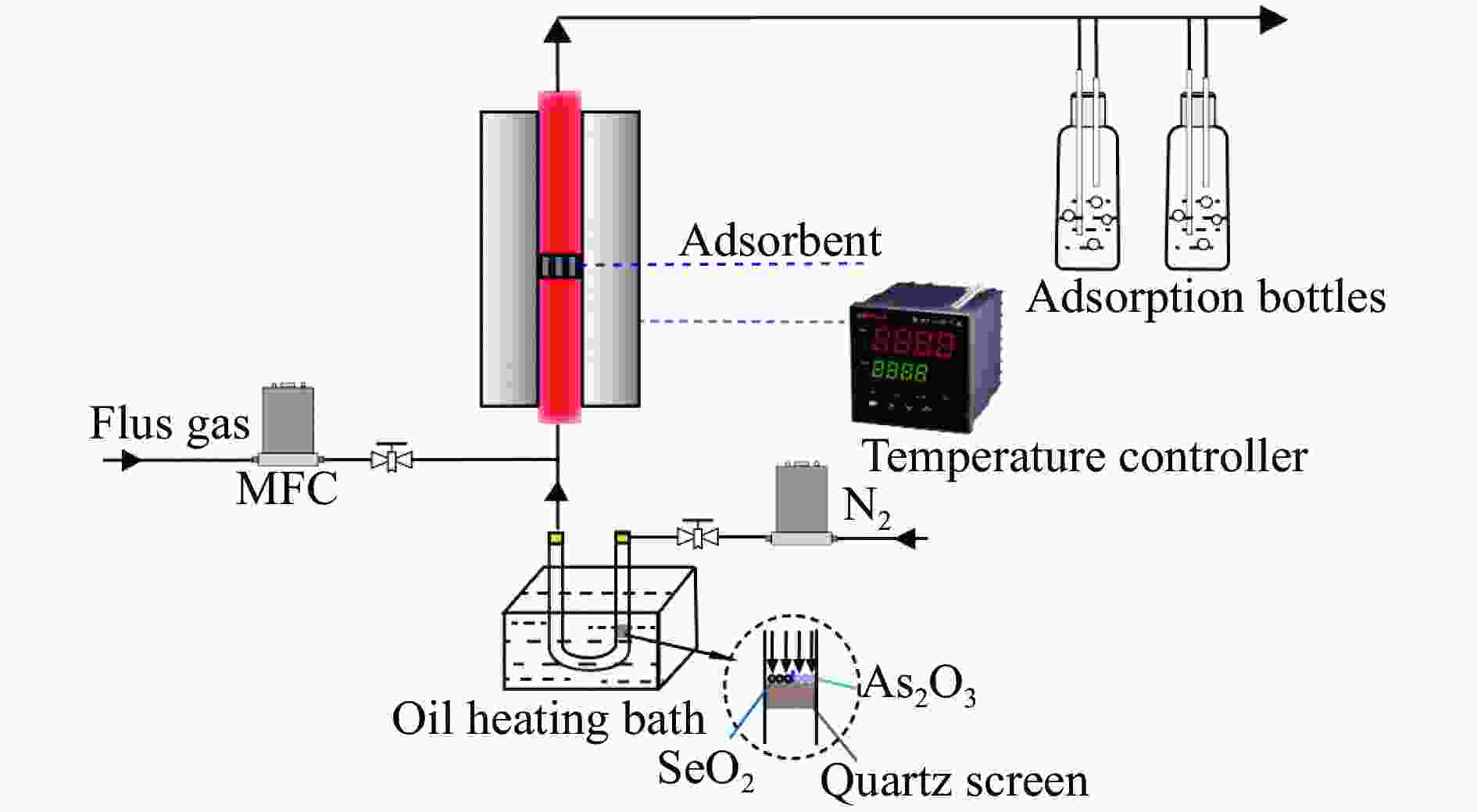
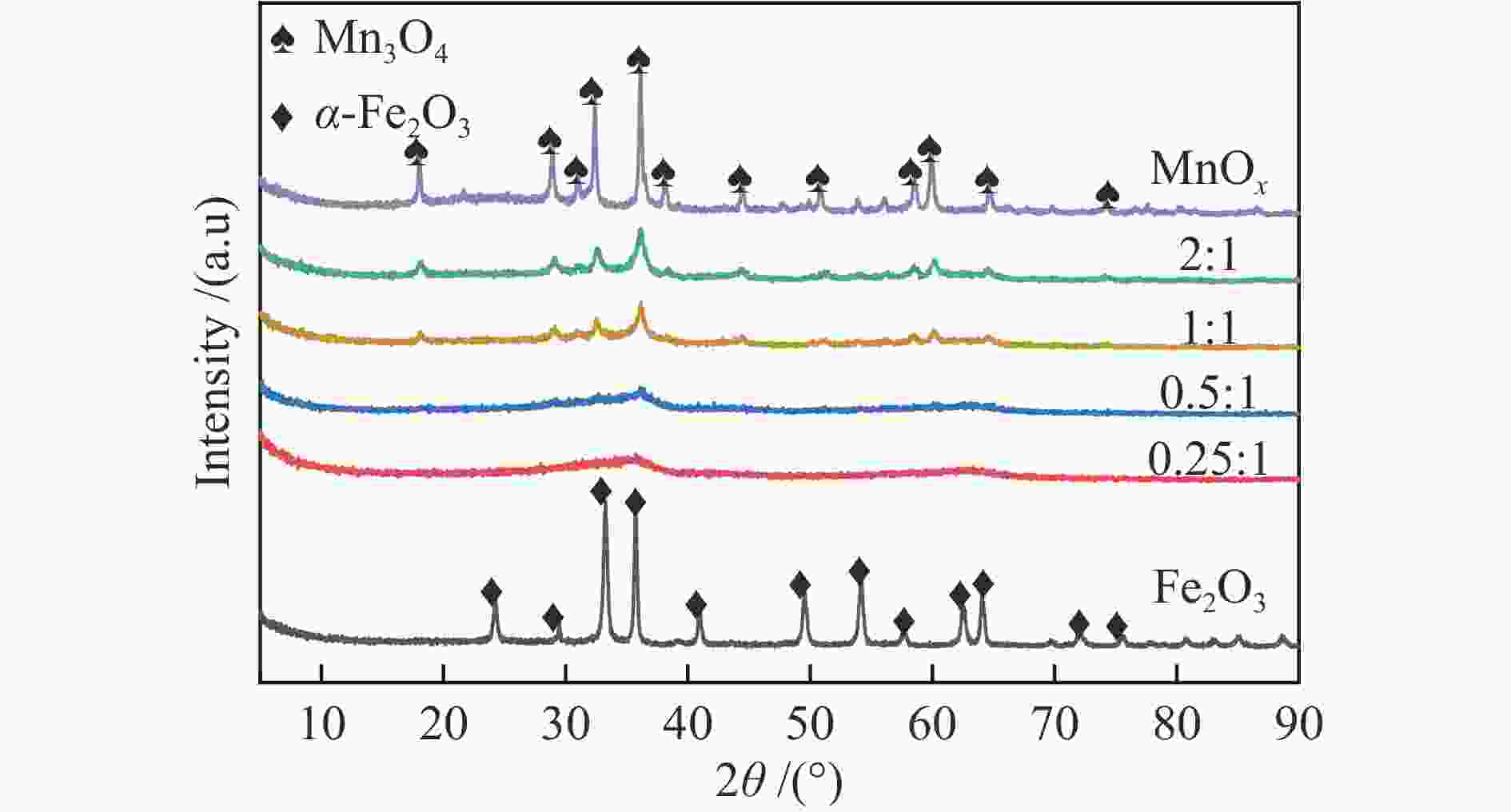
Experimental study on adsorption of arsenic and selenium in coal fired flue gas by Mn-Fe binary oxide
-
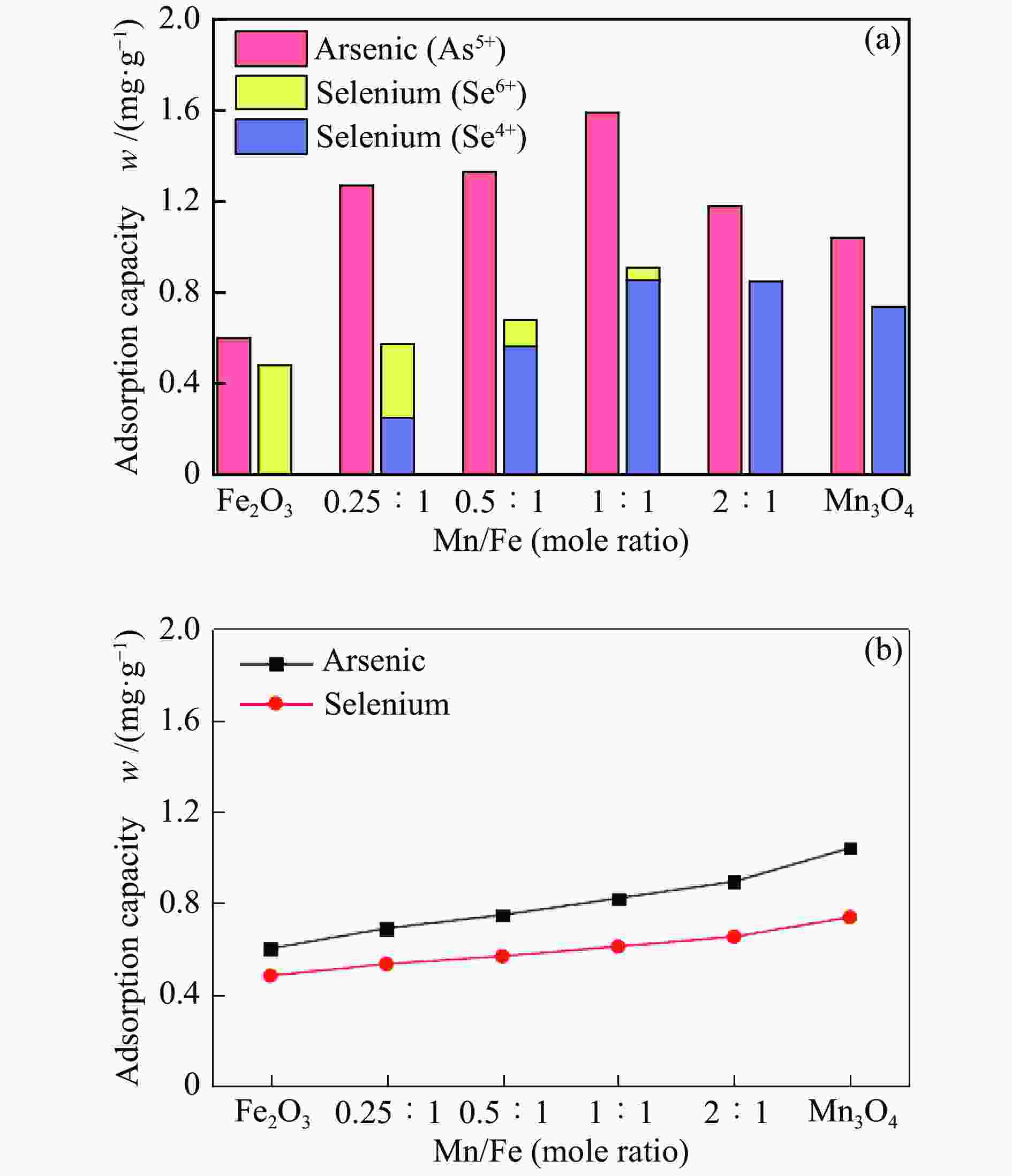
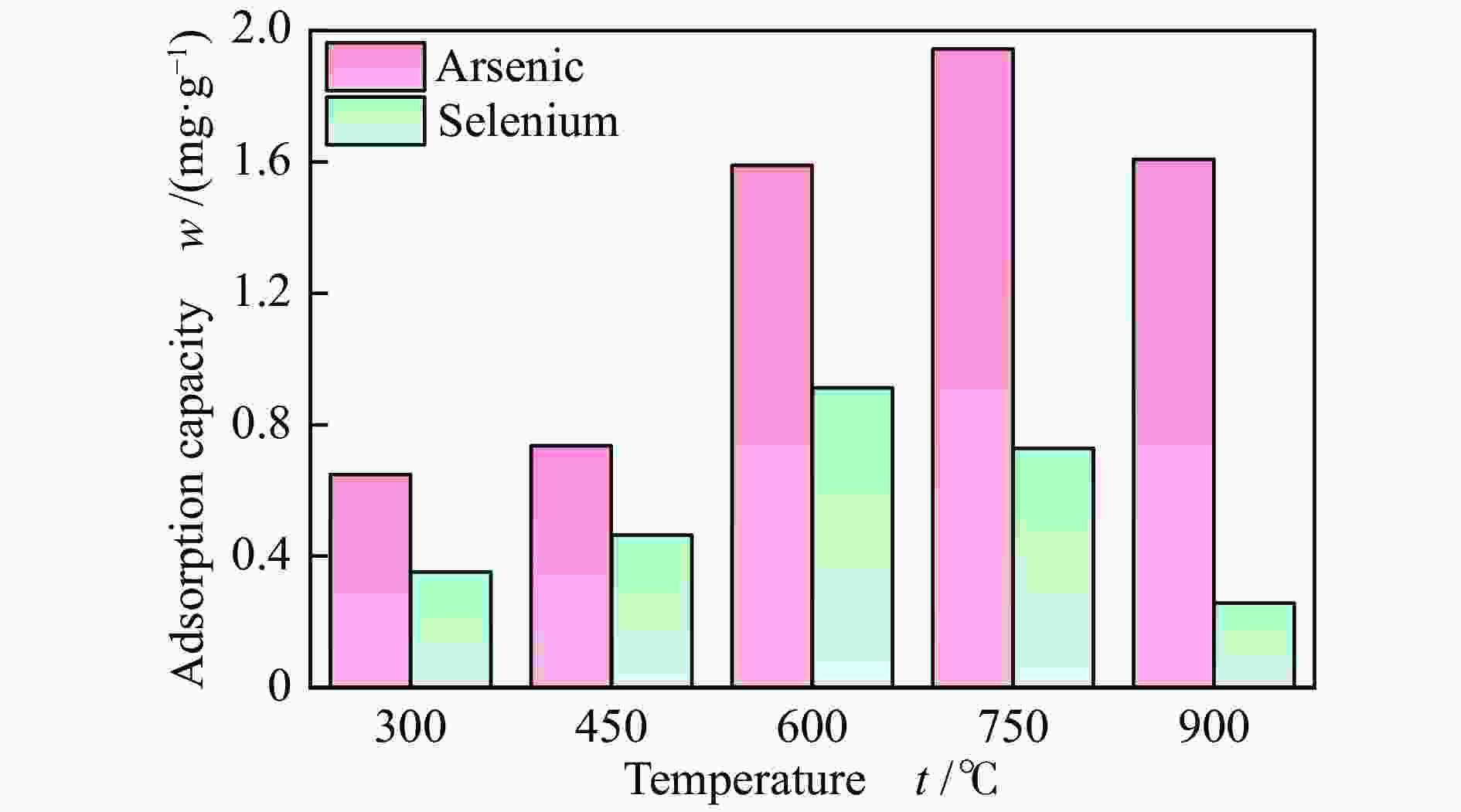
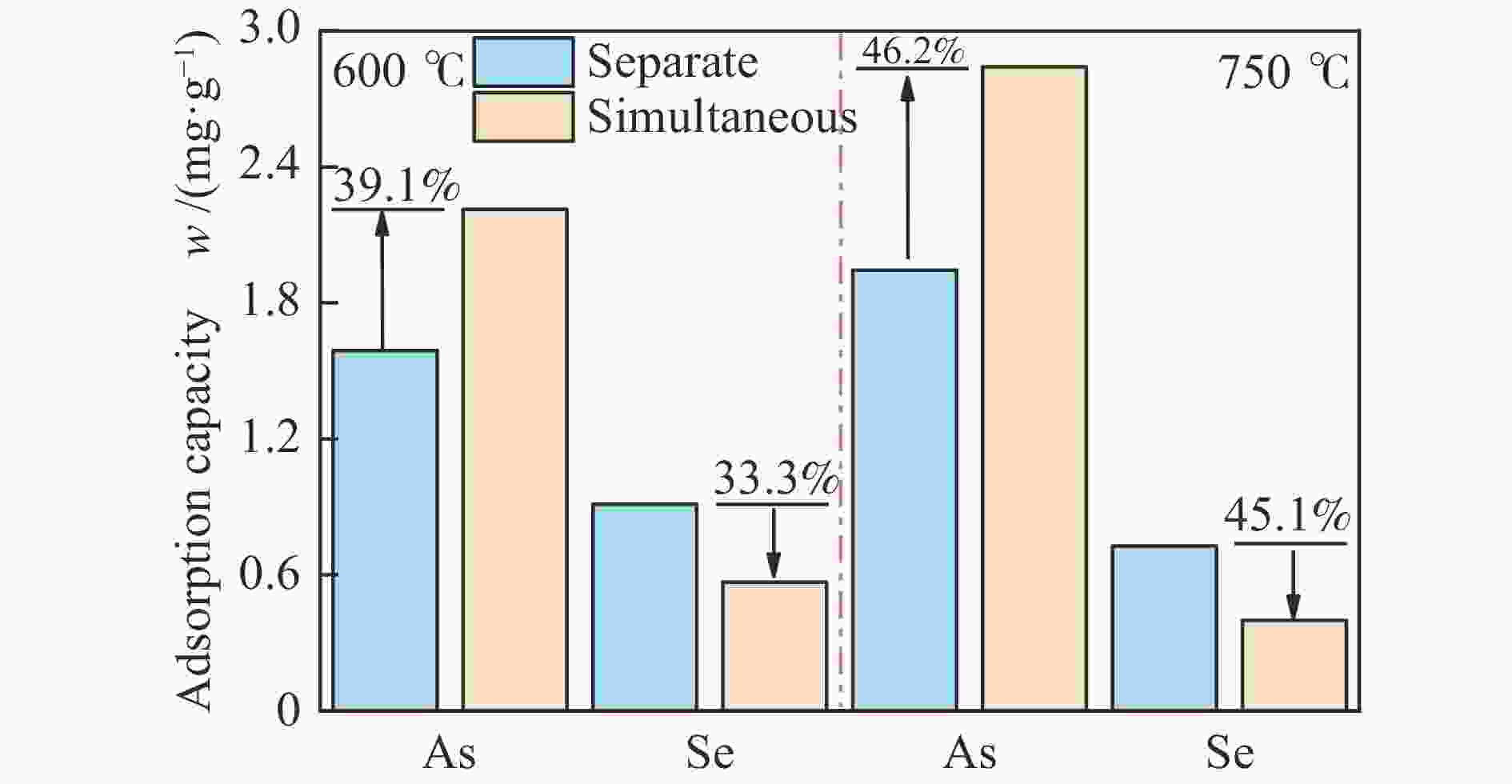
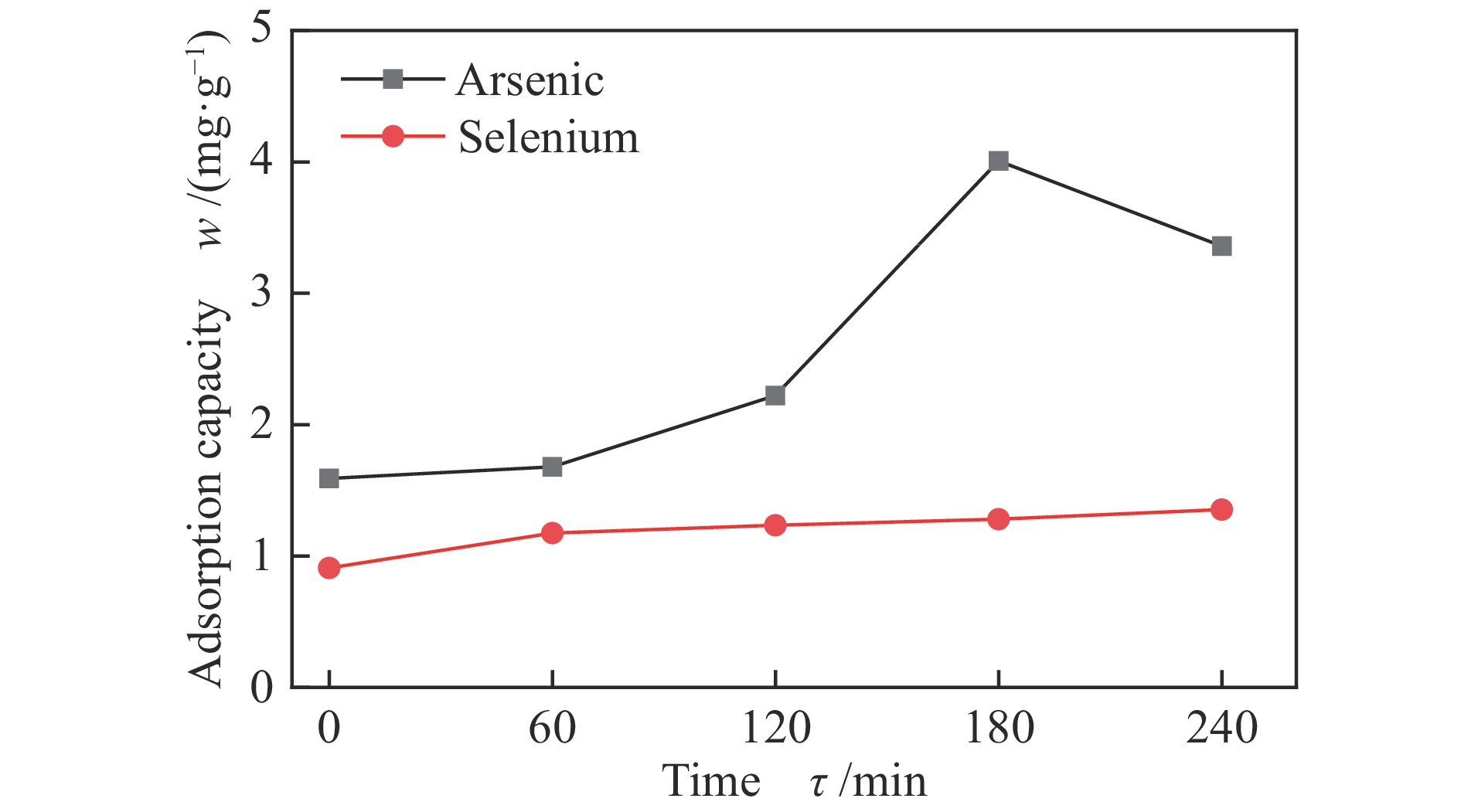
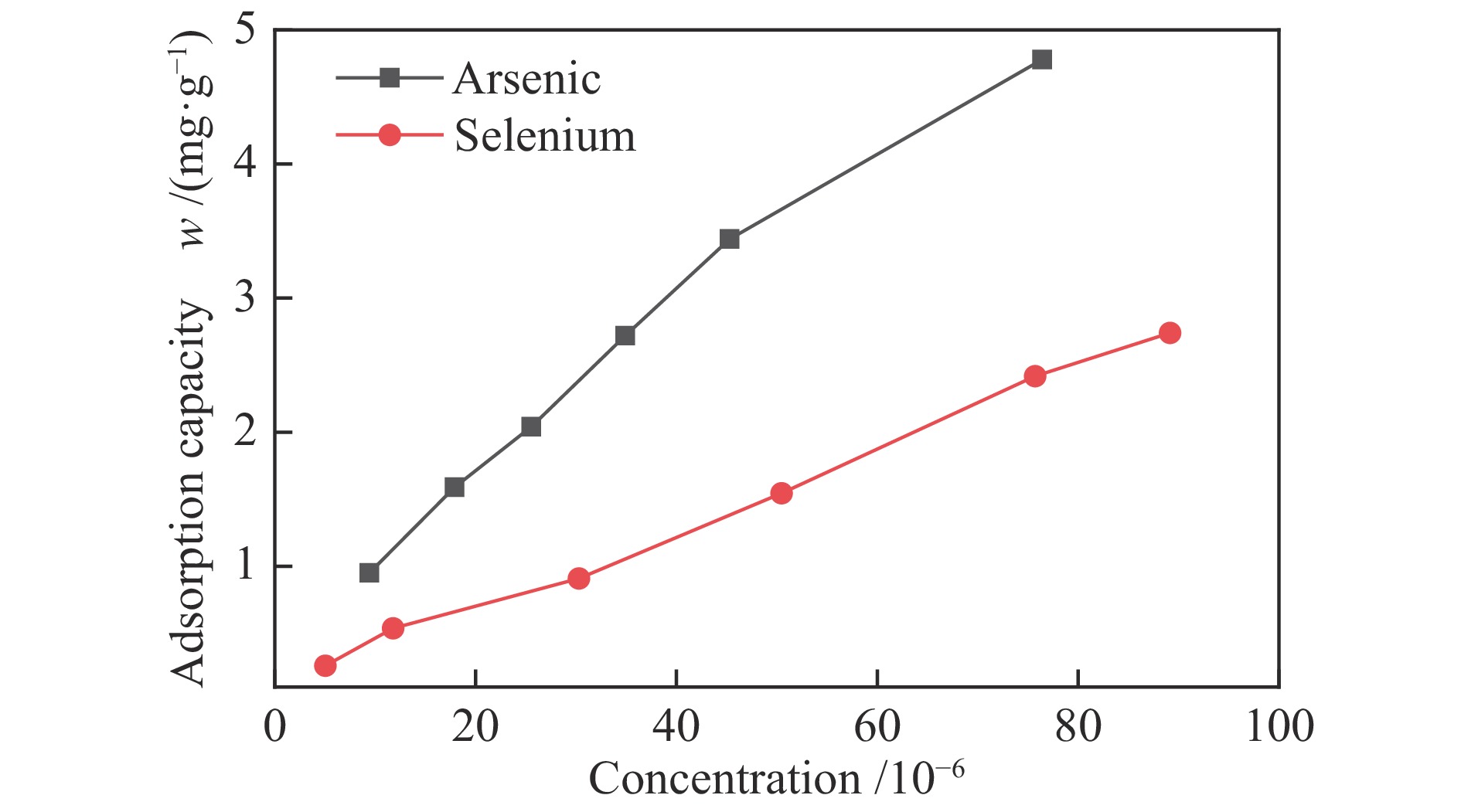
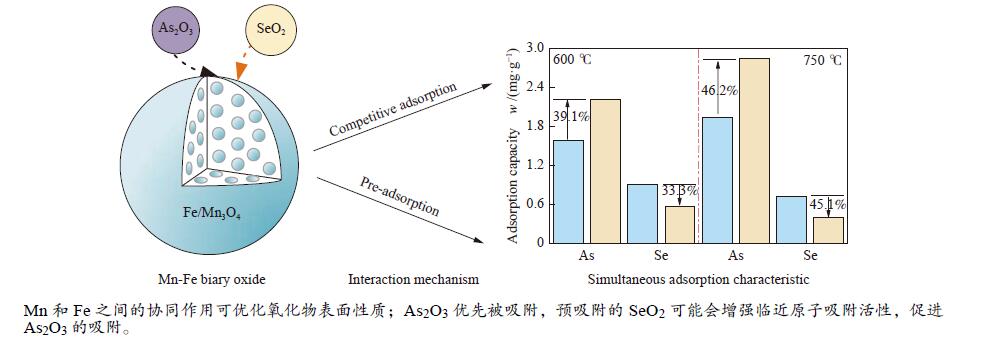
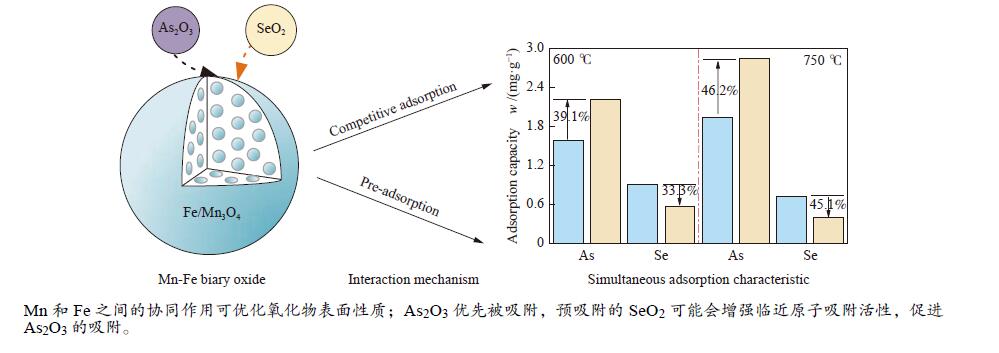
摘要: 采用共沉淀法制备了一系列Mn-Fe复合氧化物,研究了物质的量比和温度对气相砷、硒吸附的影响,考察了As2O3和SeO2双组分气体的同时吸附特性,对吸附产物中砷、硒的稳定性进行测定。结果表明,As2O3和SeO2在Mn-Fe复合氧化物表面的吸附量随Mn含量增加呈先增大后减小的趋势,当Mn/Fe物质的量比为1∶1时吸附量达到最大;两者的最佳吸附温度分别为750和600 ℃;双组分气体同时吸附时,两者存在竞争作用,Mn-Fe复合氧化物会优先吸附As2O3,SeO2的吸附受到抑制;此外,预吸附的SeO2会增强临近原子吸附活性,促进As2O3的吸附;吸附后的Mn-Fe复合氧化物浸出液中砷和硒的质量浓度均低于控制限值,在随粉煤灰进行资源化利用(如作为混凝土成分、水泥原料等)过程中不会产生二次污染。
-
关键词:
- Mn-Fe复合氧化物 /
- 吸附剂 /
- 砷 /
- 硒 /
- 同时吸附
Abstract: A series of Mn-Fe binary oxides with different molar ratios were synthesized by coprecipitation method, and their adsorption behaviors of gaseous arsenic and selenium in flue gas at different temperature were investigated. The simultaneous adsorption characteristics of As2O3 and SeO2 on Mn-Fe binary oxide were studied. To avoid secondary pollution for adsorbent utilization, As/Se leaching properties in Mn-Fe binary oxides were evaluated. Results show that the adsorption capacity of As2O3 and SeO2 on the Mn-Fe binary oxides increases firstly and then decreases with the increasing Mn content. The adsorption capacity reaches the maximum when the molar ratio is 1∶1. The optimum adsorption temperatures of As2O3 and SeO2 are 750 and 600 ℃, respectively. During the process for As2O3 and SeO2 adsorption on Mn-Fe binary oxide simultaneously, Mn-Fe binary oxides preferentially adsorb As2O3, while SeO2 is inhibited. In addition, the pre-adsorption SeO2 can enhance the adsorption activities of adjacent atoms, which is beneficial for As2O3 adsorption. The concentration of arsenic and selenium in the leaching solution of spent Mn-Fe binary oxide is far lower than the control limit, which will be no secondary pollution in the process of utilization with fly ash.-
Key words:
- Mn-Fe binary oxide /
- adsorbent /
- arsenic /
- selenium /
- simultaneous adsorption
-
表 1 吸附剂的元素含量、比表面积和孔结构参数
Table 1 Contents of Mn and Fe elements, specific surface area and pore structure parameters of adsorbents
Mn/Fe
(mole
ratio)Mn/% Fe/% Surface
area/
(m2·g−1)Pore
volume/
(cm3·g−1)Pore
diameter/
nmFe2O3 0.00 100.00 16.37 0.11 22.10 0.25∶1 20.54 79.46 201.31 0.37 5.94 0.5∶1 35.39 64.61 160.06 0.32 6.28 1∶1 51.52 48.48 117.13 0.30 9.37 2∶1 69.41 30.59 76.48 0.25 11.68 Mn3O4 100.00 0.00 19.02 0.13 20.71 表 2 Mn-Fe复合氧化物中砷和硒的浸出特性
Table 2 Leaching characteristics of As and Se on spent Mn-Fe binary oxide
Leached As/Se Separate Simultaneous As Se As Se Amount /(mg·L−1) 0.0216 0.1078 0.0126 0.0525 Proportion /% 0.271 2.372 0.114 1.733 -
[1] 吴江, Humphroy Muzvidziwa, 关昱, 张文博, 徐凯. 燃煤烟气污染物脱除技术研究进展[J]. 上海电力学院学报,2018,34(3):277−282.WU Jiang, Humphroy Muzvidziwa, GUAN Yu, ZHANG Wen-bo, XU Kai. Research progress on removal of pollutants from coal-fired flue gas[J]. J Shanghai Univ Electr Pow,2018,34(3):277−282. [2] 陈光升. 现阶段我国煤炭价格与能源消费结构的关系分析[J]. 发展研究,2021,3:29−30.CHEN Guang-sheng. Analysis of the relationship between coal price and energy consumption structure in China[J]. Devel Res,2021,3:29−30. [3] 黄永达, 胡红云, 龚泓宇, 刘慧敏, 付彪, 李帅, 罗光前, 姚洪. 燃煤电厂砷, 硒, 铅的排放与控制技术研究进展[J]. 燃料化学学报,2020,48(11):1281−1297.HUANG Yong-da, HU Hong-yun, GONG Hong-yu, LIU Hui-min, FU Biao, LI Shuai, LUO Guang-qian, YAO Hong. Research progress on emission and control technologies of arsenic, selenium and lead in coal-fired power plants[J]. J Fuel Chem Technol,2020,48(11):1281−1297. [4] 段钰锋, 朱纯, 佘敏, 姚婷, 赵士林, 汤红健, 黄天放, 刘猛. 燃煤电厂汞排放与控制技术研究进展[J]. 洁净煤技术,2019,25(2):1−17.DUAN Yu-feng, ZHU Chun, SHE Min, YAO Ting, ZHAO Shi-lin, TANG Hong-jian, HUANG Tian-fang, LIU Meng. Research progress on mercury emission and control technologies in coal-fired power plants[J]. Clean Coal Technol,2019,25(2):1−17. [5] WU Y W, ZHOU X Y, MI T G, HU Z H, XU M X, ZHANG B, ZHAO L, LU Q. First-principles insights into the adsorption and interaction mechanism of selenium on selective catalytic reduction catalyst[J]. Chemosphere,2021,275:130057. doi: 10.1016/j.chemosphere.2021.130057 [6] 陆强, 裴鑫琦, 徐明新, 王涵啸, 吴亚昌, 欧阳昊东. SCR脱硝催化剂抗砷中毒改性优化与再生研究进展[J]. 化工进展,2021,40(5):2365−2374.LU Qiang, PEI Xin-qi, XU Ming-xin, WANG Xiao-han, WU Ya-chang, OUYANG-Hao-dong. Progress in the development and regeneration of SCR catalysts for anti-arsenic poisoning[J]. Chem Ind Eng Prog,2021,40(5):2365−2374. [7] 赵重阳, 李国波, 眭华军, 刘初明, 王玲, 张亚平. 砷中毒商业V2O5-WO3/TiO2催化剂再生研究[J]. 分子催化,2020,34(5):407−414.ZHAO Chong-yang, LI Guo-bo, GUI Hua-jun, LIU Chu-ming, WANG Ling, ZHANG Ya-ping. Regeneration of commercial V2O5-WO3/TiO2 catalyst for arsenic poisoning[J]. J Mol Catal,2020,34(5):407−414. [8] SHEN F H, LIU J, ZHANG Z, DAI J X. On-Line analysis and kinetic behavior of arsenic release during coal combustion and pyrolysis[J]. Environ Sci Technol,2015,49(22):13716−13723. [9] RIESS M, MU LLER M. High temperature sorption of arsenic in gasification atmosphere[J]. Energy Fuels,2010,25(4):1438−1443. [10] 张月, 王春波, 刘慧敏, 孙喆, 李文瀚, 张永生, 潘伟平. 金属氧化物吸附剂干法脱除气相As2O3实验研究[J]. 燃料化学学报,2015,43(4):476−482. doi: 10.3969/j.issn.0253-2409.2015.04.016ZHANG Yue, WANG Chun-bo, LIU Hui-min, SUN Zhe, LI Wen-han, ZHANG Yong-sheng, PAN Wei-ping. Removal of gas-phase As2O3 in dry process by metal oxide adsorbents[J]. J Fuel Chem Technol,2015,43(4):476−482. doi: 10.3969/j.issn.0253-2409.2015.04.016 [11] 余圣辉, 张成, 袁昌乐, 马仑, 方庆艳, 陈刚. 矿物质氧化物对燃煤烟气中砷/铅的吸附特性研究[J]. 燃料化学学报,2020,48(11):1345−1355. doi: 10.3969/j.issn.0253-2409.2020.11.008YU Sheng-hui, ZHANG Cheng, YUAN Chang-le, MA Lun, FANG Qing-yan, CHEN Gang. Study on arsenic /lead adsorption characteristics by mineral oxides in coal-fired flue gas[J]. J Fuel Chem Technol,2020,48(11):1345−1355. doi: 10.3969/j.issn.0253-2409.2020.11.008 [12] HUANG Y D, HU H Y, GONG H Y, XING H X, YUAN B, FU B, LI A J, YAO H. Mechanism study of selenium retention by iron minerals during coal combustion[J]. Proc Combust Inst,2021,38(3):4189−4197. doi: 10.1016/j.proci.2020.08.006 [13] WANG Y, YU J L, WANG Z H, LIU Y X, ZHAO Y C. A review on arsenic removal from coal combustion: Advances, challenges and opportunities[J]. Chem Eng J,2021,414:128785. doi: 10.1016/j.cej.2021.128785 [14] 曹辉. 二氧化钛负载铈锰氧化物脱除煤气汞及再生的机理研究[D]. 杭州: 浙江大学, 2020.CAO Hui. Mechanism study on the mercury removal from coal gas by titanium dioxide supported cerium manganese oxides and its regeneration[D]. Hangzhou: Zhejiang University, 2020. [15] HE K Q, YUAN C G, JIANG Y H, DUAN X L, LI Y, SHI M D. Synergistic effects of Fe-Mn binary oxide for gaseous arsenic removal in flue gas[J]. Ecotox Environ Safe,2021,207:111491. doi: 10.1016/j.ecoenv.2020.111491 [16] 刘翔, 张月, 邢佳颖, 郭雨生, 许桐, 王春波. Mn改性Fe2O3/γ-Al2O3脱除气相As2O3实验研究[J]. 中国电机工程学报,2021,41(15):5250−5258.LIU Xiang, ZHANG Yue, XING Jia-ying, GUO Yu-sheng, XU Tong, WANG Chun-bo. Experimental study on removal of gas-phase As2O3 by Mn modified Fe2O3/γ-Al2O3[J]. Proc CSEE,2021,41(15):5250−5258. [17] 唐强. SO2和NOx混合气体竞争吸附实验研究[D]. 西安: 西安交通大学, 2003.TANG Qiang. Experimental investigation of the SO2 and NOx binary gas competitive adsorption[D]. Xian: Jiaotong University, 2003. [18] 胡长兴. 燃煤电站汞排放及活性炭稳定吸附机理研究[D]. 杭州: 浙江大学, 2007.HU Chang-xing. Mechanism of mercury emission and stable adsorption of activated carbon from coal-fired power station[D]. Hangzhou: Zhejiang University, 2007. [19] 张明光, 李海龙, 赵永椿, 张军营. 纳米硫化锌吸附剂中汞的稳定性研究[J]. 工程热物理学报,2018,39(7):1630−1634.ZHANG Ming-guang, LI Hai-long, ZHAO Yong-chun, ZHANG Jun-ying. Study on the stability of mercury on nano-ZnS adsorbents[J]. J Eng Therm,2018,39(7):1630−1634. [20] 孙文博. Mn基复合氧化物低温选择性催化还原NO性能及机理研究[D]. 大连: 大连理工大学, 2019.SUN Wen-bo. Selective catalytic reduction of NO over Mn-based mixed oxides and reaction mechanism studies[D]. Dalian: Dalian University of Technology, 2019. [21] 曹静, 刘盛余, 杨杰, 能子礼超, 杨淑清. 锰铁复合磁性材料制备及其对零价汞吸附性能研究[J]. 环境科学学报,2021,41(9):3745−3752.CAO Jing, LIU Sheng-yu, YANG Jie, NENGZI Li-chao, YANG Shu-qing. Study on the preparation of manganese-Fe composite magnetic materials and their capability for gaseous elemental mercury capture[J]. Acta Sci Circ,2021,41(9):3745−3752. [22] 谢江坤. 锰基复合氧化物及其对零价汞的吸附性能研究[D]. 上海: 上海交通大学, 2013.XIE Jiang-kun. Study of manganese-based binary metal oxides and their capability for gaseous elemental mercury capture[D]. Shanghai: Shanghai Jiao Tong University, 2013. [23] 姚挺. 锰基吸附剂的再生及循环脱汞机理研究[D]. 南京: 东南大学, 2020.YAO Ting. Mechanism study on regeneration and cyclic mercury removal of manganese based sorbent[D]. Nanjing: Southeast University, 2020. [24] SONG B, SONG M, CHEN D D, CAO Y, MENG F Y, WEI Y X. Retention of arsenic in coal combustion flue gas at high temperature in the presence of CaO[J]. Fuel,2020,259(1):116241−116249. [25] KONG F H, QIU J R, LIU H, ZHAO R, ZENG H C. Effect of NO/SO2 on elemental mercury adsorption by nano-Fe2O3[J]. Proc CSEE,2010,30(35):43−48. [26] XING J Y, WANG C B, HUANG Y L, LI S, ZHANG S Y. A comprehensive exploration about the effects of O2, SO2 and NO on As2O3 adsorption over Cu/γ-Al2O3 SCR catalyst: A DFT study[J]. Chem Eng Sci,2022,248:117260. doi: 10.1016/j.ces.2021.117260 [27] SCHWARTZ G E, HOWER J C, PHILLIPS A L, NELSON R, AVNER V, HEILEEN H K. Ranking coal ash materials for their potential to leach arsenic and selenium: relative importance of ash chemistry and site biogeochemistry[J]. Environ Eng Sci,2018,35(7):728−738. doi: 10.1089/ees.2017.0347 -




 下载:
下载: