Effect of preparation conditions on the morphology of Cu-SSZ-13 zeolites and their performance in the selective catalytic reduction of NOx by NH3
-
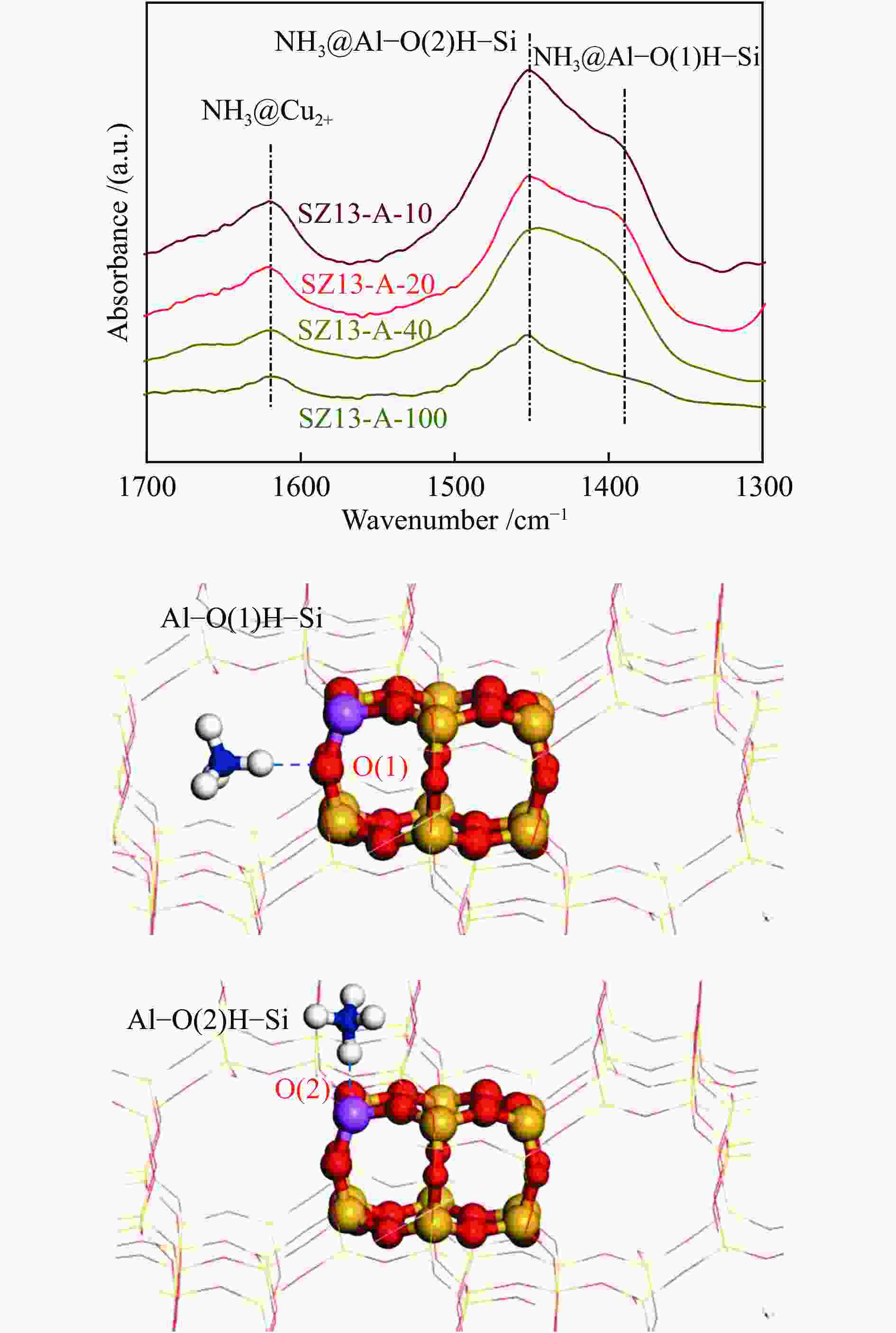
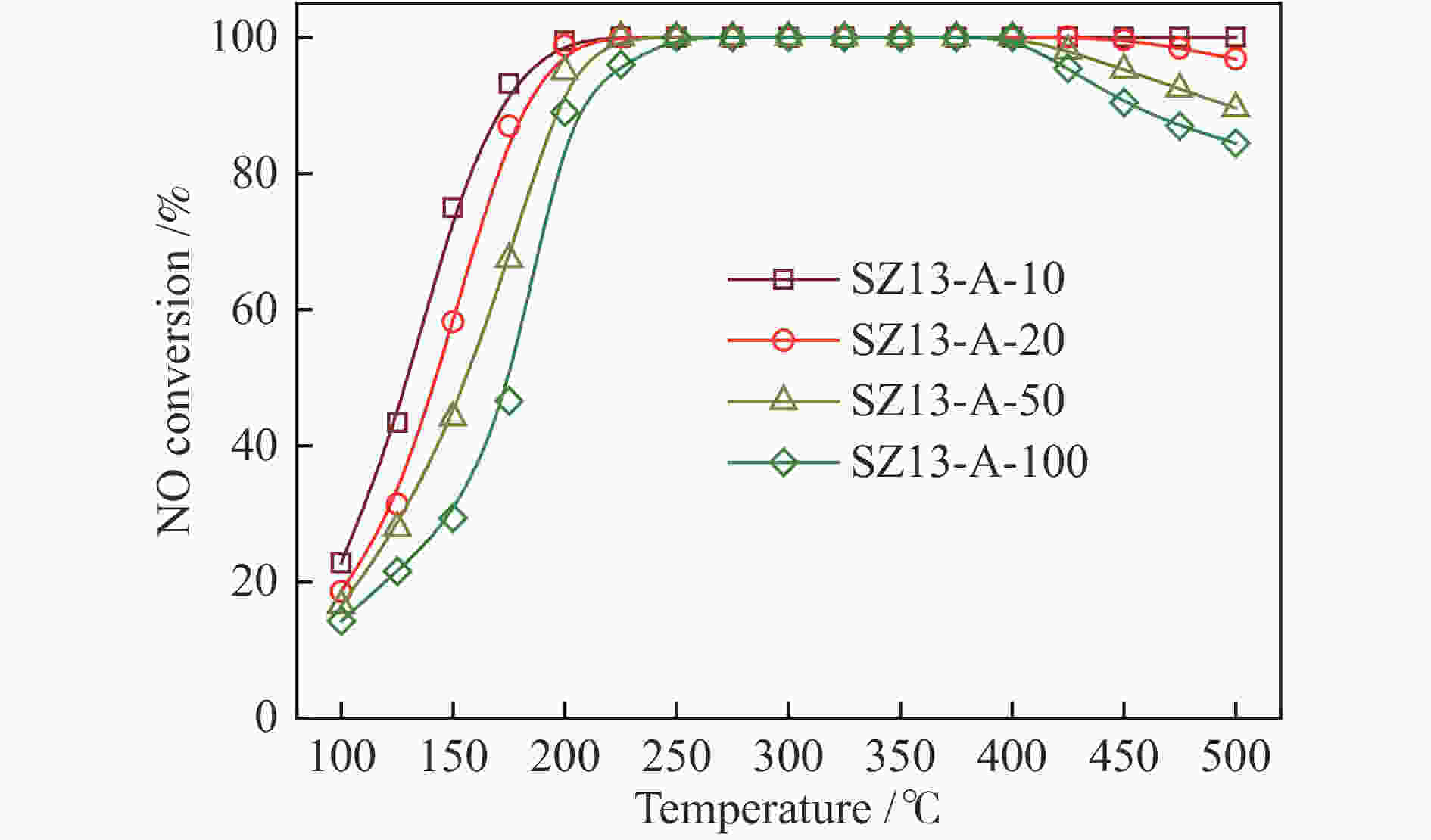
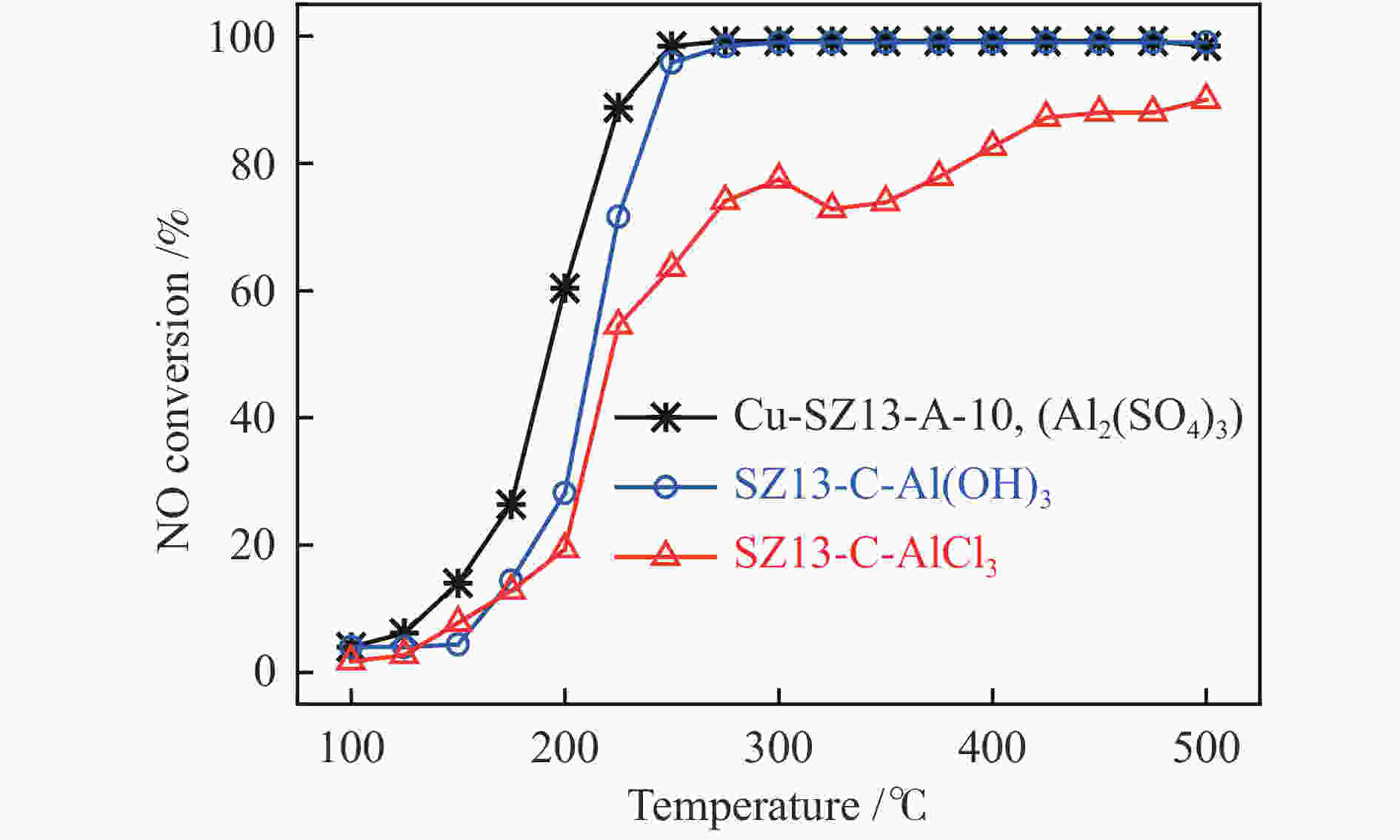
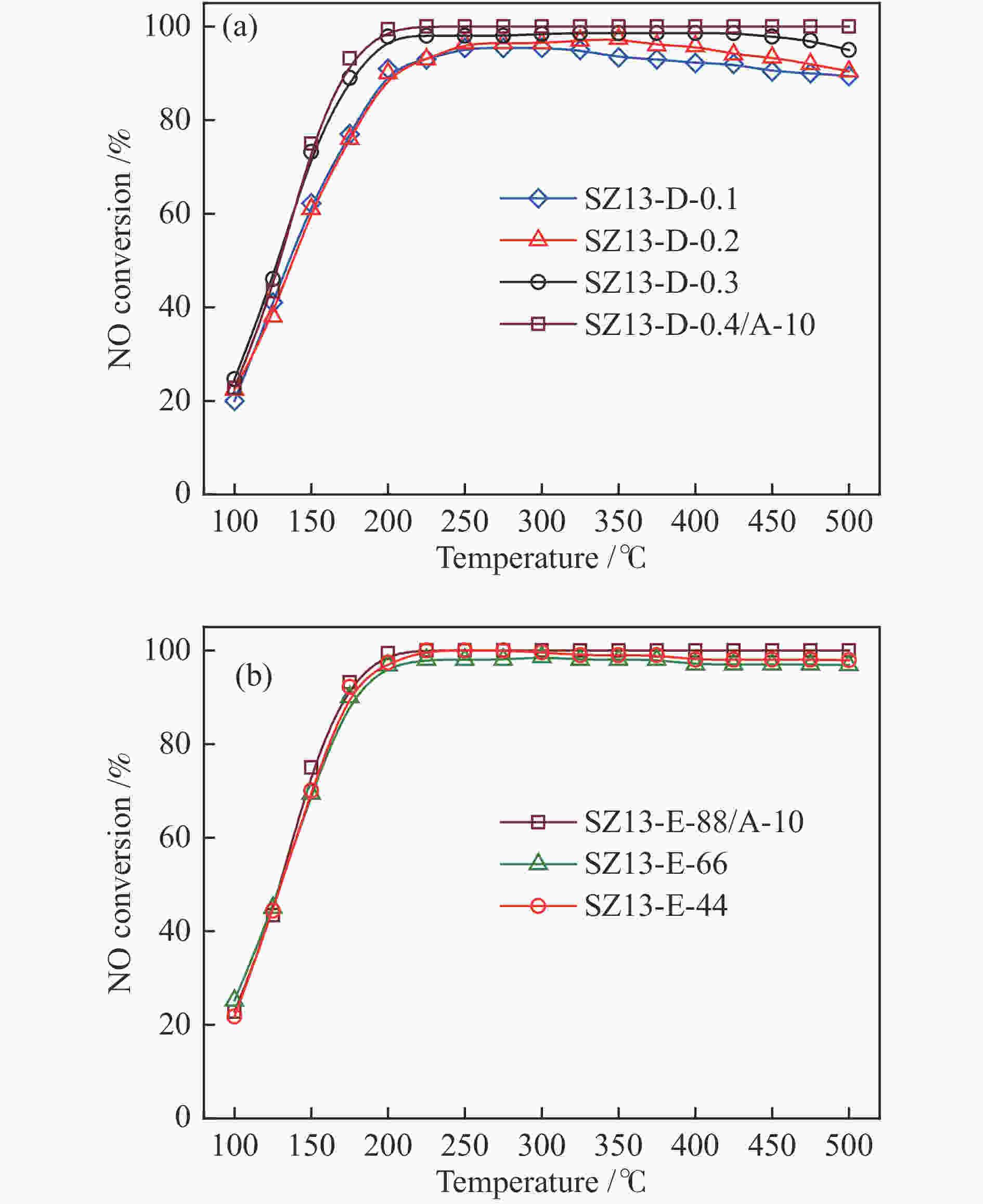
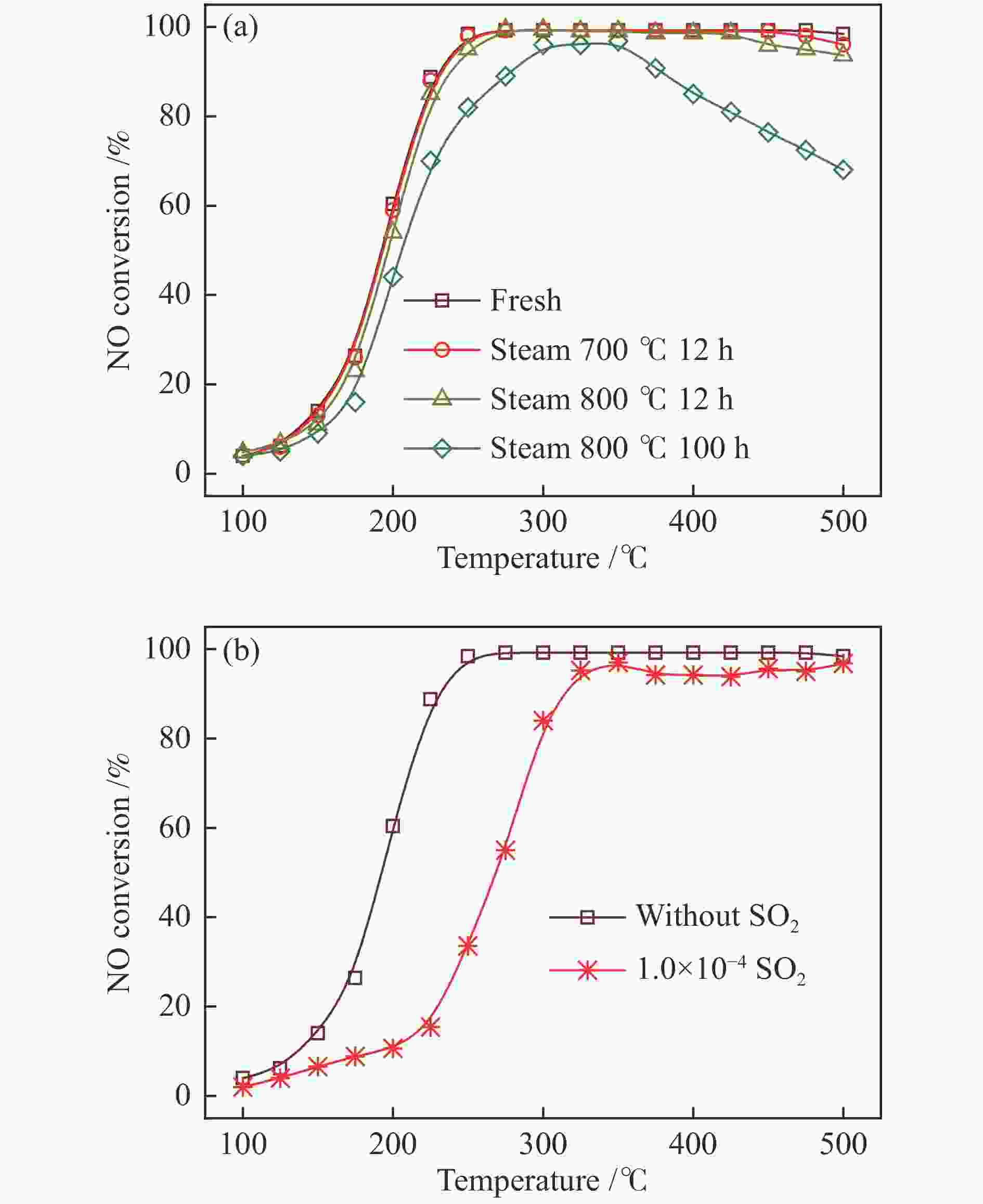
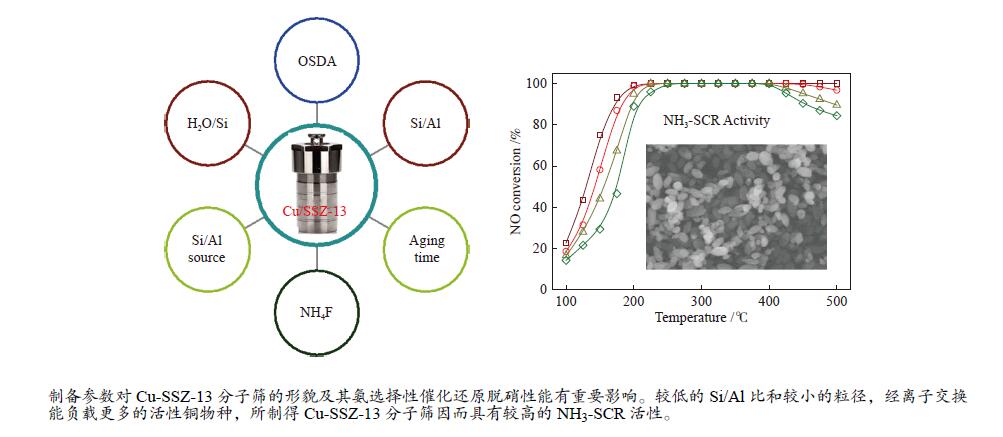
摘要: 采用传统水热法合成SSZ-13分子筛,铜离子交换法制备Cu-SSZ-13分子筛催化剂,用于氨选择性催化还原(NH3-SCR)脱硝,研究了有机模板剂(OSDA)用量、硅源、铝源、水硅比、硅铝比以及老化时间等制备参数对Cu-SSZ-13分子筛形貌、晶粒大小、酸性、Cu2+位点以及NH3-SCR催化性能的影响。结果表明,在一定范围内,SSZ-13分子筛的硅铝比越高,其晶粒越大,所负载的活性铜含量越低,NH3-SCR催化活性也越差。以硅溶胶为硅源、硫酸铝为铝源所合成的SSZ-13分子筛晶粒较小,结晶度最高,所得到的Cu-SSZ-13催化剂的NH3-SCR活性也最好。较高的水硅比所合成的分子筛晶粒较大,而增加模板剂的用量有利于提高SSZ-13分子筛的结晶度,降低其晶粒尺寸。适当延长老化时间,能降低SSZ-13分子筛晶粒尺寸,提升Cu-SSZ-13的NH3-SCR催化活性。其中,Si/Al比为10、硅溶胶为硅源、硫酸铝为铝源、OSDA/Si比为0.4、水硅比为88、老化2 h得到的Cu-SZ-A10催化剂最优,在240000 h−1的高空速下,200 ℃时NO转化率达60%,且在中高温区NO转化率保持为100%。该工作对SSZ-13分子筛形貌的调控以及高性能NH3-SCR脱硝催化剂的制备具有重要的参考价值。Abstract: SSZ-13 zeolites were synthesized by conventional hydrothermal method and the Cu-SSZ-13 zeolite catalysts were then prepared by Cu ion-exchange and used in the selective catalytic reduction of NOx with NH3 (NH3-SCR); the effect of various preparation parameters including the organic template agent (OSDA) dosage, silicon source, aluminum source, H2O/Si ratio, Si/Al ratio and aging time on the morphology, crystal size, acidity, state of Cu2+ sites and the catalytic performance of Cu-SSZ-13 in NH3-SCR were investigated. The results indicate that silica sol (JN25) and Al2(SO4)3 are appropriate as the silicon and aluminum sources, respectively, to prepare SSZ-13 zeolites with small crystals, high crystallinity and high activity in NH3-SCR. With the increase of Si/Al ratio, the crystal size of SSZ-13 increases and the copper content loaded on Cu-SSZ-13 decreases, leading to the degradation of NH3-SCR activity. A high H2O/Si ratio of 88 is conducive to forming larger SSZ-13 crystals, whilst increasing the OSDA dosage is beneficial to improving the crystallinity, reducing the crystal size, and accordingly enhancing the catalytic activity of Cu-SSZ-13 in NH3-SCR. In addition, a relatively longer aging time can also reduce the crystal size and raise the catalytic activity of Cu-SSZ-13. In particular, the Cu-SZ13-A10 zeolite catalyst synthesized with a Si/Al ratio of 10 (with the gel composition of 1SiO2 : 0.01Al2(SO4)3 : 0.3NaOH : 0.4SDA : 88H2O; JN25 as silicon source and aging for 2 h) exhibits high activity in NH3-SCR; under a high GHSV of 240000 h−1, the NO conversion reaches 60% at 200 ℃ and keeps at 100% in the moderate-high temperature range. These results should be useful for the regulation of SSZ-13 zeolite morphology and the preparation of high efficient Cu-SSZ-13 catalysts for NH3-SCR.
-
Key words:
- SSZ-13 zeolite /
- hydrothermal synthesis /
- crystal size /
- morphology /
- Cu-SSZ-13 /
- NH3-SCR
-
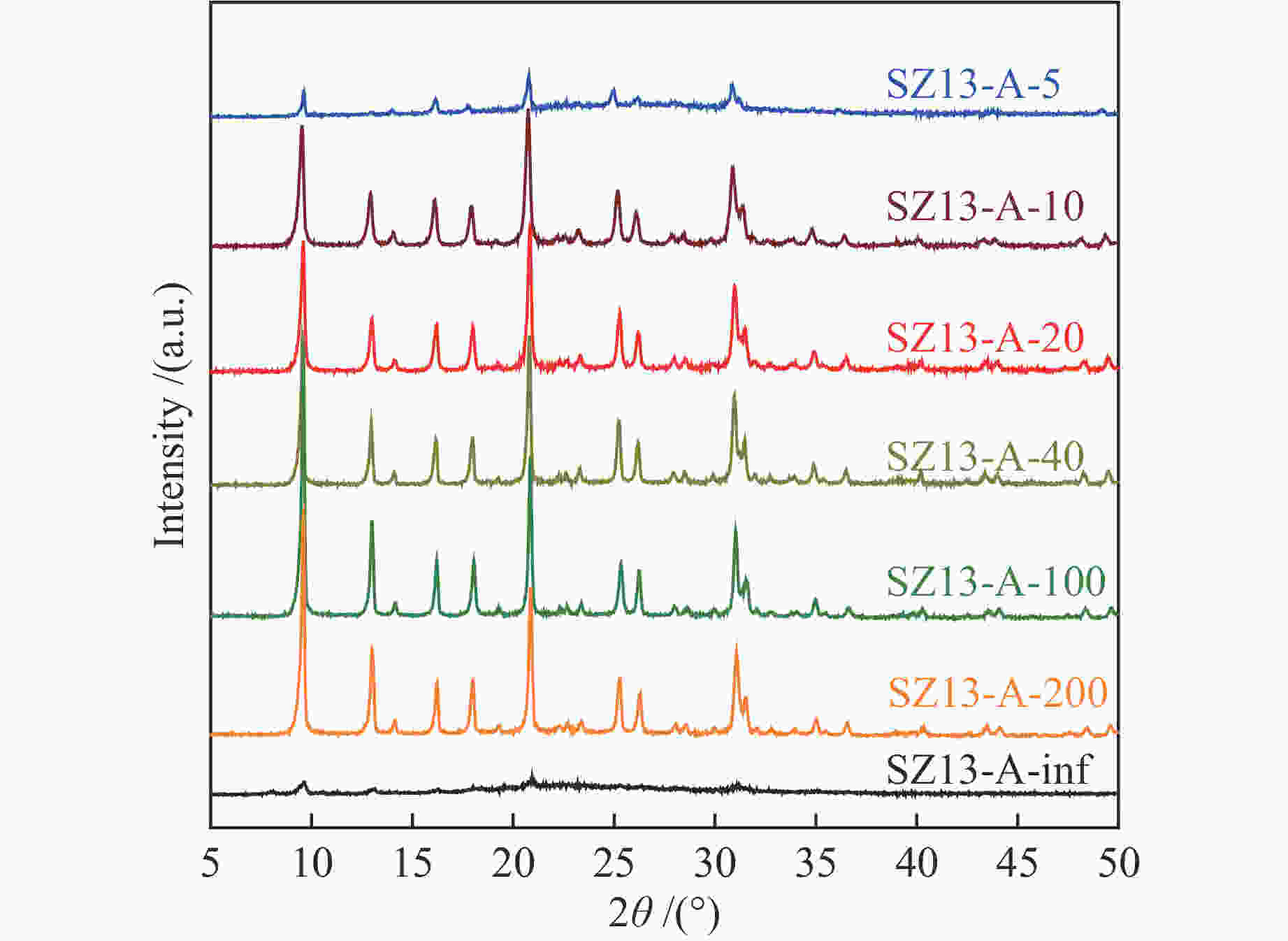
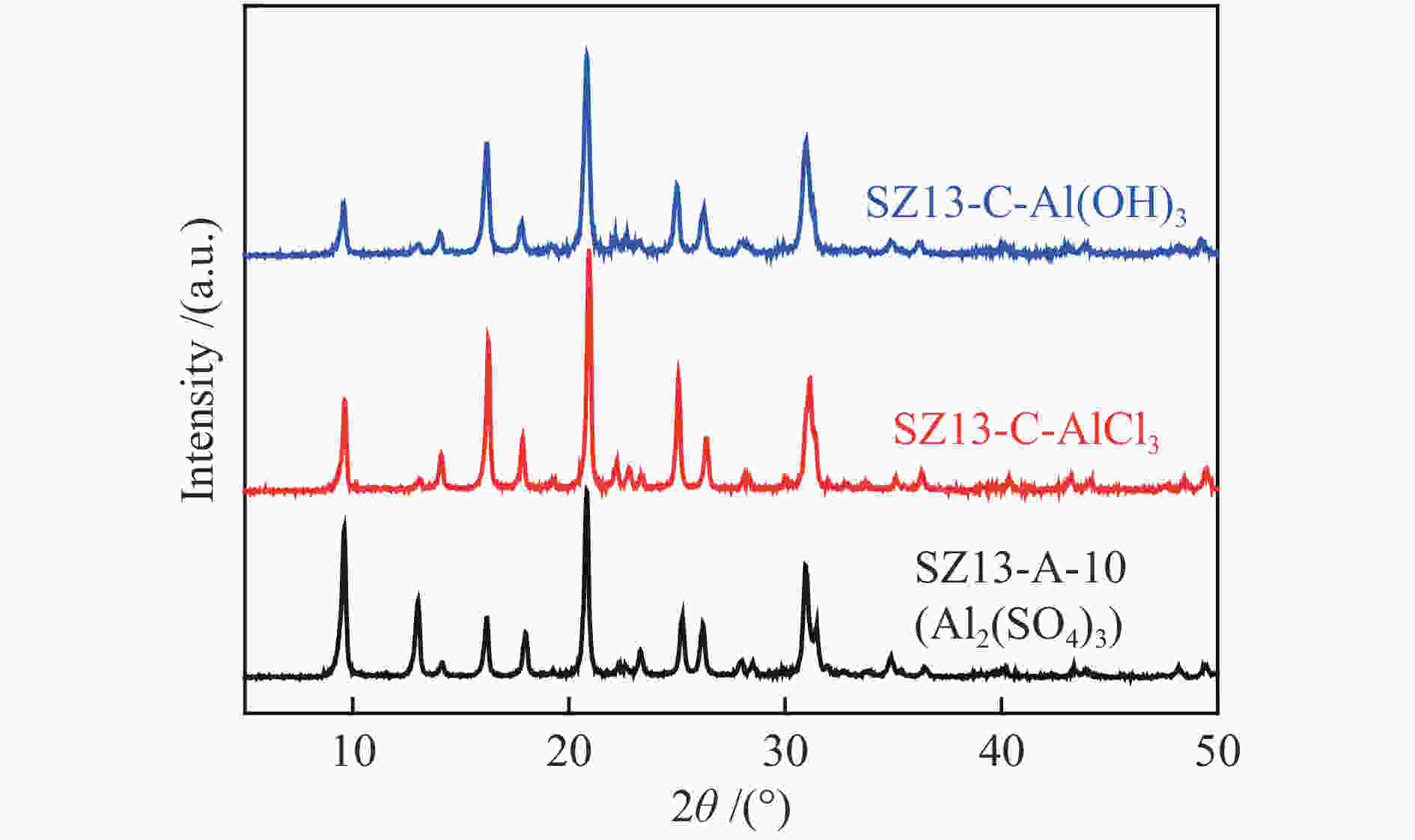
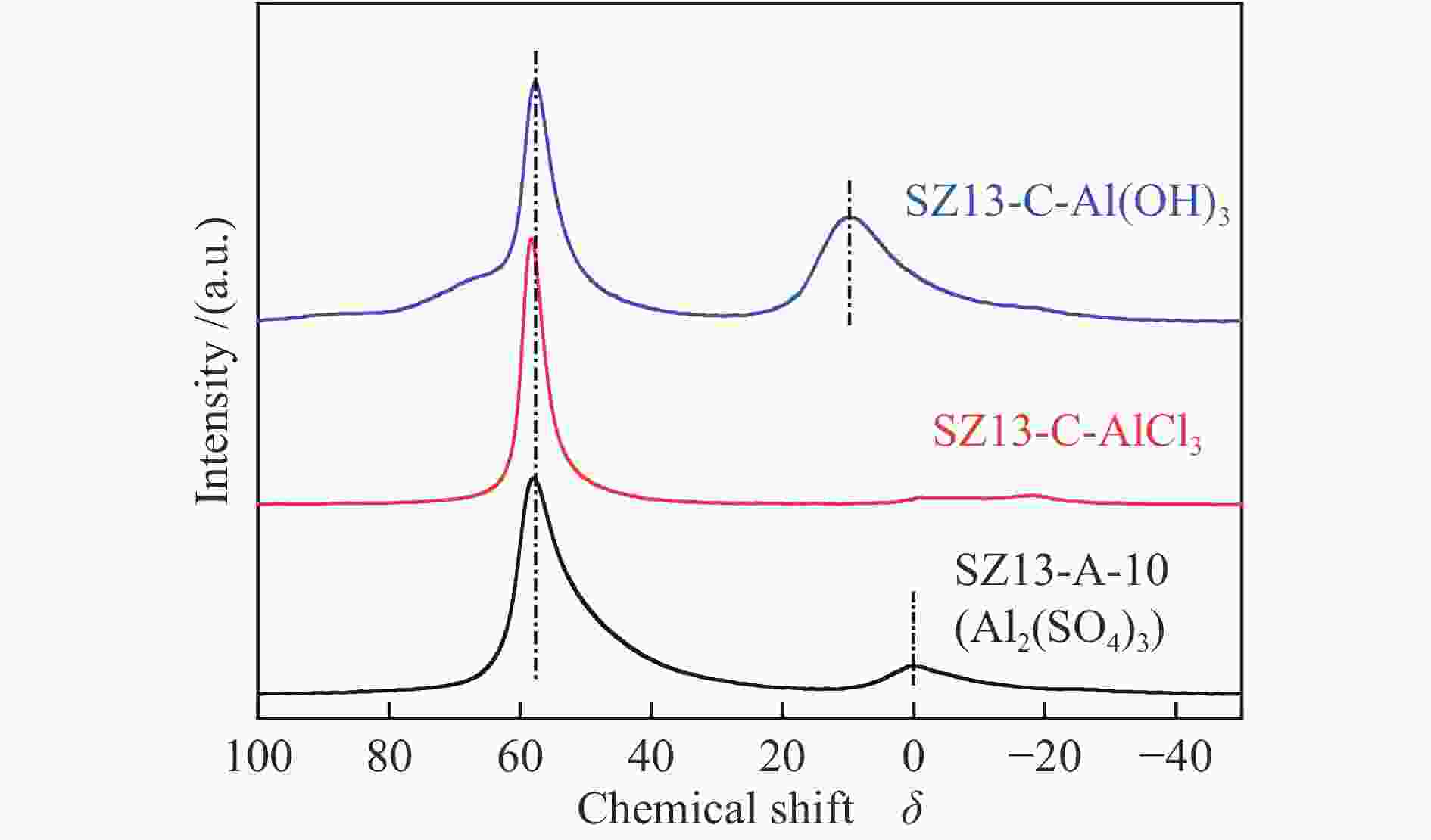
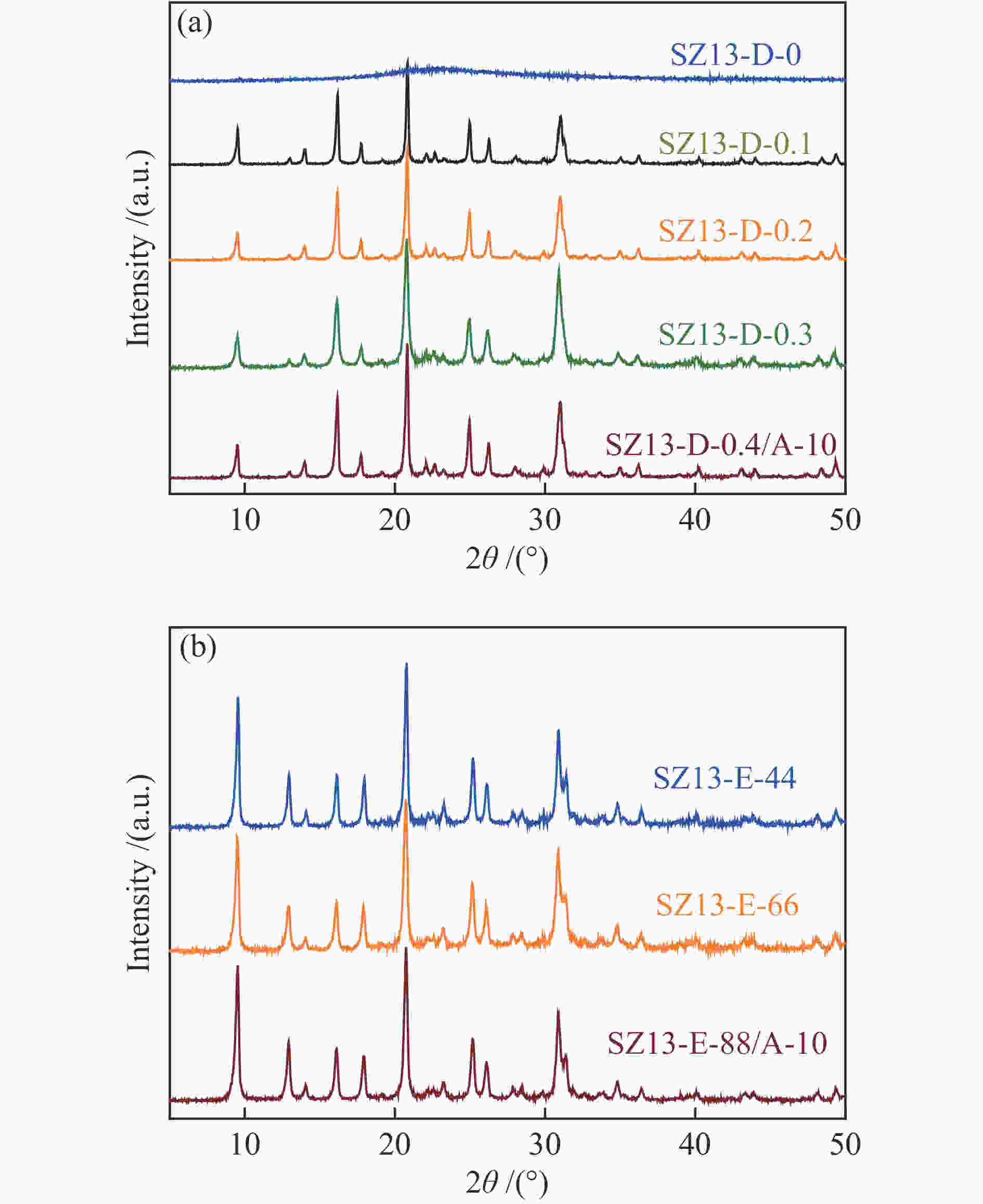
表 1 不同SSZ-13分子筛的化学组成及织构性质
Table 1 Compositional and textural properties of various SSZ-13 zeolites
Sample Si/Algel Si
sourceAl
sourceSDA/Si H2O/Si taging/
hF/Si Si/Al Cryst./
%dave/
μmCu/Al LCu/
%SBET/
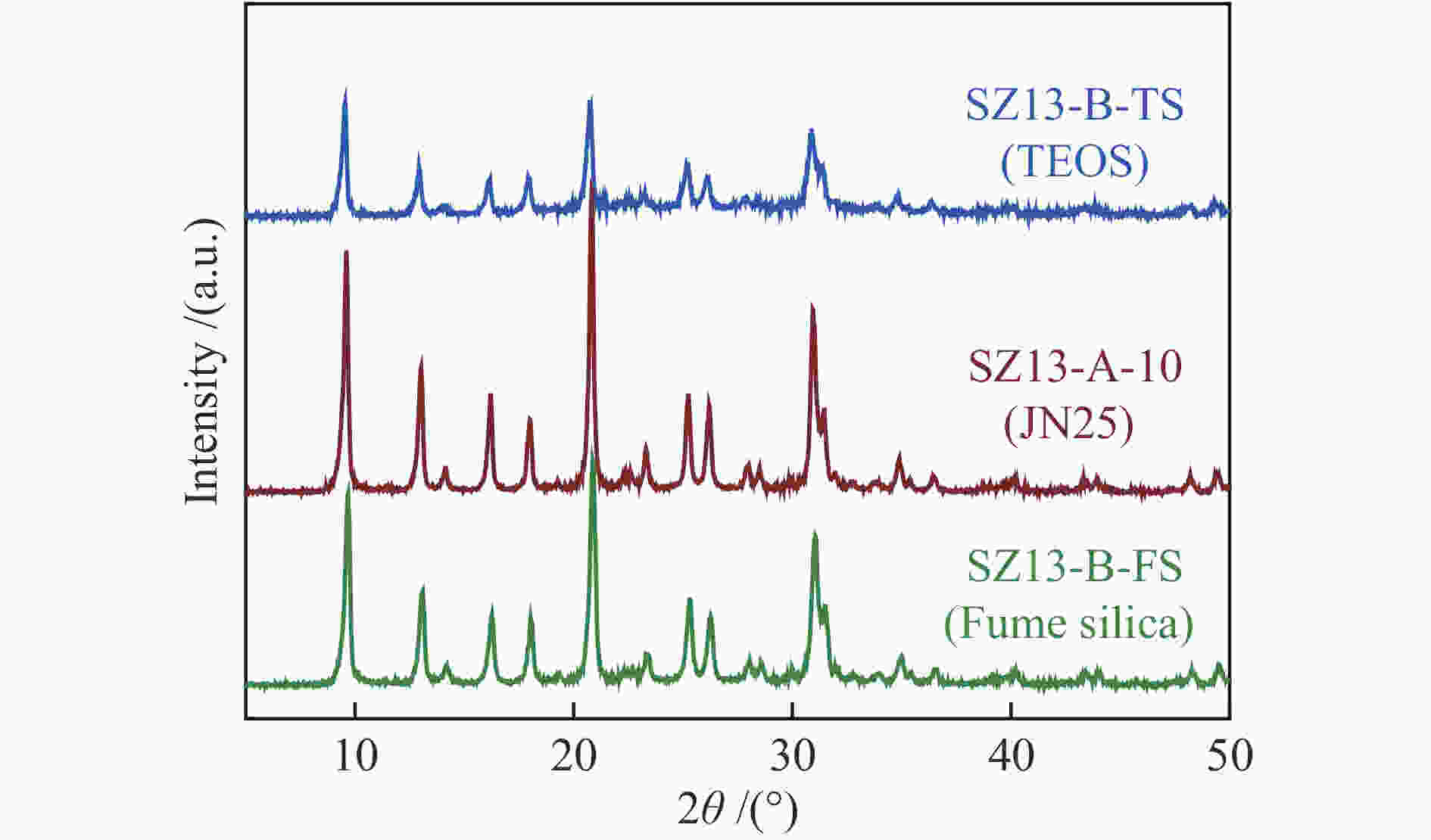
(m2·g−1)SZ13-A-5 5 JN25 Al2(SO4)3 0.4 88 2 0 <10 SZ13-A-10 10 JN25 Al2(SO4)3 0.4 88 2 0 10 94 0.7 0.19 1.8 563 SZ13-A-20 20 JN25 Al2(SO4)3 0.4 88 2 0 18 88 0.8 0.31 1.7 519 SZ13-A-40 40 JN25 Al2(SO4)3 0.4 88 2 0 27 82 5.4 0.35 1.2 506 SZ13-A-100 100 JN25 Al2(SO4)3 0.4 88 2 0 40 100 8.6 0.22 0.6 458 SZ13-A-200 200 JN25 Al2(SO4)3 0.4 88 2 0 52 93 7.1 0.15 0.4 447 SZ13-A-inf ∞ JN25 Al2(SO4)3 0.4 88 2 0 <5 SZ13-B-TS 10 TEOS Al2(SO4)3 0.4 88 2 0 9 59 1.2 0.11 1 477 SZ13-B-FS 10 Fume
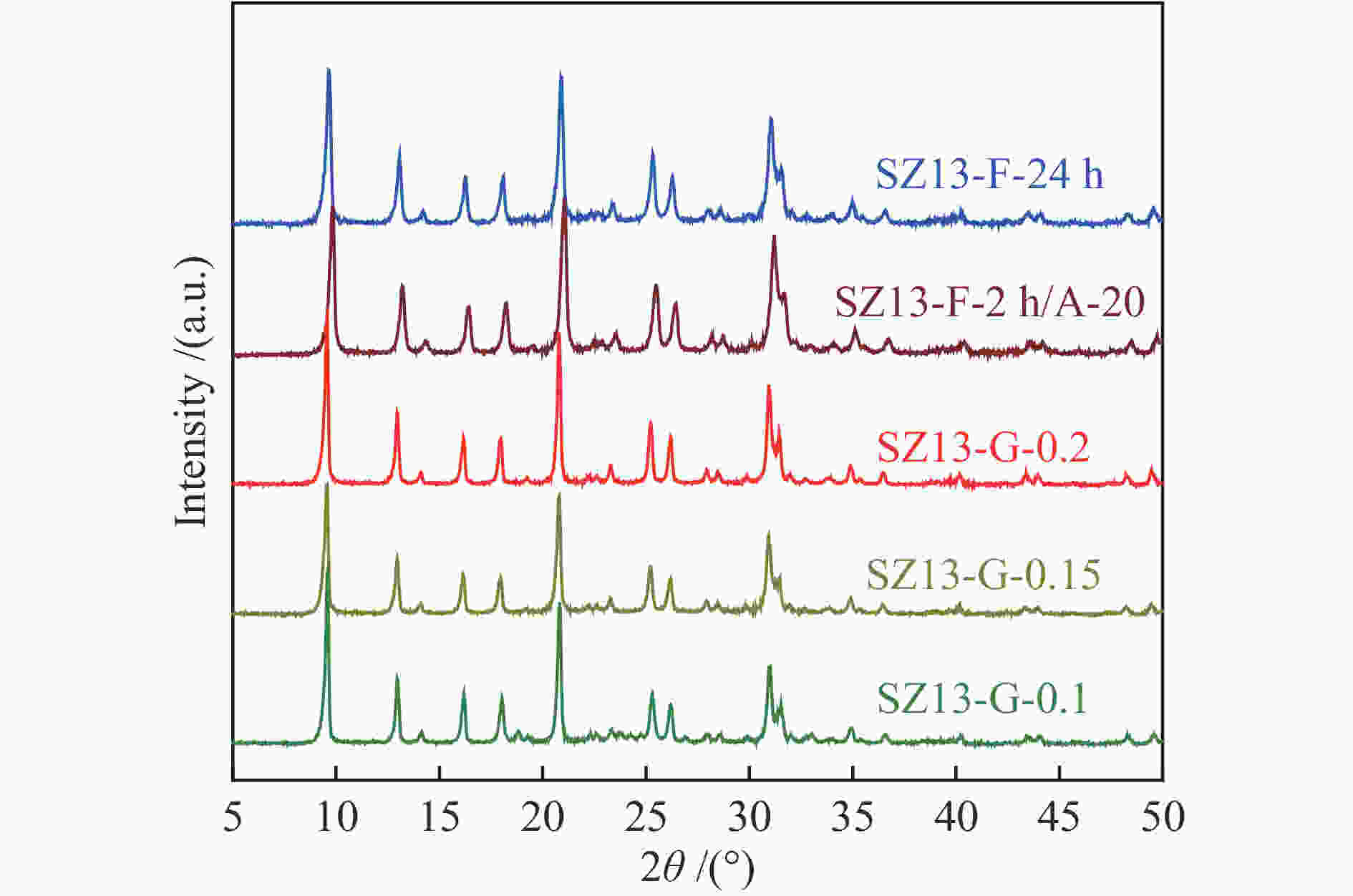
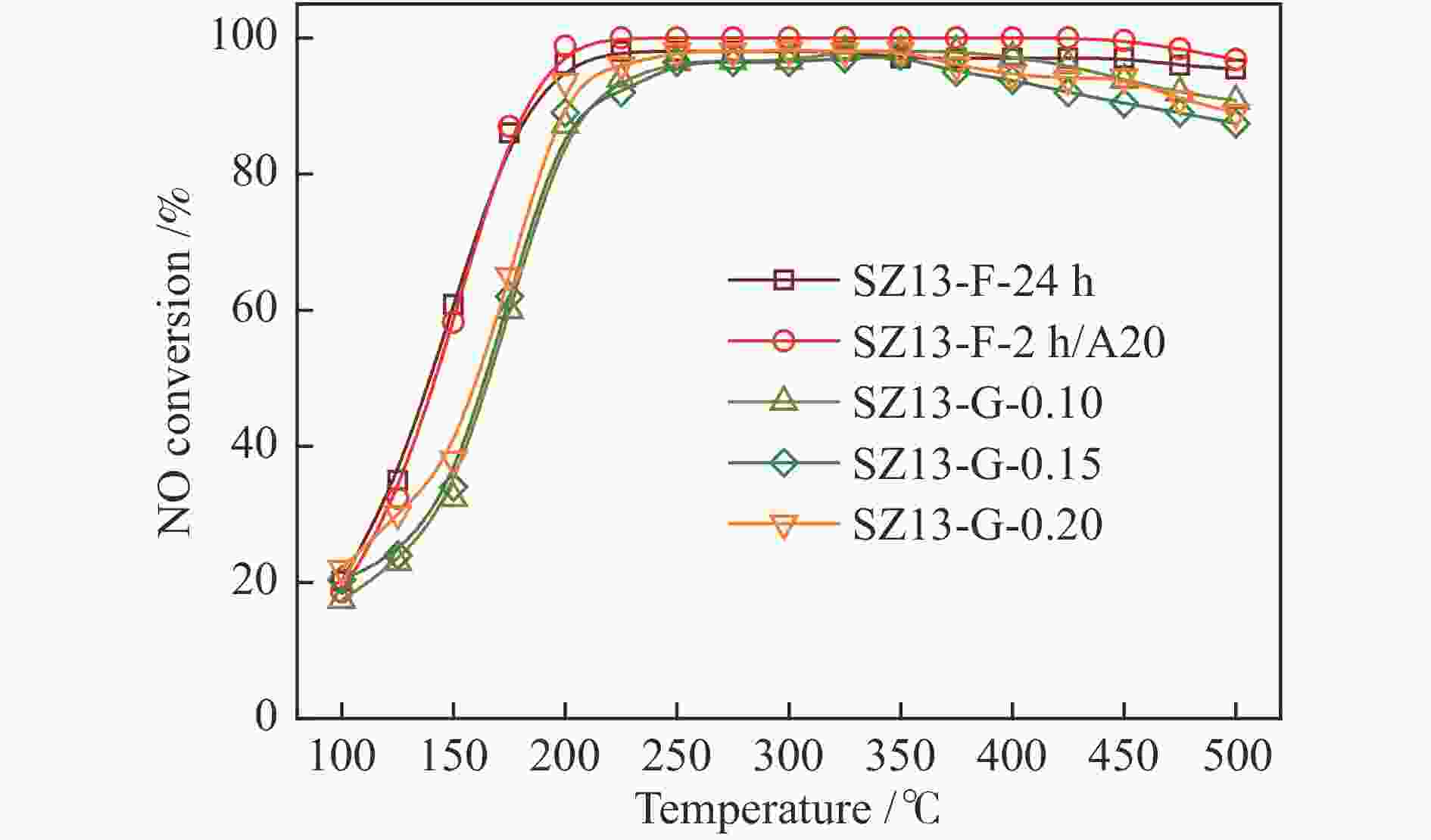
silicaAl2(SO4)3 0.4 88 2 0 8 88 0.7 0.17 1.6 487 SZ13-C-AlCl3 10 JN25 AlCl3 0.4 88 2 0 11 89 14.3 0.29 1.4 470 SZ13-C-Al(OH)3 10 JN25 Al(OH)3 0.4 88 2 0 18 87 0.9 0.18 1.6 480 SZ13-D-0 10 JN25 Al2(SO4)3 0 88 2 0 −0 SZ13-D-0.1 10 JN25 Al2(SO4)3 0.1 88 2 0 10 49 2.1 0.14 1.4 512 SZ13-D-0.2 10 JN25 Al2(SO4)3 0.2 88 2 0 9 61 0.9 0.16 1.6 527 SZ13-D-0.3 10 JN25 Al2(SO4)3 0.3 88 2 0 9 73 0.8 0.19 1.8 577 SZ13-E-44 10 JN25 Al2(SO4)3 0.4 44 2 0 10 74 0.4 0.18 1.7 571 SZ13-E-66 10 JN25 Al2(SO4)3 0.4 66 2 0 10 81 0.5 0.19 1.8 552 SZ13-F-24h 20 JN25 Al2(SO4)3 0.4 88 24 0 16 94 0.4 0.24 1.6 589 SZ13-G-0.2 20 JN25 Al2(SO4)3 0.4 88 2 0.2 20 83 1.1 0.25 1.3 547 SZ13-G-0.15 20 JN25 Al2(SO4)3 0.4 88 2 0.15 20 71 2.1 0.24 1.3 512 SZ13-G-0.1 20 JN25 Al2(SO4)3 0.4 88 2 0.1 19 77 4.8 0.27 1.5 533 Notes: The relative crystallinity (cryst.) was estimated by comparing the intensity of peaks at 9.6°, 20.6° and 30.6° in the XRD patterns of each zeolite sample to that of SZ13-A-100. The BET surface area (SBET) was determined from nitrogen sorption isotherms. The Si/Al and Cu/Al ratios and Cu loadings (LCu) were measured by ICP-AES. The average crystal size (dave) was estimated from the SEM images -
[1] KASPAR J, FORNASIERO P, HICKEY N. Automotive catalytic converters: current status and some perspectives[J]. Catal Today,2003,77:419−449. doi: 10.1016/S0920-5861(02)00384-X [2] 杨喻博, 刘世通, 徐威风. 车用NH3-SCR催化剂研究现状[J]. 内燃机与配件,2020,12(1):66−69.YANG Yu-bo, LIU Shi-tong, XU Wei-feng. Application research of NH3-SCR catalyst for diesel vehicle[J]. Int Combust Eng Parts,2020,12(1):66−69. [3] BEALE A, GAO F, LEZCANO G, PEDEN C, SZANYI J. Recent advances in automotive catalysis for NOx emission control by small-pore microporous materials[J]. Chem Soc Rev,2015,44:7371−7405. [4] 章凌, 段宏昌, 谭争国, 吴勤明, 孟祥举, 肖丰收. 用于柴油车尾气消除反应(NH3-SCR)的八元环沸石分子筛研究进展[J]. 高等学校化学学报,2020,41(1):19−27.ZHANG Ling, DUAN Hong-chang, TAN Zheng-guo, WU Qin-ming, MENG Xiang-ju, XIAO Feng-shou. Recent advances in the preparation of 8MR zeolites for the selective catalytic reduction of NO NH3-SCR in diesel engines[J]. Chem J Chin Univ,2020,41(1):19−27. [5] FICKEL D, ADDIO E, LAUTERBACH J, LOBO R. The ammonia selective catalytic reduction activity of copper-exchanged small-pore zeolites[J]. Appl Catal B: Environ,2011,102(1):441−448. [6] DEIMUND M, HARRISON L, LUNN J, LIU Y, MALEK A, SHAYIB R, DAVIS M. Effect of heteroatom concentration in SSZ-13 on the methanol to olefins reaction[J]. ACS Catal,2015,6:542−550. [7] LI Z, NAVARRO M T, TRIGUERO J M, YU J, CORMA A. Synthesis of nano-SSZ-13 and its application in the reaction of methanol to olefins[J]. Catal Sci Technol,2016,6:5856−5863. doi: 10.1039/C6CY00433D [8] XIE L, LIU F, REN L, SHI X, XIAO F, HE H. Excellent performance of one-pot synthesized Cu-SSZ-13 catalyst for the selective catalytic reduction of NOx with NH3[J]. Environ Sci Technol,2014,48:566−572. doi: 10.1021/es4032002 [9] KWAK J, TONKYN R, KIM D, SZANYI J, PEDEN C. Excellent activity and selectivity of Cu-SSZ-13 in the selective catalytic reduction of NOx with NH3[J]. J Catal,2010,275:187−190. doi: 10.1016/j.jcat.2010.07.031 [10] MAO Y, TIAN P, LIU Z. DMTO: A sustainable methanol-to-olefins technology[J]. Engineering,2021,7:17−21. doi: 10.1016/j.eng.2020.12.001 [11] WANG C, WANG J, YU T, SHEN M, WANG W, LI W. The effect of sulfate species on the activity of NH3-SCR over Cu/SAPO-34[J]. Appl Catal B: Environ,2017,204:239−249. doi: 10.1016/j.apcatb.2016.11.033 [12] XUE J, WANG X, QI G, WANG J, SHEN M, LI W. Characterization of copper species over Cu/SAPO-34 in selective catalytic reduction of NOx with ammonia: Relationships between active Cu sites and de-NOx performance at low temperature[J]. J Catal,2013,297:56−64. doi: 10.1016/j.jcat.2012.09.020 [13] WANG L, LI W, QI G, WENG D. Location and nature of Cu species in Cu/SAPO-34 for selective catalytic reduction of NO with NH3[J]. J Catal,2012,289:21−29. doi: 10.1016/j.jcat.2012.01.012 [14] ZONES S I. Zeolite SSZ-13 and its method of preparation: US, 4544538[P]. 1985-10-1. [15] REN L, ZHU L, YANG C, CHEN Y, SUN Q, ZHANG H, LI C, NAWAZ F, MENG X, XIAO F. Designed copper-amine complex as an efficient template for one-pot synthesis of Cu-SSZ-13 zeolite with excellent activity for selective catalytic reduction of NOx by NH3[J]. Chem Commun,2011,47:9789−9791. [16] KWAK J, TRAN D, BURTON S, SZANYI J, LEE J, PEDEN C. Effects of hydrothermal aging on NH3-SCR reaction over Cu/zeolites[J]. J Catal,2012,287:203−209. doi: 10.1016/j.jcat.2011.12.025 [17] LV W, MENG P, QIN Z, LI J, DONG M, WANG J, FAN W. A comparison of Al-rich Cu-SSZ-13 zeolites synthesized by different methods in their Al distribution, hydrothermal stability and catalytic performance in the selective catalytic reduction of NOx with NH3[J]. Microporous Mesoporous Mater,2021,313:110851. doi: 10.1016/j.micromeso.2020.110851 [18] IVANOVA S, LEBRUN C, VANHAECKE E, PHAM-HUU C, LOUIS B. Influence of the zeolite synthesis route on its catalytic properties in the methanol to olefin reaction[J]. J Catal,2009,265(1):1−7. doi: 10.1016/j.jcat.2009.03.016 [19] PRODINGER S, DEREWINSK M, WANG Y, WASHTON N, WALTER E, SZANYI J, GAO F, WANG Y, PEDEN C. Sub-micron Cu/SSZ-13: Synthesis and application as selective catalytic reduction (SCR) catalysts[J]. App Catal B: Environ,2017,201:461−469. doi: 10.1016/j.apcatb.2016.08.053 [20] 秦萱, 尹德嘉, 余丽泽, 张涛, 常化振, 朱燕, 刘国华, 李俊华. 硅铝比对Cu/SSZ-13 SCR活性位的影响[J]. 中国环境科学,2020,40(2):591−599. doi: 10.3969/j.issn.1000-6923.2020.02.015QIN Xuan, YIN De-jia, YU Li-zhe, ZHANG Tao, CHANG Hua-zhen, ZHU Yan, LIU Guo-hua, LI Jun-hua. Effect of Si/Al ratio on the SCR active sites of Cu/SSZ-13[J]. China Environ Sci,2020,40(2):591−599. doi: 10.3969/j.issn.1000-6923.2020.02.015 [21] GAO F, WASHTON N, WANG Y, KOLLAR M, SZANYI J, PEDEN C. Effects of Si/Al ratio on Cu/SSZ-13 NH3-SCR catalysts: Implications for the active Cu species and the roles of Bronsted acidity[J]. J Catal,2015,331:25−38. doi: 10.1016/j.jcat.2015.08.004 [22] LEZCANO-GONZALEZ I, DEKA U, ARSTAD B, VAN YPEREN-DE DEYNE A, HEMELSOET K, WAROQUIER M, VAN SPEYBROECK V, WECKHUYSEN B, BEALE A. Determining the storage, availability and reactivity of NH3 within Cu-chabazite-based ammonia selective catalytic reduction systems[J]. Phys Chem Chem Phys,2014,16:1639−1650. doi: 10.1039/C3CP54132K [23] SUZUKI K, NODA T, KATADA N, NIWA M. IRMS-TPD of ammonia: Direct and individual measurement of Brønsted acidity in zeolites and its relationship with the catalytic cracking activity[J]. J Catal,2001,250:151−160. [24] GAO F, WALTER E, KARP E, LUO J, TONKYN R, KWAK J, SZANYI J, PEDEN C. Structure-activity relationships in NH3-SCR over Cu-SSZ-13 as probed by reaction kinetics and EPR studies[J]. J Catal,2013,300:20−29. doi: 10.1016/j.jcat.2012.12.020 [25] LEE H, SONG I, JEON S, KIM D. Inter-particle migration of Cu ions in physically mixed Cu-SSZ-13 and H-SSZ-13 treated by hydrothermal aging[J]. React Chem Eng,2019,4:1059−1066. doi: 10.1039/C8RE00281A [26] 张伟, 张堃, 雍晓静, 王峰, 温鹏宇. 不同硅源合成ZSM-5分子筛及其MTP反应催化性能[J]. 天然气化工,2015,1:13−17.ZHANG Wei, ZHANG Kun, YONG Xiao-jing, WANG Feng, WEN Peng-yu. Effect of silica soures on synthesis of ZSM-5 and their catalytic properties for MTP reaction[J]. Nat Gas Chem Ind,2015,1:13−17. [27] 徐如人, 庞文琴, 于吉红, 霍启升, 陈接胜. 分子筛与多孔材料化学[M]. 1版. 北京: 科学出版社, 2004.XU Ru-ren, PANG Wen-qin, YU Ji-hong, HUO Qi-sheng, CHEN Jie-sheng. Chemistry Zeolites and Porous Materials[M]. First ed. Beijing: Science Press, 2004. [28] LV W, WANG S, WANG P, LIU Y, HUANG Z, LI J, DONG M, WANG J, FAN W. Regulation of Al distributions and Cu2+ locations in SSZ-13 zeolites for NH3-SCR of NO by different alkali metal cations[J]. J Catal,2021,393:190−201. doi: 10.1016/j.jcat.2020.11.027 [29] GAO, F, WANG Y, WASHTON N, KOLLAR M, SZANYI J, PEDEN C. Effects of alkali and alkaline earth cocations on the activity and hydrothermal stability of Cu/SSZ-13 NH3-SCR catalysts[J]. ACS Catal,2015,5:6780−6791. doi: 10.1021/acscatal.5b01621 [30] ZHOU H, MOUZON J, FARZANEH A, ANTZUTKIN O, GRAHN M, HEDLUND J. Colloidal defect-free silicalite-1 single crystals: Preparation, structure characterization, adsorption, and separation properties for alcohol/water mixtures[J]. Langmuir,2015,31:8488−8494. doi: 10.1021/acs.langmuir.5b02520 [31] TZANIS L, TRZPIT M, SOULARD M, PATARIN J. Energetic performances of STT-type zeosil: Influence of the nature of the mineralizing agent used for the synthesis[J]. J Phys Chem C,2012,116:4802−4808. doi: 10.1021/jp211819p [32] LI S, LI J, DONG M, FAN S, ZHAO T, WANG J, FAN W. Strategies to control zeolite particle morphology[J]. Chem Soc Rev,2019,48:885−907. doi: 10.1039/C8CS00774H [33] WIJAYANTI K, XIE K, KUMARB A, KAMASAMUDRAMB K, OLSSONAl L. Effect of gas compositions on SO2 poisoning over Cu/SSZ-13 used for NH3-SCR[J]. Appl Catal B: Environ,2017,219:142−154. -




 下载:
下载: