-
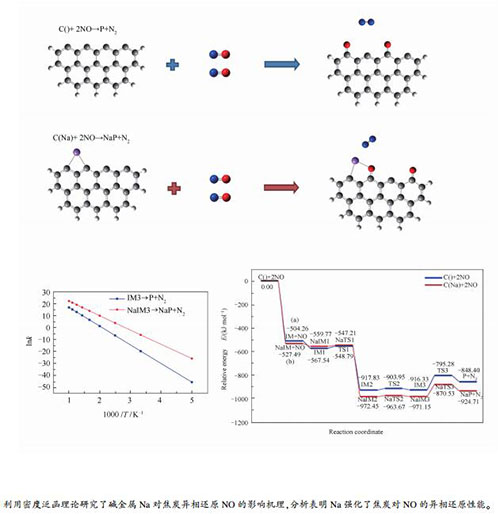
摘要: 基于密度泛函理论和过渡态理论,在分子水平上对焦炭异相还原NO以及碱金属钠的作用机理进行探究。结合单点能的零点能校正以及过渡态的虚频验证,发现钠能够有效促进焦炭对于第一个NO分子的吸附。尽管钠不能改变反应步骤,但可将焦炭异相还原NO决速步的活化能由121.04 kJ/mol降至100.62 kJ/mol;钠的存在使焦炭异相还原NO的指前因子增大且反应速率加快,增加了焦炭边缘的活性位点,强化了焦炭对于NO的异相还原性能。Abstract: A molecular modeling study based on density functional theory (DFT) and transition state theory (TST) was performed to investigate the effect of Na on the NO heterogeneous reduction by char; zero point energy correction was considered and the transition states was confirmed by frequency analysis. The results show that Na can effectively promote the adsorption of first NO molecule on to the char. The presence of Na cannot change the reaction steps, but reduce the activation energies of rate-determining steps from 121.04 kJ/mol to 100.62 kJ/mol. Moreover, the presence of Na can increase the pre-exponential factors as well as the reaction rate, meaning more active sites and enhanced catalytic performance of char in NO reduction.
-
Key words:
- biomass reburning /
- heterogeneous reduction /
- sodium /
- molecular simulation /
- kinetic analysis
-
表 1 反应路径各驻点能量及过渡态振动虚频
Table 1 Energies of various compounds and imaginary frequency of transition states
Species E/(kJ·mol-1) Frequency/cm-1 Species E/(kJ·mol-1) Frequency /cm-1 IM -2856326.30 - NaIM2 -3624341.19 - IM1 -3197558.53 - NaIM3 -3624339.89 - IM2 -3197908.82 - NaP+N2 -3624293.46 - IM3 -3197907.32 - NaTS1 -3623920.36 -263.48 P+N2 -3197839.39 - NaTS2 -3624328.91 -233.86 TS1 -3197483.39 -475.11 NaTS3 -3624234.95 -165.53 TS2 -3197895.36 -264.72 NO -341168.95 - TS3 -3197768.49 -131.19 N2 -287649.68 - NaIM -3282727.29 - R -2514653.09 - NaIM1 -3623928.51 - Na -426013.72 - 表 2 模型平均键长的计算值和文献值对比
Table 2 Calculated average bond length for the model used in this work, compared with those reported in the literature
表 3 NO在焦炭及Na修饰焦炭边缘吸附的Mulliken电荷布局分析
Table 3 Mulliken atomic charge populations for NO adsorption on the edge of char and Na-decorated char
Atom Charge /(a.u.) NO/R IM NaIM N1 0.041 -0.170 -0.186 O1 -0.041 -0.346 -0.503 C2 -0.029 0.302 0.328 C3 -0.024 0.172 0.178 表 4 反应动力学参数
Table 4 Reaction kinetic parameters
Reaction Pre-exponential factor A/s-1 Activation energy Ea/(kJ·mol-1) Arrhenius equation IM3→P+N2 1.50×1014 130.29 k=1.50×1014e-15670.84/T NaIM3→NaP+N2 7.32×1014 100.20 k=7.32×1014e-12050.92/T -
[1] 牛胜利, 韩奎华, 路春美.生物质先进再燃脱硝特性研究[J].燃料化学学报, 2010, 38(6):745-751. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract17669.shtmlNIU Sheng-li, HAN Kui-hua, LU Chun-mei. Study on nitrogen oxides reduction by advanced biomass reburning process[J]. J Fuel Chem Technol, 2010, 38(6):745-751. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract17669.shtml [2] 张国祥, 陈晓晖.富氧条件下金属催化CO还原NO的研究进展[J].化工进展, 2018, 37(12):4654-4661. http://d.old.wanfangdata.com.cn/Periodical/hgjz201812016ZHANG Guo-xiang, CHEN Xiao-hui. Research progress in metal catalysts for catalytic reduction of NO by CO with excess oxygen[J]. Chem Ind Eng Prog, 2018, 37(12):4654-4661. http://d.old.wanfangdata.com.cn/Periodical/hgjz201812016 [3] 阮丹, 齐砚勇, 李会东.高温无催化剂条件下CO还原NO数值模拟研究[J].硅酸盐通报, 2016, 35(6):1674-1681. http://d.old.wanfangdata.com.cn/Periodical/gsytb201606005RUAN Dan, QI Yan-yong, LI Hui-dong. Numerical simulation research of the reduction of NO by CO at high temperature without catalyst[J]. Ball Chin Ceram Soc, 2016, 35(6):1674-1681. http://d.old.wanfangdata.com.cn/Periodical/gsytb201606005 [4] LÓPEZ D, CALO J. The NO carbon reaction:The influence of potassium and CO on reactivity and populations of oxygen surface complexes[J]. Energy Fuels, 2007, 21(4):1872-1877. doi: 10.1021/ef070034t [5] LU P, HAO J T, YU W, ZHU X M, D X. Effects of water vapor and Na/K additives on NO reduction through advanced biomass reburning[J]. Fuel, 2015, 170:60-66. https://www.researchgate.net/publication/288020502_Effects_of_water_vapor_and_NaK_additives_on_NO_reduction_through_advanced_biomass_reburning [6] WU X, SONG Q, ZHAO H B, YAO Q. Catalytic mechanism of inherent potassium on the Char-NO reaction[J]. Energy Fuels, 2015, 29(11):7566-7571. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=8df78f24ab76897926b973f6d9ef876c [7] 周昊, 刘瑞鹏, 刘子豪, 程明, 岑可法.碱金属对焦炭燃烧过程中NOx释放的影响[J].煤炭学报, 2015, 40(5):1160-1164. http://d.old.wanfangdata.com.cn/Periodical/mtxb201505028ZHOU Hao, LIU Rui-peng, LIU Zi-hao, CHENG Ming, CEN Ke-fa. Influence of alkali metal on the evolution of NOx during coke combustion[J]. J China Coal Soc, 2015, 40(5):1160-1164. http://d.old.wanfangdata.com.cn/Periodical/mtxb201505028 [8] 刘银河, 刘艳华, 车得福, 徐通模.煤中灰分和钠添加剂对煤燃烧中氮释放的影响[J].中国电机工程学报, 2005, 25(4):136-141. http://d.old.wanfangdata.com.cn/Periodical/zgdjgcxb200504025LIU Yin-he, LIU Yan-hua, CHE De-fu, XU Tong-mo. Effects of minerals and sodium addition on nitrogen release during coal combustion[J]. Proc CSEE, 2005, 25(4):136-141. http://d.old.wanfangdata.com.cn/Periodical/zgdjgcxb200504025 [9] ZHAO Z B, LI W, QIU J S, WANG X Z, LI B Q. Influence of Na and Ca on the emission of NOx during coal combustion[J]. Fuel, 2006, 85(5):601-606. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=933eda0d9c7a6fefa840c1d93a243cb7 [10] ZHANG X X, LIN R Y. Effect of alkali metal elements on nitric oxide chemisorption at the edge of char:A DFT study[J]. Energy Procedia, 2019, 158:4805-4809. https://www.sciencedirect.com/science/article/pii/S1876610219307532 [11] 温正城, 周俊虎, 王智化, 岑可法.碱金属对煤焦异相还原NO的催化机理:量子化学研究[J].浙江大学学报(工学版), 2008, 42(8):1452-1457. http://d.old.wanfangdata.com.cn/Periodical/zjdxxb-gx200808033WEN Zheng-cheng, ZHOU Jun-hu, WANG Zhi-hua, CEN Ke-fa. Catalytic mechanism of alkali metal on NO heterogeneous reduction by char:quantum chemistry study[J]. J Zhejiang Univ-Sci(ES), 2008, 42(8):1452-1457. http://d.old.wanfangdata.com.cn/Periodical/zjdxxb-gx200808033 [12] PERRY S T, HAMBLY E M, FLETCHER T H, SOLUM M S, PUGMIRE R J. Solid-state 13C NMR characterization of matched tars and chars from rapid coal devolatilization[J]. Proc Combust Inst, 2000, 28(2):2313-2319. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=CC028008586 [13] GAO Z, YANG W, DING X, DING Y, YAN W P. Theoretical research on heterogeneous reduction of N2O by char[J]. Appl Therm Eng, 2017, 126:28-36. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=39883cac9b1d097d475518a0bf79d159 [14] 许紫阳, 岳爽, 王春波, 刘瑞琪.焦炭催化CO还原NO的反应机理研究[J].燃料化学学报, 2020, 48(3):266-274. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract29530.shtmlXU Zi-yang, YUE Shuang, WANG Chun-bo, LIU Rui-qi. Reaction mechanism of NO reduction with CO catalyzed by char[J]. J Fuel Chem Technol, 2020, 48(3):266-274. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract29530.shtml [15] 章勤, 张秀霞, 周俊虎, 周志军, 张彦威, 刘建忠, 岑可法. NO在焦炭表面的吸附特性[J].煤炭学报, 2013, 38(9):1651-1655. http://d.old.wanfangdata.com.cn/Periodical/mtxb201309023ZHANG Qin, ZHANG Xiu-xia, ZHOU Jun-hu, ZHOU Zhi-jun, ZHANG Yan-wei, LIU Jian-zhong, CEN Ke-fa. Characteristics of NO chemisorption on surface of char[J]. J China Coal Soc, 2013, 38(9):1651-1655. http://d.old.wanfangdata.com.cn/Periodical/mtxb201309023 [16] 赵鹏飞, 郭欣, 郑楚光.活性炭及氯改性活性炭吸附单质汞的机制研究[J].中国电机工程学报, 2010, 30(23):40-44. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zgdjgcxb201023007ZHAO Peng-fei, GUO Xin, ZHENG Chu-guang. Investigating the mechanism of elemental mercury binding on activated carbon and chlorine-embedded activated carbon[J]. Proc CSEE, 2010, 30(23):40-44. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zgdjgcxb201023007 [17] 钟俊, 高正阳, 丁艺, 余岳溪, 杨维结. Zigzag煤焦表面异相还原N2O反应[J].煤炭学报, 2017, 42(11):3028-3034. http://www.cnki.com.cn/Article/CJFDTotal-MTXB201711031.htmZHONG Jun, GAO Zheng-yang, DING Yi, YU Yue-xi, YANG Wei-jie. Heterogeneous reduction reaction of N2O by char based on Zigzag carbonaceous model[J]. J China Coal Soc, 2017, 42(11):3028-3034. http://www.cnki.com.cn/Article/CJFDTotal-MTXB201711031.htm [18] 朱恒毅, 孙保民, 信晶, 尹书剑, 肖海平.富氧燃烧环境下CO对煤焦异相还原NO的量子化学研究[J].煤炭学报, 2015, 40(7):1641-1647. http://d.old.wanfangdata.com.cn/Periodical/mtxb201507026ZHU Heng-yi, SUN Bao-min, XIN Jing, YIN Shu-jian, XIAO Hai-ping. Quantum chemistry research on NO heterogeneous reduction by char with the participation of CO under oxy-fuel combustion atmosphere[J]. J China Coal Soc, 2015, 40(7):1641-1647. http://d.old.wanfangdata.com.cn/Periodical/mtxb201507026 [19] ZHANG H, LIU J X, SHEN J, JIANG X M. Thermodynamic and kinetic evaluation of the reaction between NO (nitric oxide) and char(N) (char bound nitrogen) in coal combustion[J]. Energy, 2015, 82:312-321. https://www.sciencedirect.com/science/article/abs/pii/S036054421500064X [20] 宁华, 刘松, 邓年进, 黎光旭, 蓝志强. Nb(100)表面吸附氮气的第一性原理研究[J].广西大学学报(自然科学版), 2013, 3:769-776. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=gxdxxb201303041NING Hua, LIU Song, DENG Nian-jin, LI Guang-xu, LAN Zhi-qiang. Nitrogen adsorption on Nb(100) surface by using First-principles investigation[J]. J Guangxi Univ-Sci(NSE), 2013, 3:769-776. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=gxdxxb201303041 [21] 李淑萍, 孟江, 王继刚. NO在金属Ben(n=2-12)团簇表面的平行吸附[J].原子与分子物理学报, 2019, 36(2):240-245. http://d.old.wanfangdata.com.cn/Periodical/yzyfzwlxb201902011LI Shu-ping, MENG Jiang, WANG Ji-gang. Parallel adsorption for NO on the surfaces of Ben (n=2-12) clusters[J]. J At Mol Phys, 2019, 36(2):240-245. http://d.old.wanfangdata.com.cn/Periodical/yzyfzwlxb201902011 [22] CHEN N, YANG R T. Ab initio molecular orbital calculation on graphite:Selection of molecular system and model chemistry[J]. Carbon, 1998, 36(7):1061-1070. https://www.sciencedirect.com/science/article/abs/pii/S0008622398000785 [23] ZHANG X X, XIE M, WU H X, LV X X, ZHOU Z J. DFT study of the effect of Ca on NO heterogeneous reduction by char[J]. Fuel, 2020, 265. https://www.sciencedirect.com/science/article/pii/S0016236119323889 [24] OYARZúN A M, RADOVIC L R, KYOTANI T. An update on the mechanism of the graphene-NO reaction[J]. Carbon, 2015, 86:58-68. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d069c0a7408dd58c88b6146aefcef4e3 [25] GONZÁLEZ J D, MONDRAGÓN F, ESPINAL J F. Effect of calcium on gasification of carbonaceous materials with CO2:A DFT study[J]. Fuel, 2013, 114:199-205. https://www.researchgate.net/publication/271615653_Effect_of_calcium_on_gasification_of_carbonaceous_materials_with_CO2_A_DFT_study [26] ZHAO D, LIU H, SUN C L, XU L F, CAO Q X. DFT study of the catalytic effect of Na on the gasification of carbon-CO2[J]. Combust Flame, 2018, 197:471-486. https://www.sciencedirect.com/science/article/pii/S0010218018303924 [27] LIU R. Adsorption and dissociation of H2O on Au(111) surface:A DFT study[J]. Comput Theor Chem, 2013, 1019:141-145. https://www.sciencedirect.com/science/article/abs/pii/S2210271X13002892 [28] GAO Z Y, LV S K, YANG W J, YANG P F, MENG X X. Quantum chemistry investigation on the reaction mechanism of the elemental mercury, chlorine, bromine and ozone system[J]. J Mol Model, 2015, 21(6):2707. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=f709e4294faf3e6a3b7ee72663418ffe [29] 张秀霞.焦炭燃烧过程中氮转化机理与低NOx燃烧技术的开发[D].杭州: 浙江大学, 2012. http://cdmd.cnki.com.cn/Article/CDMD-10335-1012488726.htmZHANG Xiu-xia. Nitrogen conversion mechanism during char combustion and develepment of low NOx technology[D]. Hangzhou: Zhejiang University, 2012. http://cdmd.cnki.com.cn/Article/CDMD-10335-1012488726.htm [30] 易秋明, 刘华财, 阴秀丽, 吴创之.生物质新生半焦与冷态半焦CO2气化活性差异分析[J].新能源进展, 2015, 3:9-16. http://d.old.wanfangdata.com.cn/Periodical/xnyjz201503002YI Qiu-ming, LIU Hua-cai, YIN Xiu-li, WU Chuang-zhi. Comparative investigation of the CO2 gasification characteristics of biomass In-situ char and Ex-situ char[J]. J Circ Syst, 2015, 3:9-16. http://d.old.wanfangdata.com.cn/Periodical/xnyjz201503002 -





 下载:
下载: