Study on the physicochemical properties and catalytic performance of Pt/SAPO-11 catalysts treated by acid and salt
-
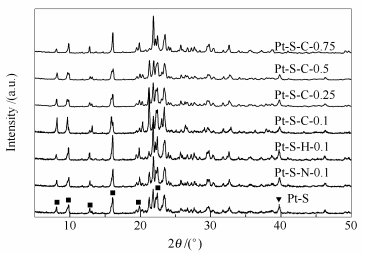
摘要: 采用不同酸和盐浸渍处理SAPO-11分子筛,然后负载Pt制成改性的Pt/SAPO-11催化剂,用XRF、XRD、N2吸附-脱附、SEM、NH3-TPD和Py-IR对催化剂进行表征,分析其物理化学性能。结果表明,酸和盐处理没有破坏SAPO-11的骨架结构,还提高了催化剂的孔容、孔径、比表面积等性质,催化剂的酸性、酸量也明显发生了改变。在固定床反应器中,评价了改性的Pt/SAPO-11催化小桐子油一步加氢制异构烷烃性能;结合催化剂表征数据表明,颗粒粒径、比表面积、孔径、酸性和酸量影响催化剂的活性及产物分布。对比发现,经0.5 mol/L柠檬酸处理的Pt/SAPO-11催化剂孔径大小、酸性、B酸和L酸量分布合适,因此,一步加氢催化处理小桐子油性能优异;其中,生物航油组分(C8-16)的收率为32.47%,异构烷烃(C8-16)选择性为53.13%。
-
关键词:
- Pt/SAPO-11催化剂 /
- 改性 /
- 异构 /
- 柠檬酸
Abstract: SAPO-11 molecular sieves were treated with different acids and salts and the modified Pt/SAPO-11 catalysts were prepared by loading platinum on the pre-treated SAPO-11. The catalyst samples were characterized with XRF, XRD, N2 adsorption-desorption, SEM, NH3-TPD and pyridine Py-IR to analyze their physicochemical properties. The results showed that the modification of acid and salt did not destroy the structure of SAPO-11, but improved the pore volume, pore size and specific surface area of the catalysts, meanwhile, the acidity and acid amount of the zeolite were also changed obviously. In a fixed-bed reactor, one-step hydrgenation of Jatropha curcas oil to iso-alkanes over modified Pt/SAPO-11 catalyst was carried out. Combined with catalyst characterization results, it was concluded that those parameters, such as particle size, specific surface area, pore diameter, acidity and acid amount, would affect the activity and product distribution of the catalysts. Among these modified catalysts, it is found that the Pt/SAPO-11 catalyst treated with 0.5 mol/L citric acid solution exhibited an excellent catalytic performance in one-step hydrogenation of Jatropha curcas oil, owing to its appropriate pore size, acidity, B acid and L acid distribution. The yield of bio-jet fuel components (C8-16) was 32.47% and the selectivity of isoparaffins was 53.13%.-
Key words:
- Pt/SAPO-11 catalyst /
- modification /
- isomerism /
- citric acid
-
表 1 改性SAPO-11的化学组成
Table 1 Chemical composition of modified SAPO-11 by XRF
Sample Composition w/% n(SiO2)/n(Al2O3) P2O5 Al2O3 SiO2 others S 46.8 42.6 10.3 0.3 0.41 S-N-0.1 47.6 40.9 11 0.5 0.46 S-H-0.1 48.0 40.4 11 0.6 0.46 S-C-0.1 47.8 38.9 12.5 0.8 0.54 S-C-0.25 46.3 38.5 14.7 0.5 0.65 S-C-0.5 42.3 37.4 19.8 0.5 0.9 S-C-0.75 40.3 36.9 22.2 0.6 1.02 表 2 改性催化剂的比表面积及孔结构
Table 2 Surface area and pore structure of modified catalysts
Sample Surface area A/(m2·g-1) Pore volume v/(cm3·g-1) Average pore size d/nm Micropore volume* v/(cm3·g-1) Micropore area* A/(m2·g-1) Pt-S 195.31 0.15 3.1 0.063 154.22 Pt-S-N-0.1 184.87 0.19 4.2 0.056 137.41 Pt-S-H-0.1 208.86 0.17 3.4 0.064 157.11 Pt-S-C-0.1 223.16 0.21 3.6 0.069 168.50 Pt-S-C-0.25 259.18 0.29 4.5 0.059 131.44 Pt-S-C-0.5 215.99 0.22 4.1 0.054 121.73 Pt-S-C-0.75 164.71 0.23 5.7 0.048 117.10 *:micropore volume, micropore area calculated by t-plot method 表 3 Py-IR获得的改性催化剂的酸型分布
Table 3 Acid type distributions of modified catalysts by Py-IR analysis
Sample Acid sites /(μmol·g-1) weak acid site (200 ℃) medium strong acid site (300 ℃) Lewis Brønsted Lewis Brønsted Pt-S 10.76 27.94 7.45 12.05 Pt-S-N-0.1 21.27 35.38 19.63 18.05 P-S-C-0.1 21.82 39.72 21.27 18.77 Pt-S-C-0.25 14.73 42.60 11.45 14.44 Pt-S-C-0.5 15.13 33.84 10.96 9.67 Pt-S-C-0.75 6.50 23.61 5.40 10.02 表 4 改性催化剂催化加氢产物分布
Table 4 Hydrocarbon distribution of modified catalysts
Sample Hydrodeoxygenation rate /% Cracking rate /% Bio-jet fuel component /% >C18/% Isoparaffin selectivity /% Aromatic hydrocarbons /% Pt-S 91.25 7.74 24.22 7.70 47.25 0.92 Pt-S-N-0.1 62.10 3.28 15.71 12.4 28.75 2.23 Pt-S-H-0.1 60.00 5.87 26.17 13.1 35.05 0.57 Pt-S-C-0.1 90.44 11.82 31.51 28.54 40.83 2.30 Pt-S-C-0.25 97.84 8.91 30.07 18.9 53.30 0 Pt-S-C-0.5 97.09 9.13 32.47 14.7 53.13 2.32 Pt-S-C-0.75 74.72 9.75 12.22 8.06 39.30 0.62 -
[1] LESTARI S, MÄKI-ARVELA P, BELTRAMINI J, LU G Q, MURZIN D Y. Transforming triglycerides and fatty acids into biofuels[J]. ChemSusChem, 2009, 2(12):1109-1119. doi: 10.1002/cssc.v2:12 [2] ZARCHIN R, RABAEV M, VIDRUK-NEHEMYA R, LANDAU M V, HERSKOWITZ M. Hydroprocessing of soybean oil on nickel-phosphide supported catalysts[J]. Fuel, 2015, 139:684-691. doi: 10.1016/j.fuel.2014.09.053 [3] GONG S, SHINOZAKI A, SHI M, QIAN E W. Hydrotreating of jatropha oil over alumina based catalysts[J]. Energy fuels, 2012, 26(4):2394-2399. doi: 10.1021/ef300047a [4] SENOL O I, VILJAVA T R, KRAUSE A O I. Hydrodeoxygenation of methyl esters on sulphided NiMo/γ-Al2O3 and CoMo/γ-Al2O3 catalysts[J]. Catal Today, 2005, 100(3/4):331-335. [5] KUMAR R, RANA B S, TIWARI R, VERMA D, KUMAR R. Hydroprocessing of jatropha oil and its mixtures with gas oil[J]. Green Chem, 2010, 12(12):2232-2239. doi: 10.1039/c0gc00204f [6] GUZMAN A, TORRES J E, PRADA L P, NUÑEZ M L. Hydroprocessing of crude palm oil at pilot plant scale[J]. Catal Today, 2010, 156(1):38-43. [7] TIWARI R, RANA B S, KUMAR R, VERMA D, KUMAR R. Hydrotreating and hydrocracking catalysts for processing of waste soya-oil and refinery-oil mixtures[J]. Catal Commun, 2011, 12(6):559-562. doi: 10.1016/j.catcom.2010.12.008 [8] KANDARAMATH HARI T, YAAKOB Z, BINITHA N N. Aviation biofuel from renewable resources:Routes, opportunities and challenges[J]. Renew Sust Energy Rev, 2015, 42:1234-1244. doi: 10.1016/j.rser.2014.10.095 [9] MILLER S J. New molecular sieve process for lube dewaxing by wax isomerization[J]. Microporous Mater, 1994, 2(5):439-449. doi: 10.1016/0927-6513(94)00016-6 [10] DWYER F G, VALYOCSIK E W. Highly siliceous porous crystalline material[P]. US:5336478, 1994-08-07. [11] WANG Y D, TAO Z C, WU B S, XU J, HUO C F, LI K, CHEN H M, YANG Y, LI Y W. Effect of metal precursors on the performance of Pt/ZSM-22 catalysts for n-hexadecane hydroisomerization[J]. J Catal, 2015, 322:1-13. doi: 10.1016/j.jcat.2014.11.004 [12] HANCSÓK J, KRÁR M, MAGYAR S, BODA L, HOLLÓ A, KALLÓ D. Investigation of the production of high quality biogasoil from pre-hydrogenated vegetable oils over Pt/SAPO-11/Al2O3[J]. Stud Surf Sci Catal, 2007, 170:1605-1610. doi: 10.1016/S0167-2991(07)81037-8 [13] VERMA D, KUMAR R, RANA B S, SINHA A K. Aviation fuel production from lipids by a single-step route using hierarchical mesoporous zeolites[J]. Energy Environ Sci, 2011, 4(5):1667-1671. doi: 10.1039/c0ee00744g [14] LIU Q, ZUO H, WANG T, MA L, ZHANG Q. One-step hydrodeoxygenation of palm oil to isomerized hydrocarbon fuels over Ni supported on nano-sized SAPO-11 catalysts[J]. Appl Catal A:Gen, 2013, 468:68-74. doi: 10.1016/j.apcata.2013.08.009 [15] WANG C, TIAN Z, WANG L, XU R, QU W, MA H, WANG B. One-step hydrotreatment of vegetable oil to produce high quality diesel-range alkanes[J]. ChemSusChem, 2012, 5(10):1974-83. doi: 10.1002/cssc.v5.10 [16] 吴伟光, 黄季焜.林业生物柴油原料麻风树种植的经济可行性分析[J].中国农村经济, 2010, 7:56-63. http://www.cnki.com.cn/Article/CJFDTOTAL-ZNJJ201007010.htmWU Wei-guang, HUANG Ji-kun. Economic feasibility analysis of jatropha plantation for biodiesel from forestry[J]. Chin Rural Economy, 2010, 7:56-63. http://www.cnki.com.cn/Article/CJFDTOTAL-ZNJJ201007010.htm [17] DUMITRIU E, ON D T, KALIAGUINE S. Isoprene by Prins condensation over acidic molecular sieves[J]. J Catal, 1997, 170(1):150-160. doi: 10.1006/jcat.1997.1745 [18] SONG H, RIOUX R M, HOEFELMEYER J D, KOMOR R, NIESZ K, GRASS, M, YANG P, SOMORJAI G A. Hydrothermal growth of mesoporous SBA-15 silica in the presence of PVP-stabilized Pt nanoparticles:Synthesis, characterization, and catalytic properties[J]. J Am Chem Soc, 2006, 128(9):3027-3037. doi: 10.1021/ja057383r [19] 李林林, 张泽凯, 张斌浩, 刘华彦, 陈银飞.柠檬酸改性SAPO-11分子筛催化叔丁醇和异丁醛合成2, 5-二甲基-2, 4-己二烯[J].工业催化, 2014, 22(1):56-60. http://www.cnki.com.cn/Article/CJFDTOTAL-GYCH201401019.htmLI Lin-lin, ZHANG Ze-kai, ZHANG Hao-bin, LIU Hua-yan, CHEN Yin-fei. Citric acid modification SAPO-11 molecular sieves for synthesis of 2, 5-dimethyl-2, 4-hexadiene from tert-butanol and isobutylene[J]. Ind Catal, 2014, 22(1):56-60. http://www.cnki.com.cn/Article/CJFDTOTAL-GYCH201401019.htm [20] ZHANG S, CHEN S L, DONG P, JI Z, ZHANO J. Synthesis, characterization and hydroisomerization catalytic performance of nanosize SAPO-11 molecular sieves[J]. Catal Lett, 2007, 118(1):109-117. https://www.researchgate.net/publication/226828342_Synthesis_characterization_and_hydroisomerization_catalytic_performance_of_nanosize_SAPO-11_molecular_sieves [21] VERMA D, RANA B S, KUMAR R, SIBI M G, SINHA A K. Diesel and aviation kerosene with desired aromatics from hydroprocessing of jatropha oil over hydrogenation catalysts supported on hierarchical mesoporous SAPO-11[J]. Appl Catal A:Gen, 2015, 490:108-116. doi: 10.1016/j.apcata.2014.11.007 [22] LIU W, SHANG T, ZHOU Q, REN J, SUN Y. Physicochemical and isomerization property of Pt/SAPO-11 catalysts promoted by rare earths[J]. J Rare Earth, 2009, 27(6):937-942. doi: 10.1016/S1002-0721(08)60366-X [23] KIM M Y, LEE K, CHOI M. Cooperative effects of secondary mesoporosity and acid site location in Pt/SAPO-11 on n-dodecane hydroisomerization selectivity[J]. J Catal, 2014, 319:232-238. doi: 10.1016/j.jcat.2014.09.001 [24] FINLEY J L, JOSHI V V, SMITH N L. Chapter 7 techniques of zeolite characterization[J]. Stud Surf Sci Catal, 1991, 58:241-283. doi: 10.1016/S0167-2991(08)63605-8 [25] 张春勇, 刘靖, 王祥生, 郭新闻, 赵文江, 卞俊杰.柠檬酸处理对β沸石结构、酸性和催化性能的影响[J].辽宁化工, 2003, 32(11):461-463. http://www.cnki.com.cn/Article/CJFDTOTAL-LNHG200311000.htmZHANG Chun-yong, LIU Jin, WANG Xiang-sheng, GUO Xin-wen, ZHAO Wen-jiang, BIAN Jun-jie. Effect of citric acid treatment on structure, acidity and catalysis of β zeolite[J]. Liaoning Chem Ind, 2003, 32(11):461-463. http://www.cnki.com.cn/Article/CJFDTOTAL-LNHG200311000.htm [26] CALEMMA V, PERATELLO S, PEREGO C. Hydroisomerization and hydrocracking of long chain n-alkanes on Pt/amorphous SiO2-Al2O3 catalyst[J]. Appl Catal A:Gen, 2000, 190(1/2):207-218. https://www.researchgate.net/publication/222234574_Hydroisomerization_and_hydrocracking_of_long_chain_n-alkanes_on_Ptamorphous_SiO2-Al2O3_catalyst [27] DELDARI H. Suitable catalysts for hydroisomerization of long-chain normal paraffins[J]. Appl Catal A:Gen, 2005, 293:1-10. doi: 10.1016/j.apcata.2005.07.008 [28] KUBIČKA D, BEJBLOVÁM, VLK J. Conversion of vegetable oils into hydrocarbons over CoMo/MCM-41 catalysts[J]. Top Catal, 2010, 53(3):168-178. https://www.researchgate.net/publication/225539793_Conversion_of_Vegetable_Oils_into_Hydrocarbons_over_CoMoMCM-41_Catalysts [29] HAN J X, SUN H, DING Y, YAN L, LUO C, WANG L, WANG B, TIAN Z. Palladium-catalyzed decarboxylation of higher aliphatic esters:towards a new protocol to the second generation biodiesel production[J]. Green Chem, 2010, 12(3):463-467. doi: 10.1039/b917690j [30] YADAV R, SAKTHIVEL A. Silicoaluminophosphate molecular sieves as potential catalysts for hydroisomerization of alkanes and alkenes[J]. Appl Catal A:Gen, 2014, 481:143-160. doi: 10.1016/j.apcata.2014.05.010 [31] 王从新, 刘千河, 刘雪斌, 阎立军, 罗琛, 王磊, 王炳春, 田志坚.反应条件对Pt/SAPO-11催化油脂一步加氢制异构烷烃的影响[J].催化剂, 2013, 34(6):1128-1138. doi: 10.1016/S1872-2067(11)60524-XWANG Cong-xin, LIU Qian-he, LIU Xue-bin, YAN Li-jun, LUO Chen, WANG Lei, WANG Bing-chun, TIAN Zhi-jian. Influence of reaction conditions on one-step hydrotreatment of lipids in the production of iso-alkanes over Pt/SAPO-11[J]. Chin J Catal, 2013, 34(6):1128-1138. doi: 10.1016/S1872-2067(11)60524-X [32] 杨娜, 王红英, 柳云骐, 刘晨光.不同链长正构烷烃在Pt/SAPO-11催化剂上的临氢转化规律研究[J].燃料化学学报, 2016, 44(1):91-98. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18766.shtmlYANG Na, WANG Hong-ying, LIU Yun-qi, LIU Chen-guang. Study on the transformation rule of different long chain alkane hydroisomerization over Pt/SAPO-11 catalyst[J]. J Fuel Chem Technol, 2016, 44(1):91-98. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18766.shtml -





 下载:
下载: