. Chem Rev,2010,110(4):2536−2572. doi: 10.1021/cr800399g [3] TANG X J, BAI Y, DUONG A, SMITH M T, LI L Y, ZHANG L P. Formaldehyde in China: Production, consumption, exposure levels, and health effects[J]. Environ Int,2009,35(8):1210−1224. doi: 10.1016/j.envint.2009.06.002 [4] LI R, HUANG Y, ZHU D D, HO W K, LEE S C, CAO J J. A review of Co3O4-based catalysts for formaldehyde oxidation at low temperature: Effect parameters and reaction mechanism[J]. Aerosol Sci Eng,2020,4:147−168. doi: 10.1007/s41810-020-00065-3 [5] NIE L H, YU J G, JARONIEC M, TAO F F. Room-temperature catalytic oxidation of formaldehyde on catalysts[J]. Catal Sci Technol,2016,6(10):3649−3669. [6] YE J W, YU Y, FAN J J, CHENG B, YU J G, HO W K. Room-temperature formaldehyde catalytic decomposition[J]. Environ Sci: Nano,2020,7:3655−3709. doi: 10.1039/D0EN00831A [7] GUO J H, LIN C X, JIANG C J, ZHANG P Y. Review on noble metal-based catalysts for formaldehyde oxidation at room temperature[J]. Appl Surf Sci,2019,475:237−255. doi: 10.1016/j.apsusc.2018.12.238 [8] BAI B Y, QIAO Q, LI J H, HAO J M. Progress in research on catalysts for catalytic oxidation of formaldehyde[J]. Chin J Catal,2016,37(1):102−122. doi: 10.1016/S1872-2067(15)61007-5 [9] ZHANG C B, HE H, TANAKA K. Catalytic performance and mechanism of a Pt/TiO2 catalyst for the oxidation of formaldehyde at room temperature[J]. Appl Catal B: Environ,2006,65:37−43. doi: 10.1016/j.apcatb.2005.12.010 [10] LI Y B, ZHANG C B, MA J Z, CHEN M, DENG H, HE H. High temperature reduction dramatically promotes Pd/TiO2 catalyst for ambient formaldehyde oxidation[J]. Appl Catal B: Environ,2017,217:560−569. doi: 10.1016/j.apcatb.2017.06.023 [11] XIANG N, HOU Y Q, HAN X J, LI Y L, GUO Y P, LIU Y J, HUANG Z G. Promoting effect and mechanism of alkali Na on Pd/SBA-15 for room temperature formaldehyde catalytic oxidation[J]. ChemCatChem,2019,11:5098−5107. doi: 10.1002/cctc.201901039 [12] 崔维怡, 王希越, 谭乃迪. 热处理温度对Pt/TiO2纳米带复合物催化HCHO氧化性能的影响[J]. 燃料化学学报,2021,49(11):1701−1708. doi: 10.1016/S1872-5813(21)60113-5CUI Wei-yi, WANG Xi-yue, TAN Nai-di. Effect of thermal treatment temperature on catalytic performance of Pt/TiO2 nanobelt composite for HCHO oxidation[J]. J Fuel Chem Technol,2021,49(11):1701−1708. doi: 10.1016/S1872-5813(21)60113-5 [13] CHEN B B, SHI C, CROCKER M, WANG Y, ZHU A M. Catalytic removal of formaldehyde at room temperature over supported gold catalysts[J]. Appl Catal B: Environ,2013,132−133:245−255. doi: 10.1016/j.apcatb.2012.11.028 [14] LI Y B, CHEN X Y, WANG C Y, ZHANG C B, HE H. Sodium enhances Ir/TiO2 activity for catalytic oxidation of formaldehyde at ambient temperature[J]. ACS Catal,2018,8(12):11377−11385. doi: 10.1021/acscatal.8b03026 [15] LU S H, WANG F, CHEN C C, HUANG F L, LI K L. Catalytic oxidation of formaldehyde over CeO2-Co3O4 catalysts[J]. J Rare Earth,2017,35(9):867−874. doi: 10.1016/S1002-0721(17)60988-8 [16] LU S H, LI K L, HUANG F L, CHEN C C, SUN B. Efficient MnOx-Co3O4-CeO2 catalysts for formaldehyde elimination[J]. Appl Surf Sci,2017,400:277−282. doi: 10.1016/j.apsusc.2016.12.207 [17] 唐彤, 卫广程, 王慧敏, 刘墨, 张秋林, 宁平. Ce/Co比对Ag/CeO2-Co3O4催化剂低温氧化降解甲醛性能的影响[J]. 燃料化学学报,2019,47(5):590−597. doi: 10.3969/j.issn.0253-2409.2019.05.010TANG Tong, WEI Guang-cheng, WANG Hui-min, LIU Mo, ZHANG Qiu-lin, NING Ping. Effect of Ce/Co ratio on the catalytic performance of Ag/CeO2-Co3O4 in the low-temperature oxidation of formaldehyde[J]. J Fuel Chem Technol,2019,47(5):590−597. doi: 10.3969/j.issn.0253-2409.2019.05.010 [18] BAI B Y, ARANDIYAN H, LI J H. Comparison of the performance for oxidation of formaldehyde on nano-Co3O4, 2D-Co3O4, and 3D-Co3O4 catalysts[J]. Appl Catal B: Environ,2013,142−143:677−683. doi: 10.1016/j.apcatb.2013.05.056 [19] ZHANG Q H, LIU X H, FAN W Q, WANG Y. Manganese-promoted cobalt oxide as efficient and stable non-noble metal catalyst for preferential oxidation of CO in H2 stream[J]. Appl Catal B: Environ,2011,102:207−214. doi: 10.1016/j.apcatb.2010.11.043 [20] DU X Y, LI C T, ZHAO L K, ZHANG J, GAO L, SHENG J J, YI Y Y, CHEN J Q, ZENG G M. Promotional removal of HCHO from simulated flue gas over Mn-Fe oxides modified activated coke[J]. Appl Catal B: Environ,2018,232:37−48. doi: 10.1016/j.apcatb.2018.03.034 [21] DU X Y, LI C T, ZHANG J, ZHAO L K, LI S H, LYU Y, ZHANG Y D, ZHU Y C, HUANG L. Highly efficient simultaneous removal of HCHO and elemental mercury over Mn-Co oxides promoted Zr-AC samples[J]. J Hazard Mater,2021,408:124830. doi: 10.1016/j.jhazmat.2020.124830 [22] CAI T, HUANG H, DENG W, DAI Q G, LIU W, WANG X Y. Catalytic combustion of 1, 2-dichlorobenzene at low temperature over Mn-modified Co3O4 catalysts[J]. Appl Catal B: Environ,2015,166−167:393−405. doi: 10.1016/j.apcatb.2014.10.047 [23] SHI C, WANG Y, ZHU A M, CHEN B B, AU C T. MnxCo3−xO4 solid solution as high-efficient catalysts for low-temperature oxidation of formaldehyde[J]. Catal Commun,2012,28:18−22. doi: 10.1016/j.catcom.2012.08.003 [24] ZHANG L, SHI L Y, HUANG L, ZHANG J P, GAO R H, ZHANG D S. Rational design of high-performance deNOx catalysts based on MnxCo3−xO4 nanocages derived from metal-organic frameworks[J]. ACS Catal,2014,4(6):1753−1763. doi: 10.1021/cs401185c [25] GUAN B Y, YU X Y, WU H B, LOU X W. Complex nanostructures from materials based on metal–organic frameworks for electrochemical energy storage and conversion[J]. Adv Mater,2017,29:1703614. doi: 10.1002/adma.201703614 [26] JIANG Z, LI Z P, QIN Z H, SUN H Y, JIAO X L, CHEN D R. LDH nanocages synthesized with MOF templates and their high performance as supercapacitors[J]. Nanoscale,2013,5:11770−11775. doi: 10.1039/c3nr03829g [27] ZHANG P, GUAN B Y, YU L, LOU X W. Facile synthesis of multi-shelled ZnS-CdS cages with enhanced photoelectrochemical performance for solar energy conversion[J]. Chem,2018,4(1):162−173. doi: 10.1016/j.chempr.2017.10.018 [28] ZHAO J H, TANG Z C, DONG F, ZHANG J Y. Controlled porous hollow Co3O4 polyhedral nanocages derived from metal-organic frameworks (MOFs) for toluene catalytic oxidation[J]. Mol Catal,2019,463:77−86. doi: 10.1016/j.mcat.2018.10.020 [29] XU W J, CHEN X, CHEN J, JIA H P. Bimetal oxide CuO/Co3O4 derived from Cu ions partly-substituted framework of ZIF-67 for toluene catalytic oxidation[J]. J Hazard Mater,2021,403:123869. doi: 10.1016/j.jhazmat.2020.123869 [30] TU S, CHEN Y, ZHANG X Y, YAO J Z, WU Y, WU H X, ZHANG J Y, WANG J L, MU B, LI Z, XIA Q B. Complete catalytic oxidation of formaldehyde at room temperature on MnxCo3−xO4 catalysts derived from metal-organic frameworks[J]. Appl Catal A: Gen,2021,611:117975. doi: 10.1016/j.apcata.2020.117975 [31] 李安明, 卫广程, 郝乔慧, 赵宾, 张秋林. Mn含量对CeO2-ZrO2-MnOx催化剂甲苯氧化净化性能的影响[J]. 燃料化学学报,2020,48(2):231−239. doi: 10.3969/j.issn.0253-2409.2020.02.013LI An-ming, WEI Guang-cheng, HAO Qiao-hui, ZHAO Bin, ZHANG Qiu-lin. Effect of Mn content on the catalytic performance of CeO2-ZrO2-MnOx in the oxidation of toluene[J]. J Fuel Chem Technol,2020,48(2):231−239. doi: 10.3969/j.issn.0253-2409.2020.02.013 [32] ZHAO W T, ZHANG Y Y, WU X W, ZHAN Y Y, WANG X Y, AU C T, JIANG L L. Synthesis of Co-Mn oxides with double-shelled nanocages for low-temperature toluene combustion[J]. Catal Sci Technol,2018,8:4494−4502. doi: 10.1039/C8CY01206G [33] DONG C, QU Z P, QIN Y, FU Q, SUN H C, DUAN X X. Revealing the highly catalytic performance of spinel CoMn2O4 for toluene oxidation: involvement and replenishment of oxygen species using in situ designed-TP techniques[J]. ACS Catal,2019,9(8):6698−6710. doi: 10.1021/acscatal.9b01324 [34] SINHA A K, SUZUKI K, TAKAHARA M, AZUMA H, NONAKA T, SUZUKI N, TAKAHASHI N. Preparation and characterization of mesostructured γ-manganese oxide and its application to VOCs elimination[J]. J Phys Chem C,2008,112:16028−16035. doi: 10.1021/jp805211z [35] ZHENG Y L, WANG W Z, JIANG D, ZHANG L. Amorphous MnOx modified Co3O4 for formaldehyde oxidation: improved low-temperature catalytic and photothermocatalytic activity.[J]. Chem Eng J,2016,284:21−27. doi: 10.1016/j.cej.2015.08.137 [36] HAN D W, MA X Y, YANG X Q, XIAO M L, SUN H, MA L J, YU X L, GE M F. Metal organic framework-templated fabrication of exposed surface defect-enriched Co3O4 catalysts for efficient toluene oxidation[J]. J Colloid Interf Sci,2021,603:695−705. doi: 10.1016/j.jcis.2021.06.139 [37] XIANG N, HAN X J, BAI Y R, LI Q Y, ZHENG J F, LI Y L, HOU Y Q, HUANG Z G. Size effect of γ-Al2O3 supports on the catalytic performance of Pd/γ-Al2O3 catalysts for HCHO oxidation[J]. Mol Catal,2020,494:111112. doi: 10.1016/j.mcat.2020.111112 [38] CHEN D, QU Z P, SUN Y H, GAO K, WANG Y. Identification of reaction intermediates and mechanism responsible for highly active HCHO oxidation on Ag/MCM-41 catalysts[J]. Appl Catal B: Environ,2013,142−143:838−848. doi: 10.1016/j.apcatb.2013.06.025 [39] ZHANG C B, LIU F D, ZHAI Y P, ARIGA H, YI N, LIU Y C, ASAKURA K, FLYTZANI-STEPHANOPOULOS M, HE H. Alkali-metal promoted Pt/TiO2 opens a more efficient pathway to formaldehyde oxidation at ambient temperatures[J]. Angew Chem Int Ed,2012,51:9628−9632. doi: 10.1002/anie.201202034 -




 下载:
下载: