Effect of hydrothermal synthesis time on the performance of Cu/Ce-Zr catalysts for catalytic water-gas shift reaction
-
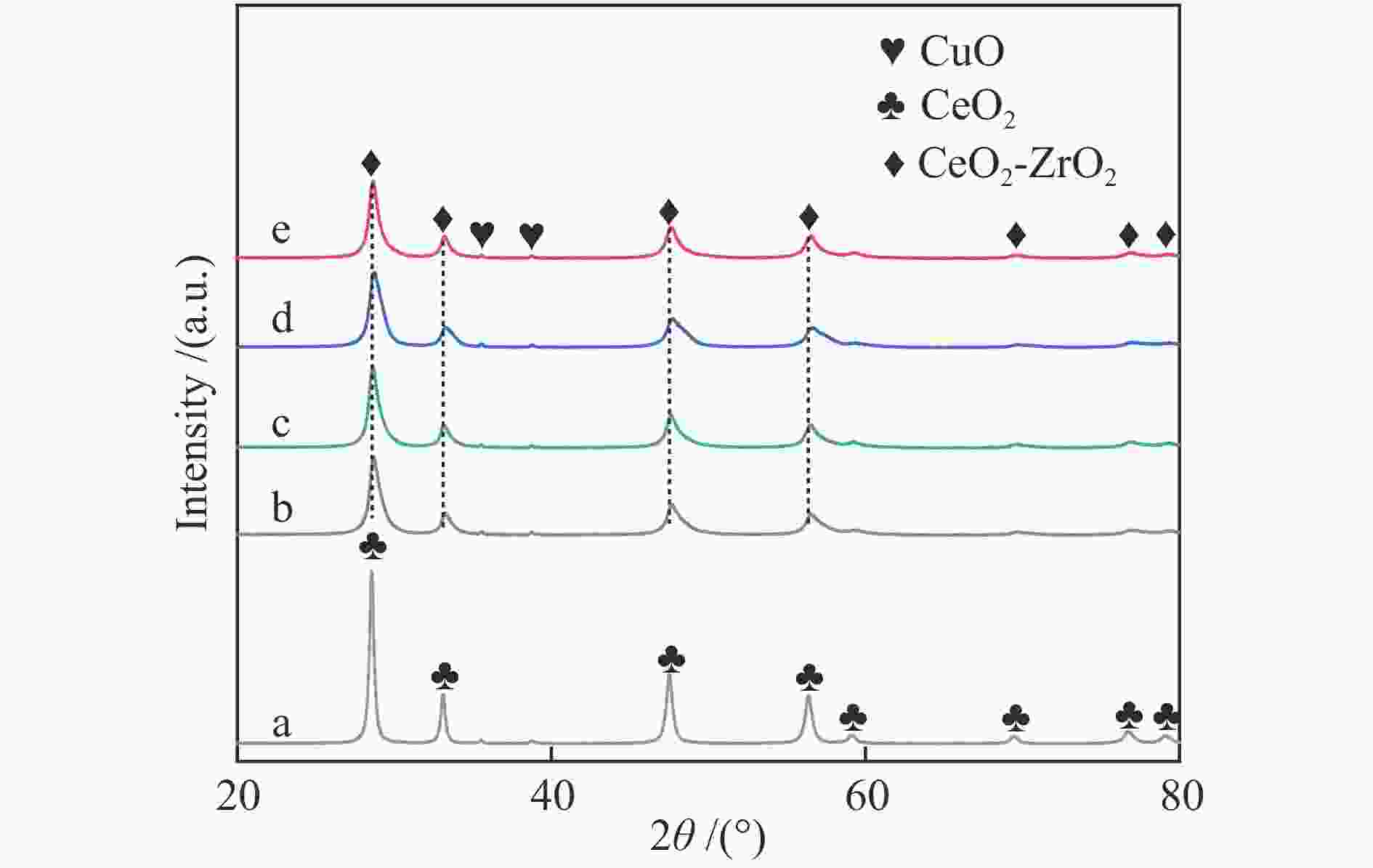
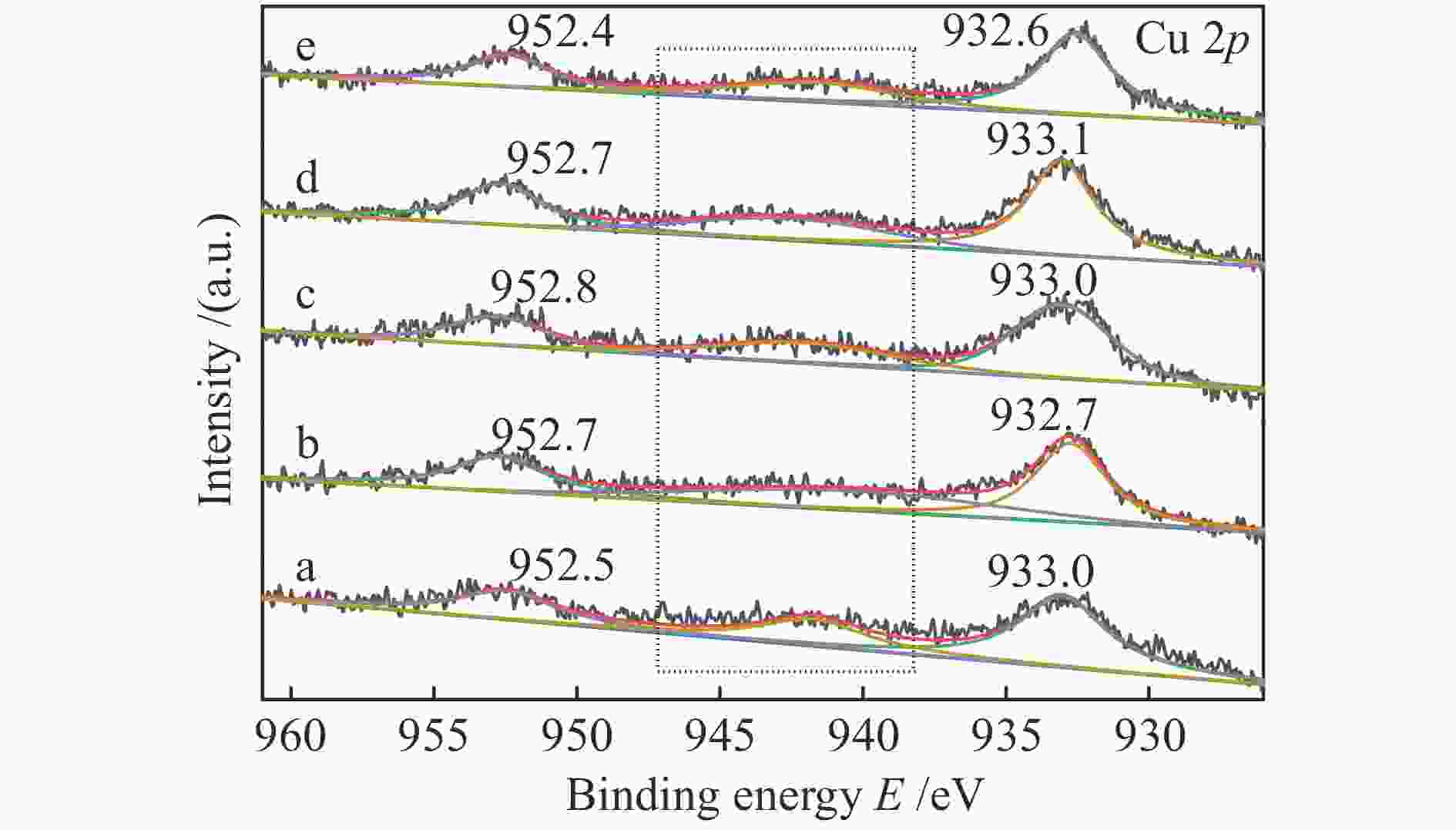
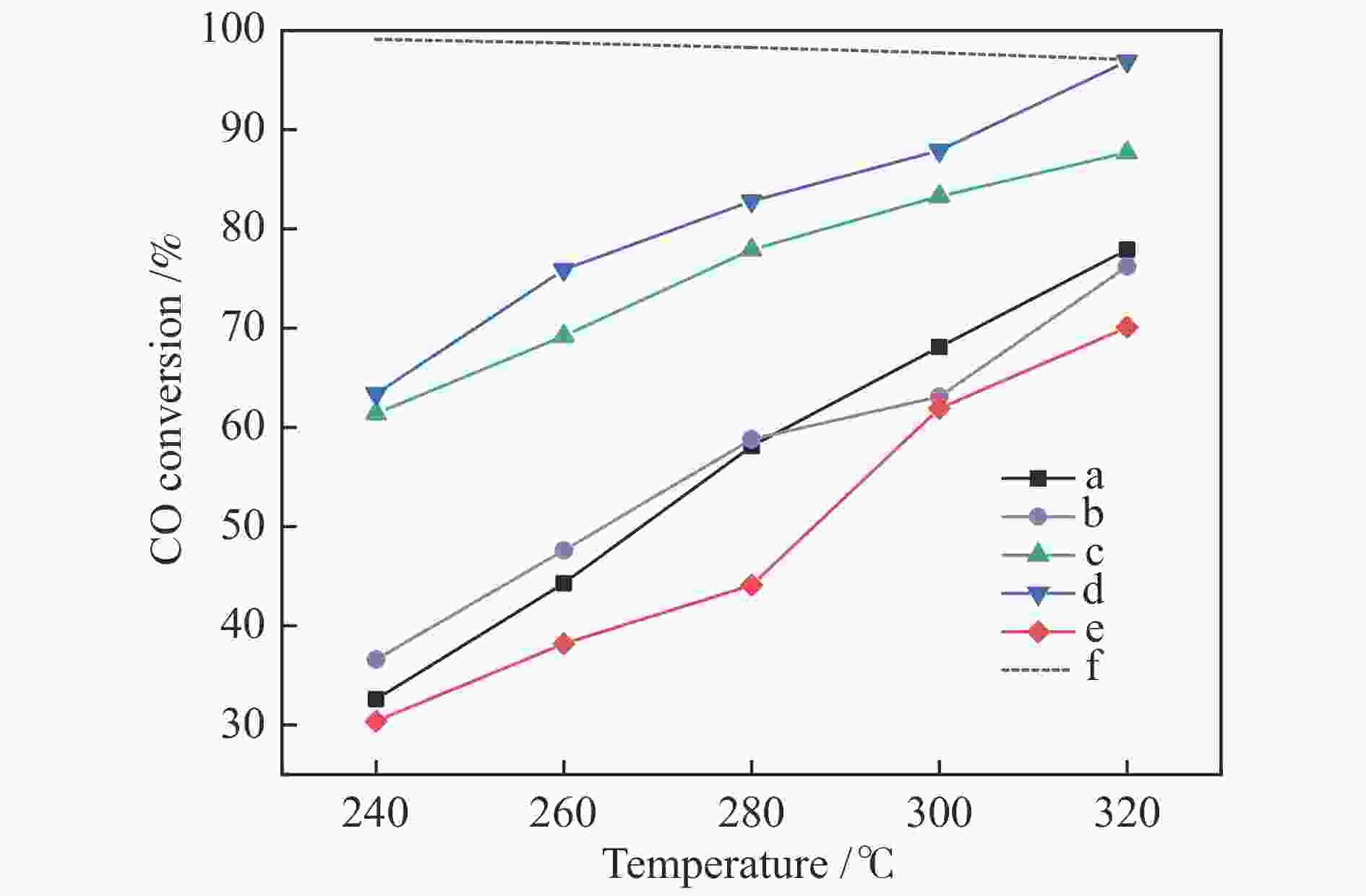
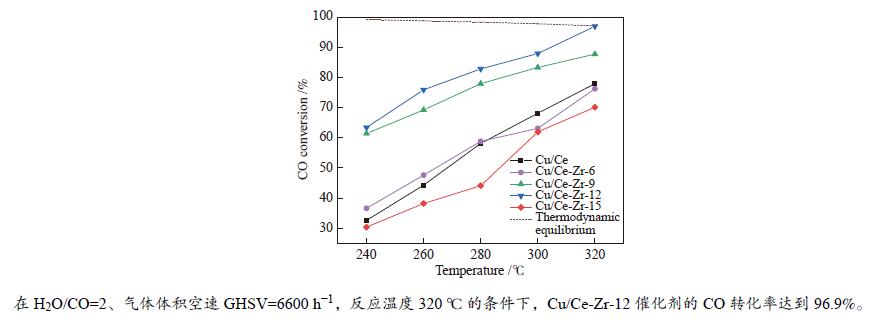
摘要: 以硝酸盐为铈、锆原料,以柠檬酸代替碱类沉淀剂,固定n(Zr∶Ce)为2∶8,采用水热法合成Ce-Zr氧化物载体,再通过浸渍法制备Cu/Ce-Zr催化剂。通过XRD、BET、H2-TPR、XPS等手段对载体和催化剂进行表征,研究水热时间对催化剂结构、性质和水气变换反应性能的影响。结果表明,催化活性主要与Cu比表面积、CuO的还原温度以及催化剂表面氧空位含量有关。其中,Cu/Ce-Zr-12催化剂的Cu比表面积较大、CuO的还原温度较低,催化剂表面的氧空位数量较多,表现出较好的催化活性。在320 ℃、水气比(W∶M)为2,体积空速GHSV=6600 h−1的反应条件下,CO转化率为96.9%,与热力学平衡值97.1%接近。
-
关键词:
- 水热法 /
- 水热时间 /
- 水气变换 /
- CeO2-ZrO2固溶体
Abstract: Ce-Zr oxide support was hydrothermally synthesized from metal nitrates of cerium and zirconium as the raw materials using citric acid instead of alkali precipitant, and then Cu/Ce-Zr catalyst was prepared by the impregnation method. The support and catalyst samples were characterized by XRD, BET, H2-TPR, XPS techniques, and the effects of different hydrothermal time on the structure, properties and performance in water-gas shift reaction were investigated. The results show that the catalyst activity is mainly related to the Cu specific surface area, reduction temperature of CuO and the number of oxygen vacancies on the catalyst surface. Among them, the Cu/Ce-Zr catalyst with hydrothermal time of 12 h has a large Cu specific surface area, a lower reduction temperature of CuO, and a large number of oxygen vacancies, so it shows a good catalytic activity. When the reaction temperature is 320 ℃, the molar ratio of water to gas (W/M) is 2, and the gas space velocity GHSV=6600 h−1, the CO conversion rate is 96.9%, which is close to the thermodynamic equilibrium value of 97.1%.-
Key words:
- hydrothermal method /
- hydrothermal time /
- water gas shift /
- CeO2-ZrO2 solid solution
-
表 1 Ce-Zr-t和Cu/Ce-Zr-t的物化性质
Table 1 Physical and chemical properties of Ce-Zr-t and Cu/Ce-Zr-t
Catalyst Cell parameters /nm SBET
/(m2·g−1)Pore volume /(mL·g−1) Cu surface areaa /(m2·g−1) CeO2 0.54075 23.9 0.08 − Ce-Zr-6 0.53881 71.5 0.13 − Ce-Zr-9 0.54017 76.8 0.14 − Ce-Zr-12 0.53875 69.2 0.15 − Ce-Zr-15 0.54026 61.8 0.10 − Cu/Ce 0.54073 21.2 0.07 1.5 Cu/Ce-Zr-6 0.53871 56.9 0.11 3.7 Cu/Ce-Zr-9 0.53944 60.6 0.12 4.9 Cu/Ce-Zr-12 0.53732 58.8 0.14 5.5 Cu/Ce-Zr-15 0.53926 50.2 0.09 3.5 a: determined by N2O experiments 表 2 CuO还原峰位置
Table 2 Reduction peak positions of CuO
Catalyst Peak position t/℃ peak α peak β Cu/Ce-Zr-6 156 182 Cu/Ce-Zr-9 155 181 Cu/Ce-Zr-12 144 171 Cu/Ce-Zr-15 157 189 表 3 催化剂Ce 3d和O 1s的XPS曲线拟合
Table 3 XPS curve fitting results of catalysts Ce 3d and O 1s
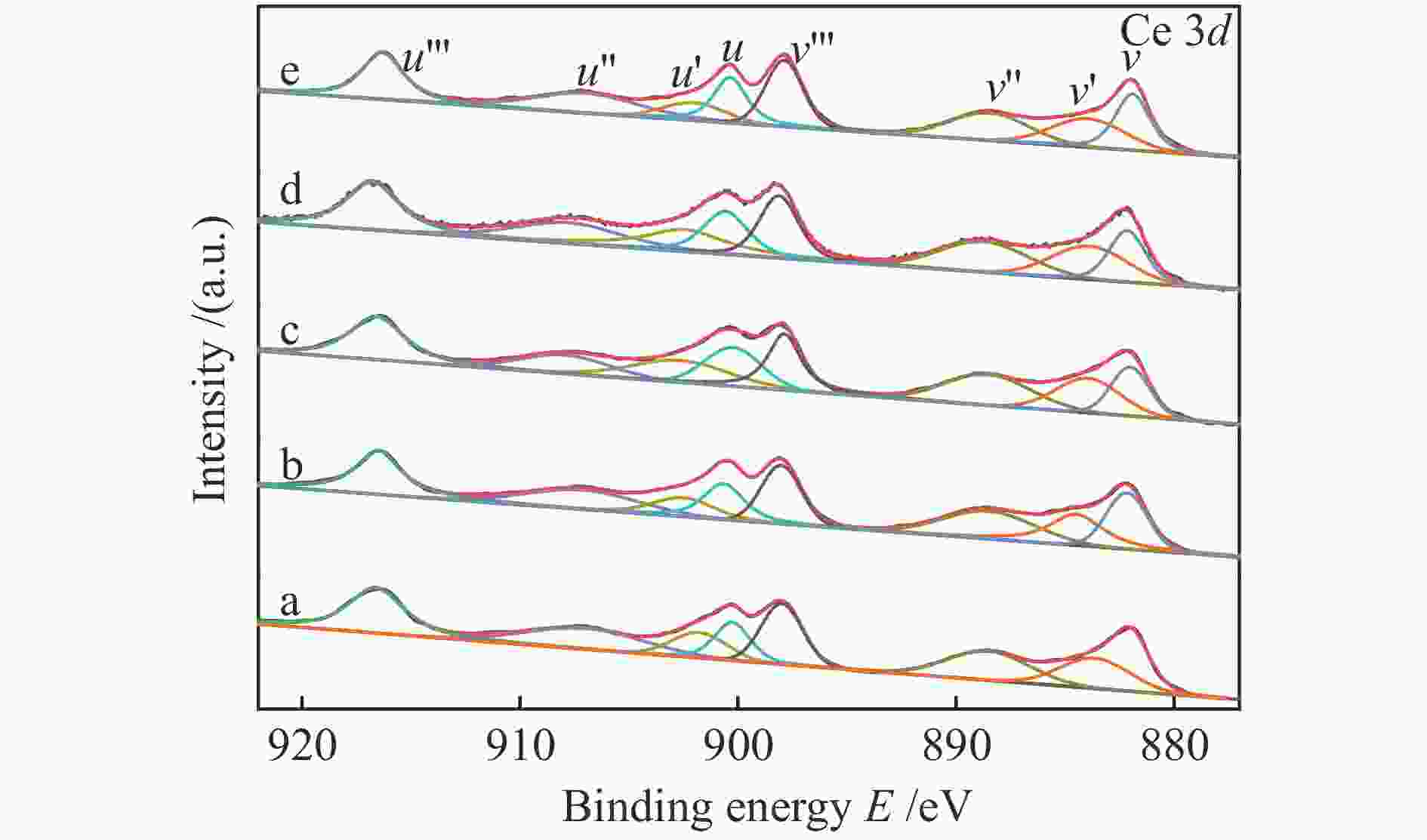
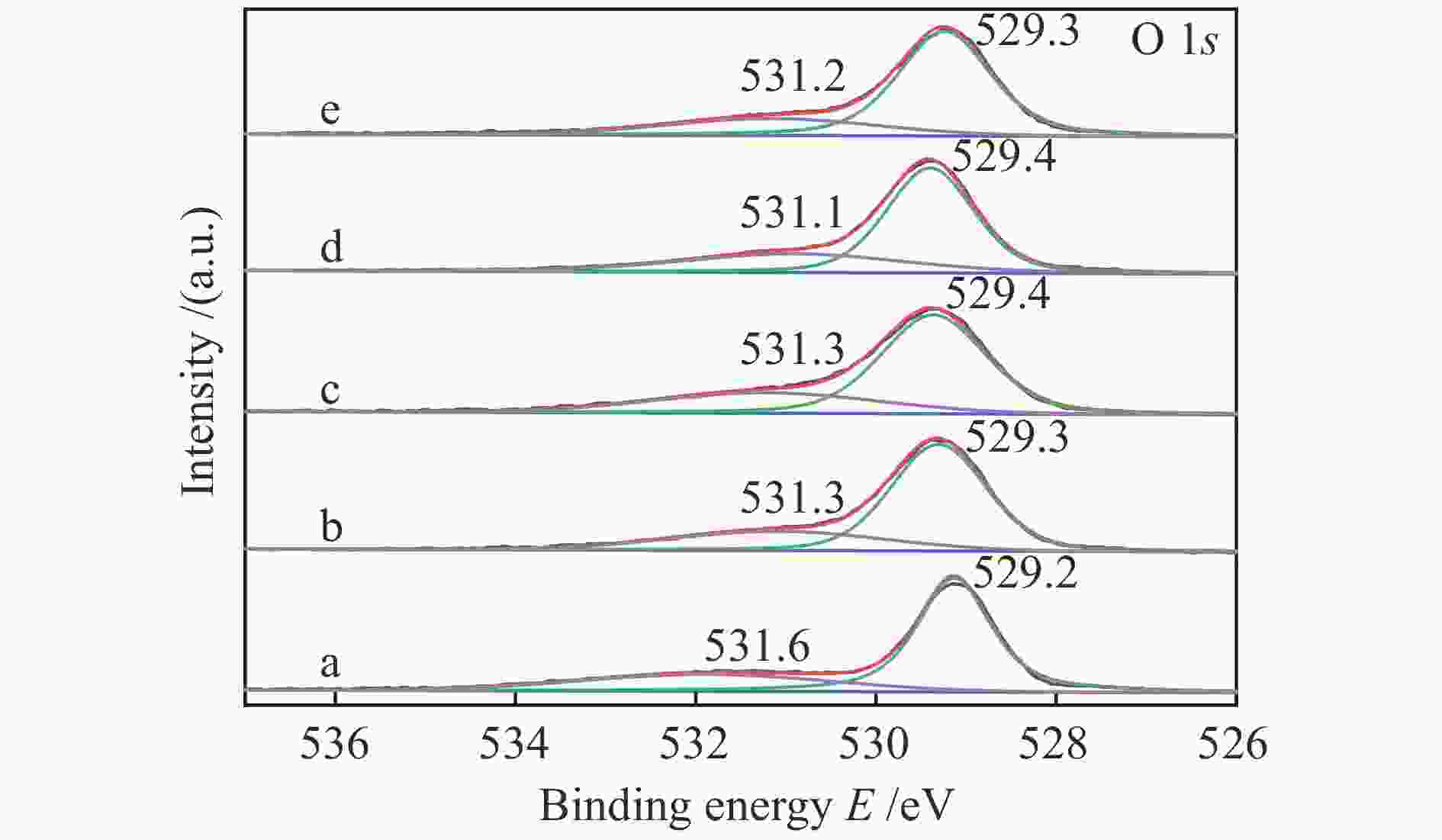
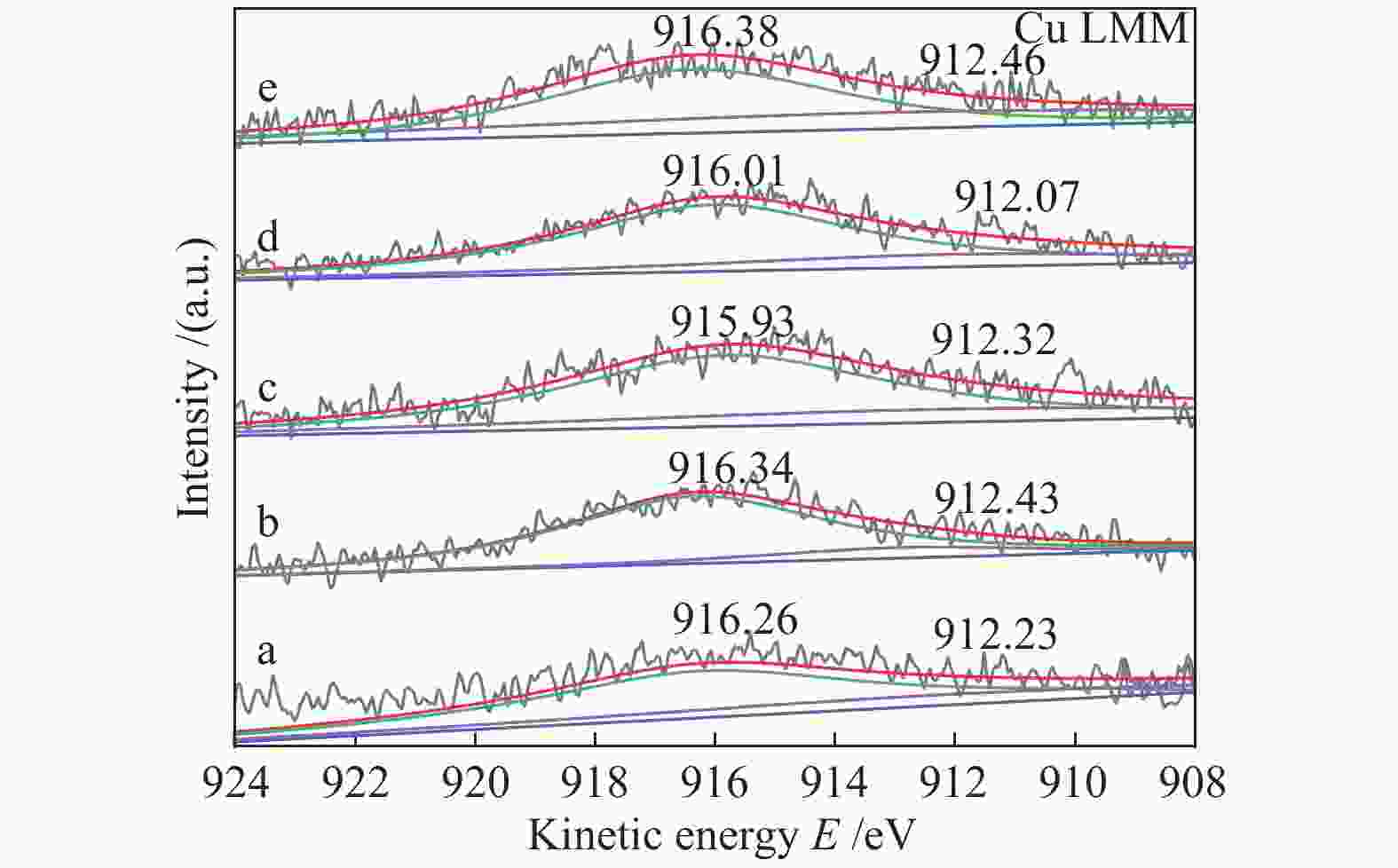
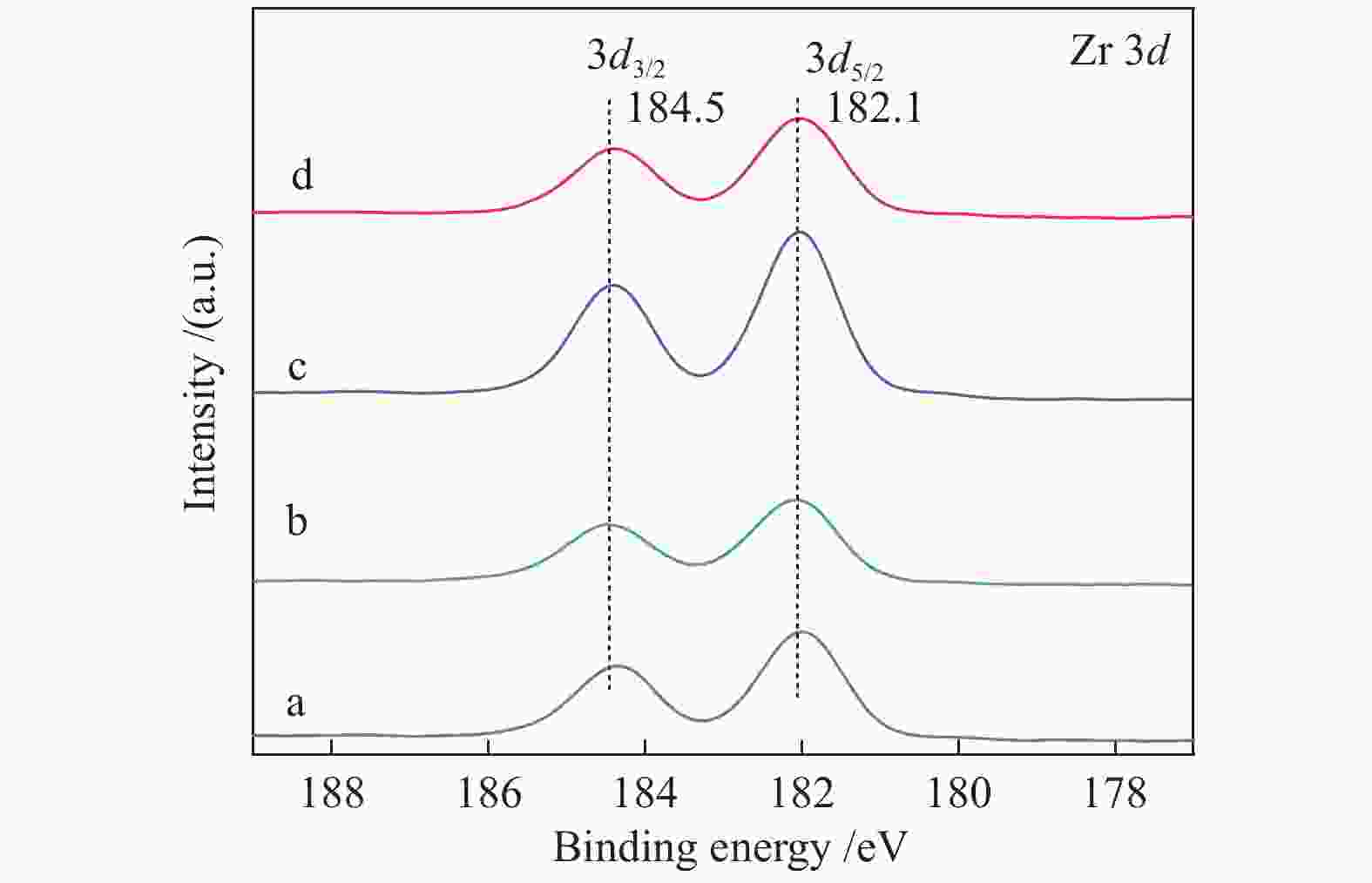
Catalyst Ce3+/(Ce3+ + Ce4+)/% Oads/(Olat + Oads)/% Cu/Ce 24.2 28.2 Cu/Ce-Zr-6 21.9 26.7 Cu/Ce-Zr-9 24.4 28.7 Cu/Ce-Zr-12 28.6 31.1 Cu/Ce-Zr-15 18.3 24.5 -
[1] LEVALLEY T L, RICHARD A R, FAN M. The progress in water gas shift and steam reforming hydrogen production technologies-A review[J]. Int J Hydrog Energy,2014,39(30):16983−17000. doi: 10.1016/j.ijhydene.2014.08.041 [2] RATNASAMY C, WAGNER J P. Water gas shift catalysis[J]. Catal Rev Sci Eng,2009,51(3):325−440. doi: 10.1080/01614940903048661 [3] YAO S Y, ZHANG X, ZHOU W, GAO R, XU W Q. Atomic-layered Au clusters on α-MoC as catalysts for the low-temperature water-gas shift reaction[J]. Science,2017,357(6349):389−393. doi: 10.1126/science.aah4321 [4] KARPENKO A, LEPPELT R, CAI J, PLZAK V, CHUVULIN A, KAISER U, BEHM R. J. Deactivation of a Au/CeO2 catalyst during the low-temperature water-gas shift reaction and its reactivation: A combined TEM, XRD, XPS, DRIFTS, and activity study[J]. J Catal,2007,250(1):139−150. doi: 10.1016/j.jcat.2007.05.016 [5] JEONG D W, POTDAR H S, SHIM J O, JANG W J, ROH H S. H2 production from a single stage water- gas shift reaction over Pt/CeO2, Pt/ZrO2, and Pt/Ce(1−x)Zr(x)O2 catalysts[J]. Int J Hydrog Energy,2013,38(11):4502−4507. doi: 10.1016/j.ijhydene.2013.01.200 [6] LEE J Y, LEE D W, LEE K Y, WANG Y. Cr-free Fe-based metal oxide catalysts for high temperature water gas shift reaction of fuel processor using LPG[J]. Catal Today,2009,146(1/2):260−264. doi: 10.1016/j.cattod.2009.01.041 [7] LEE Y L, JHA A, JANG W J, SHIM J O, JEON K W, NA H S, KIM H M, LEE D W. Optimization of Cobalt Loading in Co-CeO2 Catalyst for the High Temperature Water-Gas Shift Reaction[J]. Top Catal,2017,60:721−726. doi: 10.1007/s11244-017-0776-2 [8] PAWEL K, MARCIN K, KATARZYNA A, WIESLAW P, PAWEL W. The effect of the precursor ageing on properties of the Cu/ZnO/Al2O3 catalyst for low temperature water-gas shift (LT-WGS)[J]. Mol Catal,2014,392(7):127−133. [9] REINA T R, IVANOVA S, CENTENO M A, ODRIOZOLA J A. The role of Au, Cu & CeO2 and their interactions for an enhanced WGS performance[J]. Appl Catal B: Environ,2016,187:98−107. doi: 10.1016/j.apcatb.2016.01.031 [10] 王润平, 毛树红, 段秀琴, 李文斌, 王齐, 池永庆. 不同载体Ni基负载型催化剂对甲烷部分氧化制合成气催化行为研究[J]. 燃料化学学报,2015,43(2):228−243. doi: 10.3969/j.issn.0253-2409.2015.02.014WANG Run-ping, MAO Shu-hong, DUAN Xiu-qin, LI Wen-bin, WANG Qi, CHI Yong-qing. Catalytic performance of nickel-based catalysts with different supports in partial oxidation of methane to synthesis gas[J]. J Fuel Chem Technol,2015,43(2):228−243. doi: 10.3969/j.issn.0253-2409.2015.02.014 [11] 王东哲, 冯旭, 张健, 陈琳, 张磊, 王宏浩, 白金, 张财顺, 张政一. 助剂M(M=Cr、Zn、Y、La)对甲醇水蒸气重整制氢CuO/CeO2催化剂的影响[J]. 燃料化学学报,2019,47(10):1251−1257. doi: 10.3969/j.issn.0253-2409.2019.10.012WANG Dong-zhe, FENG Xu, ZHANG Jian, CHEN Lin, ZHANG Lei, WANG Hong-hao, BAI Jin, ZHANG Cai-shun, ZHANG Zheng-yi. Effect of promoter M(M=Cr, Zn, Y, La) on CuO/CeO2 catalysts for hydrogen production from steam reforming of methanol[J]. J Fuel Chem Technol,2019,47(10):1251−1257. doi: 10.3969/j.issn.0253-2409.2019.10.012 [12] ZHANG L, PAN L W, NI C J, SUN T J, ZHAO S S, WANG S D, WANG A J, HU Y K. CeO2-ZrO2-promoted CuO/ZnO catalyst for methanol steam reforming[J]. Int J Hydrog Energy,2013,38:4397−4406. doi: 10.1016/j.ijhydene.2013.01.053 [13] JEONG D W, NA H S, SHIM J O, JANG W J, ROH H S. A crucial role for the CeO2-ZrO2 support for the low temperature water gas shift reaction over Cu-CeO2-ZrO2 catalysts[J]. Catal Sci Technol,2015,5:3706−3713. doi: 10.1039/C5CY00499C [14] SHI L M, GAO C L, GUO F H, WANG Y J, ZHANG T B. Catalytic performance of Zr-doped CuO-CeO2 oxides for CO selective oxidation in H2-rich stream[J]. J Rare Earths,2019,37:720−725. doi: 10.1016/j.jre.2019.01.003 [15] GUO X L, ZHOU R X. Identification of the nano/micro structure of CeO2 (rod) and the essential role of interfacial copper-ceria interaction in CuCe(rod) for selective oxidation of CO in H2-rich streams[J]. J. Power Sources,2017,361(sep.1):39−53. [16] 刘玉娟, 许骥, 佟宇飞, 张娜, 张磊, 刘道胜, 韩蛟, 张财顺. 氧化铈纳米材料合成方法的研究进展[J]. 辽宁石油化工大学学报,2017,37(5):8−12 + 37. doi: 10.3969/j.issn.1672-6952.2017.05.002LIU Yu-juan, XU Ji, TONG Yu-fei, ZHANG Na, ZHANG Lei, LIU Dao-sheng, HAN Jiao, ZHANG Cai-shun. Progress in research of the synthesis methods of nanometer ceria[J]. J Liaoning Petrochem Univ,2017,37(5):8−12 + 37. doi: 10.3969/j.issn.1672-6952.2017.05.002 [17] 王丽宝, 王宏浩, 张磊, 庆绍军, 刘冬梅, 高志贤, 张海娟, 官国清. 柠檬酸量对水热合成CuO/Ce0.8Zr0.2O2催化水气变换制氢性能的影响[J]. 燃料化学学报,2022,50(3):337−345.WANG Li-bao, WANG Hong-hao, ZHANG Lei, QING Shao-jun, LIU Dong-mei, GAO Zhi-xian, ZHANG Hai-juan, GUAN Guo-qing. Effect of citric acid content on the hydrothermal synthesis of CuO/Ce0.8Zr0.2O2 catalytic water gas shift hydrogen production performance[J]. J Fuel Chem Technol,2022,50(3):337−345. [18] 焦桐, 许雪莲, 张磊, 翁幼云, 翁玉冰, 高志贤. CuO/CeO2-ZrO2/SiC整体催化剂催化甲醇水蒸气重整制氢的研究[J]. 化学学报,2021,79(4):513−519. doi: 10.6023/A20120562JIAO Tong, XU Xue-lian, ZHANG Lei, WENG You-yun, WENG Yu-bing, GAO Zhi-xian. Research on CuO/CeO2-ZrO2/SiC monolithic catalysts for hydrogen production by methanol steam reforming[J]. Acta Chim Sin,2021,79(4):513−519. doi: 10.6023/A20120562 [19] 贺建平, 张磊, 陈琳, 杨占旭, 佟宇飞. CeO2改性Cu/Zn-Al水滑石衍生催化剂对甲醇水蒸气重整制氢性能的影响[J]. 高等学校化学学报,2017,38(10):1822−1828. doi: 10.7503/cjcu20170158HE Jian-ping, ZHANG Lei, CHEN Lin, YANG Zhan-xu, TONG Yu-fei. Effect of CeO2 on Cu/Zn-Al catalysts derived from hydrotalcite precursor for methanol steam reforming[J]. Chem J Chin Univ,2017,38(10):1822−1828. doi: 10.7503/cjcu20170158 [20] 张宣娇, 孙羽, 刘明, 郝书敏, 杨涛, 张磊, 白金, 韩蛟. CeO2形貌结构对催化湿式空气氧化苯酚性能的影响[J]. 中国环境科学,2020,40(10):4330−4334. doi: 10.3969/j.issn.1000-6923.2020.10.016ZHANG Xuan-jiao, SUN Yu, LIU Ming, HAO Shu-min, YANG Tao, ZHANG Lei, BAI Jin, HAN Jiao. Effect of morphology on the performance of CeO2 for catalytic wet air oxidation of phenol[J]. Chin Environ Sci,2020,40(10):4330−4334. doi: 10.3969/j.issn.1000-6923.2020.10.016 [21] 肖国鹏, 乔韦军, 张磊, 庆绍军, 张财顺, 高志贤. 钙钛矿型甲醇水蒸气重整制氢催化材料的研究[J]. 化学学报,2021,79(1):100−107. doi: 10.6023/A20080374XIAO Guo-peng, QIAO Wei-jun, ZHANG Lei, QING Shao-jun, ZHANG Cai-shun, GAO Zhi-xian. Study on hydrogen production catalytic materials for perovskite methanol steam reforming[J]. Acta Chim Sin,2021,79(1):100−107. doi: 10.6023/A20080374 [22] YANG S Q, ZHOU F, LIU Y J, ZHANG L, CHEN Y, WANG H H, TIAN Y, ZHANG C S, LIU D S. Morphology effect of ceria on the performance of CuO/CeO2 catalysts for hydrogen production by methanol steam reforming[J]. Int J Hydrog Energy,2019,44(14):7252−7261. doi: 10.1016/j.ijhydene.2019.01.254 [23] YANG K, LIU J F, SI R R, CHEN X, DAI W X, FU X Z. Comparative study of Au/TiO2 and Au/Al2O3 for oxidizing CO in the presence of H2 under visible light irradiation[J]. J Catal,2014,317:229−239. doi: 10.1016/j.jcat.2014.06.005 [24] BIESINGER M C. Advanced analysis of copper X-ray photoelectron spectra[J]. Surf Interface Anal,2017,49:1325−1334. doi: 10.1002/sia.6239 -





 下载:
下载: