Mechanisms of Brönsted and Lewis acids of zeolite on pentenes conversion by in situ DRIFTS
-
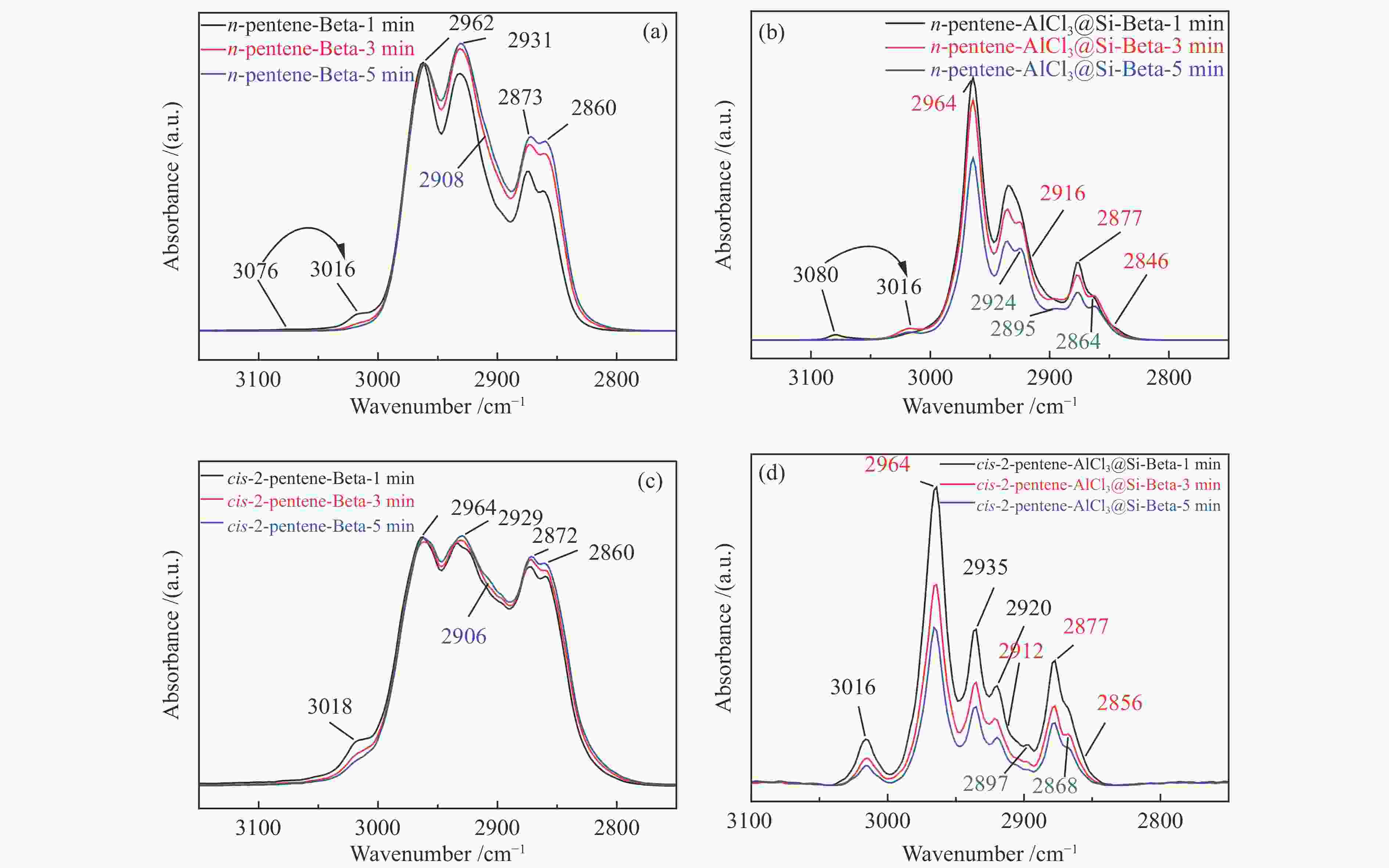
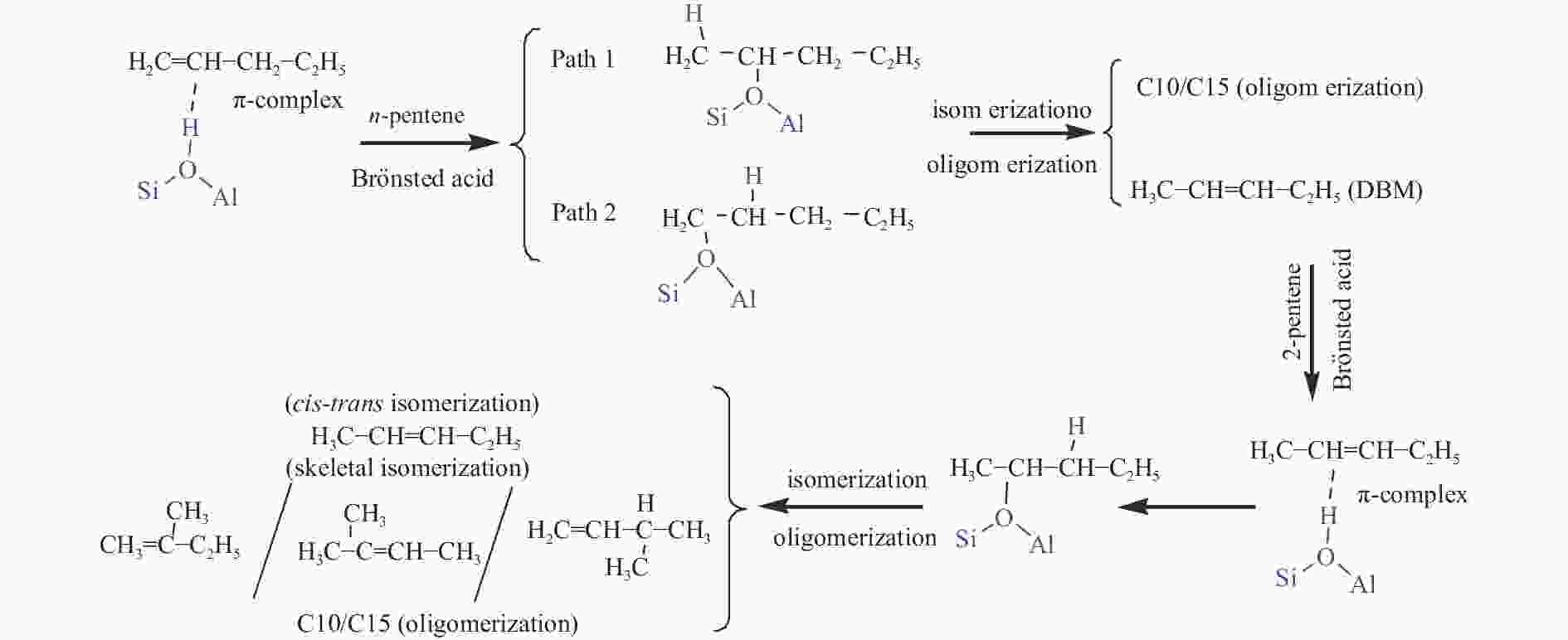
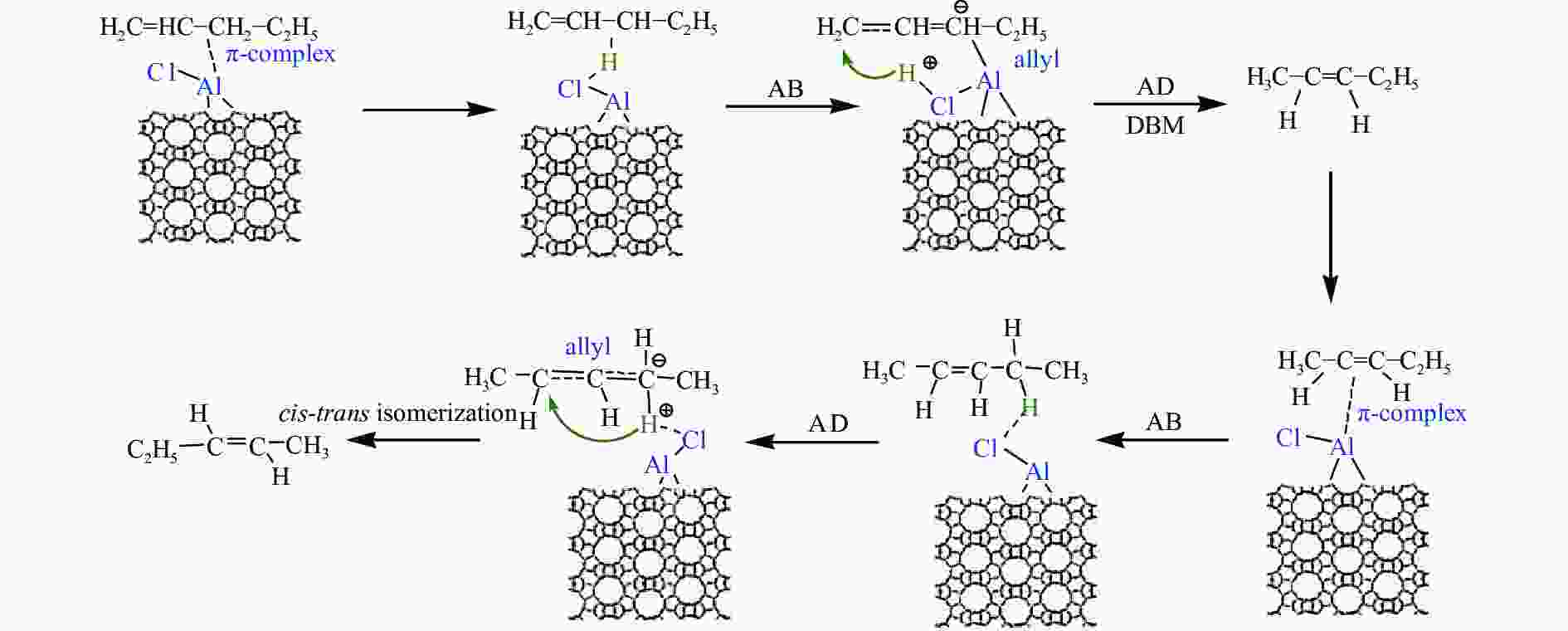
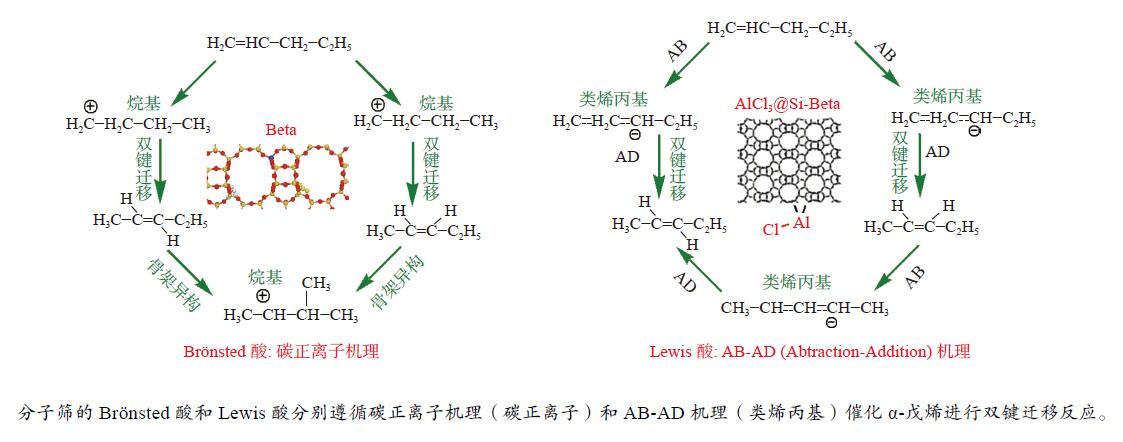
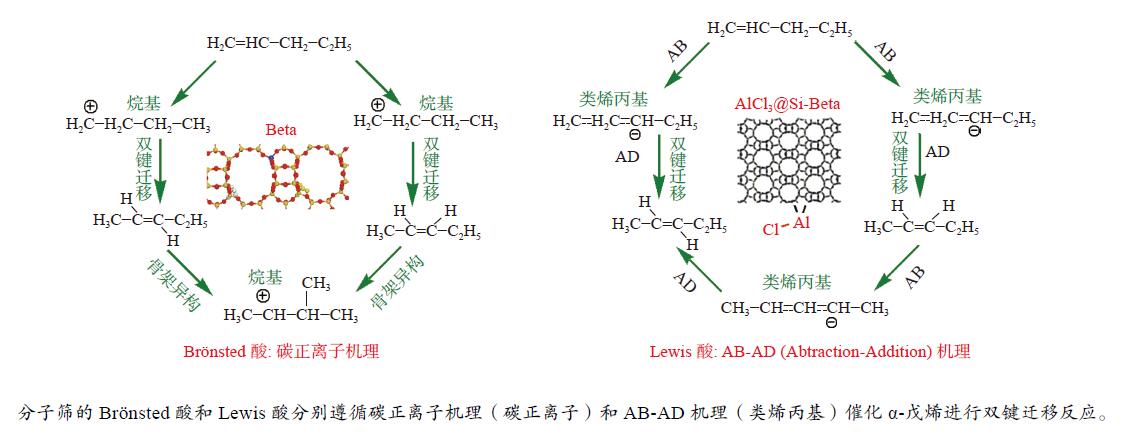
摘要: 本研究基于原位水热合成和气相浸渍的方法分别合成了含有Brönsted/Lewis酸的Beta分子筛和只含有Lewis酸的AlCl3@Si-Beta样品,并通过原位漫反射红外技术对比研究了Brönsted酸和Lewis酸催化直链和侧链戊烯同分异构体转化作用机理的差异。结果表明,同时含有Brönsted/Lewis酸的Beta分子筛中,Brönsted酸起主要的活化作用,催化戊烯进行异构和叠合反应时均遵循经典的碳正离子机理;而AlCl3@Si-Beta中的Lewis酸不含氢质子或羟基,催化α-戊烯进行双键迁移和2-戊烯的顺反异构反应时遵循AB-AD机理,以类烯丙基物种作为中间体,无法催化戊烯进行骨架异构和叠合反应。Abstract: Zeolites have been extensively used in the chemical and petrochemical industries owing to their tunable acidities and unique pore structures. Beta zeolite with Brönsted and Lewis acids and AlCl3@Si-Beta with only Lewis acid were prepared by hydrothermal synthesis and gas-phase impregnation methods, respectively. Mechanisms differences of Brönsted and Lewis acids on four pentene isomers transformation were investigated by in situ diffraction infrared Fourier transform spectrum (DRIFTS). The results suggested that Brönsted in Beta played a main role in isomerization and oligomerization reactions which all followed classical carbenium ion mechanisms. On the contrary, Lewis acid in AlCl3@Si-Beta without hydrogen proton or hydroxyl catalyzed α-pentene double-bond migration to produce β-pentene and cis-trans isomerization reaction of 2-pentene by AB-AD mechanism with allyl-like species as intermediates, and could not catalyze pentenes skeletal isomerization and oligomerization reactions.
-
Key words:
- zeolite /
- Brönsted acid /
- lewis acid /
- pentene isomers /
- mechanism
-
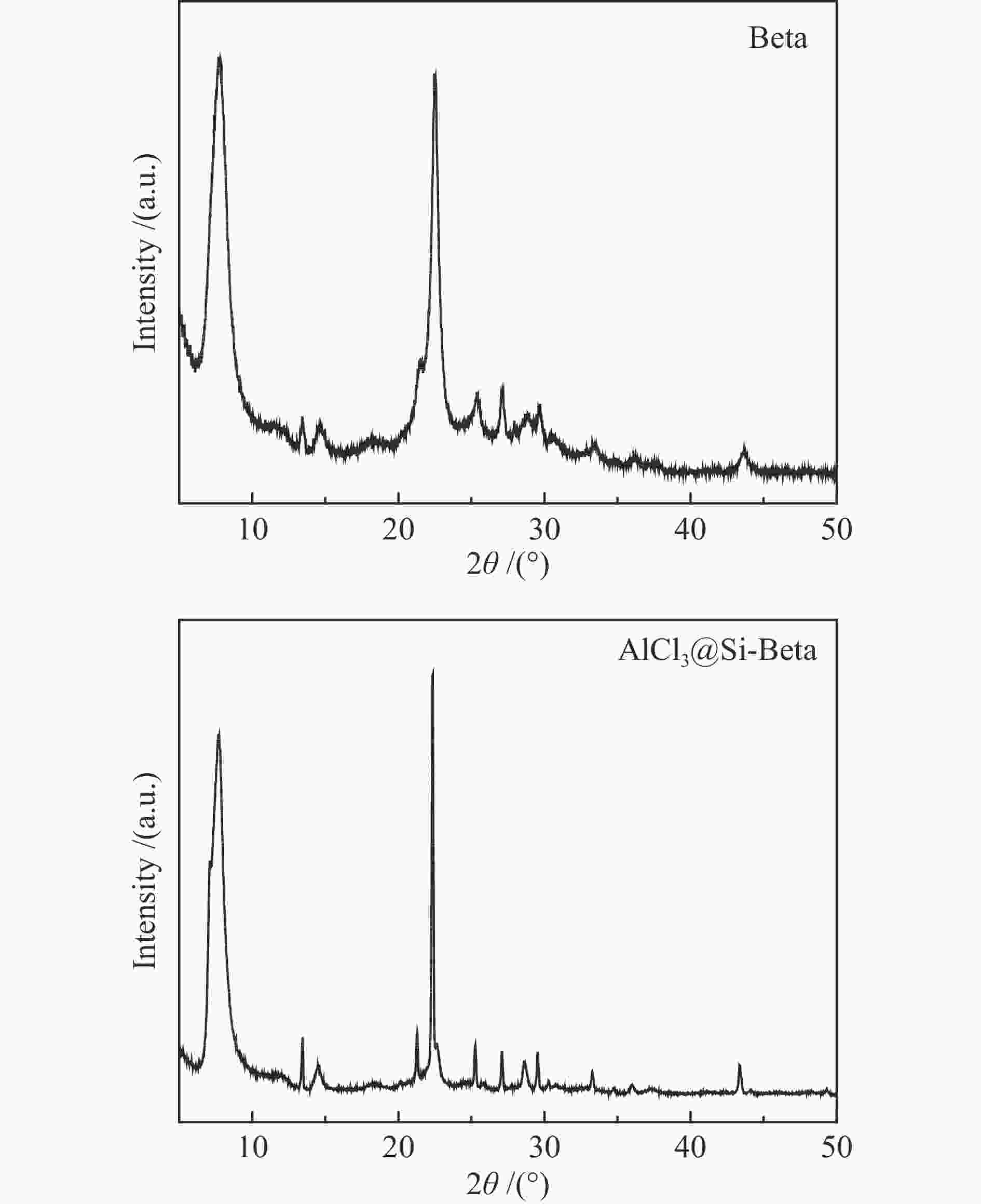
表 1 不同样品的织构性质
Table 1 Textual properties of two samples.
Sample Surface area/(m2·g−1) Volume b/
(cm3·g−1)SBET a Smic b Sext c Beta 592.1 335.1 256.9 0.13 AlCl3@Si-Beta 465.7 432.9 32.8 0.17 a: SBET obtained by the BET method, b: Calculated from the t-plot method, c: Calculated from difference value of SBET and Smic 表 2 不同样品的酸性质
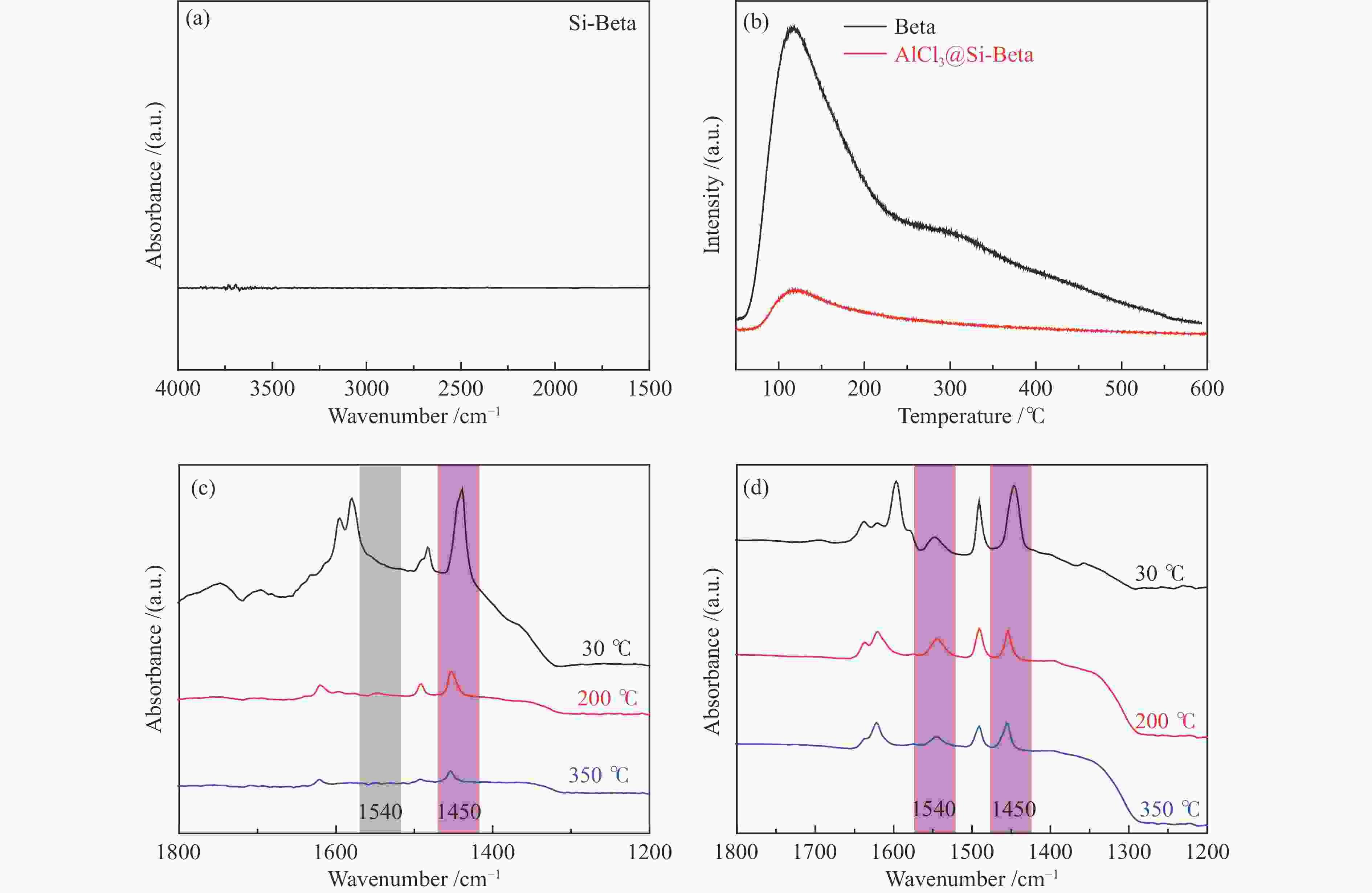
Table 2 Acidic properties of samples
Sample Acid types/(μmol Py·g−1) a Brönsted acid b Lewis acid c 30 ℃ 200 ℃ 350 ℃ 30 ℃ 200 ℃ 350 ℃ Beta 160.4 156.1 82.7 445.8 124.9 120.1 AlCl3@Si-Beta 0.0 0.0 0.0 160.9 32.4 12.7 a: Calculated from the band areas of the FT-IR spectra obtained by pyridine desorption at 30, 200, and 350 ℃, b: SBrönsted acid =1.88*A*0.65*0.65/M (A represents peak area at about 1540 cm−1; M represents mass of catalyst), c: SLewis acid = 1.42*A*0.65*0.65/M (A represents peak area at about 1450 cm−1; M represents mass of catalyst) 表 3 不同酸性分子筛催化戊烯反应的产物分布[29]
Table 3 Product distribution of pentene isomers reactions on two zeolites with different acids[29]
Sample Reactant Product distribution n-pentene cis-2-pentene trans-2-pentene 2-methyl-1-butene 2-methyl-2-butene C6 + Beta n-pentene × √ √ − − √ cis-2-pentene √ × √ − − − 2-methyl-1-butene − − √ × √ √ 2-methyl-2-butene − − − √ × √ AlCl3@Si-Beta n-pentene × √ √ × × × cis-2-pentene √ × √ × × × 2-methyl-1-butene × × × × √ × 2-methyl-2-butene × × × √ × × Reaction condition: 10% pentenes in helium and reaction temperature 100 ℃, “√” represents a large amount of product, “×” represents none, “−” represents a very small amount of product which can be ignored -
[1] BUSCA G. Acid catalysts in industrial hydrocarbon chemistry[J]. Chem Rev,2007,107(11):5366−5410. doi: 10.1021/cr068042e [2] FAN S, WANG H, HE S, YUAN K, WANG P, LI J, WANG S, QIN Z, DONG M, FAN W, WANG J. Formation and evolution of methylcyclohexene in the initial period of methanol to olefins over H-ZSM-5[J]. ACS Catal,2022,12(20):12477−12487. doi: 10.1021/acscatal.2c03410 [3] YI F, XU D, TAO Z, HU C, BAI Y, ZHAO G, CHEN H, CAO J P, YANG Y. Correlation of Brønsted acid sites and Al distribution in ZSM-5 zeolites and their effects on butenes conversion[J]. Fuel,2022,320:123729. doi: 10.1016/j.fuel.2022.123729 [4] HAN F, SUN H, ZHAO Z, XU Y, DONG H, LIU W, SUN L, WANG Z, HOU G, KITANO M, LI W, SHEN M, CHEN H. Selective catalytic reduction of NOx by methanol on metal-free zeolite with Brønsted and Lewis acid pair[J]. ACS Catal,2022,12(4):2403−2414. doi: 10.1021/acscatal.1c05624 [5] CHU Y, YI X, LI C, SUN X, ZHENG A. Brønsted/Lewis acid sites synergistically promote the initial C-C bond formation in the MTO reaction[J]. Chem Sci,2018,9(31):6470−6479. doi: 10.1039/C8SC02302F [6] KATADA N, SUZUKI K, NODA T, SASTRE G, NIWA M. Correlation between Brønsted acid strength and local sructure in zeolites[J]. J Phys Chem C,2009,113(44):19208−19217. doi: 10.1021/jp903788n [7] MÜLLER S, LIU Y, KIRCHBERGER F M, TONIGOLD M, SANCHEZSANCHEZ M, LERCHER J A. Hydrogen transfer pathways during zeolite catalyzed methanol conversion to hydrocarbons[J]. J Am Chem Soc,2016,138(49):15994−16003. doi: 10.1021/jacs.6b09605 [8] YOKOI T, MOCHIZUKI H, NAMBA S, KONDO J N, TATSUMI T. Control of the Al distribution in the framework of ZSM-5 zeolite and its evaluation by solid-state NMR technique and catalytic properties[J]. J Phys Chem C,2015,119(27):15303−15315. doi: 10.1021/acs.jpcc.5b03289 [9] ZHAO R R, ZHAO Z, LI S, ZHANG W. Insights into the correlation of aluminum distribution and Brönsted acidity in H-Beta zeolites from solid-state NMR spectroscopy and DFT calculations[J]. J Phys Chem Lett,2017,8(10):2323−2327. doi: 10.1021/acs.jpclett.7b00711 [10] CHEN Z, LIU S, WANG H, NING Q, ZHANG H, YUN Y, REN J, LI Y W. Synthesis and characterization of bundle-shaped ZSM-22 zeolite via the oriented fusion of nanorods and its enhanced isomerization performance[J]. J Catal,2018,361:177−185. doi: 10.1016/j.jcat.2018.02.019 [11] 庹杰, 李石擎, 徐浩, 关业军, 吴鹏. 分子筛结构设计及酸性调控在合成气催化转化中的应用进展[J]. 燃料化学学报,2023,51(1):1−19.TUO Jie, LI Shi-qing, XU Hao, GUAN Ye-jun, WU Peng. A progress of structure design and acidity tunning of zeolites in catalytic syngas conversion[J]. J Fuel Chem Technol,2023,51(1):1−19. [12] HANSFORD R C. A mechanism of catalytic cracking[J]. Ind Eng Chem,2002,39(7):849−852. [13] KONDO J N, LIQUN S, WAKABAYASHI F, DOMEN K. IR study of adsorption and reaction of 1-butene on H-ZSM-5[J]. Catal Lett,1997,47(2):129−133. doi: 10.1023/A:1019096703066 [14] SPOTO G, BORDIGA S, RICCHIARDI G, SCARANO D, ZECCHINA A, BORELLO E. IR study of ethene and propene oligomerization on H-ZSM-5: Hydrogen-bonded precursor formation, initiation and propagation mechanisms and structure of the entrapped oligomers[J]. J Chem Soc, Faraday Trans,1994,90:2827−2835. [15] HÖLDERICH W F, RÖSELERseler J, HEITMANN G, LIEBENS A T. The use of zeolites in the synthesis of fine and intermediate chemicals[J]. Catal Today,1997,37(4):353−366. doi: 10.1016/S0920-5861(97)81094-2 [16] BRODU N, MANERO M H, ANDRIANTSIFERANA C, PIC J S, VALDÉS H. Role of Lewis acid sites of ZSM-5 zeolite on gaseous ozone abatement[J]. Chem Eng J,2013,231(17):281−286. [17] MARQUES J P, GENER I, AYRAULT P, BORDADO J C, LOPES J M, RIBEIRO F R, GUISNET M. Infrared spectroscopic study of the acid properties of dealuminated BEA zeolites[J]. Microporous Mesoporous Mater,2003,60(1):251−262. [18] 郑健, 李强, 秦玉才, 宋丽娟. HFAU分子筛非骨架铝物种对Brønsted酸性质影响的理论研究[J]. 燃料化学学报,2022,50(7):849−858. doi: 10.19906/j.cnki.jfct.2022009ZHENG Jian, LI Qiang, QIN Yu-cai, SONG Li-juan. Impact of the extra-framework aluminum species on the properties of Brønsted acid sites in HFAU zeolites by using thiophene probe molecule: A periodic DFT study[J]. J Fuel Chem Technol,2022,50(7):849−858. doi: 10.19906/j.cnki.jfct.2022009 [19] BUSCA G. The surface acidity of solid oxides and its characterization by IR spectroscopic methods. An attempt at systematization. Phys Chem 1: 723-736[J]. Phys Chem Chem Phys,1999,1(5):723−736. doi: 10.1039/a808366e [20] GABRIENKO A A, ARZUMANOV S S, TOKTAREV A V, STEPANOV A G. Solid-state NMR characterization of the structure of intermediates formed from olefins on metal oxides (Al2O3 and Ga2O3)[J]. J Phys Chem C,2012,116(40):21430−21438. doi: 10.1021/jp3071103 [21] YAMAMOTO Y. From σ- to π-electrophilic Lewis acids. Application to selective organic transformations[J]. J Org Chem,2007,72(21):7817−7831. doi: 10.1021/jo070579k [22] 张立伟, 张怀科, 陈志强, 刘粟侥, 任杰. ZSM-5分子筛骨架铝落位对甲醇转化制芳烃催化性能影响[J]. 燃料化学学报,2019,47(12):1468−1475. doi: 10.3969/j.issn.0253-2409.2019.12.007ZHANG Li-wei, ZHANG Huai-ke, CHEN Zhi-qiang, LIU Su-yao, REN Jie. Effect of framework Al siting on catalytic performance in methanol to aromatics over ZSM-5 zeolites[J]. J Fuel Chem Technol,2019,47(12):1468−1475. doi: 10.3969/j.issn.0253-2409.2019.12.007 [23] LARMIER K, CHIZALLET C, MAURY S, CADRAN N, ABBOUD J, MARCEAU E. Isopropanol dehydration on amorphous silica-alumina: synergy of Brønsted and Lewis acidities at pseudo-bridging silanols[J]. Angew Chem,2016,56(1):1−6. [24] BERNAUER M, TABOR E, PASHKOVA V, KAUCKÝ D, SOBALÍK Z, WICHTERLOVÁ B, DEDECEK J. Proton proximity-Newkey parameter controlling adsorption, desorption and activity in propene oligomerization over H-ZSM-5 zeolites[J]. J Catal,2016,344:157−172. doi: 10.1016/j.jcat.2016.09.025 [25] Davydov A. Molecular Spectroscopy of Oxide Catalyst Surfaces[M]. Chichister: John Wiley & Sons, 2003: 313–318. [26] 马健波, 高卓然, 胡亚琼, 蔡海军, 朱小春, 张学军. ZSM-35分子筛上戊烯骨架异构化[J]. 应用化工,2022,51(1):76−79. doi: 10.3969/j.issn.1671-3206.2022.01.017MA Jian-bo, GAO Zhuo-ran, HU Ya-qiong, CAI Hai-jun, ZHU Xiao-chun, ZHANG Xue-jun. The skeletal isomerization of pentene over ZSM-35[J]. Appl Chem Ind,2022,51(1):76−79. doi: 10.3969/j.issn.1671-3206.2022.01.017 [27] IVANOV P, PAPP H. In situ FT-IR study on the reaction path of skeletal isomerization of n-butene over different zeolites[J]. Appl Surf Sci,2001,179(1):234−239. [28] 刘宗俨, 黄星亮, 董乐, 田洪锋, 彭文宇, 高玥, 代小平. 不同形状催化剂对戊烯骨架异构化性能的影响[J]. 工业催化,2019,27(11):44−49.LIU Zong-yan, HUANG Xing-liang, DONG Le, TIAN Hong-feng, PENG Wen-yu, GAO Yue, DAI Xiao-ping. Effect of catalysts with different shapes on the isomerization of pentene skeleton[J]. Ind Catal,2019,27(11):44−49. [29] YI F, CAO J, XU D, TAO Z, HU C, BAI Y, ZHAO G, CHEN H, YANG Y, LI Y. Mechanisms of double-bond migration reactions of pentene isomers on different Lewis acids[J]. Appl Surf Sci,2022,589:152970. doi: 10.1016/j.apsusc.2022.152970 [30] YI F, HE P, CHEN H, HE Y, TAO Z, LI T, ZHAO G, YUN Y, WEN X, YANG Y, LI Y. Mechanisms of double-bond isomerization reactions of n-butene on different Lewis acids[J]. ACS Catal,2021,11(17):11293−11304. doi: 10.1021/acscatal.1c02846 [31] BUSCA G, RAMIS G, LORENZELLI V. Adsorption and oligomerization of isobutene on oxide catalyst surfaces. A Fourier-transform infrared study[J]. J Chem Soc, Faraday Trans,1989,85(1):137−146. doi: 10.1039/f19898500137 [32] YI F, CHEN H, HUANG L, HU C, WANG J, LI T, WANG H, TAO Z, YANG Y, LI Y. Effects of the acidity and shape selectivity of dealuminated zeolite beta on butene transformations[J]. Fuel,2021,300:120694. doi: 10.1016/j.fuel.2021.120694 [33] 安源, 钱梁华, 朱岳中, 华建英. 2-甲基-1-丁烯异构为2-甲基-2-丁烯的研究[J]. 上海化工,2002,27(22):23−29. doi: 10.16759/j.cnki.issn.1004-017x.2002.22.005AN Yuan, QIAN Liang-hua, ZHU Yue-zhong, HUA Jian-ying. Isomeirzation from 2-methyl-1-butene to 2-methyl-2-butene[J]. Shanghai Chem Ind,2002,27(22):23−29. doi: 10.16759/j.cnki.issn.1004-017x.2002.22.005 -




 下载:
下载: