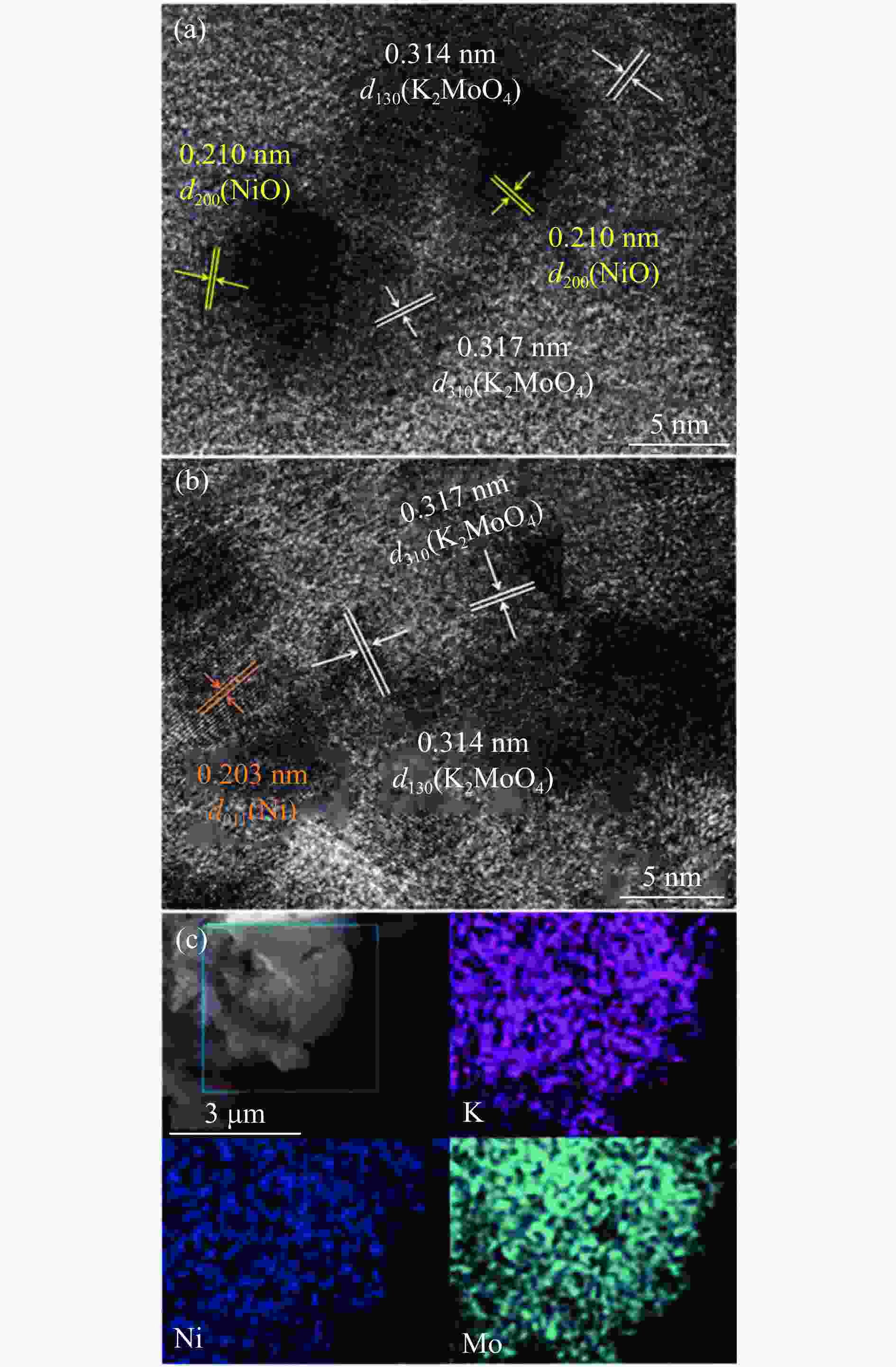
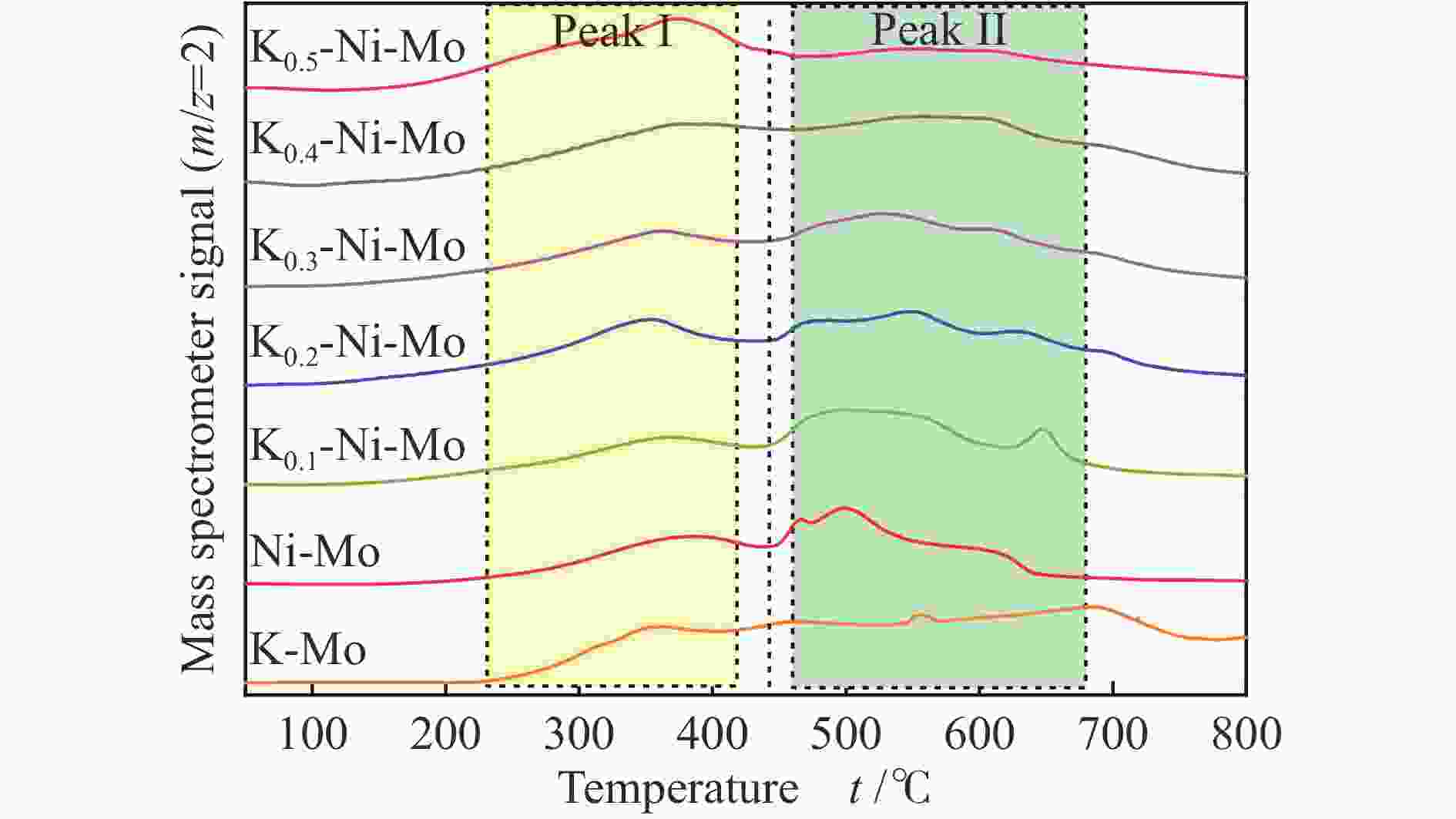
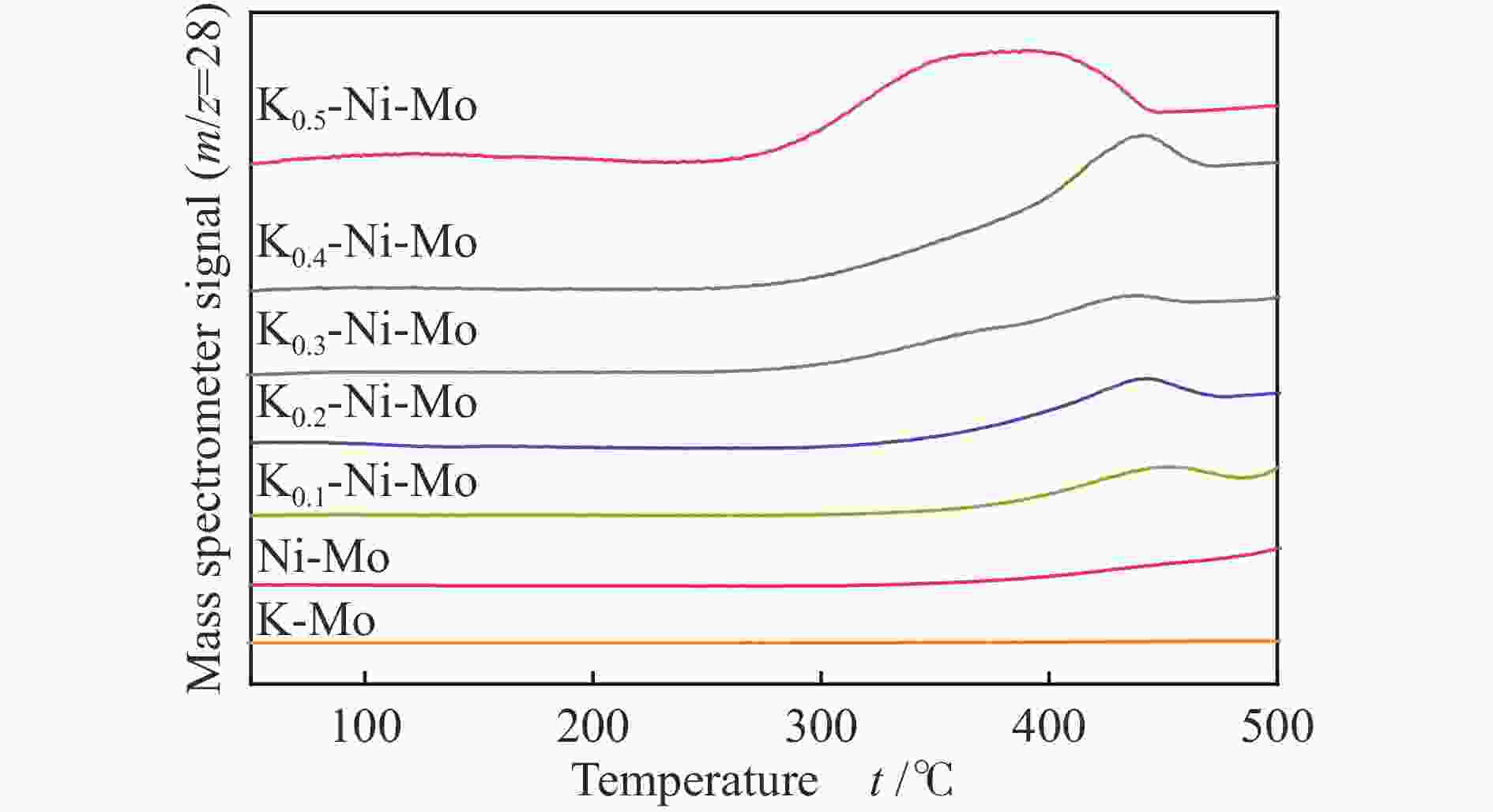
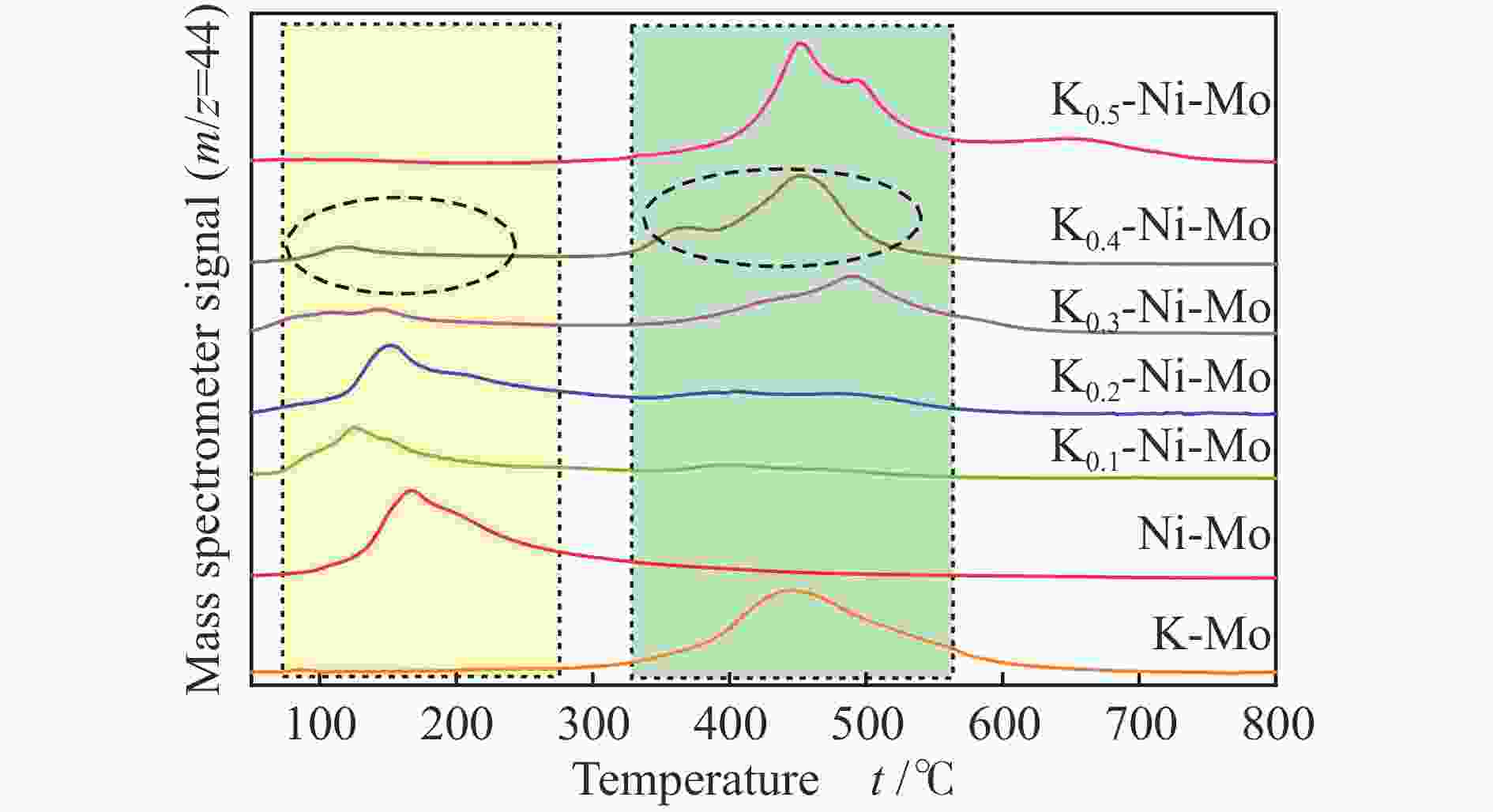
Hydrothermal reduction synthesis of K-Ni-Mo-based catalyst and its catalytic performance for higher alcohol synthesis from syngas
-
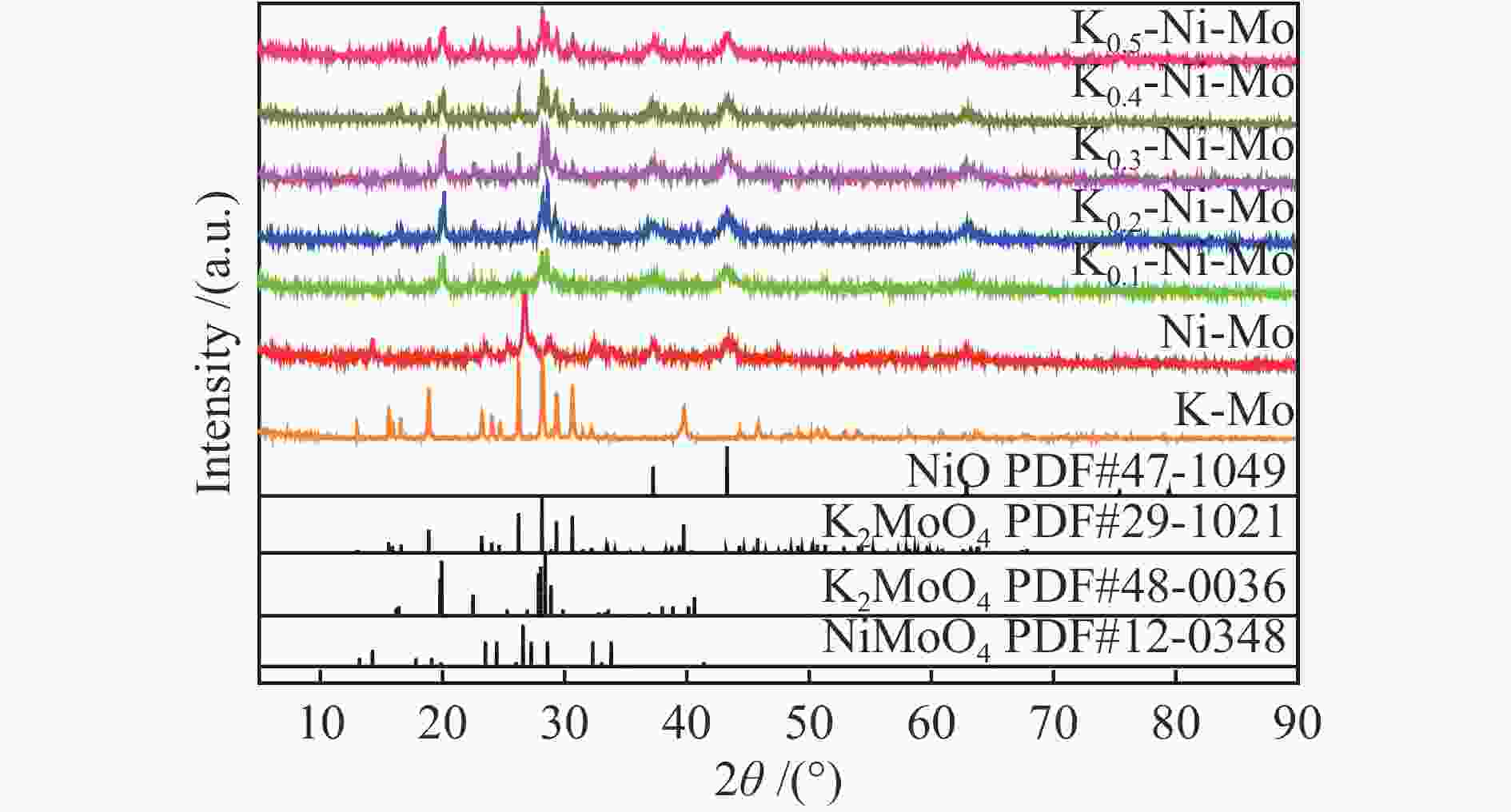
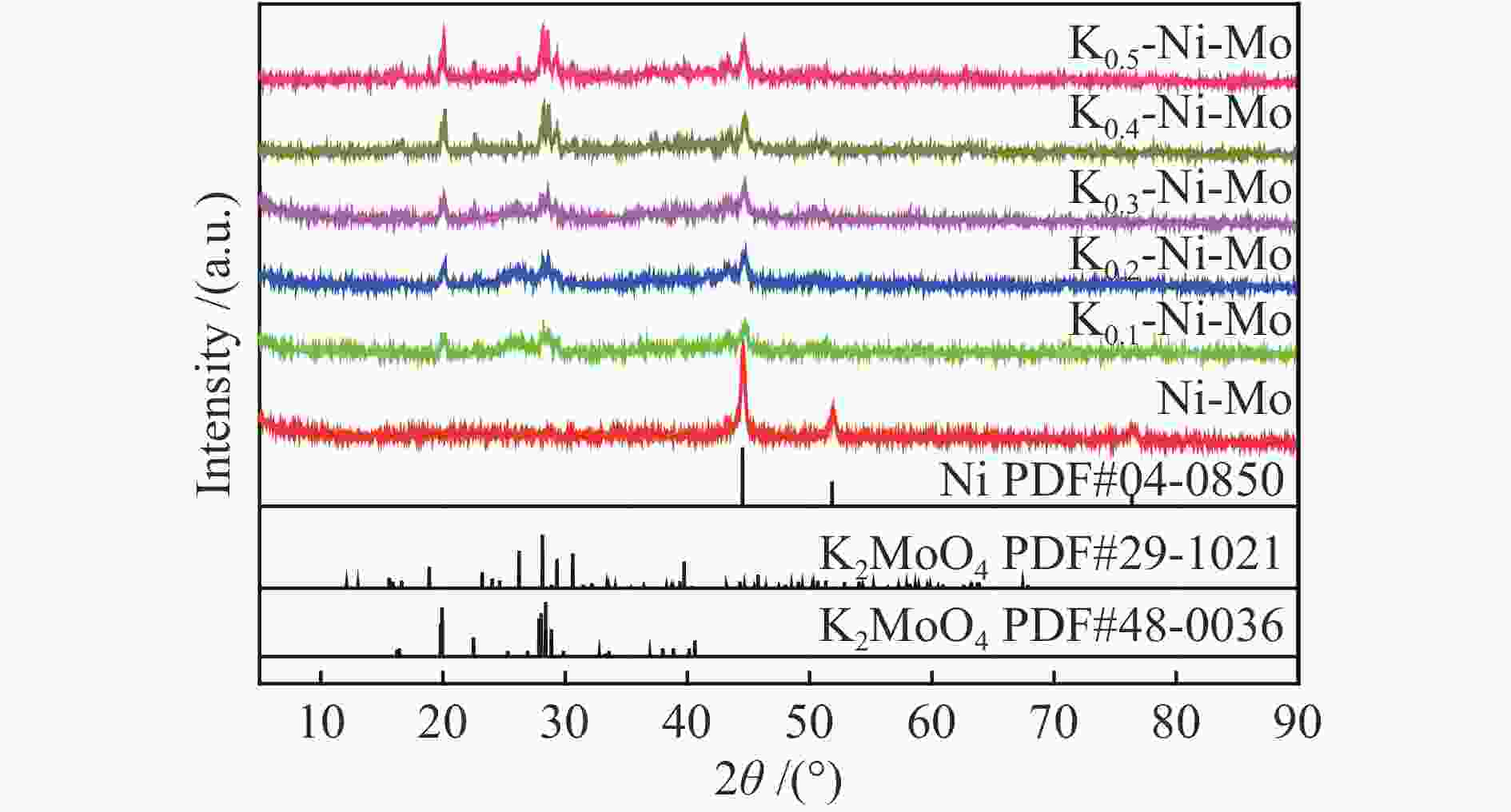
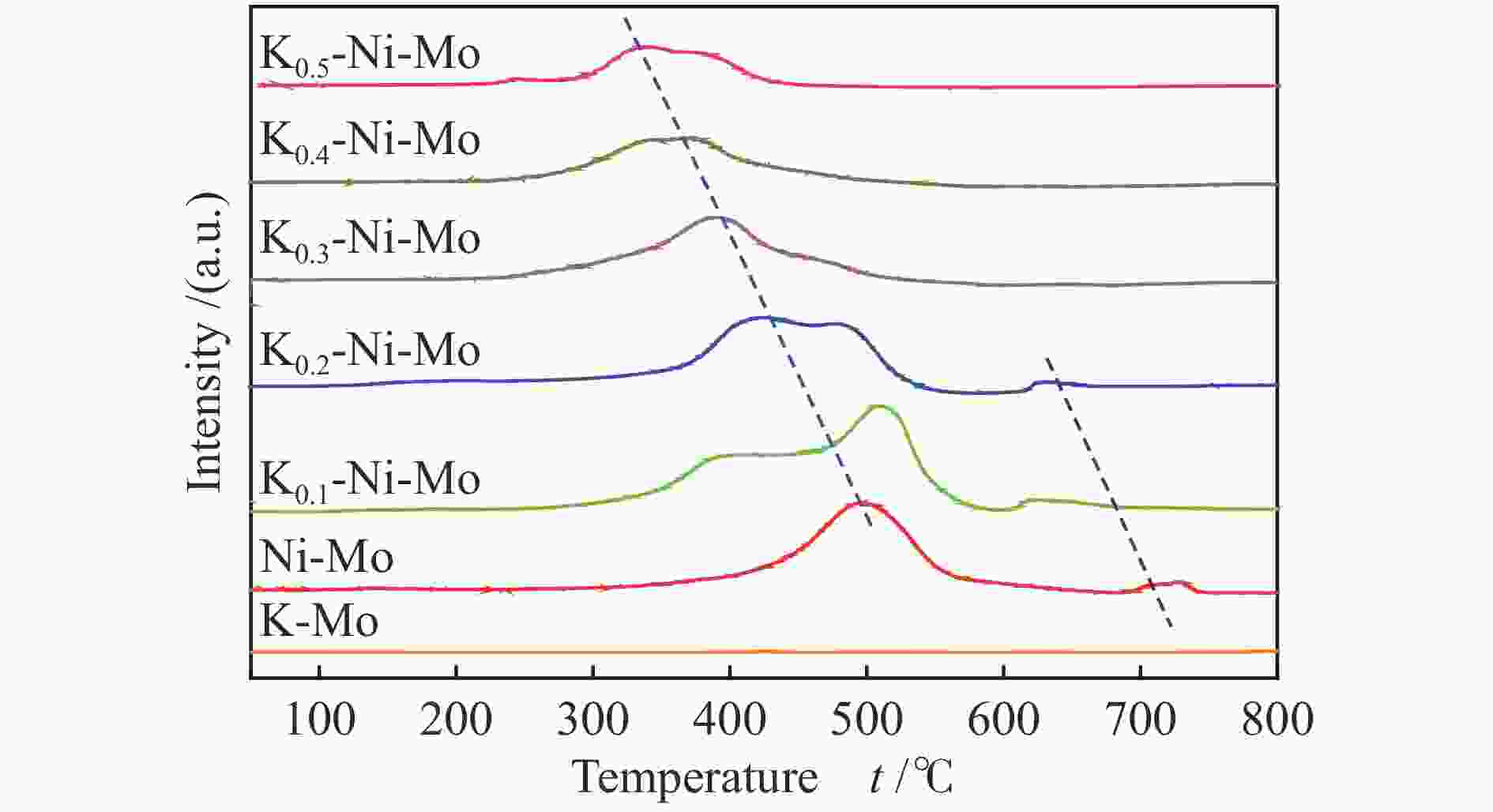
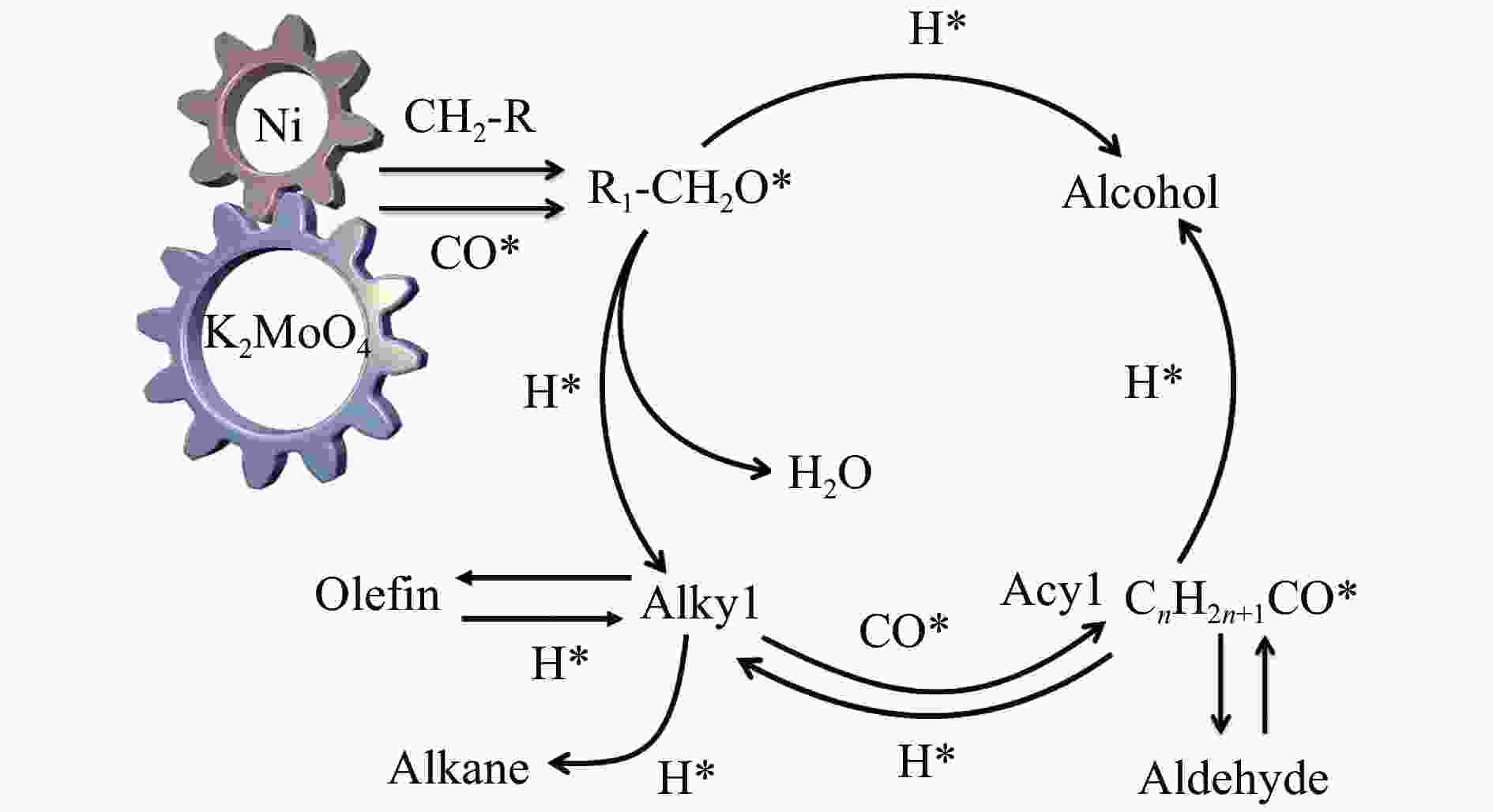
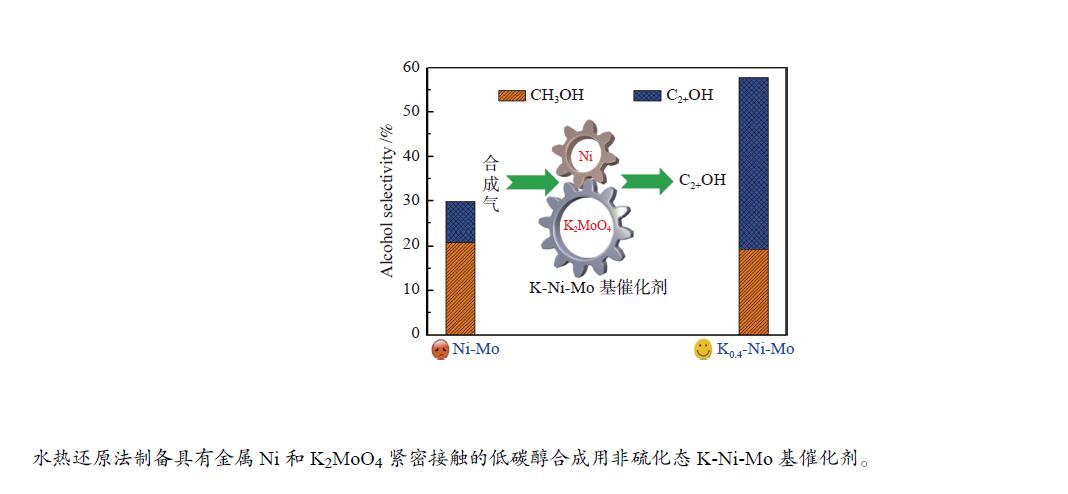
摘要: 采用水热还原法制备出具有金属Ni和K2MoO4紧密接触的合成低碳醇用非硫化态K-Ni-Mo基催化剂。通过XRD、N2物理吸附-脱附、H2-TPR、HR-TEM、SEM-EDS、XPS、H2-TPD、CO-TPD和CO2-TPD等手段对所合成催化剂进行了分析表征。研究结果显示,向Ni-Mo基催化剂中引入K可产生K2MoO4相,同时伴随NiMoO4相含量的降低,显著提升了K-Ni-Mo基催化剂上CO非解离吸附活化能力,从而促进了CO转化和醇类产物的形成。此外,同时增加的催化剂表面碱性可提高催化剂上碱性羟基基团数量,进而有效降低烃类选择性,表现出优异的合成低碳醇性能。其中,K0.4-Ni-Mo基催化剂具有最优的催化性能,在5 MPa、240 ℃、空速5000 h−1的反应条件下,CO转化率达到19.6%,总醇选择性57.8%,其中,总醇中C2+醇选择性为66.5%。Abstract: A series of non-sulfurized K-Ni-Mo-based catalysts with close contact between Ni and K2MoO4 were prepared by hydrothermal reduction for higher alcohol synthesis from syngas. The as-prepared catalysts were characterized by XRD, N2 adsorption-desorption, H2-TPR, HR-TEM, SEM-EDS, XPS, H2-TPD, CO-TPD and CO2-TPD techniques. The results indicate that the introduction of K facilitates the formation of the K2MoO4 phase while brings about a decrease of NiMoO4. It can significantly assist the non-dissociative activation of CO for insertion and subsequently alcohol formation. Moreover, the addition of K increases the surface basicity, which leads to more amount of basic hydroxy groups on the catalytic surface. The catalytic basicity ameliorates the production of alcohols. In particular, the K0.4-Ni-Mo catalyst shows the best catalytic behavior with CO conversion of 19.6%, total alcohol selectivity of 57.8% and C2+ alcohol selectivity in the total alcohols of 66.5% at GHSV of 5000 h−1, 240 °C, and 5 MPa.
-
图 1 合成气制低碳醇反应装置示意图
1:气瓶;2:系统前定压器;3:热电偶;4:加热炉;5:催化剂床层;6:反应器;7:热阱;8:冷阱;9:系统后定压器;10:气相色谱
Figure 1 Schematic diagram of reaction device for higher alcohols synthesis from syngas
1: Gas cylinder; 2: Pre-reaction pressure regulator; 3: Thermocouple; 4: Heating furnace; 5: Catalytic bed; 6: Reactor; 7: Hot trap; 8: Cold trap; 9: Back-pressure regulator; 10: Gas chromatography
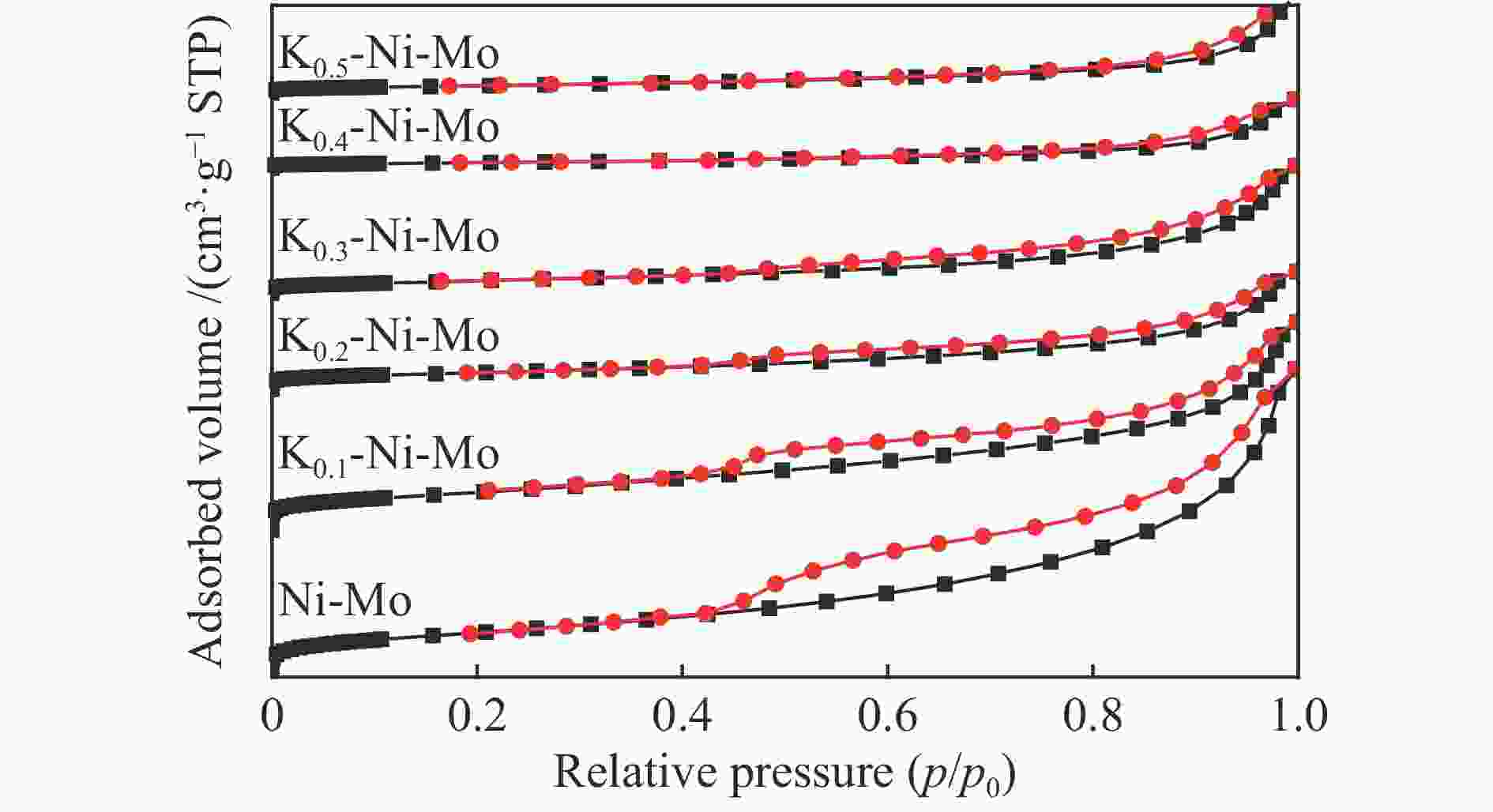
表 1 不同K含量的K-Ni-Mo基催化剂的织构参数
Table 1 Texture parameters of the K-Ni-Mo-based catalysts
Catalyst SBET/(m2·g−1) vmic/(cm3·g−1) Dave/nm Ni-Mo 72.6 0.21 11.7 K0.1-Ni-Mo 65.9 0.15 9.3 K0.2-Ni-Mo 31.2 0.09 12.1 K0.3-Ni-Mo 24.5 0.10 16.8 K0.4-Ni-Mo 10.0 0.05 20.8 K0.5-Ni-Mo 9.3 0.05 22.4 SBET: BET surface area; vmic: pore volume; Dave: pore diameter 表 2 不同K含量的K-Ni-Mo基催化剂的Ni/K2MoO4比
Table 2 Ni/K2MoO4 ratios of the K-Ni-Mo-based catalysts
Catalyst Ni/K2MoO4 ratio a K0.1-Ni-Mo 0.43 K0.2-Ni-Mo 1.23 K0.3-Ni-Mo 1.46 K0.4-Ni-Mo 1.58 K0.5-Ni-Mo 0.89 a: by XPS 表 3 不同K-Ni-Mo基催化剂上低碳醇合成反应性能a
Table 3 Catalytic performances of the K-Ni-Mo-based catalystsa
Catalyst CO conversion/% STYROH
/(mg·g−1·h−1)Product selectivity w/%b Alcohol distribution C/% ROHc CHnd CH4 MeOH C2+OHe Ni-Mo 28.1 52 29.9 70.1 49.6 69.7 30.3 K0.1-Ni-Mo 25.4 38 38.9 61.1 59.4 46.7 53.3 K0.2-Ni-Mo 21.6 37 40.1 59.9 55.9 39.8 60.2 K0.3-Ni-Mo 20.5 31 43.2 56.8 57.5 37.6 62.4 K0.4-Ni-Mo 19.6 71 57.8 42.2 41.3 33.5 66.5 K0.5-Ni-Mo 19.7 69 53.6 46.4 57.8 36.7 63.3 a: Reaction conditions: t =240 °C, p=5 MPa, GHSV=5000 h−1, H2/CO=2; b: CO2 free; c: ROH=total alcohol; d: CHn=total hydrocarbon;
e: Alcohol with carbon numbers of 2–5 (mainly ethanol, 1-propanol, 1-butanol, 1-pentanol, and 2-propanol) -
[1] BAO J, YANG G, YONEYAMA Y, TSUBAKI N. Significant advances in C1 catalysis: Highly efficient catalysts and catalytic reactions[J]. ACS Catal,2019,9(4):3026−3053. doi: 10.1021/acscatal.8b03924 [2] DAMMA D, SMIRNIOTIS P G. Recent advances in the direct conversion of syngas to oxygenates[J]. Catal Sci Technol,2021,11(16):5412−5431. doi: 10.1039/D1CY00813G [3] LIU H, AN J, CHEN T, WANG G. Low temperature conversion of syngas to ethanol in liquid phase over Fe modified CuZn catalyst: The promoting effect of Fe[J]. J Energy Inst,2019,92(6):1736−1742. doi: 10.1016/j.joei.2019.01.001 [4] LIU J, LIU Y, YAN W, YANG D, FAN J, HUANG W. Effect of zinc source on the ethanol synthesis from syngas over a slurry CuZnAl catalyst[J]. Int J Hydrogen Energy,2020,45(43):22469−22479. doi: 10.1016/j.ijhydene.2020.06.166 [5] 王慧君, 武应全, 田少鹏, 王立言, 弓娜娜, 解红娟, 谭猗生. F-T组分改性KCuZrO2催化剂上CO加氢合成异丁醇的性能研究[J]. 燃料化学学报,2020,48(3):302−310. doi: 10.3969/j.issn.0253-2409.2020.03.006WANG Hui-jun, WU Ying-quan, TIAN Shao-peng, WANG Li-yan, GONG Na-na, XIE Hong-juan, TAN Yi-sheng. Study on the performance of F-T component modified KCuZrO2 catalyst for CO hydrogenation to isobutanol[J]. J Fuel Chem Technol,2020,48(3):302−310. doi: 10.3969/j.issn.0253-2409.2020.03.006 [6] ZHAO L, DUAN J, ZHANG Q, LI Y, FANG K. Preparation, structural characteristics, and catalytic performance of Cu-Co alloy supported on Mn-Al oxide for higher alcohol synthesis via syngas[J]. Ind Eng Chem Res,2018,57(44):14957−14966. doi: 10.1021/acs.iecr.8b03304 [7] ZHAO L, MU X, LIU T, FANG K. Bimetallic Ni-Co catalysts supported on Mn-Al oxide for selective catalytic CO hydrogenation to higher alcohols[J]. Catal Sci Technol,2018,8(8):2066−2076. doi: 10.1039/C7CY02555F [8] LI Z, ZENG Z, YAO D, FAN S, GUO S, LV J, HUANG S, WANG Y, MA X. High-performance CoCu catalyst encapsulated in KIT-6 for higher alcohol synthesis from syngas[J]. ACS Sustainable Chem Eng,2020,8(1):200−209. doi: 10.1021/acssuschemeng.9b05133 [9] 郭海军, 李清林, 张海荣, 熊莲, 彭芬, 姚时苗, 陈新德. 凹凸棒石负载Cu-Fe-Co基催化剂组合体系用于CO加氢制备低碳醇[J]. 燃料化学学报,2019,47(11):1346−1356. doi: 10.3969/j.issn.0253-2409.2019.11.009GUO Hai-jun, LI Qing-lin, ZHANG Hai-rong, XIONG Lian, PENG Fen, YAO Shi-miao, CHEN Xin-de. Attapulgite supported Cu-Fe-Co based catalyst combination system for CO hydrogenation to lower alcohols[J]. J Fuel Chem Technol,2019,47(11):1346−1356. doi: 10.3969/j.issn.0253-2409.2019.11.009 [10] ZENG Z, LI Z, GUAN T, GUO S, HU Z, WANG J, RYKOV A, LV J, HUANG S, WANG Y, MA X. CoFe alloy carbide catalysts for higher alcohols synthesis from syngas: Evolution of active sites and Na promoting effect[J]. J Catal,2022,405:430−444. doi: 10.1016/j.jcat.2021.12.024 [11] 杨展董, 马恩娟, 张乾, 栾春晖, 黄伟. 氮掺杂碳纳米管担载CuCoCe对合成气制低碳醇的催化性能[J]. 燃料化学学报, 2020, 48(7): 804–812.YANG Zhan-dong, MA En-juan, ZHANG Qian, LUAN Chun-hui, HUANG Wei. Catalytic performance of CuCoCe supported on nitrogen-doped carbon nanotubes for the synthesis of higher alcohols from syngas[J] J Fuel Chem Technol, 2020, 48(7): 804–812. [12] ZHANG L, BALL M R, LIU Y, KUECH T F, HUBER G W, MAVRIKAKIS M, HERMANS I, DUMESIC J A. Synthesis gas conversion over Rh/Mo catalysts prepared by atomic layer deposition[J]. ACS Catal,2019,9(3):1810−1819. doi: 10.1021/acscatal.8b04649 [13] ASUNDI A S, HOFFMAN A S, BOTHRA P, BOUBNOV A, VILA F D, YANG N, SINGH J A, ZENG L, RAIFORD J A, ABILD-PEDERSEN F, BARE S R, BENT S F. Understanding structure-property relationships of MoO3-promoted Rh catalysts for syngas conversion to alcohols[J]. J Am Chem Soc,2019,141(50):19655−19668. doi: 10.1021/jacs.9b07460 [14] ZHAO L, LI Y, LIU X, FANG K. Low-temperature synthesis of high-performance nano-MoS2-based catalyst via non-thermal plasma for higher alcohol synthesis from syngas[J]. Catal Today,2020,355:17−25. doi: 10.1016/j.cattod.2019.01.069 [15] LI J, HU R, QU H, SU Y, WANG N, SU H, GU X. Radio-frequency thermal plasma-induced novel chainmail-like core-shell MoO2 as highly stable catalyst for converting syngas to higher alcohols[J]. Appl Catal B: Environ,2019,249:63−71. doi: 10.1016/j.apcatb.2019.02.060 [16] 熊烨. 合成气制醇类的钼基催化剂的研究进展[J]. 天然气化工(C1化学与化工),2019,44(3):117−121.XIONG Ye. Research progress in Mo-based catalysts for synthesis of alcohols from syngas[J]. Nat Gas Chem Ind,2019,44(3):117−121. [17] YANG Y, LIN T, QI X, YU F, AN Y, LI Z, DAI Y, ZHONG L, WANG H, SUN Y. Direct synthesis of long-chain alcohols from syngas over CoMn catalysts[J]. Appl Catal A: Gen,2018,549:179−187. doi: 10.1016/j.apcata.2017.09.037 [18] ZHANG F, LI Y, GAO S, FANG H, LIANG X, YUAN Y. Synthesis of higher alcohols by CO hydrogenation on a K-promoted Ni-Mo catalyst derived from Ni-Mo phyllosilicate[J]. Catal Sci Technol,2018,8(16):4219−4228. doi: 10.1039/C8CY01095A [19] ISHIDA T, YANAGIHARA T, LIU X, OHASHI H, HAMASAKI A, HONMA T, OJI H, YOKOYAMA T, TOKUNAGA M. Synthesis of higher alcohols by Fischer-Tropsch synthesis over alkali metal-modified cobalt catalysts[J]. Appl Catal A: Gen,2013,458:145−154. doi: 10.1016/j.apcata.2013.03.042 [20] MA W, KUGLER E L, WRIGHT J, DADYBURJOR D B. Mo-Fe catalysts supported on activated carbon for synthesis of liquid fuels by the Fischer-Tropsch process: Effect of Mo addition on reducibility, activity, and hydrocarbon selectivity[J]. Energy Fuels,2006,20(6):2299−2307. doi: 10.1021/ef0602372 [21] ASUNDI A S, HOFFMAN A S, CHI M, NATHAN S S, BOUBNOV A, HONG J, BARE S R, BENT S F. Enhanced alcohol production over binary Mo/Co carbide catalysts in syngas conversion[J]. J Catal,2020,391:446−458. doi: 10.1016/j.jcat.2020.09.003 [22] ZHANG L, BALL M R, RIVERA-DONES K R, WANG S-C, KUECH T F, HUBER G W, HERMANS I, DUMESIC J A. Synthesis gas conversion over molybdenum-based catalysts promoted by transition metals[J]. ACS Catal,2020,10(1):365−374. doi: 10.1021/acscatal.9b03968 [23] WANG N, HU R, LI J, BAI F, ZHANG Y, SU H, GU X. Insight into the promotion mechanism of K and Ni in sulfide molybdenum-based catalysts for higher alcohols synthesis from syngas[J]. Catal Commun,2017,91:57−61. doi: 10.1016/j.catcom.2016.12.015 [24] BOAHENE P E, DALAI A K. Higher alcohols synthesis: Experimental and process parameters study over a CNH-supported KCoRhMo catalyst[J]. Ind Eng Chem Res,2017,56(46):13552−13565. doi: 10.1021/acs.iecr.7b02146 [25] LIAKAKOU E T, HERACLEOUS E, TRIANTAFYLLIDIS K S, LEMONIDOU A A. K-promoted NiMo catalysts supported on activated carbon for the hydrogenation reaction of CO to higher alcohols: Effect of support and active metal[J]. Appl Catal B: Environ,2015,165:296−305. doi: 10.1016/j.apcatb.2014.10.027 [26] XIE W, ZHOU J, JI L, SUN S, PAN H, ZHU J, GAO C, BAO J. Targeted design and synthesis of a highly selective Mo-based catalyst for the synthesis of higher alcohols[J]. RSC Adv,2016,6(45):38741−38745. doi: 10.1039/C6RA05332G [27] 李莹, 赵璐, 刘晓展, 曾春新, 房克功. 低温等离子体制备低碳醇合成用KNiMo基催化剂及其结构性能表征[J]. 燃料化学学报,2019,47(5):513−522. doi: 10.3969/j.issn.0253-2409.2019.05.001LI Ying, ZHAO Lu, LIU Xiao-zhan, ZENG Chun-xin, FANG Ke-gong. Preparation of KNiMo-based catalysts by using non-thermal plasma and their catalytic performance in the synthesis of higher alcohols from syngas[J]. J Fuel Chem Technol,2019,47(5):513−522. doi: 10.3969/j.issn.0253-2409.2019.05.001 [28] MA C, LI H, LIN G, ZHANG H. Ni-decorated carbon nanotube-promoted Ni-Mo-K catalyst for highly efficient synthesis of higher alcohols from syngas[J]. Appl Catal B: Environ,2010,100(1/2):245−253. doi: 10.1016/j.apcatb.2010.07.040 [29] ZHANG X, LUAN X, DAI X, REN Z, CUI M, ZHAO H, NIE F, HUANG X. Enhanced higher alcohol synthesis from CO hydrogenation on Zn-modified MgAl-mixed oxide supported KNiMoS-based catalysts[J]. Ind Eng Chem Res,2020,59(4):1413−1421. doi: 10.1021/acs.iecr.9b04047 [30] ASLAM W, BELTRAMINI J N, ATANDA L A, BATALHA N R, SCHULLI T U, KONAROVA M. The catalytic activity of KMoCo carbon spheres for higher alcohols synthesis from syngas[J]. Appl Catal A: Gen,2020,605:117803. doi: 10.1016/j.apcata.2020.117803 [31] FERRARI D, BUDRONI G, BISSON L, RANE N J, DICKIE B D, KANG J H, ROZEVELD S J. Effect of potassium addition method on MoS2 performance for the syngas to alcohol reaction[J]. Appl Catal A: Gen,2013,462:302−309. [32] YONG J, LUAN X, DAI X, ZHANG X, QIAO H, YANG Y, HUANG X. Tuning the metal-support interaction in supported K-promoted NiMo catalysts for enhanced selectivity and productivity towards higher alcohols in CO hydrogenation[J]. Catal Sci Technol,2017,7(18):4206−4215. doi: 10.1039/C7CY01295K [33] ZHANG M, ZHANG W, XIE W, QI Z, WU G, LV M, SUN S, BAO J. Effects of cobalt promoter and reduction temperature on the surface species and syngas adsorption of K-Co-Mo/C catalyst for mixed alcohols synthesis[J]. J Mol Catal A: Chem,2014,395:269−275. doi: 10.1016/j.molcata.2014.08.029 [34] ZHANG X, KIM D, LEE L Y S. Copper-doped ZnS with internal phase junctions for highly selective CO production from CO2 photoreduction[J]. ACS Appl Energy Mater,2021,4(3):2586−2592. doi: 10.1021/acsaem.0c03163 [35] KRYSHTAB T, KHOMCHENKO V S, ANDRACA-ADAME J A, RODIONOV V E, KHACHATRYAN V B, TZYRKUNOV Y A. The influence of doping element on structural and luminescent characteristics of ZnS thin films[J]. Superlattices Microstruct,2006,40(4/6):651−656. doi: 10.1016/j.spmi.2006.09.001 [36] FAN X, LIU D, ZHAO Z, LI J, LIU J. Influence of Ni/Mo ratio on the structure-performance of ordered mesoporous Ni-Mo-O catalysts for oxidative dehydrogenation of propane[J]. Catal Today,2020,339:67−78. doi: 10.1016/j.cattod.2019.02.036 [37] WANGER C D, RIGGS W M, DAVIS L E, MOULDER J F, MUILENBERG G E. Handbook of X-Ray photoelectron Spectroscopy[M]. Eden Prairie, Minn: Perkin-Elmer Corporation, Physical Electronics Division, 1979. [38] ZHAO L, MU X, YU M, FANG K. A novel catalyst for higher alcohol synthesis from syngas: Co-Zn supported on Mn-Al oxide[J]. Fuel Process Technol,2018,177:16−29. doi: 10.1016/j.fuproc.2018.04.006 [39] SHI X, YU H, GAO S, LI X, FANG H, LI R, LI Y, ZHANG L, LIANG X, YUAN Y. Synergistic effect of nitrogen–doped carbon-nanotube-supported Cu-Fe catalyst for the synthesis of higher alcohols from syngas[J]. Fuel,2017,210:241−248. doi: 10.1016/j.fuel.2017.08.064 [40] 张文文, 栾春晖, 黄伟. CuCoAl催化剂中各金属在合成气制备低碳醇反应中的作用[J]. 天然气化工(C1化学与化工),2021,46(5):43−48.ZHANG Wen-wen, LUAN Chun-hui, HUANG Wei. Role of metals in CuCoAl catalysts in synthesis of lower alcohols from syngas[J]. Nat Gas Chem Ind,2021,46(5):43−48. [41] XI X, ZENG F, CAO H, CANNILLA C, BISSWANGER T, DE GRAAF S, PEI Y, FRUSTERI F, STAMPFER C, PALKOVITS R, HEERES H J. Enhanced C3+ alcohol synthesis from syngas using KCoMoSx catalysts: Effect of the Co-Mo ratio on catalyst performance[J]. Appl Catal B: Environ,2020,272:118950. doi: 10.1016/j.apcatb.2020.118950 [42] LIAKAKOU E T, HERACLEOUS E. Transition metal promoted K/Mo2C as efficient catalysts for CO hydrogenation to higher alcohols[J]. Catal Sci Technol,2016,6(4):1106−1119. doi: 10.1039/C5CY01173F [43] HUANG C, ZHU C, ZHANG M, LU Y, WANG Q, QIAN H, CHEN J, FANG K. Direct conversion of syngas to higher alcohols over a CuCoAl| t-ZrO2 multifunctional catalyst[J]. ChemCatChem,2021,13(13):3184−3197. doi: 10.1002/cctc.202100293 [44] BAO J, SUN Z, FU Y, BIAN G, ZHANG Y, TSUBAKI N. Mixed alcohol synthesis from syngas on K-Co-Mo/C catalyst prepared by a sol-gel method[J]. Top Catal,2009,52(6/7):789−794. doi: 10.1007/s11244-009-9200-x [45] ZAMAN S, SMITH K J. A review of molybdenum catalysts for synthesis gas conversion to alcohols: Catalysts, mechanisms and kinetics[J]. Catal Rev,2012,54(1):41−132. doi: 10.1080/01614940.2012.627224 [46] AO M, PHAM G H, SUNARSO J, TADE M O, LIU S. Active centers of catalysts for higher alcohol synthesis from syngas: A review[J]. ACS Catal,2018,8(8):7025−7050. doi: 10.1021/acscatal.8b01391 -




 下载:
下载: