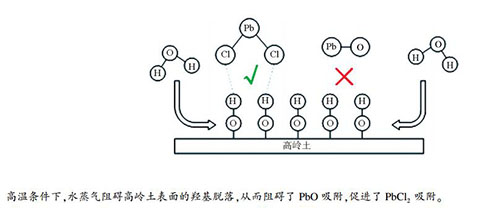
| [1] |
中华人民共和国国家统计局.中国统计年鉴2019[M].北京:中国统计出版社, 2019.National Bureau of Statistics. China Statistical Yearbook 2019[M]. Beijing: China Statistics Press, 2019.
|
| [2] |
付彪.煤加工利用过程中有害微量元素的迁移转化行为研究[D].安徽: 中国科学技术大学, 2019. http://kns.cnki.net/KCMS/detail/detail.aspx?dbcode=CDFD&filename=1019048781.nhFU Biao. The partitioning and transformation behavior of hazardous trace elements during coal utilization[D]. Anhui: University of Science and Technology of China, 2019. http://kns.cnki.net/KCMS/detail/detail.aspx?dbcode=CDFD&filename=1019048781.nh
|
| [3] |
MEIJ R, WINKEL B H. Trace elements in world steam coal and their behaviour in dutch coal-fired power stations: A review[J]. Int J Coal Geol, 2009, 77(3): 289-93. http://www.sciencedirect.com/science/article/pii/S0166516208001444
|
| [4] |
王昕晔.垃圾焚烧过程中铅和镉的挥发特性及其排放控制研究[D].南京: 东南大学, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10286-1016215491.htmWANG Xin-ye. Volatilization characteristics and emissions control of lead and cadmium during waste incineration[D]. Nanjing: Southest University, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10286-1016215491.htm
|
| [5] |
张英英. PM2.5暴露诱导心脏毒性及其相关分子机制研究[D].太原: 山西大学, 2019. http://cdmd.cnki.com.cn/Article/CDMD-10108-1019245914.htmZHANG Ying-ying. The cardiactoxicity effect and molecular mechanism in response to atmospheric PM2.5 inhalation[D]. Taiyuan: Shanxi University, 2019. http://cdmd.cnki.com.cn/Article/CDMD-10108-1019245914.htm
|
| [6] |
方婷.煤矿区中铅的环境地球化学研究[D].合肥: 中国科学技术大学, 2015.FANG Ting. Environmental geochemistry of lead in coal mining area[D]. Hefei: University of Science and Technology of China, 2019.
|
| [7] |
白向飞, 李文华, 陈亚飞, 姜英.中国煤中微量元素分布基本特征[J].煤质技术, 2007, (1): 1-4. doi: 10.3969/j.issn.1007-7677.2007.01.001BAI Xiang-fei, LI Wen-hua, CHEN Ya-fei, JIANG Ying. The general distributions of trace elements in Chinese coals[J]. Coal Qual Technol, 2007, (1): 1-4 doi: 10.3969/j.issn.1007-7677.2007.01.001
|
| [8] |
白向飞.中国煤中微量元素分布赋存特征及其迁移规律试验研究[D].北京: 煤炭科学研究总院, 2003. http://d.wanfangdata.com.cn/Thesis/Y578127BAI Xiang-fei. The distributions, modes of occurrenxe and volatility of trace elements in coals of China[D]. Beijing: China Coal Research Institute, 2003. http://d.wanfangdata.com.cn/Thesis/Y578127
|
| [9] |
LU K, LU J, CHEN L. Lead distribution in permo-carboniferous coal from the North China plate, China[J]. Environ Geochem Health, 2005, 27(1): 31-7. http://europepmc.org/abstract/MED/15688128
|
| [10] |
LIU K, QING M, CHANG Y. Sources and health risks of heavy metals in PM2.5 in a campus in a typical suburb area of taiyuan, North China[J]. Atmosphere, 2018, 9(2): 10. http://www.researchgate.net/publication/323237433_Sources_and_Health_Risks_of_Heavy_Metals_in_PM25_in_a_Campus_in_a_Typical_Suburb_Area_of_Taiyuan_North_China
|
| [11] |
董世豪, 谢扬, 皇甫延琦, 史旭荣, 易睿, 史国良, 冯银厂.扬州市PM2.5中重金属来源及潜在健康风险评估[J].环境科学, 2019, 40(2): 540-547. http://www.cnki.com.cn/Article/CJFDTotal-HJKZ201902004.htmDONG Shi-hao, XIE Yang, HUANGFU Yan-qi, SHI Xu-rong, YI Rui, SHI Guo-liang, FENG Ying-chang. Source apportionment and heath risk quantification of heavy metals in PM2.5 in Yangzhou, China[J]. Environ Sci, 2019, 40(2): 540-547. http://www.cnki.com.cn/Article/CJFDTotal-HJKZ201902004.htm
|
| [12] |
杨静, 陈龙, 刘敏, 孟祥周, 张希.上海典型燃烧源铅和汞大气排放趋势分析[J].环境科学, 2018, 39(9): 3987-3994. http://www.cqvip.com/QK/91181X/201809/676218931.htmlYANG Jing, CHEN Long, LIU Min, MENG Xiang-zhou, ZHANG Xi. Historical trends of atmospheric Pb and Hg emissions from fossil fuel combustion in Shanghai[J]. Environ Sci, 2018, 39(9): 3987-3994. http://www.cqvip.com/QK/91181X/201809/676218931.html
|
| [13] |
Sources of atmospheric lead (Pb) in and around an Indian megacity[J]. Atmos Environ, 2018, 193(33): 57-65.
|
| [14] |
HUANG Y, WANG X, LIU C, WANG Y, DONG L. Kaolinite induced control of particulate lead and cadmium emissions during fluidized bed waste incineration[J]. Asia-Pac J Chem Eng, 2017, 12(2): 321-31. doi: 10.1002/apj.2075
|
| [15] |
PUNJAK W A, SHADMAN F. Aluminosilicate sorbents for control of alkali vapors during coal combustion and gasification[J]. Energy Fuels, 1988, 2(3): 702-708. doi: 10.1021/ef00011a017
|
| [16] |
SCOTTO M V, UBEROI M, PETERSON T W, SHADMAN F, WENDT J O.L. Metal capture by sorbents in combustion processes[J]. Fuel Process Technol, 1994, 39(1/3): 357-372. http://www.sciencedirect.com/science/article/pii/0378382094901929
|
| [17] |
程运, 王昕晔, 吕文婷, 黄亚继, 谢浩, 郭若军, 朴桂林.高岭土高温吸附重金属和碱金属的研究进展[J].化工进展, 2019, 38(8): 3852-3865. http://d.wanfangdata.com.cn/periodical/hgjz201908042CHENG Yun, WANG Xin-ye, LV Wen-ting, HUANG Ya-ji, XIE Hao, GUO Ruo-jun, PIAO Gui-lin. A review on heavy and alkali metals adsorption by kaolin at high temperature[J]. Chem Ind Eng Prog, 2019, 38(8): 3852-3865. http://d.wanfangdata.com.cn/periodical/hgjz201908042
|
| [18] |
王浩, 刘小伟, 徐义书, 张煜枫, 王小朋.煤粉燃烧中添加改性高岭土脱除Pb和V实验研究[J].中国电机工程学报, 2019, 39(6): 1692-1699+1865. http://www.cqvip.com/QK/90021X/20196/7001836368.htmlWANG Hao, LIU Xiao-wei, XU Yi-shu, ZHANG Yu-feng, WANG Xiao-peng. Study on the emission characteristics of Pb and V during pulverized coal combustion by adding kaolin and modified kaolin[J]. Proc CSEE, 2019, 39(6): 1692-1699+1865. http://www.cqvip.com/QK/90021X/20196/7001836368.html
|
| [19] |
马杨杨, 仲兆平, 赖旭东.矿物添加剂对煤燃烧过程中重金属的富集[J].化工进展, 2020, 39(6): 2479-2486. http://d.wanfangdata.com.cn/periodical/hgjz202006043MA Yang-yang, ZHONG Zhao-ping, LAI Xu-dong. Enrichment of heavy metals during coal combustion by mineral additives[J]. Chem Ind Eng Prog, 2020, 39(6): 2479-2486. http://d.wanfangdata.com.cn/periodical/hgjz202006043
|
| [20] |
YAO H, NARUSE I. Using sorbents to control heavy metals and particulate matter emission during solid fuel combustion[J]. Particuology, 2009, 7(6): 477-482. http://www.sciencedirect.com/science/article/pii/S1674200109000790
|
| [21] |
YAO H, NARUSE I. Control of trace metal emissions by sorbents during sewage sludge combustion[J]. Proc Combust Inst, 2005, 30(2): 3009-3016. http://www.sciencedirect.com/science/article/pii/S0082078404000505
|
| [22] |
WANG Xinye, HUANG Yaji, ZHONG Zhaoping, YAN Yupeng, NIU Miaomiao, WANG Yongxing. Control of inhalable particulate lead emission from incinerator using kaolin in two addition modes[J]. Fuel Process Technol. 2014, 119: 228-235. http://www.sciencedirect.com/science/article/pii/S0378382013003640
|
| [23] |
史燕红, 吴华成.燃煤电厂重金属铅排放特性研究进展[J].热力发电, 2016, 45(1): 1-8. doi: 10.3969/j.issn.1002-3364.2016.01.001SHI Yan-hong, WU Hua-cheng. Emission characteristics of Pb in coal-fired plants: Research development[J]. Therm Power Gen, 2016, 45(1): 1-8. doi: 10.3969/j.issn.1002-3364.2016.01.001
|
| [24] |
CHENG Y, XING J, BU C, ZHANG J, PIAO G, HUANG Y, XIE H, WANG X. Dehydroxylation and structural distortion of kaolinite as a high-temperature sorbent in the furnace[J]. Minerals, 2019, 9(10): 587. http://www.researchgate.net/publication/336111338_Dehydroxylation_and_Structural_Distortion_of_Kaolinite_as_a_High-Temperature_Sorbent_in_the_Furnace
|
| [25] |
WENDT J O L, LEE S. High-temperature sorbents for Hg, Cd, Pb, and other trace metals: Mechanisms and applications[J]. Fuel, 2010, 89(4): 894-903. http://www.ingentaconnect.com/content/el/00162361/2010/00000089/00000004/art00016
|
| [26] |
GALE T K, WENDT J O L. High-temperature interactions between multiple-metals and kaolinite[J]. Combust Flame, 2002, 131(3): 299-307. http://www.sciencedirect.com/science/article/pii/S0010218002004042
|
| [27] |
YOO J I, SHINAGAWA T, WOOD J P, LINAK W P, SANTOIANNI D A, KING C J, SEO Y C, WENDT J O L. High-temperature sorption of cesium and strontium on dispersed kaolinite powders[J]. Environ Sci Technol, 2005, 39(13): 5087-5094. http://www.ncbi.nlm.nih.gov/pubmed/16053114
|
| [28] |
GALE T K, WENDT J O L. In-furnace capture of cadmium and other semi-volatile metals by sorbents[J]. Proc Combust Inst, 2005, 30(2): 2999-3007. http://www.sciencedirect.com/science/article/pii/S0082078404002498
|
| [29] |
WANG G, JENSEN P A, WU H, FRANDSEN F J, LAXMINARAYAN Y, SANDER B, GLARBORG P. Potassium capture by kaolin, part 2: K2CO3, KCl, and K2SO4[J]. Energy Fuels, 2018, 32(3): 3566-3578. doi: 10.1021/acs.energyfuels.7b04055
|
| [30] |
WANG G, JENSEN P A, WU H, FRANDSEN F J, SANDER B, GLARBORG P. Potassium capture by kaolin, part 1: KOH[J]. Energy Fuels, 2018, 32(2): 1851-1862. doi: 10.1021/acs.energyfuels.7b03645
|
| [31] |
WANG G, JENSEN P A, WU H, FRANDSEN F J, Bøjer M, GLARBORG P. Entrained Flow Reactor Study of K-Capture by Solid Additives[C]. Proceedings of the 24th European Biomass Conference & Exhibition, 2016.
|
| [32] |
XU Y, LIU X, WANG H, ZENG X, ZHANG Y, HAN J, XU M, PAN S. Influences of in-furnace kaolin addition on the formation and emission characteristics of PM2.5 in a 1000 MW coal-fired power station[J]. Environ Sci Technol, 2018, 52(15): 8718-8724. http://europepmc.org/abstract/MED/29965744
|
| [33] |
GALE T K, WENDT J O L. High-temperature interactions between multiple-metals and kaolinite[J]. Combust Flame, 2002, 131(3): 299-307. http://www.sciencedirect.com/science/article/pii/S0010218002004042
|
| [34] |
ZHANG X, LIU H, XING H, WANG G, LI H, XIAO K, LIU W, YU Y, YAO H. Investigation of potassium vapor time-resolved adsorption and potassium-sodium competitive adsorption by modified kaolinite[J]. Fuel, 2019, 258(15): 166124. http://www.sciencedirect.com/science/article/pii/S0016236119314784
|
| [35] |
XING H, LIU H, ZHANG X, DENG H, HU H, YAO H. Enhanced sodium adsorption capacity of kaolinite using a combined method of thermal pre-activation and intercalation-exfoliation: Alleviating the problems of slagging and fouling during the combustion of Zhundong coal[J]. Fuel, 2019, 239(3): 312-319. http://www.zhangqiaokeyan.com/academic-journal-foreign_other_thesis/0204112774452.html
|
| [36] |
ZHANG X, LIU H, XING H, WANG G, DENG H, HU H, LI X, YAO H. Correlations between the sodium adsorption capacity and the thermal behavior of modified kaolinite during the combustion of Zhundong coal[J]. Fuel, 2019, 237(2): 170-177. http://www.sciencedirect.com/science/article/pii/S0016236118316685
|
| [37] |
YU M, HUANG Y, XIA W, ZHU Z, FAN C, LIU C, DONG L, XU L, LIU L, ZHA J, WANG X. PbCl2 capture by kaolin and metakaolin under different influencing factors of thermal treatment[J]. Energy Fuels, 2020, 34(2): 2284-2292. http://www.researchgate.net/publication/338522517_PbCl_2_Capture_by_Kaolin_and_Metakaolin_under_Different_Influencing_Factors_of_Thermal_Treatment
|
| [38] |
GALE T K, WENDT J O L. Mechanisms and models describing sodium and lead scavenging by a kaolinite aerosol at high temperatures[J]. Aerosol Sci Technol, 2003, 37(11): 865-876. doi: 10.1080/02786820300929
|
| [39] |
WANG X, HUANG Y, ZHONG Z, PAN Z, LIU C. Theoretical investigation of cadmium vapor adsorption on kaolinite surfaces with DFT calculations[J]. Fuel, 2016, 166(2): 333-339. http://europepmc.org/abstract/MED/25880048
|
| [40] |
WANG X, HUANG Y, PAN Z, WANG Y, LIU C. Theoretical investigation of lead vapor adsorption on kaolinite surfaces with DFT calculations[J]. J Hazard Mater, 2015, 295(9): 43-54. http://europepmc.org/abstract/MED/25880048
|





 下载:
下载: