Pyrolysis characteristics and kinetics of co-pyrolysis of microalgae and plastics
-
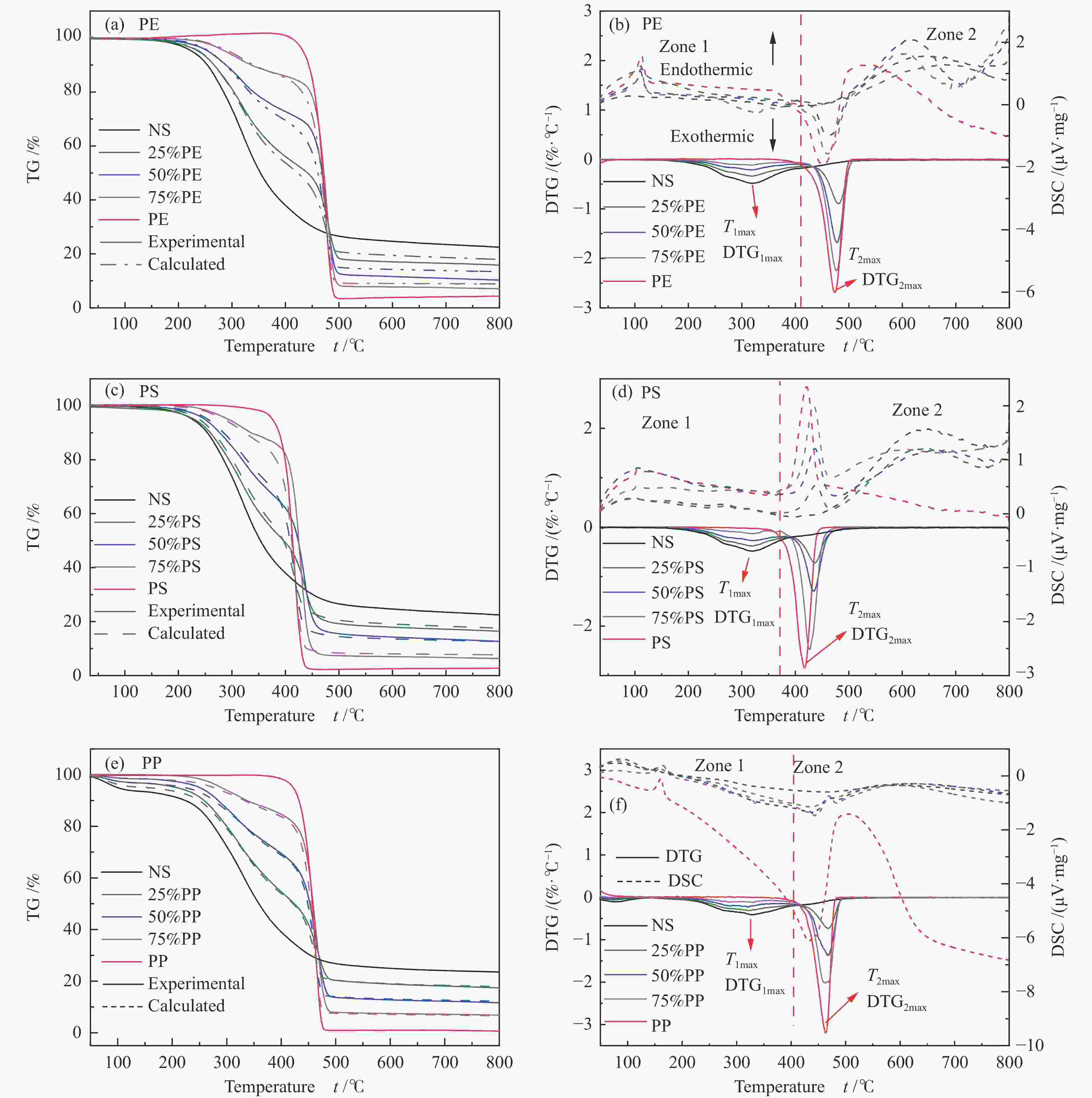
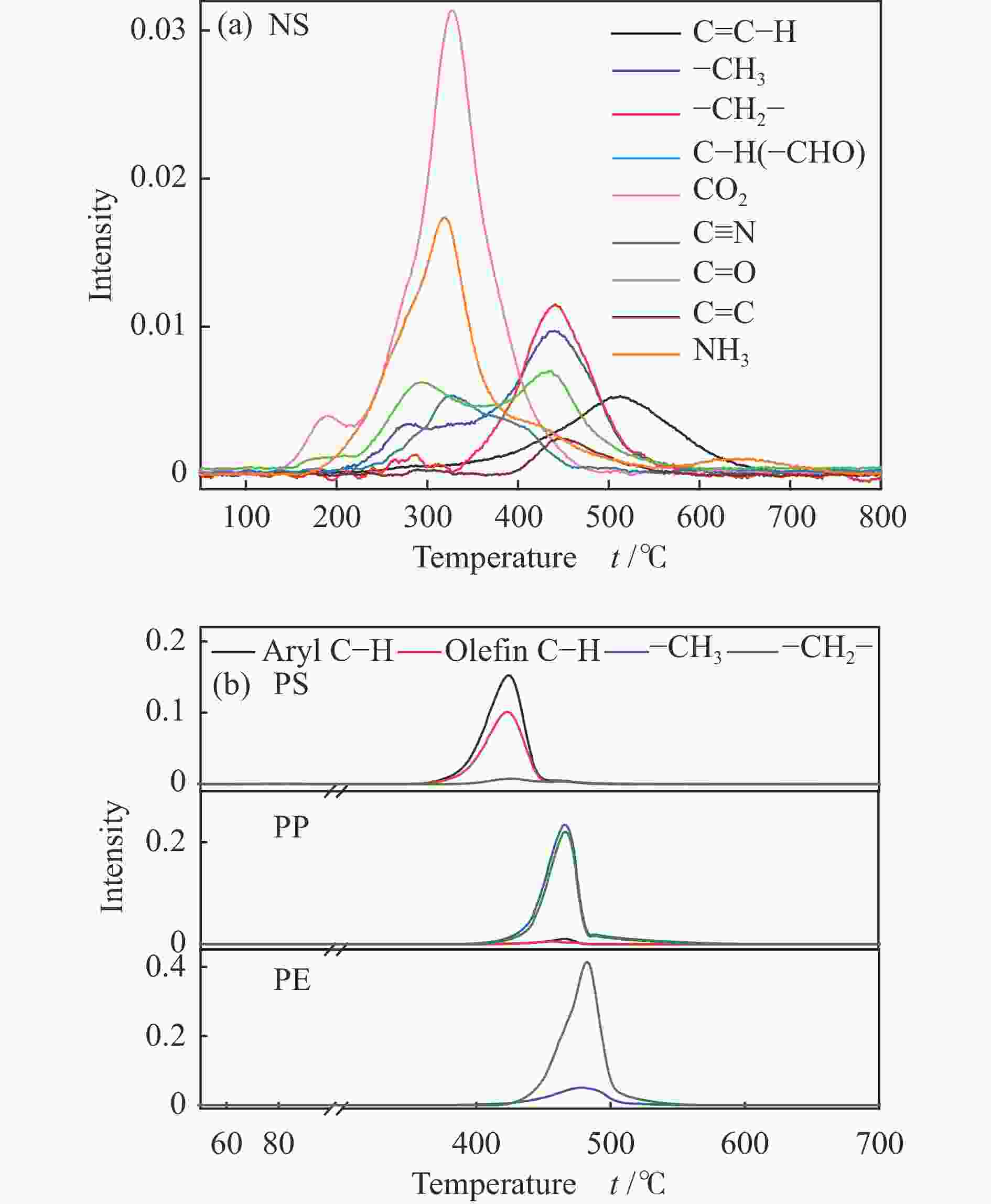
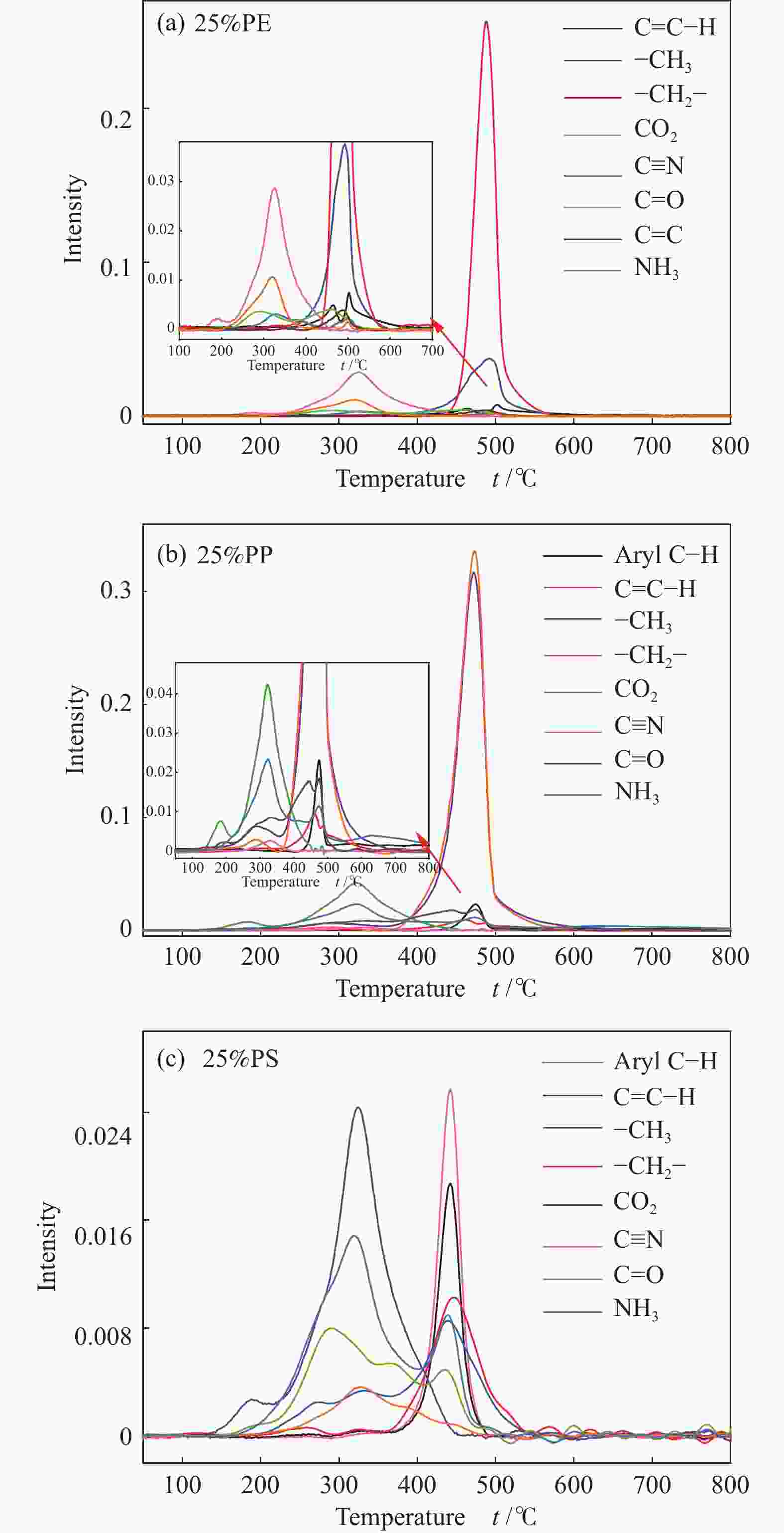
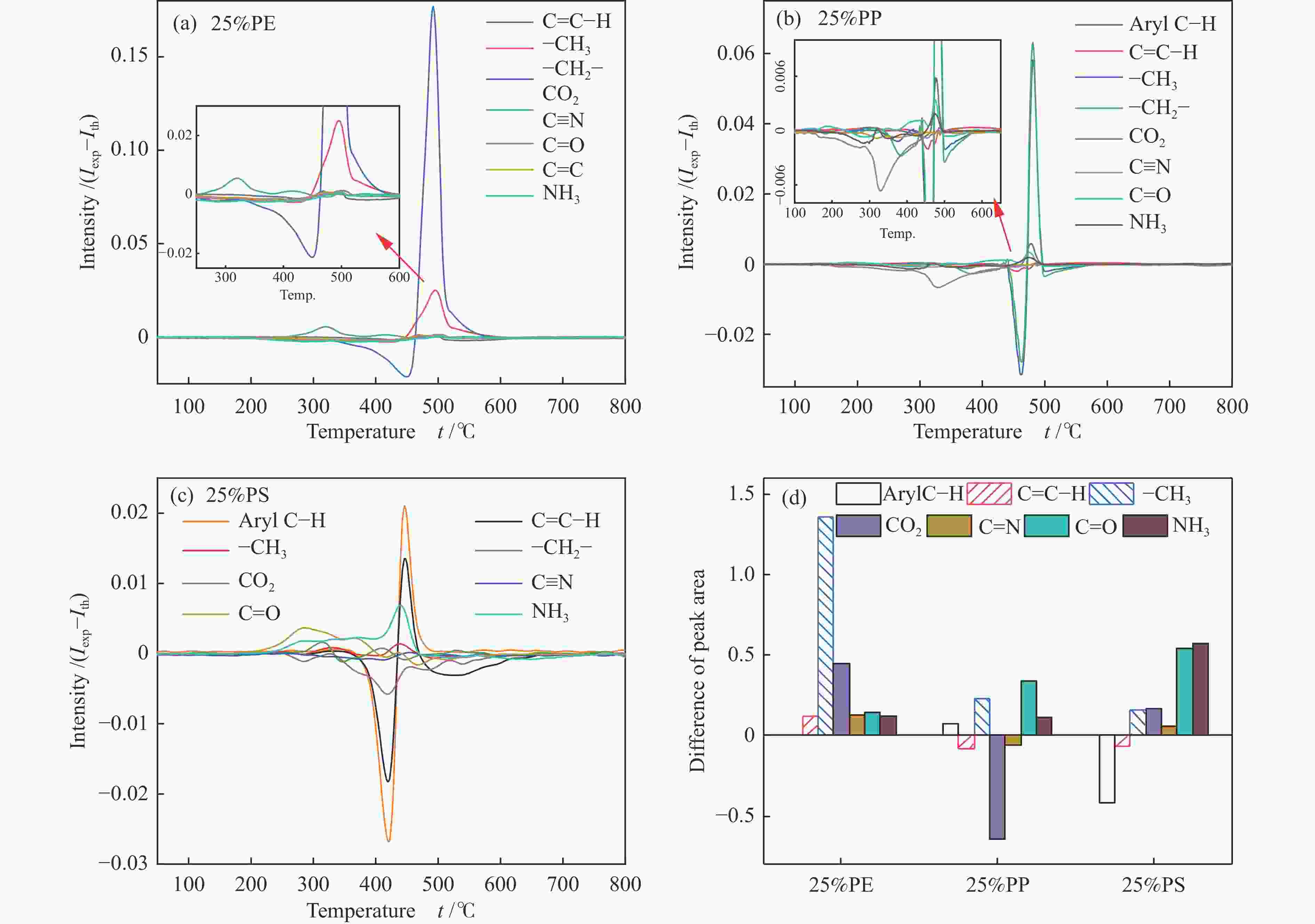
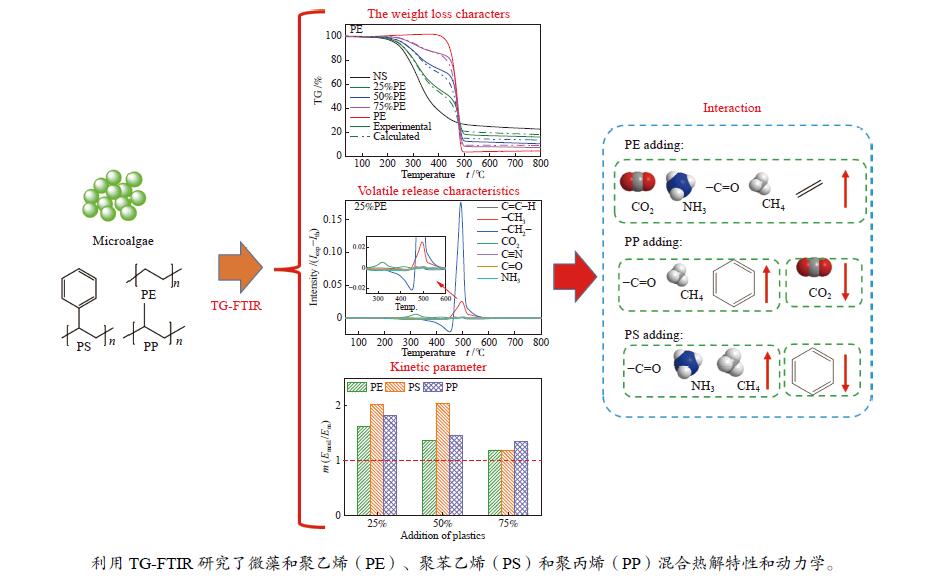
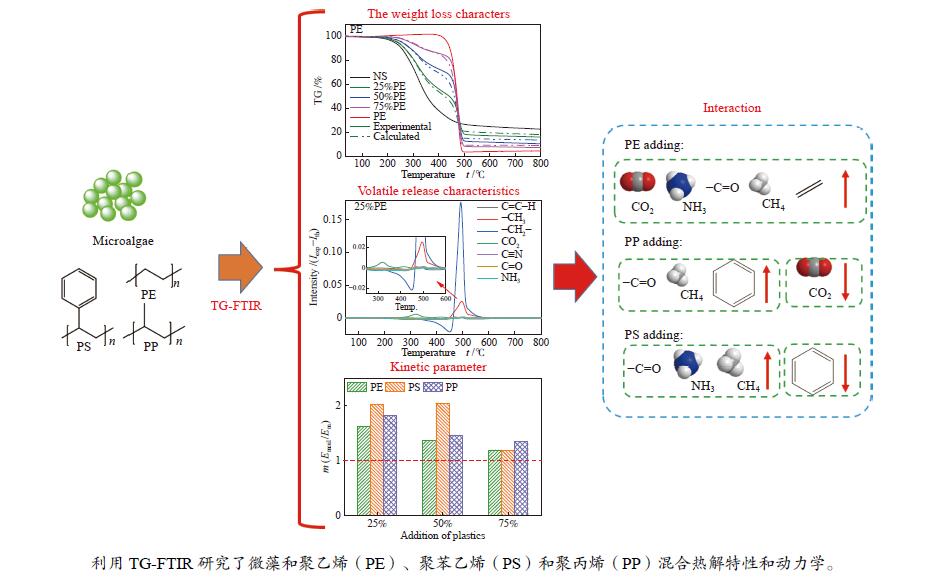
摘要: 本研究利用TG-FTIR研究了微藻和聚乙烯(PE)、聚苯乙烯(PS)和聚丙烯(PP)混合热解特性,探讨了塑料添加比及塑料结构对热解失重和挥发分释放特性以及动力学的影响。微藻与塑料混合热解存在交互作用,使塑料的热解温度升高,并抑制焦炭形成,其中,PE能有效地减少残余物产率,而PS混合热解时其热解温度明显升高。并且微藻与塑料混合热解能降低热解过程中的平均活化能,且在较低比例塑料添加时作用较强。此外,微藻与聚乙烯混合会促进CO2的释放以及聚乙烯断键生成–CH3和C=C–H。微藻与聚丙烯混合却抑制CO2的释放,加剧了聚丙烯生成芳烃C–H。而微藻与聚苯乙烯混合会轻微促进C=O和NH3生成,并加剧微藻的氢转移和聚苯乙烯苯环断裂释放–CH3。Abstract: Plastics as co-feeding can effectively improve the quality of microalgae pyrolysis oil. The effects of co-pyrolysis of microalgae and polyethylene (PE), polystyrene (PS) and polypropylene (PP) on pyrolysis characteristics and kinetics were investigated by TG-FTIR. There is an interaction between microalgae and plastic, which increases the pyrolysis temperature of plastic and inhibits the formation of coke. PE can effectively reduce the residual yield, while the pyrolysis temperature of PS obviously transfers to high temperature during co-pyrolysis. In addition, co-pyrolysis reduces the average activation energy, especially low proportion of plastic. Besides, the co-pyrolysis of microalgae with PE promotes C−C fracture to release CO2 and −CH3 and C=C−H. However, the co-pyrolysis of microalgae and PP inhibits the release of CO2, and intensifies the formation of aromatic hydrocarbon C−H from polypropylene. Moreover, the co-pyrolysis of microalgae and PS can slightly promote the formation of C=O and NH3, and aggravate the hydrogen transfer of microalgae to break aromatic ring of polystyrene and release CH3.
-
Key words:
- microalgae /
- plastics /
- co-pyrolysis /
- TG-FTIR /
- kinetics
-
表 1 样品的工业分析和元素分析
Table 1 Proximate, ultimate analysis of samples and biochemical constituents of microalgae
Sample Elemental analysis wd/% Proximate analysis wd/% C H N O① M V FC A NS 50.6 7.3 6.7 29.4 4.0 79.6 10.4 6.0 PE 84.3 15.5 −② 0.2 − 100.0 − − PS 91.4 7.5 − 1.1 − 97.2 2.8 − PP 84.9 14.7 − 0.6 − 99.9 0.1 − ①: calculated by difference; ②: undetected 表 2 基于Doyle 和Coats-Redfern法计算的微藻和塑料混合热解的动力学参数
Table 2 Kinetic parameters for the co-pyrolysis of microalgae and plastics via Doyle and Coats-Redfern method
Sample Temperature /℃ Coats-Redfern Doyle Em /
(kJ·mol−1)E/(kJ·mol−1) A/s R2 E/(kJ·mol−1) A/s R2 NS 216–357 47.91 2.7×10 0.9969 54.31 3.4×102 0.9987 37.04 357–526 16.15 2.0×10−2 0.9856 26.38 2.4 0.9964 25%PE 231–361 41.42 3.6 0.9889 48.31 6.0×10 0.9935 66.21 361–458 9.65 1.7×10−3 0.9752 19.95 4.5×10−1 0.9943 458–495 149.76 2.3×108 0.9862 154.27 5.9×108 0.9882 50%PE 252–345 37.97 9.2×10−1 0.9892 45.11 1.8×10 0.9937 130.79 345–442 7.14 4.1×10−4 0.9766 17.34 1.5×10−1 0.9968 442–494 205.48 1.9×1012 0.9796 207.13 2.7×1012 0.9817 75%PE 285–341 32.99 1.4×10−1 0.9872 40.62 3.8 0.9928 208.20 341–432 7.59 2.2×10−4 0.9785 17.64 7.4×10−2 0.9962 432–492 267.09 4.3×1016 0.9862 265.65 3.5×1016 0.9873 PE 432–486 321.04 3.4×1020 0.9960 316.75 1.9×1020 0.9963 318.89 25%PS 221–352 42.54 5.7 0.9951 49.26 8.9×10 0.9972 46.58 352–418 16.44 1.3×10−2 0.9869 26.04 1.4 0.9949 418–475 77.71 2.6×103 0.9790 85.21 2.2×104 0.9843 50%PS 244–337 43.16 4.1 0.9909 49.91 6.4×10 0.9944 74.29 337–413 18.65 1.3×10−2 0.9874 27.97 1.1 0.9945 413–472 125.05 1.1×107 0.9851 130.13 3.5×107 0.9876 75%PS 290–337 39.31 5.8×10−1 0.9965 46.64 1.1×10 0.9979 176.53 337–391 16.80 2.8×10−3 0.9776 26.04 2.7×10 0.9908 391–448 221.33 2.7×1014 0.9831 221.39 2.8×1014 0.9846 PS 375–431 267.91 2.0×1018 0.9919 265.33 1.4×1018 0.9925 266.62 25%PP 174–353 47.90 1.5×10 0.9980 53.89 1.8×102 0.9988 61.55 354–453 17.52 1.3×10−2 0.9920 27.17 1.3 0.9970 465–504 118.07 1.7×106 0.9929 123.97 6.6×106 0.9941 50%PP 197–337 49.37 1.3×10 0.9978 55.22 1.4×102 0.9984 124.06 354–442 15.65 4.7×10−3 0.9925 25.23 5.3×10−1 0.9971 459–504 179.67 4.7×1010 0.9912 182.46 8.2×1010 0.9922 75%PP 229–337 56.23 2.9×10 0.9963 61.63 2.4×102 0.9976 186.92 342–427 14.83 1.7×10−3 0.9899 24.31 2.1×10−1 0.9961 449–498 236.05 5.2×1014 0.9911 235.98 5.3×1014 0.9918 PP 419–486 321.74 9.5×1020 0.9932 317.14 5.0×1020 0.9936 319.44 -
[1] CHEN X, LI S J, LIU Z H, CHEN Y Q, YANG H P, WANG X H, CHE Q F, CHEN W, CHEN H P. Pyrolysis characteristics of lignocellulosic biomass components in the presence of CaO[J]. Bioresour Technol,2019,287:7. [2] ROSS, A B, P BILLER, M L KUBACKI, H LI, A LEA-LANGTON, J M JONES. Hydrothermal processing of microalgae using alkali and organic acids[J]. Fuel,2010,89(9):2234−2243. doi: 10.1016/j.fuel.2010.01.025 [3] LI F H, SRIKANTH C S, SANKAR B. A review on catalytic pyrolysis of microalgae to high-quality bio-oil with low oxygeneous and nitrogenous compounds[J]. Renewable Sustainable Energy Rev,2019,108:481−497. doi: 10.1016/j.rser.2019.03.026 [4] BACH, QUANG-VU, CHEN W H. Pyrolysis characteristics and kinetics of microalgae via thermogravimetric analysis (TGA): A state-of-the-art review[J]. Bioresour Technol,2017,246:88−100. doi: 10.1016/j.biortech.2017.06.087 [5] AYSU T, MAROTO-VALER M M, SANNA A. Ceria promoted deoxygenation and denitrogenation of Thalassiosira weissflogii and its model compounds by catalytic in-situ pyrolysis[J]. Bioresour Technol,2016,208:140−148. doi: 10.1016/j.biortech.2016.02.050 [6] AZIZI K, MOSTAFA K M, HAMED A N. A review on bio-fuel production from microalgal biomass by using pyrolysis method[J]. Renewable Sustainable Energy Rev,2018,82:3046−3059. doi: 10.1016/j.rser.2017.10.033 [7] 张泽, 赵洪君, 孟洁, 洪晨, 李益飞. 生物质的热解及生物油提质的研究进展[J]. 环境工程,2021,39(3):161−171. doi: 10.13205/j.hjgc.202103023ZHANG Ze, ZHAO Hong-Jun, MENG Jie, HONG Chen, LI Yi-Fei. Reseach progess of biomass pyrolysis and bio oil upgrading[J]. Environ Eng,2021,39(3):161−171. doi: 10.13205/j.hjgc.202103023 [8] CAMPANELLA A, HAROLD M P. Fast pyrolysis of microalgae in a falling solids reactor: Effects of process variables and zeolite catalysts[J]. Biomass Bioenergy,2012,46:218−232. doi: 10.1016/j.biombioe.2012.08.023 [9] 王文燕, 张光义, 孟辉波, 朱新宇, 张建岭, 许光文. 糠醛渣热解特性及热解挥发产物对其燃烧烟气原位控氮作用[J]. 化工学报,2021,72(11):5770−5778. doi: 10.11949/0438-1157.20211028WANG Wen-yan, ZHANG Guang-yi, MENG Hui-bo, ZHU Xin-yu, ZHANG Jian-ling, XU Guang-wen. Furfural residue pyrolysis characteristics and the effect of its pyrolysis products on in-situ control of NOx emission from its combustion flue gas[J]. CIESC J,2021,72(11):5770−5778. doi: 10.11949/0438-1157.20211028 [10] NANDAKUMAR T, DWIVEDI U, PANT K K. KUMAR S, BALARAMAN E. Wheat straw/HDPE co-reaction synergy and enriched production of aromatics and light olefins via catalytic co-pyrolysis over Mn, Ni, and Zn metal modified HZSM-5[J]. Catal Today,2023,408:111−126. [11] TIAN F J, YU J L, MCKENZIE L J, J HAYASHI, LI C Z. Conversion of fuel-N into HCN and NH3 during the pyrolysis and gasification in steam: A comparative study of coal and biomass[J]. Energy Fuels,2007,21(2):517−521. doi: 10.1021/ef060415r [12] RANJEET K M, KAUSTUBHA M. Co-pyrolysis of waste biomass and waste plastics (polystyrene and waste nitrile gloves) into renewable fuel and value-added chemicals[J]. Carbon Res Conv,2020,3:145−155. doi: 10.1016/j.crcon.2020.11.001 [13] 毛俏婷, 胡俊豪, 赵雨佳, 闫舒航, 杨海平, 陈汉平. 生物质和废塑料混合热解协同特性研究[J]. 燃料化学学报,2020,48(3):286−292. doi: 10.3969/j.issn.0253-2409.2020.03.004(MAO Qiao-ting, HU Jun-hao, ZHAO Yu-jia, YAN Shu-hang, YANG Hai-ping, CHEN Han-ping. Synergistic effect during biomass and waste plastics co-pyrolysis[J]. J Fuel Chem and Technol,2020,48(3):286−292. doi: 10.3969/j.issn.0253-2409.2020.03.004 [14] RAHMAN, M H, P R BHOI, A SAHA, V PATIL, S ADHIKARI. Thermo-catalytic co-pyrolysis of biomass and high-density polyethylene for improving the yield and quality of pyrolysis liquid[J]. Energy,2021,225:120231. [15] CHEN W M, SHI S K, CHEN M Z, ZHOU X Y. Fast co-pyrolysis of waste newspaper with high-density polyethylene for high yields of alcohols and hydrocarbons[J]. Waste Manage,2017,67:155−162. doi: 10.1016/j.wasman.2017.05.032 [16] YUAN H R, FAN H G, SHAN R, HE M Y, GU J, CHEN Y. Study of synergistic effects during co-pyrolysis of cellulose and high-density polyethylene at various ratios[J]. Energy Conv Manag,2018,157:517−526. doi: 10.1016/j.enconman.2017.12.038 [17] TANG Z Y, CHEN W, HU J H, LI S Q, CHEN Y Q, YANG H P, CHEN H P. Co-pyrolysis of microalgae with low-density polyethylene (LDPE) for deoxygenation and denitrification[J]. Bioresour Technol,2020,311:123502. doi: 10.1016/j.biortech.2020.123502 [18] DUAN P G, JIN B B, XU Y P, WANG F. Co-pyrolysis of microalgae and waste rubber tire in supercritical ethanol[J]. Chem Eng J, 2015. 269(Supplement C): 262−271. [19] XU S N, CAO B, B B UZOEJINWA, E A ODEY, WANG S, SHANG H, LI C H, HU Y M, WANG Q, J N NWAKAIRE. Synergistic effects of catalytic co-pyrolysis of macroalgae with waste plastics[J]. Process Saf Environ Protect,2020,137:34−48. doi: 10.1016/j.psep.2020.02.001 [20] WU X Y, WU Y L, WU K J, CHEN Y, HU H S, YANG M D. Study on pyrolytic kinetics and behavior: The co-pyrolysis of microalgae and polypropylene[J]. Bioresour Technol,2015,192:522−528. doi: 10.1016/j.biortech.2015.06.029 [21] CHEN R, ZHANG S, YANG X, LI G, ZHOU H, LI Q, ZHANG Y. Thermal behaviour and kinetic study of co-pyrolysis of microalgae with different plastics[J]. Waste Manage,2021,126:331−339. doi: 10.1016/j.wasman.2021.03.001 [22] TANG Z Y, CHEN W, CHEN Y Q, YANG H P, CHEN H P. Co-pyrolysis of microalgae and plastic: Characteristics and interaction effects[J]. Bioresour Technol,2019,274:145−152. doi: 10.1016/j.biortech.2018.11.083 [23] ABOUL-ENEIN, ATEYYA A, FATHI S S, MOHAMED A B. Co-production of hydrogen and carbon nanomaterials using NiCu/SBA15 catalysts by pyrolysis of a wax by-product: Effect of Ni-Cu loading on the catalytic activity[J]. Int J Hydrog Energy,2019,44(59):31104−31120. doi: 10.1016/j.ijhydene.2019.10.042 [24] HU Q, TANG Z Y, YAO D D, YANG H P, SHAO J A, CHEN H P. Thermal behavior, kinetics and gas evolution characteristics for the co-pyrolysis of real-world plastic and tyre wastes[J]. J Clean Prod,2020,260:121102. [25] WANG X, SHENG L L, YANG X Y. Pyrolysis characteristics and pathways of protein, lipid and carbohydrate isolated from microalgae Nannochloropsis sp[J]. Bioresour Technol,2017,229:119−125. doi: 10.1016/j.biortech.2017.01.018 [26] WANG X, TANG X H, YANG X Y. Pyrolysis mechanism of microalgae Nannochloropsis sp. based on model compounds and their interaction[J]. Energy Conv Manag,2017,140:203−210. doi: 10.1016/j.enconman.2017.02.058 [27] CHEN H P, XIE Y P, CHEN W, XIA M W, LI K X, CHEN Z Q, CHEN Y Q, YANG H P. Investigation on co-pyrolysis of lignocellulosic biomass and amino acids using TG-FTIR and Py-GC/MS[J]. Energy Conv Manag,2019,196:320−329. doi: 10.1016/j.enconman.2019.06.010 -




 下载:
下载: