Effects of phosphating process of MoP catalyst on hydrogenation of acetic acid to ethanol
-
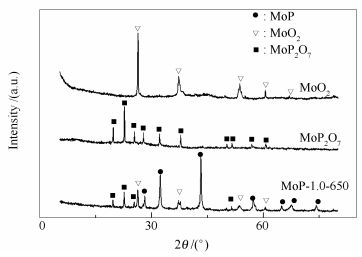
摘要: 制备了系列磷化钼催化剂用于醋酸加氢合成乙醇活性评价,并采用XRD、XPS和SEM等技术对催化剂进行表征。结果表明,催化剂除含有MoP外,还有MoP2O7和MoO2等物种,催化醋酸生成乙醇的活性物种是MoP,或者是MoP与MoP2O7、MoO2协同起催化作用。磷化温度一定程度上影响催化剂的形成和活性组分的分布,磷化温度太低,MoP形成量少,磷化温度太高,MoP发生团聚和烧结,磷化温度为650℃时制备的催化剂活性最高。磷钼物质的量比为1.0时催化剂的乙醇合成性能最高。Abstract: A series of molybdenum phosphide (MoP) catalysts for the hydrogenation of acetic acid to ethanol were successfully synthesized and identified by means of X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS) and Scanning electron microscope (SEM). The results reveal that MoP2O7 and MoO2 exist on the catalyst surface together with MoP. MoP or the synergistic effects of MoP2O7 and MoO2 species play roles in hydrogenation of acetic acid to ethanol. Phosphating temperature significantly affects the formation and dispersion of phosphide. A low phosphating temperature is not sufficient for the formation of MoP, but a high phosphating temperature leads to the agglomeration of MoP. The catalyst reduced at 650℃ has the highest hydrogenation activity and its P/Mo molar ratio is 1.0.
-
Key words:
- molybdenum phosphide /
- acetic acid /
- hydrogenation /
- ethanol
-
表 1 MoP-1.0-650、MoP2O7和MoO2催化醋酸加氢的活性
Table 1 Acetic acid conversion and product distribution of MoP-1.0-650, MoP2O7 and MoO2
Catalyst CH3COOH conversion x/% Selectivity s/% CH3CH2OH CH3COOCH2CH3 CH3OH CH3CHO C2H4 CH3 C2H6 MoP-1.0-650 93.75 91.23 4.76 0.12 0.91 0.32 0.15 2.51 MoP2O7 94.03 16.79 14.87 0.06 0.47 58.11 0.41 9.29 MoO2 88.93 12.35 22.51 0.05 0.87 6.44 0.05 57.73 t=260 ℃, p=4.0 MPa, GHSV=8 760 h-1, LHSV=0.6 h-1 表 2 不同磷钼物质的量比催化剂的催化性能
Table 2 Activity of catalysts synthesized with different P/Mo molar ratio
Catalyst t/℃ CH3COOH conversion x/% Selectivity s/% CH3CH2OH CH3COOCH2CH3 CH3OH CH3CHO C2H4 CH3 C2H6 MoP-0.8-650 240 79.72 73.52 12.95 0.25 0.68 2.05 0.95 9.6 260 87.41 80.53 6.19 0.21 0.82 1.89 0.72 9.64 MoP-1.0-650 240 86.62 82.67 13.51 0.13 0.7 0.75 0.21 2.03 260 93.75 91.23 4.76 0.12 0.91 0.32 0.15 2.51 MoP-1.2-650 240 81.53 80.75 11.83 0.13 0.73 0.71 1.02 4.83 260 86.36 85.23 6.36 0.13 0.84 0.71 1.77 4.96 p=4.0 MPa, GHSV=8 760 h-1, LHSV=0.6 h-1 -
[1] ZHOU M C, ZHANG H T, MA H F, YING W Y. The catalytic properties of K modified PtSn/Al2O3 catalyst for acetic acid hydrogenation to ethanol[J]. Fuel, 2016, 144(1):115-123. doi: 10.1016/j.fuproc.2015.12.022 [2] ONYESTYAK G. Ni/silica based bimetallic catalysts by solid-state co-reduction of admixed metal oxides for acetic acid hydroconversion to ethanol[J]. Res Chem Intermed, 2007, 38(2):1-4. https://www.researchgate.net/publication/276597075_Nisilica-based_bimetallic_catalysts_by_solid-state_co-reduction_of_admixed_metal_oxides_for_acetic_acid_hydroconversion_to_ethanol [3] 解建国, 王安杰, 李翔, 胡永康. MCM-41作载体制备磷化钼加氢脱硫催化剂[J].石油炼制与化工, 2009, 5(4):496-502. http://www.cnki.com.cn/Article/CJFDTOTAL-SYLH200702000.htmXIE Jian-guo, WANG An-jie, LI Xiang, HU Yong-kang. Preparation of MCM-41 supported molybdenum phosphide hydrodesulfurization catalyst[J]. Petro Process Petrochem, 2009, 5(4):496-502. http://www.cnki.com.cn/Article/CJFDTOTAL-SYLH200702000.htm [4] PEYMAN F, LIU J. Impact of volatile fatty acid recovery on economics of ethanol production from brown algae via mixed alcohol synthesis[J]. Chem Eng Res Des, 2015, 98(1):107-122. https://www.researchgate.net/publication/275653390_Impact_of_volatile_fatty_acid_recovery_on_economics_of_ethanol_production_from_brown_algae_via_mixed_alcohol_synthesis [5] 李凤艳, 李庆杰, 赵天波, 孙桂大, 李翠清.喹啉和二苯并噻吩在磷化钼催化剂上的HNN和HDS性能[J].石油化工高等学校学报, 2006, 19(2):001-004. http://www.cnki.com.cn/Article/CJFDTOTAL-SYHX200602000.htmLI Feng-yan, LI Qing-jie, ZHAO Tian-bo, SUN Gui-da, LI Cui-qing. Characteristic of quinoline hydrodenitrogenation and dibenzothiophene hydrodesulfurization over molubdenum phosphide catalysts[J]. J Petrochem Univ, 2006, 19(2):001-004. http://www.cnki.com.cn/Article/CJFDTOTAL-SYHX200602000.htm [6] ZHANG S B, DUAN X P, YE L M, LIN H Q, XIE Z X, YUAN Y Z. Production of ethanol by gas phase hydrogenation of acetic acid over carbon nanotube-supported Pt-Sn nanoparticles[J]. Catal Today, 2013, 215(1):260-266. https://www.researchgate.net/publication/277536790_Production_of_ethanol_by_gas_phase_hydrogenation_of_acetic_acid_over_carbon_nanotube-supported_Pt-Sn_nanoparticles [7] CLARK P, WANG X, OYANA S T. Characterization of silica supported molybdenum and tungsten phosphide hydroprocessing catalysts by 31P nuclear magnetic resonance spectroscopy[J]. J Catal, 2002, 207(1):256-265. https://www.researchgate.net/publication/244394030_Characterization_of_Silica-Supported_Molybdenum_and_Tungsten_Phosphide_Hydroprocessing_Catalysts_by_31P_Nuclear_Magnetic_Resonance_Spectroscopy [8] 鄢景森, 王安杰, 李翔, 鲁墨弘, 胡永康.以SiO2为载体的磷化钼催化剂上喹啉的加氢脱氮反应[J].石油学报 (石油加工), 2006, 22(3):28-32. http://www.cnki.com.cn/Article/CJFDTotal-SXJG200603000.htmYAN Jing-sen, WANG An-jie, LI Xiang, LU Mo-hong, HU Yong-kang. Hydrodenitrogenation of quinolone catalyzed by SiO2-supported molybdenum phosphide[J]. Acta Pet Sin (Pet Process Set), 2006, 22(3):28-32. http://www.cnki.com.cn/Article/CJFDTotal-SXJG200603000.htm [9] FENG Z C, LIANG C H, WU W C, WU Z L, SANTENE R A V, LI C. Carbon monoxide adsorption on molybdenum phosphides:Fourier transform infrared spectroscopic and density functional theory studies[J]. Phys Chem, 2003, 107(1):13698-13702. https://www.researchgate.net/publication/231636627_Carbon_Monoxide_Adsorption_on_Molybdenum_Phosphides_Fourier_Transform_Infrared_Spectroscopic_and_Density_Functional_Theory_Studies [10] 林迎春, 黎先财, 杨爱军, 郭辉瑞.制备条件对磷化钼催化剂重整性能的影响[J].南昌大学学报, 2012, 34(4):312-315. http://www.cnki.com.cn/Article/CJFDTOTAL-NCDG201204003.htmLIN Yin-chun, LI Xian-cai, YANG Ai-jun, GUO Hui-rui. Effect of preparation condition to molybdenum phosphide catalysts for CO2 reforming of CH4[J]. J Nanchang Univ, 2012, 34(4):312-315. http://www.cnki.com.cn/Article/CJFDTOTAL-NCDG201204003.htm [11] OYAMA S T, CLARK P, LEDEN E J, REQUEJO F G. XAFS characterization of highly active alumina-supported molybdenum phosphide catalysts (MoP/Al2O3) for hydrotreating[J]. Phys Chem, 2001, 105(1):4961-4966. doi: 10.1021/jp004500q [12] 曲本连, 柴永明, 相春娥, 张景成, 刘晨光.磷化镍和磷化钼催化剂的原位XRD研究[J].石油学报 (石油加工), 2009, 25(4):496-502. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG200904005.htmQU Ben-lian, CHAI Yong-ming, XIANG Chun-e, ZHANG Jing-cheng, LIU Chen-guang. In-situ XRD study of nickel phosphide and molybdenum phosphide catalysts[J]. Acta Pet Sin (Pet Process Set), 2009, 25(4):496-502. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG200904005.htm [13] PEREZ-ROMO P, POTVIN C, MANOLI J M, CHEHIMI M, EGA-MARIADASSOU G D. Phosphorus-doped molybdenum oxynitrides and oxygen-modified molybdenum carbides:Synthesis, characterization and determination of turnoverrates for propene hydrogenation[J]. J Catal, 2002, 208(1):187-196. doi: 10.1006/jcat.2002.3564 [14] STINNER C, PRINS R, WEBER T. Formation, structure, and HDN activity of unsupported molybdenum phosphide[J]. J Catal, 2000, 191(2):438-444. doi: 10.1006/jcat.1999.2808 [15] ITO Y, KAWAMOTO H, SAKA S. Efficient and selective hydrogenation of aqueous acetic acid on Ru-Sn/TiO2 for bioethanol production from lignocellulosics[J]. Fuel, 2016, 178(1):118-123. https://www.researchgate.net/publication/299073977_Efficient_and_selective_hydrogenation_of_aqueous_acetic_acid_on_Ru-SnTiO2_for_bioethanol_production_from_lignocellulosics [16] CLARK P A, OYAMA S T. Alumina-supported molybdenum phosphide hydroprocessing catalysts[J]. J Catal, 2003, 218(1):78-87. doi: 10.1016/S0021-9517(03)00086-1 [17] CHENG R H, SHU Y Y, LI L, SUN J, WANG X D, ZHANG T. CO adsorption on highly dispersed MoP/Al2O3 prepared with citric acid[J]. Thermochim Acta, 2006, 450(1):42-46. doi: 10.1016/j.tca.2006.07.007 [18] OMYESTYA'K G, HARNOS S, BADARI C A, KLE'BERT S, ASZONYI A, VALYON J. Hydroconversion of acetic acid over indiumand phosphorus modified nickel/laponite catalysts[J]. React Kinet Mech Catal, 2015, 115(1):201-216. doi: 10.1007/s11144-014-0825-9 [19] 周桂林, 王普光, 蒋宗轩, 应品良, 李灿. MoP催化剂上乙炔选择性催化加氢[J].催化学报, 2011, 32(1):27-30. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA201101006.htmZHOU Gui-lin, WANG Pu-guang, JIANG Zong-xuan, YING Pin-liang, LI Can. Selective hydrogenation of acetylene over a MoP catalyst[J]. Chin J Catal, 2011, 32(1):27-30. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA201101006.htm [20] 龚树文, 张达, 邢生凯, 刘丽君, 李考真, 崔庆新.制备条件对磷化钼加氢脱硫催化活性的影响[J].燃料化学学报, 2009, 37(4):464-467. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17475.shtmlGONG Shu-wen, ZHANG Da, XING Sheng-kai, LIU Li-jun, LI Kao-zhen, CUI Qing-xin. Influence of the synthesis on HDS activity of molybdenum phosphide catalysts[J]. J Fuel Chem Technol, 2009, 37(4):464-467. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17475.shtml [21] MONTESINOS-CACELLANOS A, ZEPEDA T A, PAWELEC B, FIERRO L J, REYES J A. Preparation, characterization cerformance of alumina supported nanostructured Mo-phosphide systems[J]. Chem Mater, 2007, 19(1):5627-5636. https://www.researchgate.net/publication/231240400_Preparation_Characterization_and_Performance_of_Alumina-Supported_Nanostructured_Mo-Phosphide_Systems [22] GUO Q H, REN L L. Hydrodechlorination of trichloroethylene over MoP/-Al2O3 catalystwith high surface area[J]. Catal Today, 2016, 264(1):158-162. [23] YAO Z W, TONG J, QIAO X, JIANG J, ZHAO Y, LIU D M, ZHANG Y C, WANG H Y. Novel synthesis of dispersed molybdenum and nickel phosphides from thermal carbonization of metal and phosphorus-containing resins[J]. Dalton Trans, 2015, 44(2):19383-19391. https://www.researchgate.net/publication/283244169_Novel_synthesis_of_dispersed_molybdenum_and_nickel_phosphides_from_thermal_carbonization_of_metal-_and_phosphorus-containing_resins [24] PHILLIPS D C, SAWHILL S J, SELF R. Synthesis, characterization and hydrodesulfurization properties of silica-supported molybdenum phosphide catalysts[J]. J Catal, 2002, 207(1):266-273. http://www.sciencedirect.com/science/article/pii/S0021951702935244 -





 下载:
下载: