Fischer-Tropsch synthesis performance of cobalt-based catalysts supported on bimodal porous SiO2 with high specific surface area
-
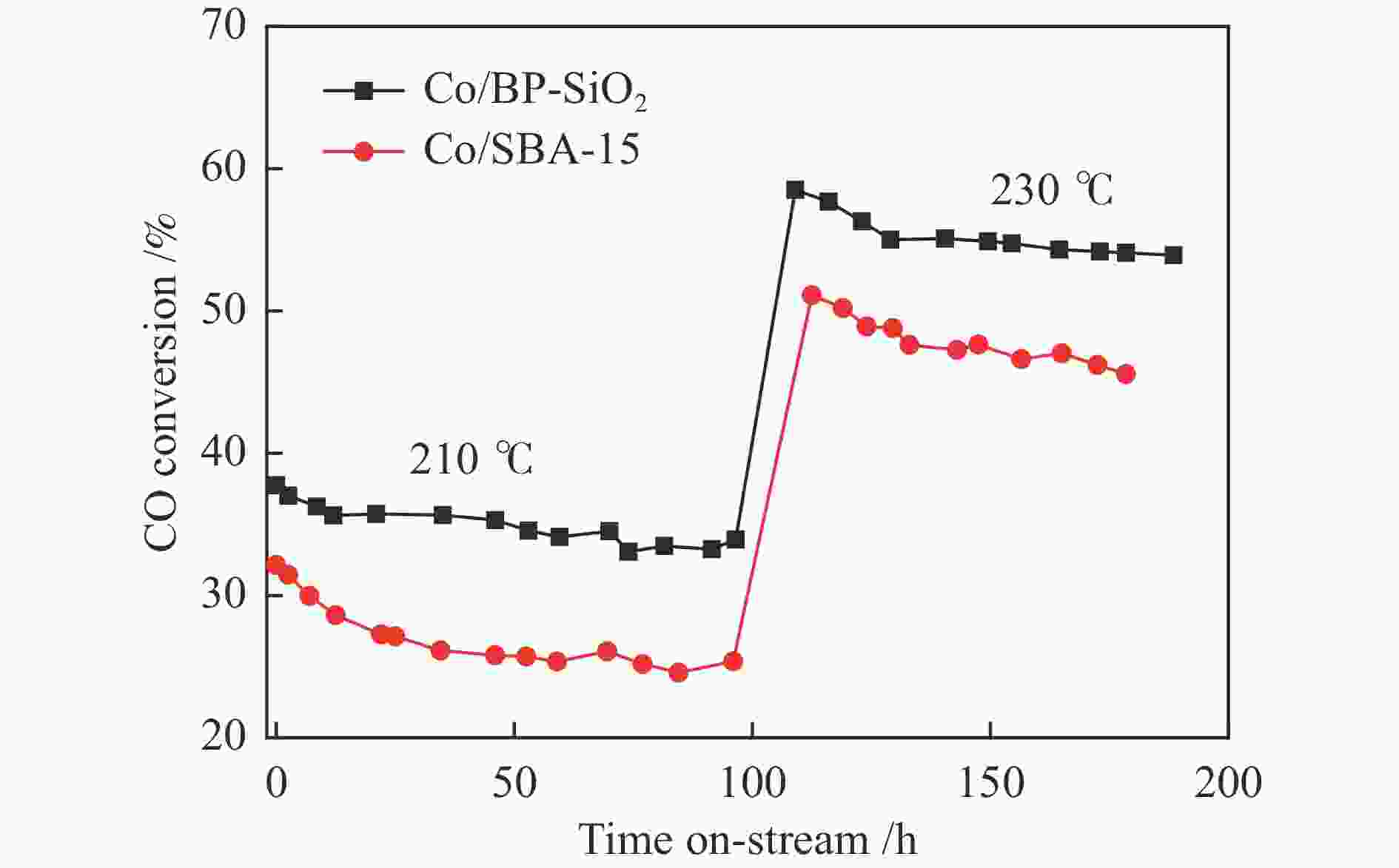
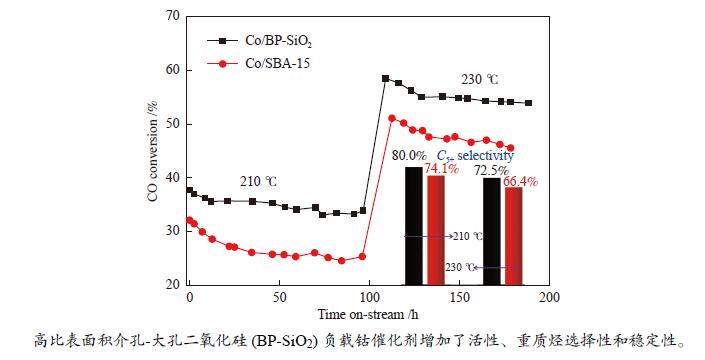
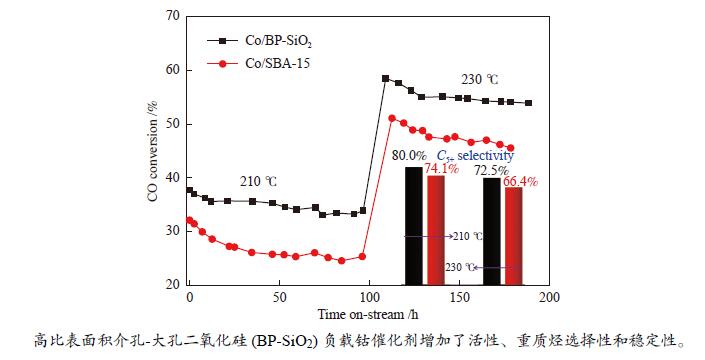
摘要: 载体的结构可以显著影响钴基费托合成催化剂的活性和产物选择性。大孔结构载体可以改善反应物和产物的传质情况,提升CO转化活性和C5+ 产物选择性;高比表面积载体有利于使负载的金属分散,提高催化剂的金属利用效率和稳定性。然而,要获得同时具备高比表面积和大孔结构特征的载体相对困难。本研究采用结构导向水解法,合成了一种比表面积达1103.2 m2/g的介孔(2.9 nm)-大孔(63.8 nm)双孔二氧化硅(BP-SiO2)载体,研究了其负载钴催化剂的费托合成反应性能。结果表明,相对规整介孔SBA-15分子筛负载的钴催化剂Co/SBA-15,210 ℃反应时,催化剂Co/BP-SiO2的CO转化率提高33.3%,CH4选择性降低30.1%,C5+ 选择性增加到80.0%,稳定性显著增强。Abstract: The structure of the supports can significantly affect the Fischer-Tropsch catalyst activity and selectivity. The porous structure can improve the mass transfer of reactants, enhance the CO conversion activity and C5+ product selectivity; the high specific surface area is beneficial to disperse the loaded metal, improve the catalyst metal utilization efficiency and catalyst stability. However, it is relatively difficult for supports to obtain high specific surface area and macropore structure characteristics simultaneously. A mesoporous (2.9 nm) -macroporous (63.8 nm) bi-porous silica (BP-SiO2) support with a high specific surface area of 1103.2 m2/g was synthesized by the structure-directed hydrolysis method, and its catalytic performance for Fischer-Tropsch synthesis was investigated. The results showed that compared to the Co/SBA-15 catalyst with equivalent mesopore diameter, the catalyst Co/BP-SiO2 showed CO conversion rate nearly increased by 33.3%, CH4 selectivity reduced by 30.1%, improved C5+ selectivity and stability.
-
Key words:
- Fischer-Tropsch synthesis /
- bimodal pore /
- cobalt based catalyst
-
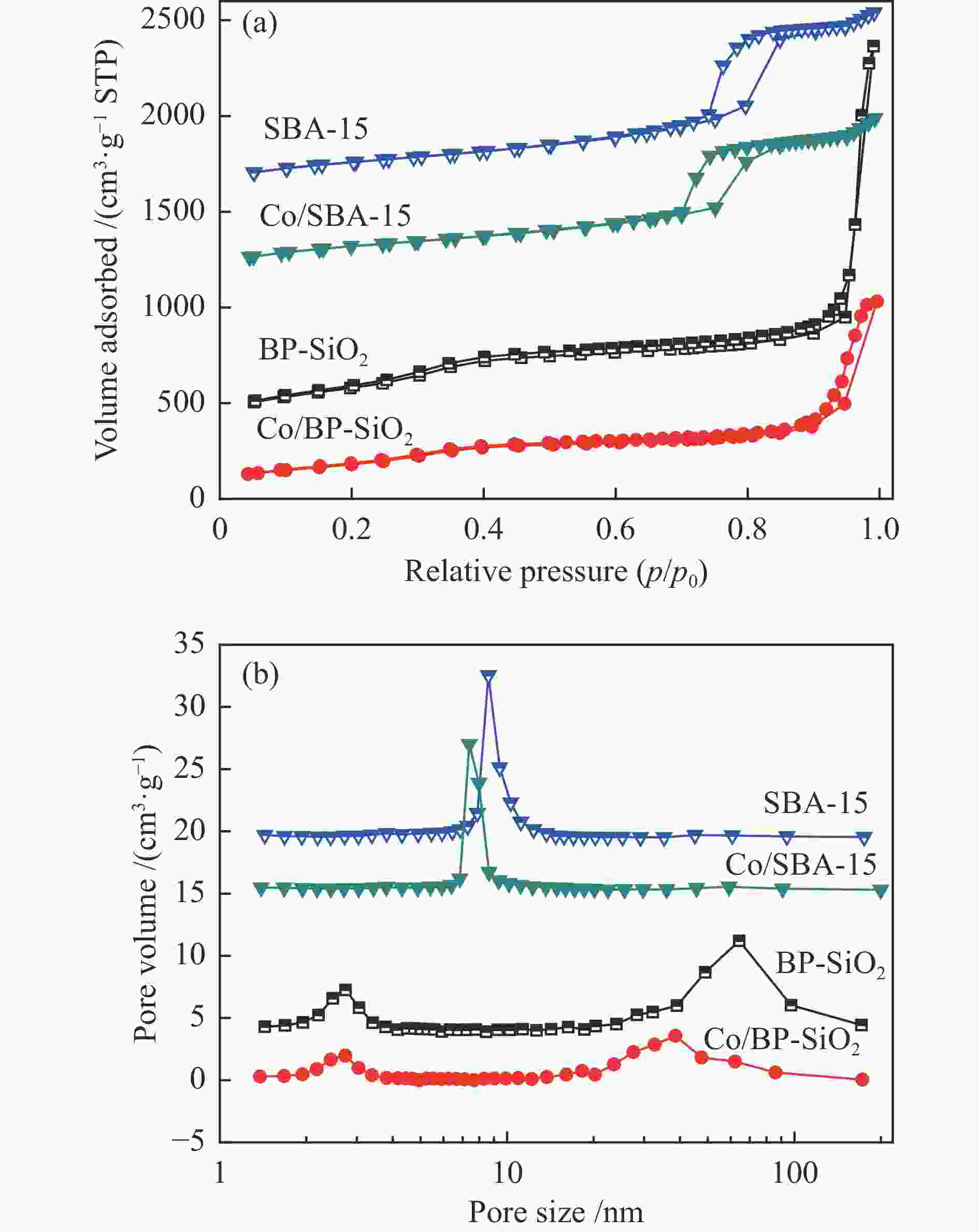
表 1 催化剂及载体的物性参数、催化剂的Co粒径参数
Table 1 Textural properties of various catalysts and support, cobalt particle size of the catalysts
Sample SBET/
(m2·g−1)Average pore
size a /nmBJH pore
size /nmPore volume /
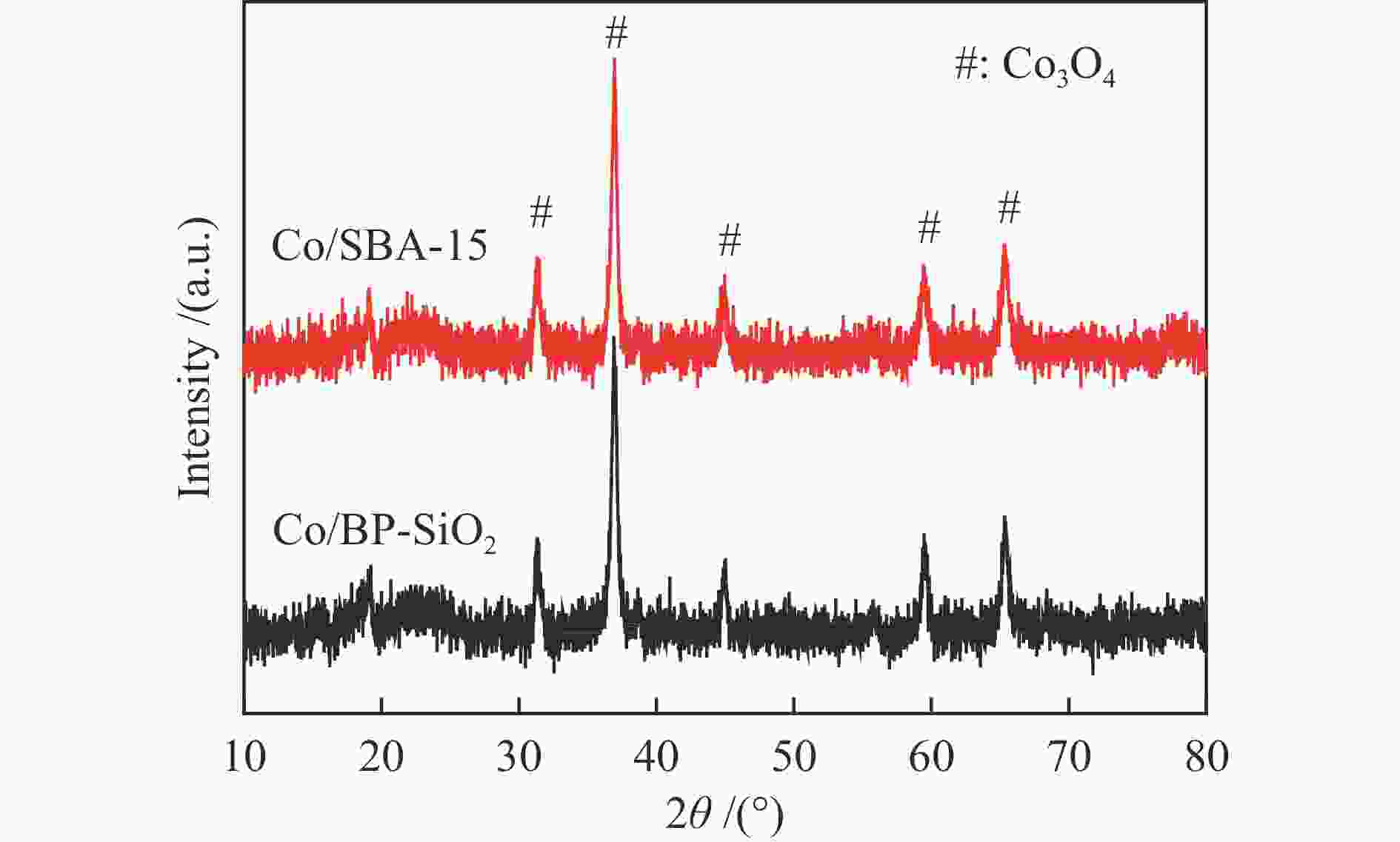
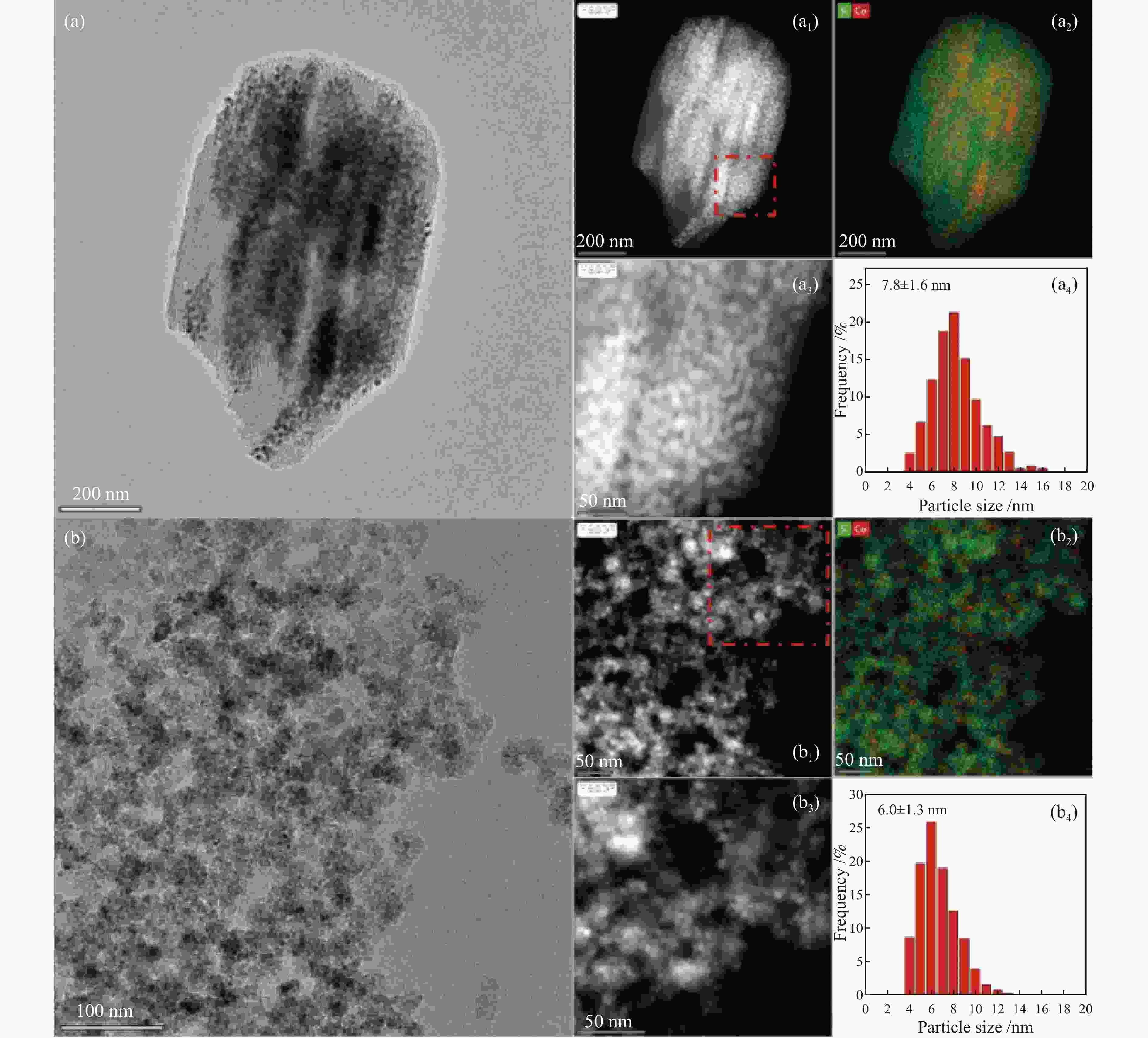
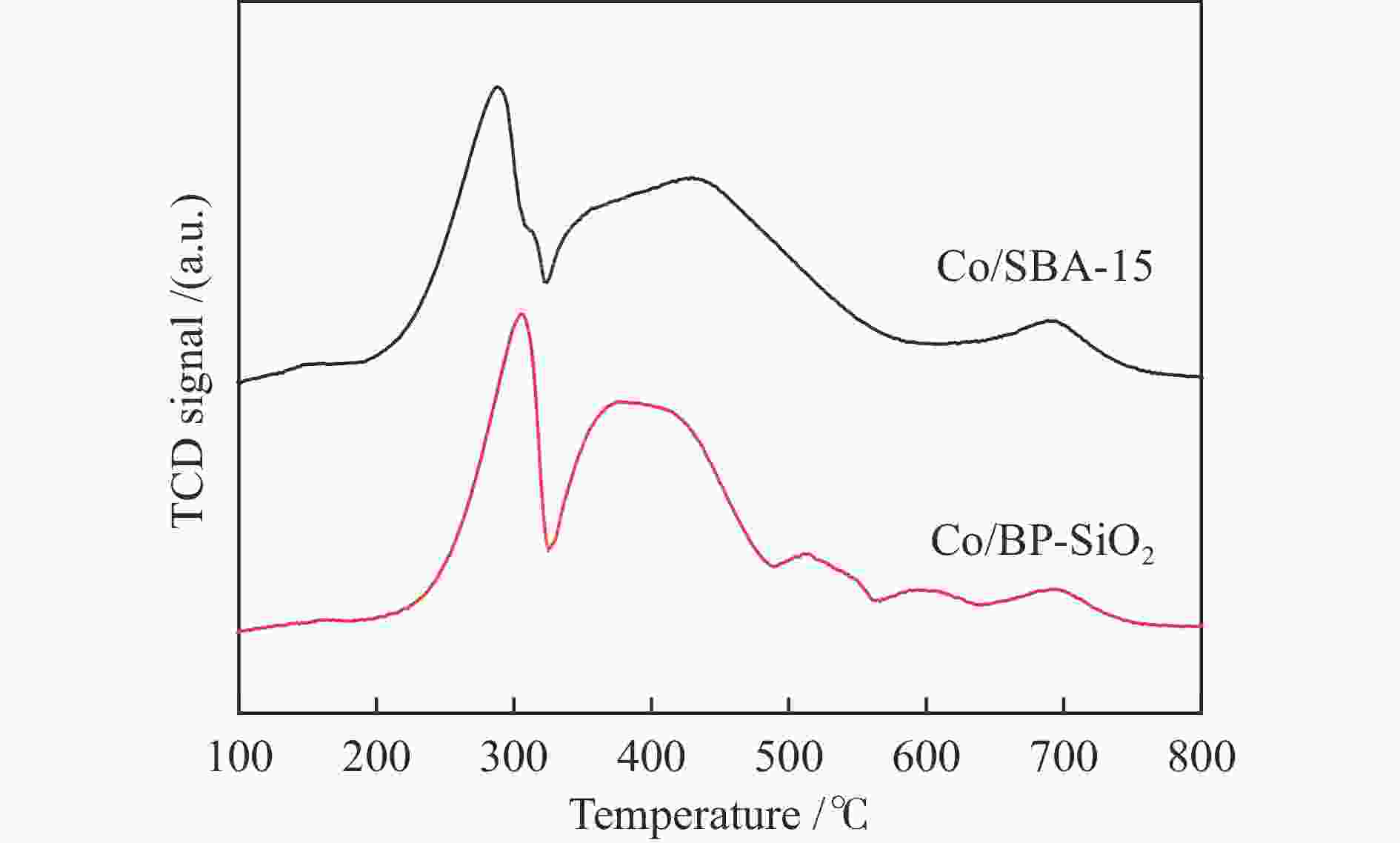
(cm3·g−1)Co particle size /nm XRD b TEM c H2-TPD d SBA-15 702.3 8.7 8.5 1.53 − − − Co/SBA-15 682.0 7.9 7.2 1.35 7.2 7.8 6.1 BP-SiO2 1103.2 11.4 2.9,63.8 3.15 − − − Co/BP-SiO2 723.6 8.9 2.7,40.2 1.61 5.8 6.0 5.3 a: Average pore size = 4 (pore volume/SBET), b: Particle size of Co3O4 was calculated by the Scherrer equation, Co metal particle size was calculated using the formulas DCo = 0.75 × DCo3O4, c: Average diameter of CoxOy crystallites obtained by TEM measurement, d: Co metal particle size was calculated by H2-TPD, Reduction at a temperature of 450 ℃ for 8 h, H2 chemisorption performance at 100 ℃ 表 2 固定床反应器上催化剂的费-托合成反应性能
Table 2 Evaluation of the Fischer-Tropsch reaction performance of the catalyst in a fixed-bed reactor a
Catalyst t/℃ CO initial
conversion /%CO steady
state conversion /%Activity lossb /% Product selectivity/mol% C1 C2 − C4 C5+ Co/BP-SiO2 210 37.7 33.8 10.3 10.6 9.4 80.0 230 59.1 54.9 7.1 14.7 11.2 74.1 Co/SBA-15 210 32.1 25.3 21.2 13.8 13.7 72.5 230 51.0 46.3 9.2 18.2 15.4 66.4 a: Reduction conditions: in pure hydrogen at 450 ℃, 0.1 MPa for 8 h; reaction conditions: H2/CO = 2, 210 and 230 ℃, 1.0 MPa, 4 L/(g·h), b: Activity loss= (CO initial conversion−CO steady state conversion)/CO initial conversion × 100% -
[1] LIN T, YU F, AN Y, QIN T, LI L, GONG K, ZHONG L, SUN Y. Cobalt carbide nanocatalysts for efficient syngas conversion to value-added chemicals with high selectivity[J]. Acc Chem Res,2021,54(8):1961−1971. doi: 10.1021/acs.accounts.0c00883 [2] 孙予罕, 陈建刚, 王俊刚, 贾丽涛, 侯博, 李德宝, 张娟. 费托合成钴基催化剂的研究进展[J]. 催化学报,2010,31(8):919−927.SUN Yu-han, CHEN Jian-gang, WANG Jun-gang, JIA Li-tao, HOU Bo, LI De-bao, ZHANG Juan. The development of cobalt-based catalysts for Fischer-Tropsch synthesis[J]. Chin J Catal,2010,31(8):919−927. [3] ZHANG Q, KANG J, WANG Y. Development of novel catalysts for Fischer-Tropsch synthesis: Tuning the product selectivity[J]. ChemCatChem,2010,2(9):1030−1058. doi: 10.1002/cctc.201000071 [4] SUN F, YANG R, XIA Z, YANG Y, ZHAO Z, GU S, WU D, DING Y, JIANG Z. Effects of cobalt carbide on Fischer-Tropsch synthesis with MnO supported Co-based catalysts[J]. J Energy Chem,2020,42:227−232. doi: 10.1016/j.jechem.2019.07.007 [5] 李娟, 吴梁鹏, 邱勇, 定明月, 王铁军, 李新军, 马隆龙. 费托合成催化剂的研究进展[J]. 化工进展,2013,S1:100−109.LI Juan, WU Liang-peng, QIU Yong, DING Ming-yue, WANG Tie-jun, LI Xin-jun, MA Long-long. Research advances in catalysts for Fischer-Tropsch synthesis[J]. Chem Ind Eng Prog,2013,S1:100−109. [6] CHENG Q, ZHAO N, LYU S, TIAN Y, GAO F, DONG L, JIANG Z, ZHANG J, TSUBAKI N, LI X. Tuning interaction between cobalt catalysts and nitrogen dopants in carbon nanospheres to promote Fischer-Tropsch synthesis[J]. Appl Catal B: Environ,2019,248:73−83. doi: 10.1016/j.apcatb.2019.02.024 [7] LIU C, CHEN Y, ZHAO Y, LYU S, WEI L, LI X, ZHANG Y, LI J. Nano-ZSM-5-supported cobalt for the production of liquid fuel in Fischer-Tropsch synthesis: Effect of preparation method and reaction temperature[J]. Fuel,2019,263:116619. [8] XU Y, LI X, GAO J, WANG J, MA G, WEN X, YANG Y, LI Y, DING M. A hydrophobic FeMn@Si catalyst increases olefins from syngas by suppressing C1 by-products[J]. Science,2021,371:610−613. doi: 10.1126/science.abb3649 [9] ZENG B, HOU B, JIA L, WANG J, CHEN C, LI D, SUN Y. The intrinsic effects of shell thickness on the Fischer-Tropsch synthesis over core-shell structured catalysts[J]. Catal Sci Technol,2013,3(12):3250−3255. doi: 10.1039/c3cy00665d [10] ZHOU W, CHENG K, KANG J, ZHOU C, SUBRAMANIAN V, ZHANG Q, WANG Y. New horizon in C1 chemistry: Breaking the selectivity limitation in transformation of syngas and hydrogenation of CO2 into hydrocarbon chemicals and fuels[J]. Chem Soc Rev,2019,48(12):3193−3228. doi: 10.1039/C8CS00502H [11] WEN X, ZHANG Y, LIU C, HONG J, WEI L, CHEN Y, LI J. Performance of hierarchical ZSM-5 supported cobalt catalyst in the Fischer-Tropsch synthesis[J]. J Fuel Chem Technol,2017,45(8):950−955. doi: 10.1016/S1872-5813(17)30045-2 [12] BUKUR D B, MANDIĆ M, TODIĆ B, NIKAČEVIĆ N. Pore diffusion effects on catalyst effectiveness and selectivity of cobalt based Fischer-Tropsch catalyst[J]. Catal Today,2020,343:146−155. doi: 10.1016/j.cattod.2018.10.069 [13] ZHONG M, WANG J, CHEN C, MA Z, JIA L, HOU B, LI D. Incorporating silicon carbide nanoparticles into Al2O3@Al to achieve an efficient support for Co-based catalysts to boost their catalytic performance towards Fischer-Tropsch synthesis[J]. Catal Sci Technol,2019,9(21):6037−6046. doi: 10.1039/C9CY01422E [14] LI X, NISA M U, CHEN Y, LI Z. Co-based catalysts supported on silica and carbon materials: effect of support property on cobalt species and Fischer-Tropsch synthesis performance[J]. Ind Eng Chem Res,2019,58(8):3459−3467. doi: 10.1021/acs.iecr.8b05451 [15] ISHIHARA D, TAO K, YANG G, HAN L, TSUBAKI N. Precisely designing bimodal catalyst structure to trap cobalt nanoparticles inside mesopores and its application in Fischer-Tropsch synthesis[J]. Chem Eng J,2016,306:784−790. doi: 10.1016/j.cej.2016.08.014 [16] LIU Z, LI J, BUETTNER M, RANGANATHAN R V, UDDI M, WANG R. Metal-support interactions in CeO2- and SiO2-supported cobalt catalysts: effect of support morphology, reducibility, and interfacial configuration[J]. ACS Appl Mater Inter,2019,11(18):17035−17049. doi: 10.1021/acsami.9b02455 [17] TSUBAKIA N, ZHANG Y, SUN S, MORI H, YONEYAMA Y, LI X, FUJIMOTO K. A new method of bimodal support preparation and its application in Fischer-Tropsch synthesis[J]. Catal Commun,2001,2(10):311−315. doi: 10.1016/S1566-7367(01)00055-3 [18] SONG D, LI J. Effect of catalyst pore size on the catalytic performance of silica supported cobalt Fischer-Tropsch catalysts[J]. J Mol Catal A: Chem,2006,247(1-2):206−212. doi: 10.1016/j.molcata.2005.11.021 [19] XIONG H, ZHANG Y, LIEW K, LI J. Fischer-Tropsch synthesis: The role of pore size for Co/SBA-15 catalysts[J]. J Mol Catal A: Chem,2008,295(1-2):68−76. doi: 10.1016/j.molcata.2008.08.017 [20] 王举汉, 文熊, 刘成超, 张煜华, 赵燕熹, 李金林. 多级孔Co/Al-SiO2催化剂制备及其费-托合成催化性能[J]. 无机材料学报,2020,35(9):999−1004. doi: 10.15541/jim20190500WANG Ju-han, WEN Xiong, LIU Cheng-chao, ZHANG Yu-hua, ZHAO Yan-xi, LI Jin-lin. Preparation and Fischer-Tropsch synthesis performance of hierarchical Co/Al-SiO2 catalyst[J]. J Inor Mater,2020,35(9):999−1004. doi: 10.15541/jim20190500 [21] WITOON T, CHAREONPANICH M, LIMTRAKUL J. Effect of hierarchical meso-macroporous silica supports on Fischer-Tropsch synthesis using cobalt catalyst[J]. Fuel Process Technol,2011,92(8):1498−1505. doi: 10.1016/j.fuproc.2011.03.011 [22] LIU Y, FANG K, CHEN J, SUN Y. Effect of pore size on the performance of mesoporous zirconia-supported cobalt Fischer-Tropsch catalysts[J]. Green Chem,2007,9(6):611−615. doi: 10.1039/B614266D [23] NIU C, LI H, XIA M, WANG J, CHEN C, MA Z, JIA L, HOU B, LI D. Mass transfer advantage of hierarchical structured cobalt-based catalyst pellet for Fischer-Tropsch synthesis[J]. AlChE J.,2021,67(6):e17226. [24] LI H, WANG J, CHEN C, JIA L, HOU B, LI D. Effects of macropores on reducing internal diffusion limitations in Fischer-Tropsch synthesis using a hierarchical cobalt catalyst[J]. RSC Advances,2017,7(16):9436−9445. doi: 10.1039/C6RA27166A [25] KOO H M, TRAN-PHU T, YI G R, SHIN C H, CHUNG C H, BAE J W. Effect of the ordered meso-macroporous structure of Co/SiO2 on the enhanced activity of hydrogenation of CO to hydrocarbons[J]. Catal Sci Technol,2016,6(12):4221−4231. doi: 10.1039/C5CY01685A [26] WEI L, ZHAO Y, ZHANG Y, LIU C, HONG J, XIONG H, LI J. Fischer-Tropsch synthesis over a 3D foamed MCF silica support: Toward a more open porous network of cobalt catalysts[J]. J Catal,2016,340:205−218. doi: 10.1016/j.jcat.2016.04.019 [27] ZHAO D, FENG J, HUO Q, MELOSH N, FREDRICKSON G H, CHMELKA B F, STUCKY G D. Triblock copolymer syntheses of mesoporous silica with periodic 50 to 300 angstrom pores[J]. Science,1998,279:548−552. doi: 10.1126/science.279.5350.548 [28] ZHANG Y, XIONG H, LIEW K, LI J. Effect of magnesia on alumina-supported cobalt Fischer-Tropsch synthesis catalysts[J]. J Mol Catal A: Chem,2005,237(1-2):172−181. doi: 10.1016/j.molcata.2005.04.057 [29] NURUNNABI M, TURN S Q. Pore size effects on Ru/SiO2 catalysts with Mn and Zr promoters for Fischer-Tropsch synthesis[J]. Fuel Process Technol,2015,130:155−164. doi: 10.1016/j.fuproc.2014.10.004 [30] MARTÍNEZ A, LÓPEZ C, MÁRQUEZ F, DÍAZ I. Fischer-Tropsch synthesis of hydrocarbons over mesoporous Co/SBA-15 catalysts: the influence of metal loading, cobalt precursor, and promoters[J]. J Catal,2003,220(2):486−499. doi: 10.1016/S0021-9517(03)00289-6 [31] GARCES L J, HINCAPIE B, ZERGER R, SUIB S L. The effect of temperature and support on the reduction of cobalt oxide: An in situ X-ray diffraction study[J]. J Phys Chem C,2015,119(10):5484−5490. doi: 10.1021/jp5124184 [32] PRIETO G, MARTÍNEZ A, CONCEPCIÓN P, MORENO-TOST R. Cobalt particle size effects in Fischer-Tropsch synthesis: Structural and in situ spectroscopic characterisation on reverse micelle-synthesised Co/ITQ-2 model catalysts[J]. J Catal,2009,266(1):129−144. doi: 10.1016/j.jcat.2009.06.001 -




 下载:
下载: