Research on CuO/CeO2-ZrO2/SiC monolithic catalysts for hydrogen production from steam reforming of methanol
-
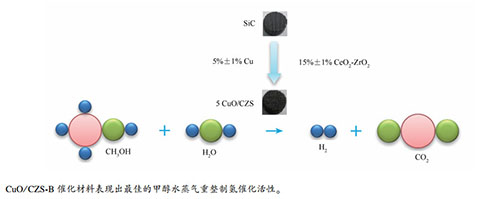
摘要: 采用浸渍法和溶胶凝胶法制备了CuO/CeO2-ZrO2/SiC整体催化剂,并将其用于甲醇水蒸气重整制氢反应中。结果表明,与CuO/CeO2-ZrO2颗粒催化剂相比,CuO/CeO2-ZrO2/SiC整体催化剂催化活性较好,产氢速率较快且重整气中CO体积分数较低。进一步探究了涂层涂覆量和CuO负载量对催化性能的影响,结果表明,当CeO2-ZrO2复合氧化物涂层涂覆量在15%±1%,CuO负载量为5%±1%时,催化性能较好;当反应温度为340℃,水醇物质的量比为1.2,甲醇水蒸气气体空速为4840 h-1时,甲醇转化率为86.0%,产氢速率为1490.0 L/(m3·s),重整气中CO体积分数为1.55%。最后通过单因素实验法探究了甲醇水蒸气气体空速、水醇物质的量比和反应温度对反应的影响。结果表明,随着气体空速变大,甲醇转化率下降,产氢速率上升,重整气中CO体积分数下降。随着水醇物质的量比增加,甲醇转化率先上升后下降,产氢速率先上升后下降,重整气中CO体积分数下降。随着反应温度的升高,甲醇转化率、产氢速率和重整气中CO体积分数均上升。Abstract: CuO/CeO2-ZrO2/SiC monolithic catalysts were prepared by the sol-gel and incipient-wetness impregnation methods, and then used in methanol steam reforming reaction for H2 production. The results indicated that CuO/CeO2-ZrO2/SiC monolithic catalysts showed better activity, higher hydrogen production rate and less CO volume fraction than the CuO/CeO2-ZrO2 bead catalysts. Then the effects of CuO content and coating amount on methanol steam reforming were explored. When the CeO2-ZrO2 mass content was 15%±1% and CuO was 5%±1%, the obtained catalyst showed the best catalytic activity. At a reaction temperature of 340 ℃, water and methanol molar ratio of 1.2, methanol and water gas hourly space velocity of 4840 h-1, methanol conversion reached 86.0%, hydrogen production rate was 1490.0 L/(m3·s), and CO content in reformed gas was 1.55%. The effects of gas hourly space velocity, water and methanol molar ratio and temperature on methanol steam reforming reaction activity were studied by the single factor experiments. The results showed that, as the gas hourly space velocity increased, methanol conversion decreased, hydrogen production rate increased, and the volume fraction of CO in the reformed gas decreased. As the molar ratio of water to methanol increased, both the methanol conversion and the hydrogen production rate increased first and then declined, and the volume fraction of CO in the reformed gas decreased. With the increase of reaction temperature, methanol conversion rate, hydrogen production rate and the content of CO in the reformed gas increased.
-
Key words:
- methanol steam reforming /
- hydrogen /
- carbon monoxide /
- monolithic catalysts
-
表 1 催化剂的甲醇转化率、CO体积分数和产氢速率
Table 1 Methanol conversion, CO volume fraction and hydrogen production rate of catalysts
Catalyst Methanol conversiona /% CO volume fractiona /% H2 production ratea /(L·m-3·s-1) 10 CuO/CZ 76.3 2.76 1302.1 10 CuO/CZS 79.9 1.85 1395.0 a: reaction condition: t=340 ℃, W/M=1.2:1, GHSV=4840 h-1 表 2 CZS催化载体涂层涂覆质量分数
Table 2 Mass coating percentage of CZS catalytic support
Catalytic support Times CZS-A CZS-B CZS-C Coating amount w/% 1 7.33 7.31 7.34 2 - 7.32 7.30 3 - - 7.30 total 7.33 14.63 21.94 -
[1] HE J P, YANG Z X, ZHANG L, LI Y, PAN L W. Cu supported on ZnAl-LDHs precursor prepared by in-situ synthesis method on γ-Al2O3 as catalytic material with high catalytic activity for methanol steam reforming[J]. Int J Hydrogen Energy, 2017, 42(15):9930-9937. doi: 10.1016/j.ijhydene.2017.01.229 [2] MEI D, FENG Y, QIAN M, CHEN Z Q. An innovative micro-channel catalyst support with a micro-porous surface for hydrogen production via methanol steam reforming[J]. Int J Hydrogen Energy, 2016, 41(4):2268-2277. doi: 10.1016/j.ijhydene.2015.12.044 [3] MA Y F, GUAN G Q, PHANTHONG P, LI X M, GAO J, HAO X G, WANG Z D, ABUDULA A. Steam reforming of methanol for hydrogen production over nanostructured wire-like molybdenum carbide catalyst[J]. Int J Hydrogen Energy, 2014, 39(33):18803-18811. doi: 10.1016/j.ijhydene.2014.09.062 [4] 刘玉娟, 王东哲, 张磊, 王宏浩, 陈琳, 刘道胜, 韩蛟, 张财顺.载体焙烧气氛对甲醇水蒸气重整制氢CuO/CeO2催化剂的影响[J].燃料化学学报, 2018, 46(8):992-999. http://www.ccspublishing.org.cn/article/id/a6e5d5bf-0e5d-4327-b0f9-f6cde8e4ef32LIU Yu-juan, WANG Dong-zhe, ZHANG Lei, WANG Hong-hao, CHEN Lin, LIU Dao-sheng, HAN Jiao, ZHANG Cai-shun. Effect of support calcination atmospheres on the activity of CuO/CeO2 catalysts for methanol steam reforming[J]. J Fuel Chem Technol, 2018, 46(8):992-999. http://www.ccspublishing.org.cn/article/id/a6e5d5bf-0e5d-4327-b0f9-f6cde8e4ef32 [5] 王东哲, 冯旭, 张健, 陈琳, 张磊, 王宏浩, 白金, 张财顺, 张政一.助剂M(M=Cr, Zn, Y, La)对甲醇水蒸气重整制氢CuO/CeO2催化剂的影响[J].燃料化学学报, 2019, 47(10):1251-1257. http://www.ccspublishing.org.cn/article/id/fc3872f1-7c21-4624-8959-0eb51860b79bWANG Dong-zhe, FENG Xu, ZHANG Jian, CHEN Lin, ZHANG Lei, WANG Hong-hao, BAI Jin, ZHANG Cai-shun, ZHANG Zheng-yi. Effect of promoter M (M=Cr, Zn, Y, La) on CuO/CeO2 catalysts for hydrogen production from steam reforming of methanol[J]. J Fuel Chem and Technol, 2019, 47(10):1251-1257. http://www.ccspublishing.org.cn/article/id/fc3872f1-7c21-4624-8959-0eb51860b79b [6] YANG S Q, ZHOU F, LIU Y J, ZHANG L, CHEN Y, WANG H H, TIAN Y, ZHANG C S, LIU D S. Morphology effect of ceria on the performance of CuO/CeO2 catalysts for hydrogen production by methanol steam reforming[J]. Int J Hydrogen Energy, 2019, 44(14):7252-7261. doi: 10.1016/j.ijhydene.2019.01.254 [7] JIANG C J, TRIMM D L, WAINWRIGHT M S. Kinetic study of steam reforming of methanol over copper-based catalysts[J]. Appl Catal A:Gen, 1993, 93(2):245-255. doi: 10.1016/0926-860X(93)85197-W [8] JIANG C J, TRIMM D L, WAINWRIGHT M S. Kinetic mechanism for the reaction between methanol and water over a Cu/ZnO/Al2O3 catalyst[J]. Appl Catal A:Gen, 1993, 97(2):145-158. doi: 10.1016/0926-860X(93)80081-Z [9] AMPHLETT J C, CREBER K A M, DAVIS J M, MANN R F, PEPPLEY B A, STOKES D M. Hydrogen production by steam reforming of methanol for polymer electrolyte fuel cells[J]. Int J Hydrogen Energy, 1994, 19(2):131-137. doi: 10.1016/0360-3199(94)90117-1 [10] LIU N, YUAN Z S, WANG S D, ZHANG C X, WANG S J, LI D Y. Characterization and performance of a ZnO-ZnCr2O4/CeO2-ZrO2 monolithic catalyst for methanol auto-thermal reforming process[J]. Int J Hydrogen Energy, 2008, 33(6):1643-1651. doi: 10.1016/j.ijhydene.2007.12.058 [11] DANWITTAYAKUL S, DUTTA J. Zinc oxide nanorods based catalysts for hydrogen production by steam reforming of methanol[J]. Int J Hydrogen Energy, 2012, 37(7):5518-5526. doi: 10.1016/j.ijhydene.2011.12.161 [12] TAHAY P, KHANI Y, JABARI M, BAHADORAN F, SAFARI N. Highly porous monolith/TiO2 supported Cu, Cu-Ni, Ru, and Pt catalysts in methanol steam reforming process for H2 generation[J]. Appl Catal A:Gen, 2018, 554:44-53. doi: 10.1016/j.apcata.2018.01.022 [13] FASANYA O O, AL-HAJRI R, AHMED O U, MYINT M T Z, ATTA A Y, JIBRIL B Y, DUTTA J. Copper zinc oxide nanocatalysts grown on cordierite substrate for hydrogen production using methanol steam reforming[J]. Int J Hydrogen Energy, 2019, 44(41):22936-22946. doi: 10.1016/j.ijhydene.2019.06.185 [14] KHANI Y, BAHADORAN F, SAFARI N, SOLTANALI S, TAHERI S A. Hydrogen production from steam reforming of methanol over Cu-based catalysts:The behavior of ZnxLaxAl1-xO4 and ZnO/La2O3/Al2O3 lined on cordierite monolith reactors[J]. Int J Hydrogen Energy, 2019, 44(23):11824-11837. doi: 10.1016/j.ijhydene.2019.03.031 [15] VERLATO E, BARISON S, CIMINO S, DERGAL F, LISI L, MANCINO G, MUSIANI M, VAZQUEZ-GOMEZ L. Catalytic partial oxidation of methane over nanosized Rh supported on Fecralloy foams[J]. Int J Hydrogen Energy, 2014, 39(22):11473-11485. doi: 10.1016/j.ijhydene.2014.05.076 [16] BENITO P, NUYTS G, MONTI M, NOLF W D, FORNASARI G, JANSSENS K, SCAVETTA E, VACCARI A. Stable Rh particles in hydrotalcite-derived catalysts coated on FeCrAlloy foams by electrosynthesis[J]. Appl Catal B:Environ, 2015, 179:321-332. doi: 10.1016/j.apcatb.2015.05.035 [17] AVILA P, MONTES M, MIRO E E. Monolithic reactors for environmental applications:A review on preparation technologies[J]. Chem Eng J, 2005, 109(1/3):11-36. http://www.sciencedirect.com/science/article/pii/S1385894705000793 [18] PALMA V, MARTINO M, MELONI E, RICCA A. Novel structured catalysts configuration for intensification of steam reforming of methane[J]. Int J Hydrogen Energy, 2017, 42(3):1629-1638. doi: 10.1016/j.ijhydene.2016.06.162 [19] LOPEZ E, DIVINS N J, ANZOLA A, SCHBIB S, BORIO D, LLORCA J. Ethanol steam reforming for hydrogen generation over structured catalysts[J]. Int J Hydrogen Energy, 2013, 38(11):4418-4428. doi: 10.1016/j.ijhydene.2013.01.174 [20] 刘娜, 袁中山, 张纯希, 王淑娟, 李德意, 王树东. Zn-Cr整体催化剂中Ce-Zr溶胶涂层的制备、表征及对甲醇自热重整反应的影响[J].催化学报, 2005, 26(12):1078-1082. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cuihuaxb200512009LIU Na, YUAN Zhong-shan, ZHANG Chun-xi, WANG Shu-juan, LI De-yi, WANG Shu-dong. Preparation, characterization and effect of Ce-Zr washcoat on Zn-Cr monolithic catalysts for methanol autothermal reforming[J]. Chin J Catal, 2005, 26(12):1078-1082. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cuihuaxb200512009 [21] 刘娜, 王树东, 袁中山, 张纯希, 王淑娟, 李德意, 付桂芝.甲醇自热重整制氢整体催化剂的制备[J].化工学报, 2004, 55(S1):90-94. http://www.cnki.com.cn/Article/CJFDTotal-HGSZ2004S1017.htmLIU Na, WANG Shu-dong, YUAN Zhong-shan, ZHANG Chun-xi, WANG Shu-juan, LI De-yi, FU Gui-zhi. Methanol autothermal reforming for hydrogen generation over monolithic catalyst[J]. CIESC J, 2004, 55(S1):90-94. http://www.cnki.com.cn/Article/CJFDTotal-HGSZ2004S1017.htm [22] LIU H T, LI S Q, ZHANG S B, WANG J M, ZHOU G J, CHEN L, WANG X L. Catalytic performance of novel Ni catalysts supported on SiC monolithic foam in carbon dioxide reforming of methane to synthesis gas[J]. Catal Commun, 2008, 9(1):51-54. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=313383b0d20bcb75449aa2eba45e0456 [23] CUI X T, KAER S K. Two-dimensional thermal analysis of radial heat transfer of monoliths in small-scale steam methane reforming[J]. Int J Hydrogen Energy, 2018, 43(27):11952-11968. doi: 10.1016/j.ijhydene.2018.04.142 [24] GOU Y Z, WANG H, JIAN K, SHAO C W, WANG X Z. Preparation and characterization of SiC fibers with diverse electrical resistivity through pyrolysis under reactive atmospheres[J]. J Eur Ceram Soc, 2017, 37(2):517-522. doi: 10.1016/j.jeurceramsoc.2016.09.023 [25] ZHANG L, PAN L W, NI C J, SUN T J, ZHAO S S, WANG S D, WANG A J, HU Y K. CeO2-ZrO2-promoted CuO/ZnO catalyst for methanol steam reforming[J]. Int J Hydrogen Energy, 2013, 38(11):4397-4406. doi: 10.1016/j.ijhydene.2013.01.053 [26] 杨淑倩, 贺建平, 张娜, 隋晓伟, 张磊, 杨占旭.稀土掺杂改性Cu/Zn-Al水滑石衍生催化剂对甲醇水蒸气重整制氢性能的影响[J].燃料化学学报, 2018, 46(2):179-188. http://www.ccspublishing.org.cn/article/id/00984ce6-5d53-4ea0-929f-0c4b80514203YANG Shu-qian, HE Jian-ping, ZHANG Na, SUI Xiao-wei, ZHANG Lei, YANG Zhan-xu. Effect of rare-earth element modification on the performance of Cu/ZnAl catalysts derived from hydrotalcite precursor in methanol steam reforming[J]. J Fuel Chem Technol, 2018, 46(2):179-188. http://www.ccspublishing.org.cn/article/id/00984ce6-5d53-4ea0-929f-0c4b80514203 [27] ZHANG X, SHI P. Production of hydrogen by steam reforming of methanol on CeO2 promoted Cu/Al2O3 catalysts[J]. J Mol Catal A:Chem, 2003, 194(1/2):99-105. http://www.sciencedirect.com/science/article/pii/S1381116902004648 [28] ZHANG X R, SHI P, ZHAO J X, ZHAO M Y, LIU C T. Production of hydrogen for fuel cells by steam reforming of methanol on Cu/ZrO2/Al2O3 catalysts[J]. Fuel Process Technol, 2003, 83(1/3):183-192. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=a32b3ad82d84942b2068be5b564bb769 -





 下载:
下载: