Effect of Fe modified Zr-based montmorillonites on the pyrolysis behavior of Xinjiang Hefeng coal
-
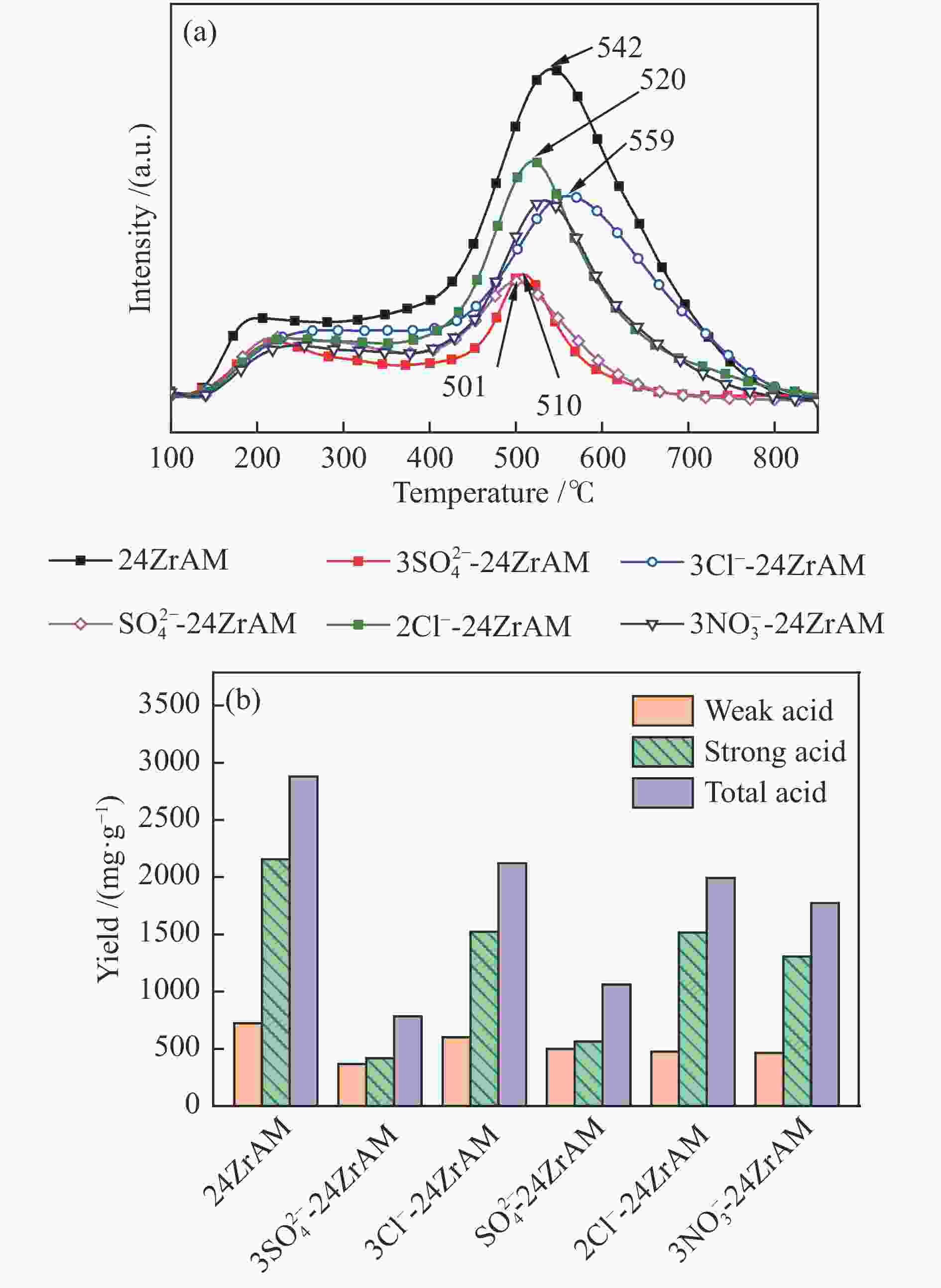
摘要: 采用机械球磨法制备不同Fe盐改性Zr基蒙脱土催化剂,通过X射线衍射仪(XRD)、物理吸附仪(BET)、化学吸附仪(NH3-TPD、H2-TPR)和X射线光电子能谱仪(XPS)分析其组成和结构。结果表明,与24ZrAM相比,以FeCl3·6H2O和FeCl2·4H2O为铁源时,3Cl−-24ZrAM和2Cl−-24ZrAM中生成了Fe–O–Zr结构。Fe的硫酸盐存在时,催化剂的比表面积显著减小。引入Fe后,催化剂的总酸量均降低,3Cl−-24ZrAM的酸量与强酸强度最高。H2-TPR显示,3Cl−-24ZrAM、2Cl−-24ZrAM和
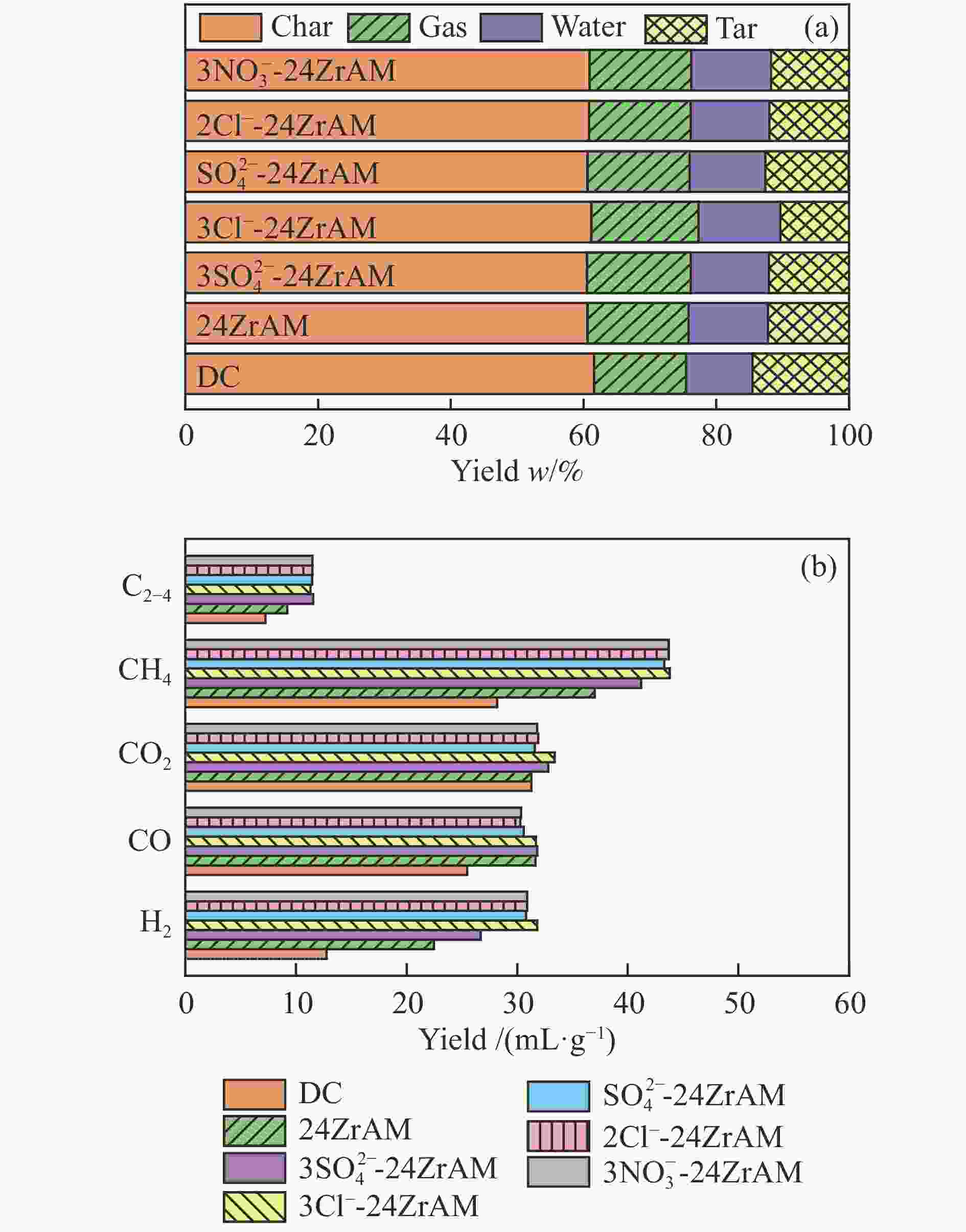
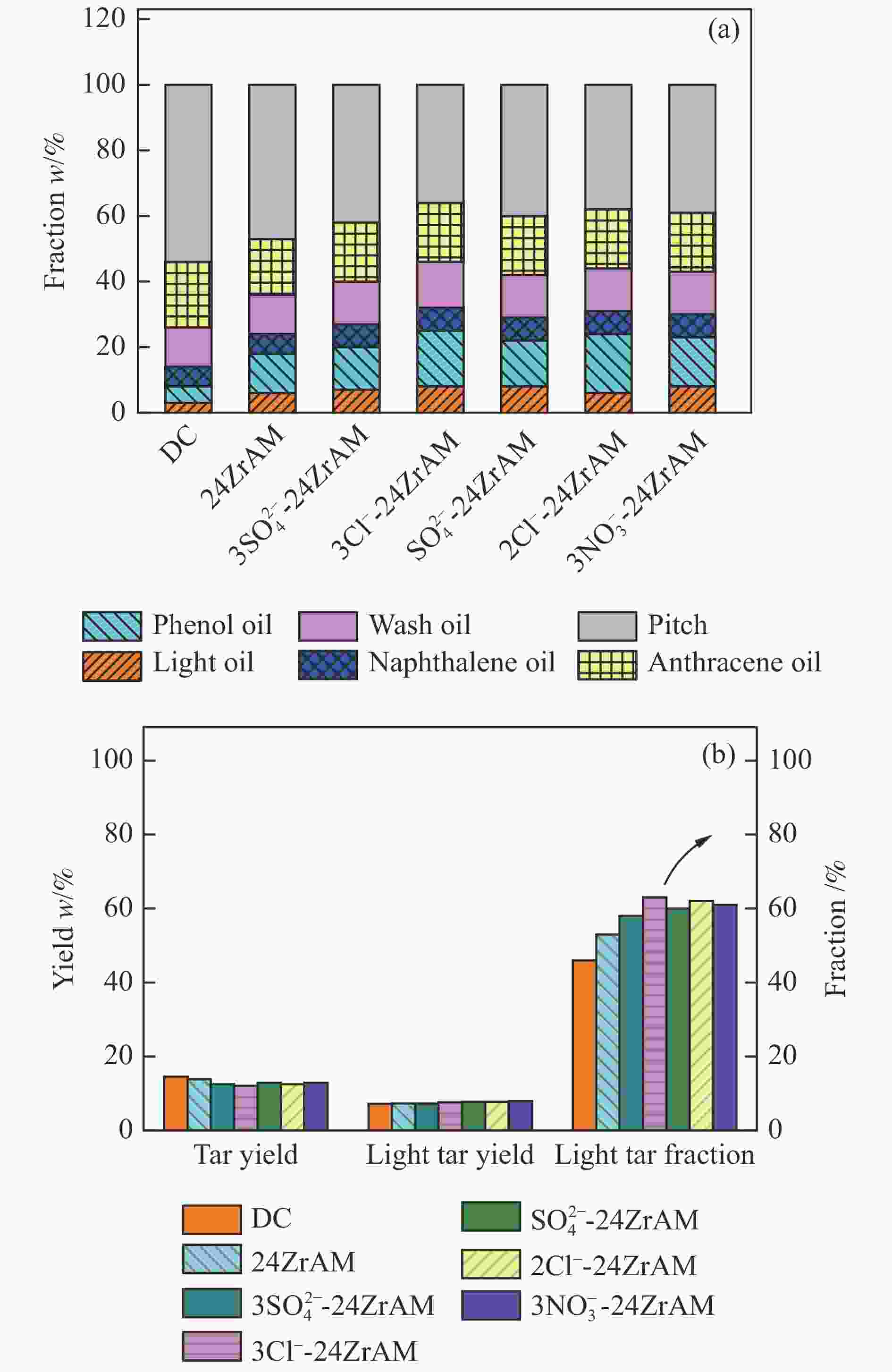
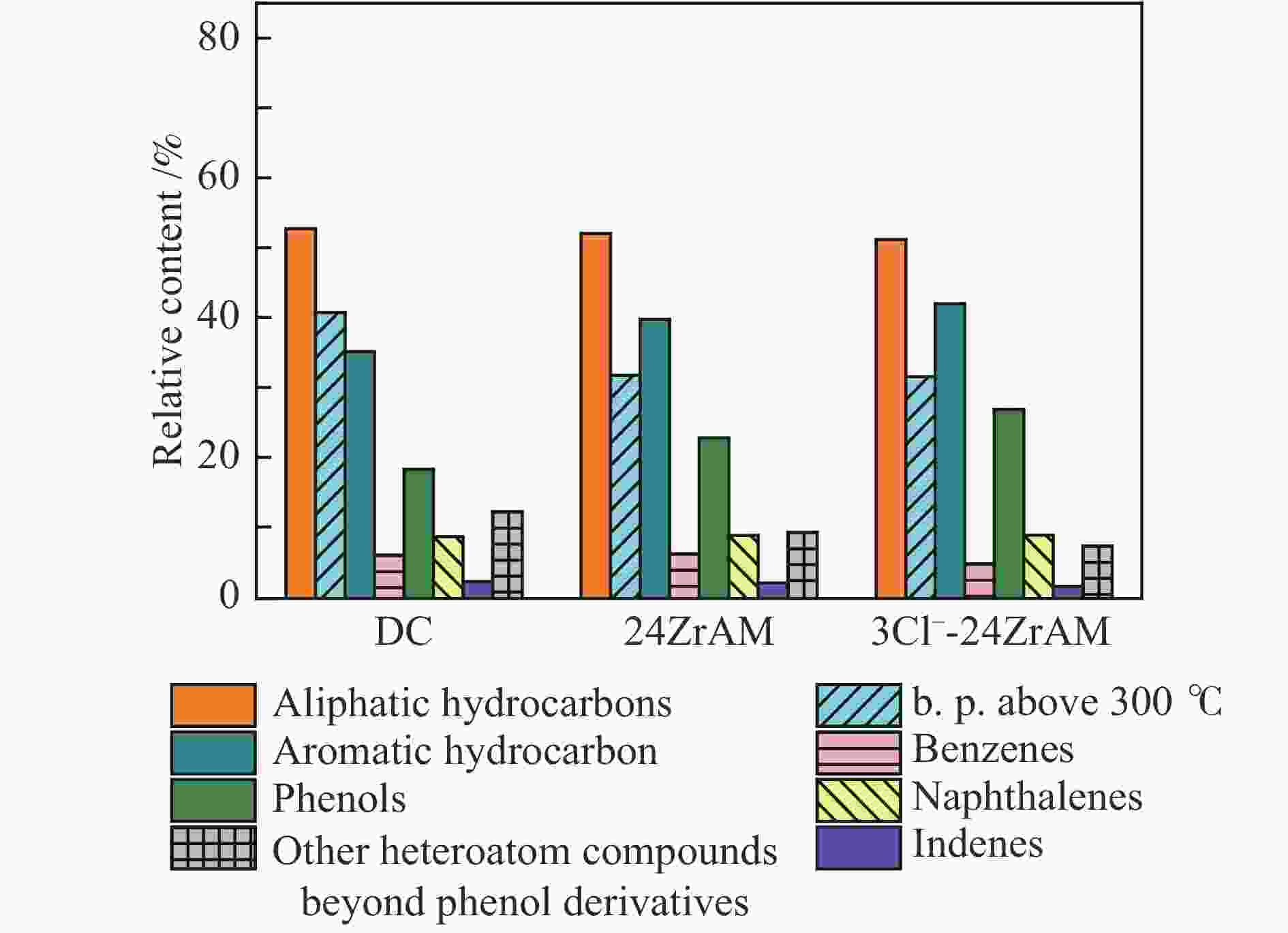
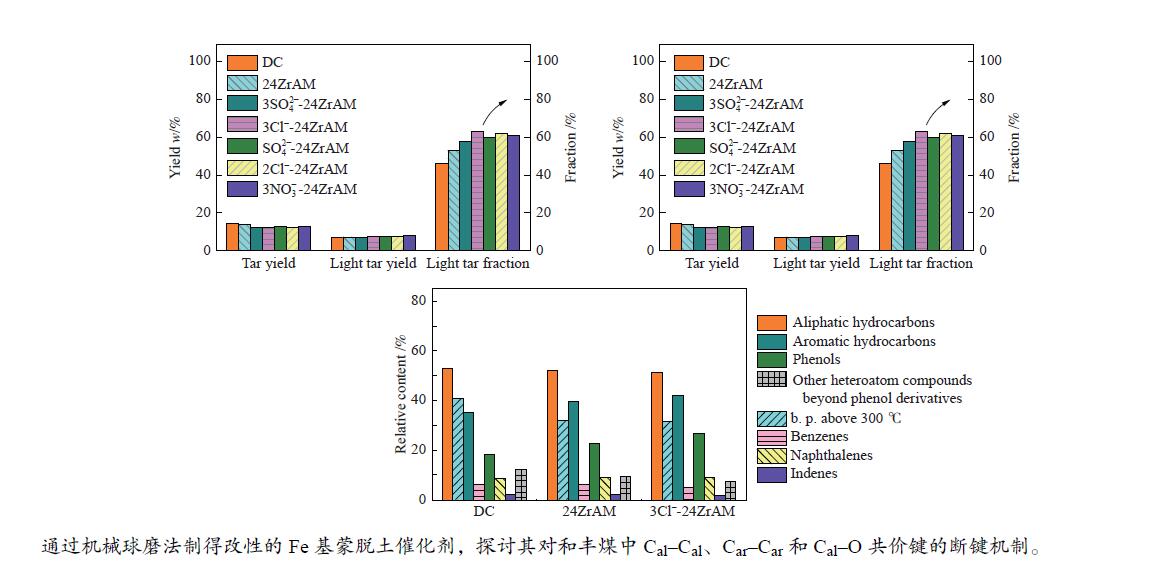
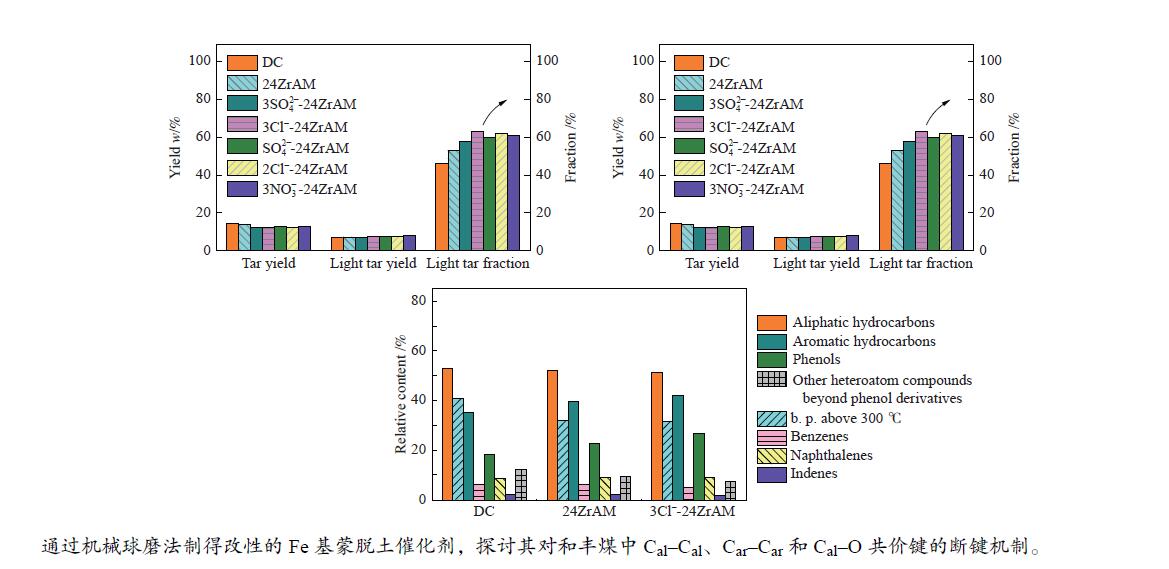
$3{\rm{NO}}_3^ - $ -24ZrAM中Fe2O3还原为Fe3O4的温度均低于500 ℃。在固定床反应器中考察了催化剂对新疆和丰煤热解行为及含不同桥键模型化合物的断键机制的影响。与24ZrAM相比,不同Fe源改性蒙脱土均能降低焦油中沥青的质量分数,其中,3Cl−-24ZrAM的裂解活性最高,轻质焦油分率增加至63%,比24ZrAM提高了18.9%,轻油和酚油质量分数分别为24ZrAM的1.3倍和1.4倍,长链烃含量进一步降低,降幅为0.7%。引入Fe后,苄基苯基醚(BPE)、联苄和联苯的转化率分别比24ZrAM提高了5%、1.6%和43.9%。-
关键词:
- Fe改性Zr基蒙脱土 /
- 新疆和丰煤 /
- 模型化合物 /
- 催化热解 /
- 焦油品质
Abstract: A serial of Fe modified Zr-based montmorillonites were prepared by mechanical ball milling and their characteristics were depicted by X-ray diffractometer (XRD), N2 adsorption-desorption instrument (BET), temperature-programmed desorption of ammonia (NH3-TPD, H2-TPD), X-ray photoelectron spectrometer (XPS). The results show that compared with 24ZrAM, when FeCl3·6H2O and FeCl2·4H2O are used as iron sources, Fe–O–Zr structure appears on the 3Cl−-24ZrAM and 2Cl−-24ZrAM catalysts. In the presence of Fe sulfate, the specific surface area of the catalysts decreases markedly. With the introducing of Fe, the total acid content of catalysts reduces. Wherein, 3Cl−-24ZrAM has the highest acid content and strongest acid strength. H2-TPR shows that the temperature for Fe2O3 reduced to Fe3O4 in 3Cl−-24ZrAM, 2Cl−-24ZrAM and$3{\rm{NO}}_3^ - $ -24ZrAM is lower than 500 ℃. Then, the effect of catalysts on the pyrolysis behavior of Xinjiang Hefeng coal and the bridge bond cleavage mechanism of different model compounds were investigated in a fixed bed reactor. It is noted that compared with 24ZrAM, the fraction of coal tar pitch all declines under the action of Fe species. Among them, 3Cl−-24ZrAM has the highest cracking activity with the light tar fraction of 63%, which is 18.9% higher than that of 24ZrAM. Meanwhile, the content of light oil and phenol oil are 1.3 times and 1.4 times higher than that of 24ZrAM, respectively. As for long-chain hydrocarbons, a further decline by 0.7% is observed. In addition, the conversion rates of benzyl phenyl ether (BPE), dibenzyl and biphenyl increase by 5%, 1.6% and 43.9%, respectively. -
表 1 煤样分析
Table 1 Analysis of coal samples
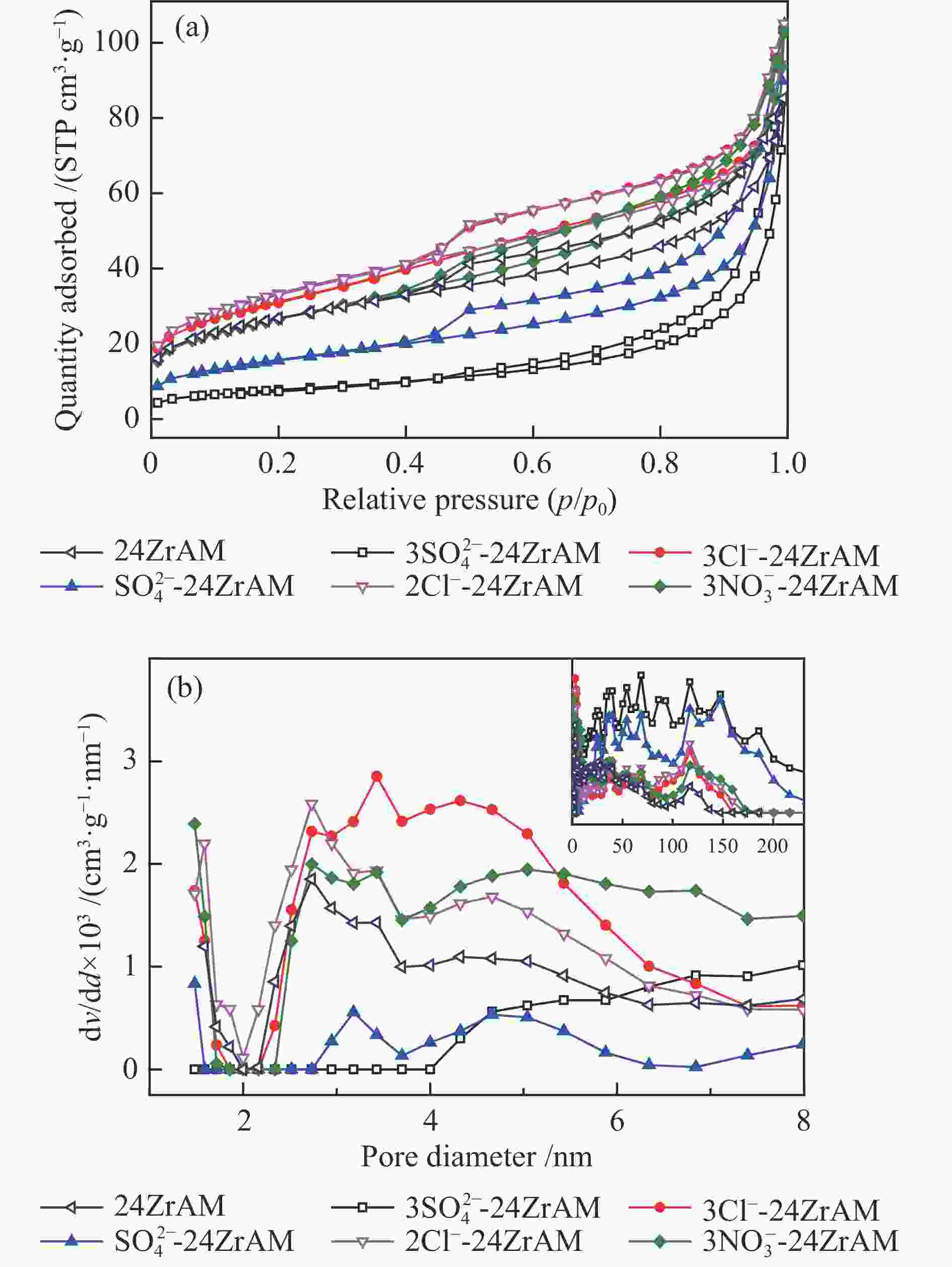
Sample Proximate analysis w/% Ultimate analysis wdaf/% Ad Vdaf FCdaf* C H O* N S Raw coal 22.83 47.47 52.53 72.06 5.64 19.07 3.00 0.23 DC 1.10 44.98 55.02 61.17 4.93 30.86 2.83 0.21 *: by difference 表 2 催化剂的孔结构特征和Fe2O3的粒径
Table 2 Pore structure characteristics of the catalysts and crystallize size of Fe2O3
Sample BET surface area/(m2·g−1) Pore volume/(cm3·g−1) Average pore size/nm Crystallize size/nm 24ZrAM 93 0.12 6.58 ${\rm{3SO}}_4^{2 - } $-24ZrAM 28 0.13 16.75 8.7 3Cl−-24ZrAM 108 0.13 6.19 6.2 ${\rm{SO}}_4^{2 - }$-24ZrAM 56 0.16 11.55 10.2 2Cl−-24ZrAM 117 0.15 6.40 9.5 ${\rm{3NO}}_3^ - $-24ZrAM 94 0.16 7.07 5.2 表 3 联苄热解产物分布
Table 3 Distribution of bibenzyl pyrolysis products
Sample Monomer aromatics content /% Dimeric aromatics content /% BTX styrene other
alkylbenzenesbiphenyl propane-1,2-
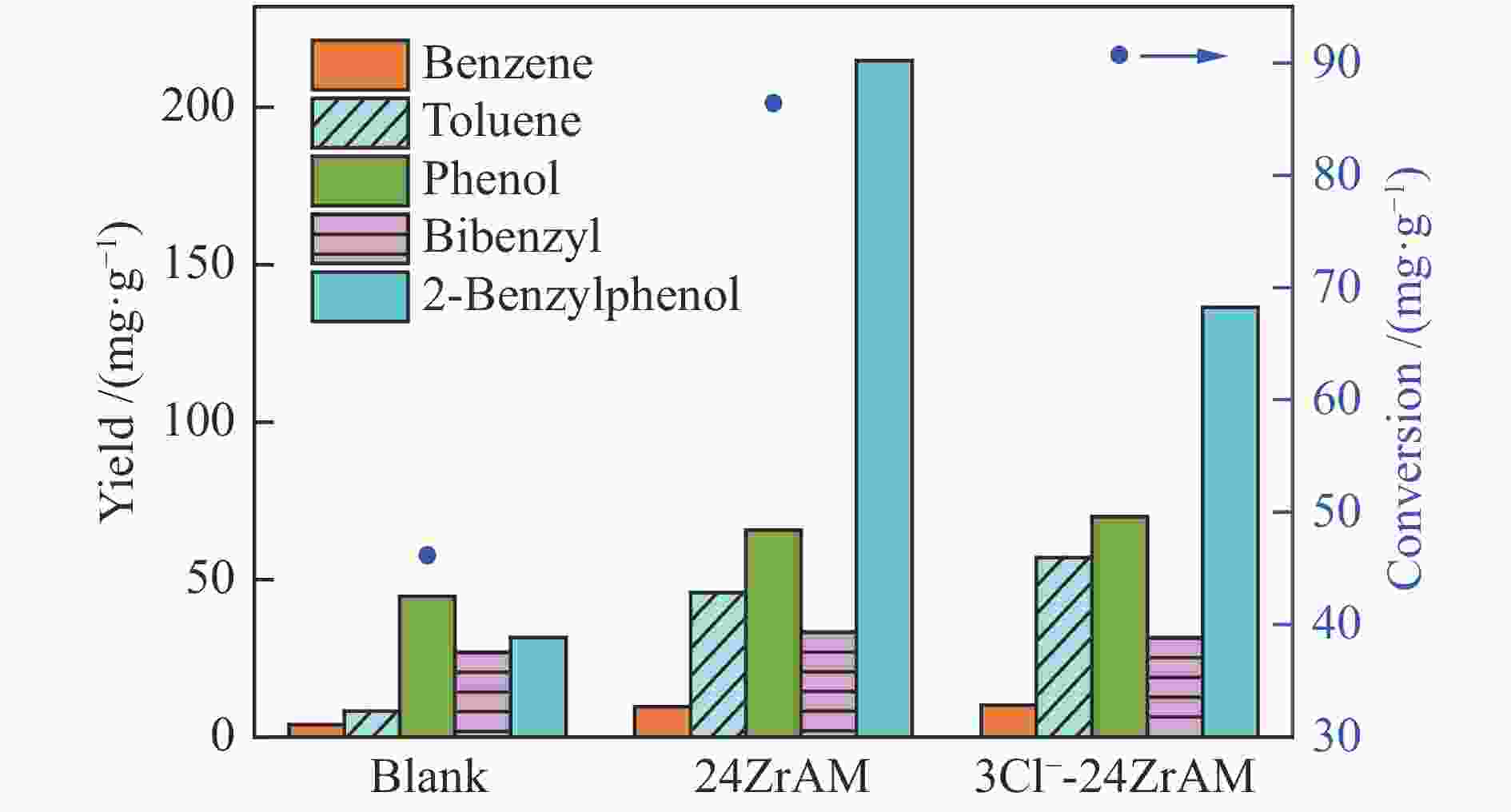
diyldibenzene1,3-diphenylpropane stilbene others Blank 3.7 0.4 0.7 13.2 34.1 30.3 15.7 1.9 24ZrAM 4.0 0.4 11.3 11.8 22.2 23.0 12.1 12.5 3Cl−-24ZrAM 7.3 3.0 19.1 24.9 14.9 16.4 3.8 10.6 表 4 联苯热解产物分布
Table 4 Distribution of biphenyl pyrolysis products
Sample Monomer aromatics content/% Dimeric aromatics content/% BTX other
alkylbenzenesnaphthalene 2-methyl-1,1’-
biphenyl4-methyl-1,1’-
biphenylbibenzyl others Blank 8.1 − 12.7 4.8 15.4 48.3 10.7 24ZrAM 1.1 1.8 0.4 36.2 42.7 1.5 16.3 3Cl−-24ZrAM 4.3 1.7 0.4 32.2 41.8 3.4 16.2 -
[1] 莫海燕, 朱平, 彭文才, 李江兵, 张建树. 石河子烟煤Fenton试剂氧化的研究[J]. 石河子大学学报(自然科学版),2017,35(1):23−27.MO Hai-yan, ZHU Ping, PENG Wen-cai, LI Jiang-bing, ZHANG Jian-shu. Oxidation behaviours of Shihezi bituminous coal by Fenton reagents[J]. J Shihezi Uni: Nat Sci,2017,35(1):23−27. [2] JORDAN C A, AKAY G. Effect of CaO on tar production and dew point depression during gasification of fuel cane bagasse in a novel downdraft gasifier[J]. Fuel Process Technol,2013,106:654−660. doi: 10.1016/j.fuproc.2012.09.061 [3] PARK D K, PH D, SONG E. Pressurized pyrolysis characteristics of two ranks of coal in a thermogravimetric analyzer[J]. J Energy Eng,2017,143(5):04017017. doi: 10.1061/(ASCE)EY.1943-7897.0000450 [4] PRETORIUS G N, BUNT J R, GRABNER M, NEOMAGUS H, WAANDERS F B, EVERSON R C, STRYDOM C A. Evaluation and prediction of slow pyrolysis products derived from coals of different rank[J]. J Anal Appl Pyrolysis,2017,128:156−167. doi: 10.1016/j.jaap.2017.10.014 [5] GAO S P, ZHAI L R, QIN Y H, WANG Z Q, ZHAO J T, FANG Y T. Investigation into the cleavage of chemical bonds induced by CO2 and its mechanism during the pressurized pyrolysis of coal[J]. Energy Fuels,2018,32(3):3243−3253. doi: 10.1021/acs.energyfuels.7b03950 [6] WANG H J, FENG Y H, ZHANG X X, LIU W, ZHAO Y L. Study of coal hydropyrolysis and desulfurization by ReaxFF molecular dynamics simulation[J]. Fuel,2015,145:241−248. doi: 10.1016/j.fuel.2014.12.074 [7] XU K, HU S, ZHANG L P, LI H J, CHEN Y F, XIONG Z, XU J, JIANG L, WANG Y, SU S, XIANG J. Effect of temperature on Shenfu coal pyrolysis process related to its chemical structure transformation[J]. Fuel Process Technol,2021,213:106662. doi: 10.1016/j.fuproc.2020.106662 [8] LI Q, WANG Z H, HE Y, SUN Q, ZHANG Y W, KUMAR S, ZHANG K, CEN K F. Pyrolysis characteristics and evolution of char structure during pulverized coal pyrolysis in drop tube furnace: Influence of temperature[J]. Energy Fuels,2017,31(5):4799−4807. doi: 10.1021/acs.energyfuels.7b00002 [9] WANG G J, HOU B L, ZHANG J, WANG H, GAO D Y, CHANG G Z, THALLADA B. Effect of pressure and H2 on the pyrolysis characteristics of lignite: Thermal behavior and coal char structural properties[J]. J Anal Appl Pyrolysis,2018,135:1−9. doi: 10.1016/j.jaap.2018.10.003 [10] ZHANG W D, ZHAO Y J, SUN S Z, FENG D D, LI P F. Effects of pressure on the characteristics of bituminous coal pyrolysis char formed in a pressurized drop tube furnace[J]. Energy Fuels,2019,33(12):12219−12226. doi: 10.1021/acs.energyfuels.9b02774 [11] FU Y, GUO Y H, ZHANG K X. Effect of three different catalysts (KCl, CaO, and Fe2O3) on the reactivity and mechanism of low-rank coal pyrolysis[J]. Energy Fuels,2016,30(3):2428−2433. doi: 10.1021/acs.energyfuels.5b02720 [12] LIU S Q, TUO K Y, WANG L P, CHEN G, MA W P, FANG M Y. Microwave-assisted metal-catalyzed pyrolysis of low-rank coal: Promising option towards obtaining high-quality products[J]. J Energy Inst,2020,93(4):1602−1614. doi: 10.1016/j.joei.2020.01.022 [13] XU L, TANG M C, DUAN L E, LIU B L, MA X X, ZHANG Y L, ARGYLE M D, FAN M H. Pyrolysis characteristics and kinetics of residue from China Shenhua industrial direct coal liquefaction plant[J]. Thermochim Acta, 2014, 589: 1–10. [14] AMIN M N, LI Y, RAZZAQ R, LU X M, LI C S, ZHANG S J. Pyrolysis of low rank coal by nickel based zeolite catalysts in the two-staged bed reactor[J]. J Anal Appl Pyrolysis,2016,118:54−62. doi: 10.1016/j.jaap.2015.11.019 [15] ZHONG M, ZOU D, XU Y B, JIN L J, PAN Y. Effect of kaolinites modified with Zr and transition metals on the pyrolysis behaviors of low-rank coal and its model compound[J]. J Energy Inst,2021,95:41−51. doi: 10.1016/j.joei.2020.11.009 [16] ZHAO H Y, LI Y H, SONG Q, MA Q X, MA L, LIU S C, SHU Xi Q. The rate-limiting step in the integrated coal tar decomposition and upgrading-iron ore reduction reaction determined by kinetic analysis[J]. J Anal Appl Pyrolysis,2020,147:104808. doi: 10.1016/j.jaap.2020.104808 [17] SONG Q, ZHAO H Y, CHANG S Q, LI Y, ZOU F, SHU X Q, ZHANG P. Study on the catalytic pyrolysis of coal volatiles over hematite for the production of light tar[J]. Fuel,2020,151:104927. [18] QI X J, GUO X, XUE L C, ZHENG C G. Effect of iron on Shenfu coal char structure and its influence on gasification reactivity[J]. J Anal Appl Pyrolysis,2014,110:401−407. doi: 10.1016/j.jaap.2014.10.011 [19] FENG J, XUE X Y, LI X H, LI W Y, GUO X F, LIU K. Products analysis of Shendong long-flame coal hydropyrolysis with iron-based catalysts[J]. Fuel Process Technol,2015,130:96−100. doi: 10.1016/j.fuproc.2014.09.035 [20] ZHAO H Y, LI Y H, SONG Q, LV J X, SHU Y F, LIANG X X, SHU Xin-qian. Effects of iron ores on the pyrolysis characteristics of a low-rank bituminous coal[J]. Energy Fuels,2016,30(5):3831−3839. doi: 10.1021/acs.energyfuels.6b00061 [21] HE L, HUI H L, LI S G, LIN W G. Production of light aromatic hydrocarbons by catalytic cracking of coal pyrolysis vapors over natural iron ores[J]. Fuel,2018,216:227−232. doi: 10.1016/j.fuel.2017.12.005 [22] BIESEKI L, BERTELL F, TREICHEL H, PENHA F G PERGHER S B C. Acid treatments of montmorillonite-rich clay for Fe removal using a factorial design method[J]. Mater Res,2013,16(5):1122−1127. doi: 10.1590/S1516-14392013005000114 [23] 钟梅, 赵渊, 李显, 马凤云. K+, Ca2+ 和 Fe3+ 对和丰煤热解产物分布, 结构及品质的影响[J]. 燃料化学学报,2018,46(9):1044−1054. doi: 10.3969/j.issn.0253-2409.2018.09.003ZHONG Mei, ZHAO Yuan, LI Xian, MA Feng-yun. Effects of K+, Ca2+ and Fe3+ on the distribution structure and quality of the pyrolysis products of Hefeng coal[J]. J Fuel Chem Technol,2018,46(9):1044−1054. doi: 10.3969/j.issn.0253-2409.2018.09.003 [24] 石磊. 煤共价键结构在热解过程中的阶段解离研究[D]. 北京: 北京化工大学, 2014.Shi L. Study on the cleavage of covalent bonds in coal pyrolysis from various temperature stages[D]. Beijing: Beijing University of Chemical Technology, 2014. [25] ZOU D, JIN L J, ZHONG M, HU H Q, MA F Y. Catalytic performance of modified kaolinite in pyrolysis of benzyl phenyl ether: A model compound of low rank coal[J]. J Energy Inst,2020,93(6):2314−2324. doi: 10.1016/j.joei.2020.07.003 [26] PETKOVA V, PELOVSKI Y. Investigation on the thermal properties of Fe2O(SO4)2. Part II[J]. J Therm Anal Calorim,2001,64(3):1037−1044. doi: 10.1023/A:1011599719230 [27] LACALAMITA M, VENTRUTI G, DELLA V G, RADICA F, MAURO D, SCHINGARO E. In Situ high-temperature X-ray powder diffraction and infrared spectroscopic study of melanterite, FeSO4·7H2O[J]. Miner,2021,11(4):392. doi: 10.3390/min11040392 [28] MULLER M, VILLALBA J C, ANAISSI J F. Thermal decomposition (TG-DTA) of iron salts [FeCl3·6H2O] and [Fe(NO3)3·9H2O] with morphologic and chemical analysis of final product[J]. Semina: Ciências Exatas e Tecnológicas,2014,35(1):9−14. [29] 车欣, 王志, 公旭中, 郭占成, 王福生. FeCl2强化浸锌渣焙烧反应性的热重解析[J]. 矿冶工程,2010,30(5):74−78. doi: 10.3969/j.issn.0253-6099.2010.05.019CHE Xin, WANG Zhi, GONG Xu-zhong, GUO Zhan-cheng, WANG Fu-sheng. Hermogravimetry analysison roasting reactivity of zinc-leaching residues strengt henedby FeCl2[J]. Mining Metall Eng,2010,30(5):74−78. doi: 10.3969/j.issn.0253-6099.2010.05.019 [30] MASSET P, POINSO J, POIGNET J. TG/DTA/MS study of the thermal decomposition of FeSO4· 6H2O[J]. J Therm Anal Calorim,2006,83(2):457−462. doi: 10.1007/s10973-005-7267-6 [31] ZHONG M, ZHAI J R, XU Y B, JIN L J, YE Y F, HU H Q, MA F Y. Catalytic cracking of coal-tar model compounds over ZrO2/Al2O3 and Ni-Ce/Al2O3 catalysts under steam atmosphere[J]. Fuel,2020,263:116763. doi: 10.1016/j.fuel.2019.116763 [32] PRADHAN S, MISHRA B G. Catalytic application of SO42−/Fe–ZrO2 nanoparticles synthesized by a urea hydrolysis method for environmentally benign one pot synthesis of 1, 8-dioxodecahydroacridines[J]. RSC Adv,2015,5(105):86179−86190. doi: 10.1039/C5RA11136F [33] 管秀丽. 元素掺杂蒙脱石固体酸的制备、表征及其芳烃开环裂解性能[D]. 安徽: 安徽工业大学, 2016.Guan Xiu-li. Preparation, characterization of solid acid of montmorillonite doped with Zr and Ti and their properties of aromatic ring opening and cracking[D]. Anhui: Anhui University of Technology, 2016. [34] LIU Y, YANG X H, YAN C J, WANG H Q, ZHOU S. Solvent-free synthesis of zeolite LTA monolith with hierarchically porous structure from metakaolin[J]. Mater Lett,2019,248:28−31. doi: 10.1016/j.matlet.2019.03.135 [35] YANG H Y, ZHOU Y, TONG D S, YANG M, FANG K, ZHOU C H, YU W H. Catalytic conversion of cellulose to reducing sugars over clay-based solid acid catalyst supported nanosized SO42−-ZrO2[J]. J Catal,2019,185:105376. [36] 晏冬霞, 王华, 李孔斋, 魏永刚, 祝星, 程显名. 铈铁锆三元复合氧化物上碳烟的催化燃烧[J]. 燃料化学学报,2011,39(3):229−235. doi: 10.3969/j.issn.0253-2409.2011.03.013YAN Dong-xia, WANG Hua, LI Kong-zhai, WEI Yong-gang, ZHU Xing, CHENG Xian-ming. Catalytic combustion of soot on Ce–Fe–Zr–O ternary mixed oxide catalysts[J]. J Fuel Chem Technol,2011,39(3):229−235. doi: 10.3969/j.issn.0253-2409.2011.03.013 [37] FANG Z H, HUANG M Y, LIU B, JIANG F, XU Y B, LIU X H. Identifying the crucial role of water and chloride for efficient mild oxidation of methane to methanol over a [Cu2 (μ-O)]2+-ZSM-5 catalyst[J]. J Catal, 2021, 405: 1−14. [38] 刘宗宽, 张磊, 江健, 边城. 煤焦油加氢精制和加氢裂化催化剂的研究进展[J]. 化工进展,2012,31(12):2672−2677.LIU Zong-kuan, ZHANG Lei, JIANG Jian, BIAN Cheng. Advances in coal tar hydro-refining and hydro-cracking catalysts[J]. Chem Ind Eng Prog,2012,31(12):2672−2677. [39] 金其奇, 谢峻林, 李凤祥, 齐凯, 方德, 何峰. 涂层组分对堇青石脱硝催化剂性能的影响[J]. 化工进展,2019,38(3):1411−1418.JIN Qi-qi, XIE Jun-lin, LI Feng-xiang, QI Kai, FANG De, HE Feng. Performance of structured cordierite catalysts with different coatings for NH3-SCR[J]. Chem Ind Eng Prog,2019,38(3):1411−1418. [40] WANG D C, JIN L J, LI Y, HU H Q. Partial oxidation of vacuum residue over Al and Zr-doped α-Fe2O3 catalysts[J]. Fuel,2017,210:803−810. doi: 10.1016/j.fuel.2017.09.008 [41] SONG Q, ZHAO H Y, CHANG S Q, LI Y, ZOU F, SHU X Q, ZHANG P. Study on the catalytic pyrolysis of coal volatiles over hematite for the production of light tar[J]. J Anal Appl Pyrolysis,2020,151:104927. doi: 10.1016/j.jaap.2020.104927 [42] 乐嘉炜. 煤中小分子化合物及催化剂对煤热解产物分布的影响[D]. 上海: 华东理工大学, 2017.LE Jia-wei. Research on the effect of low molecular compounds and catalysts on the coal pyrolysis products distribution[D]. Shanghai: East China University of Science and Technology, 2017. -






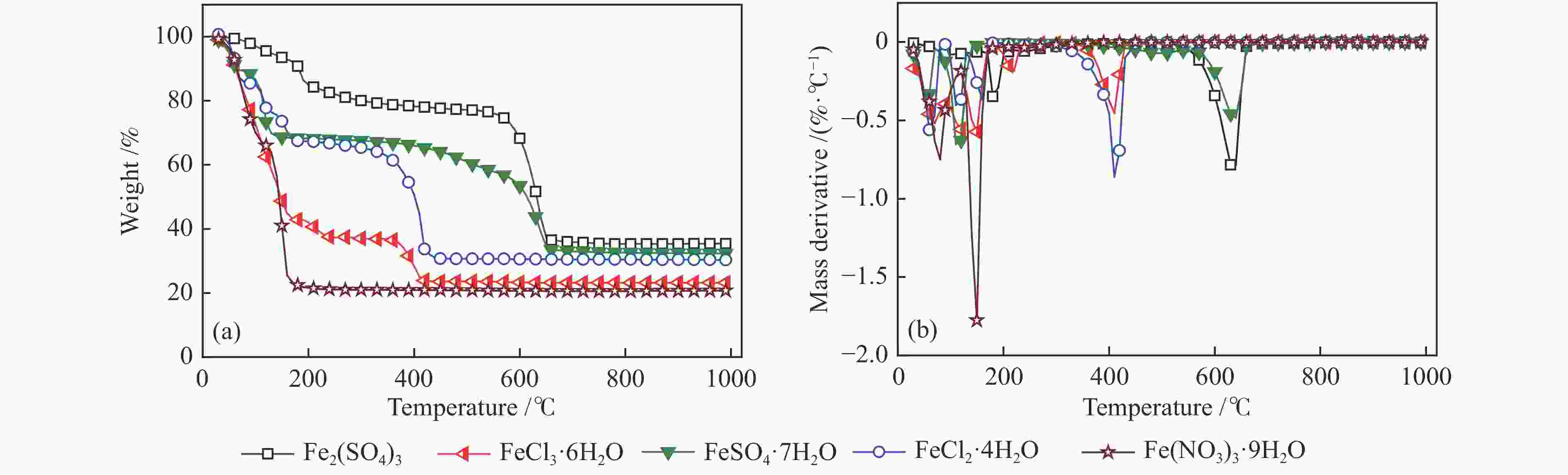
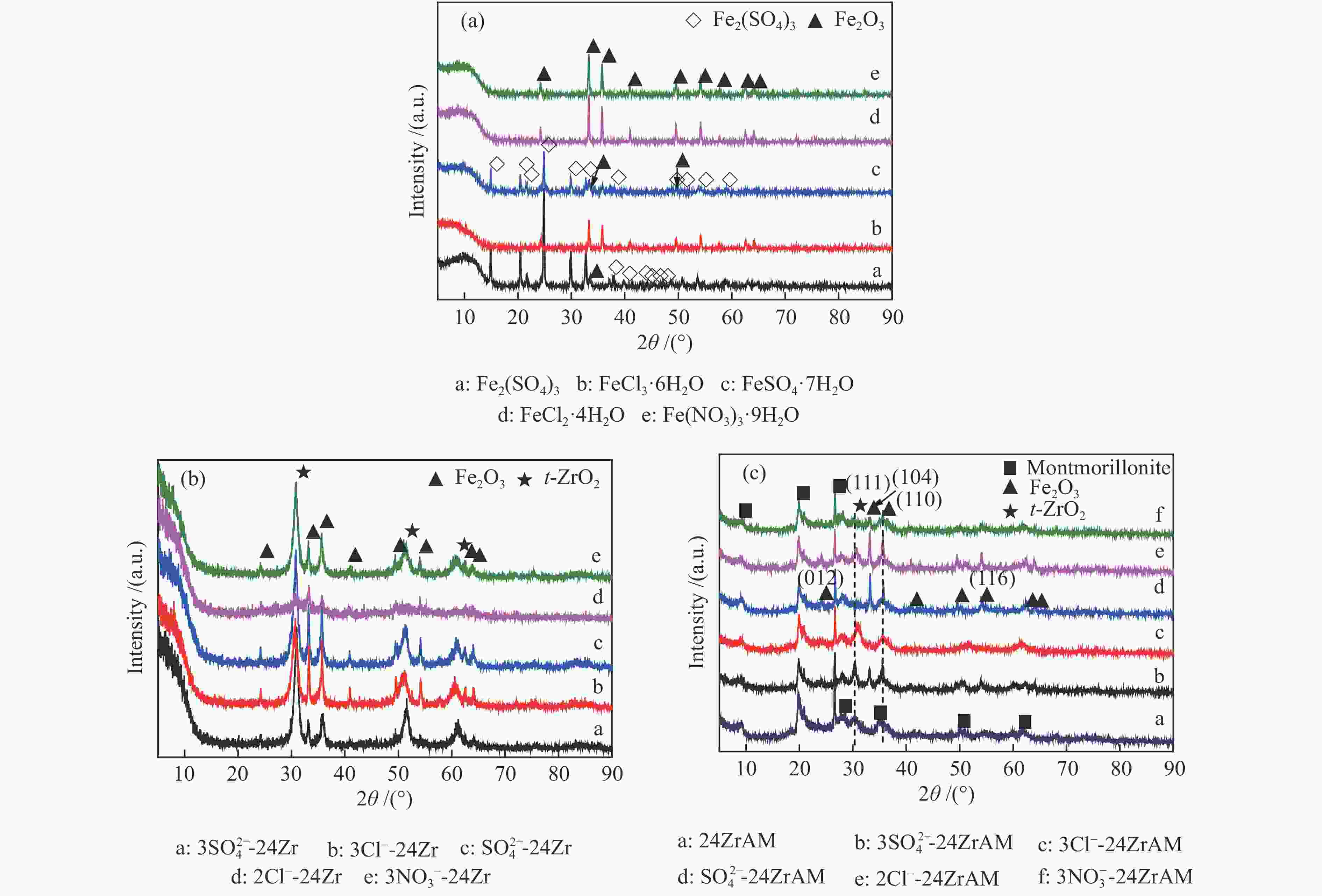
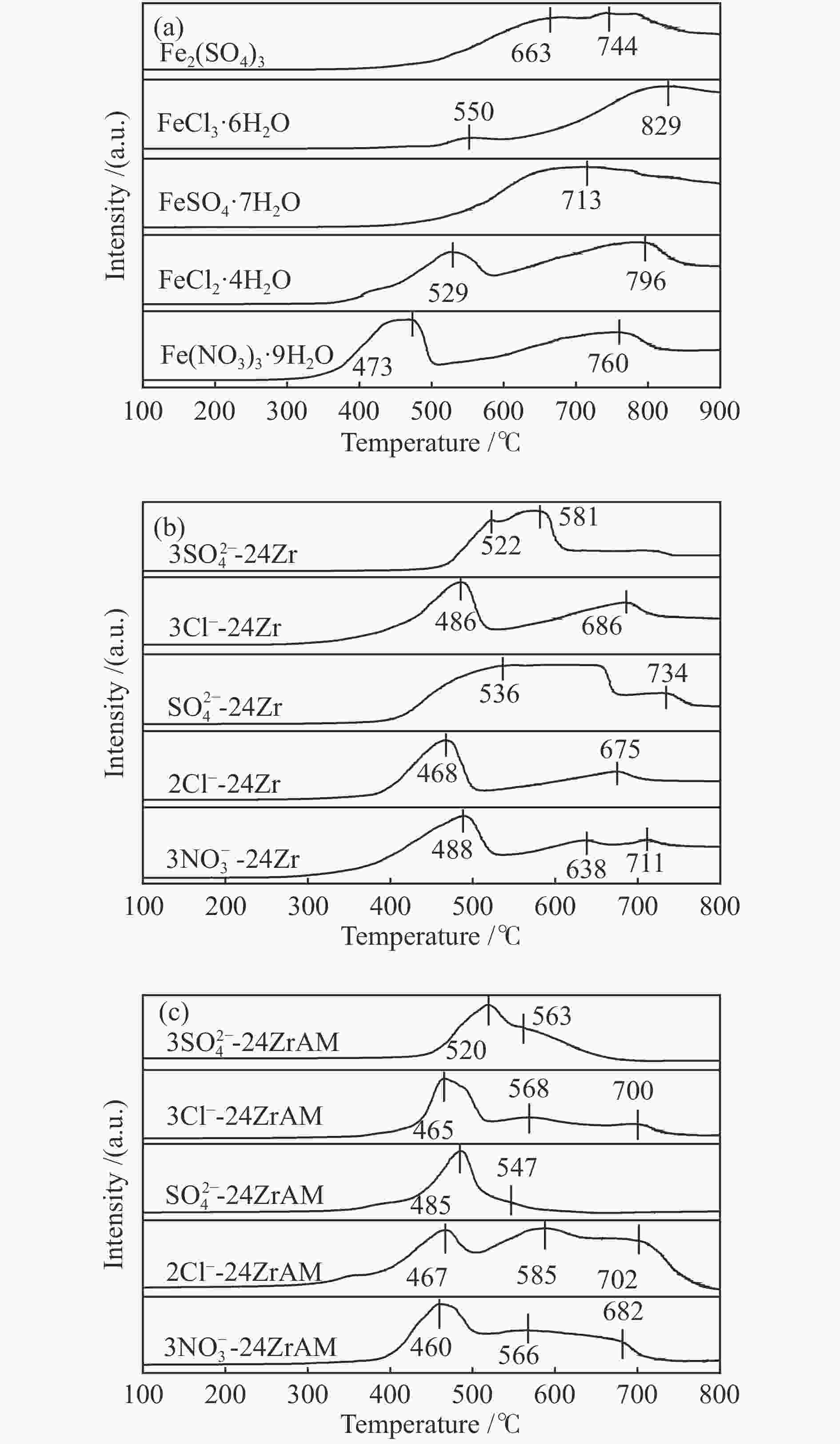
 下载:
下载: