Study on high temperature pyrolysis process and sulfur transformation property of high sulfur petroleum coke
-
摘要: 为深入了解高硫石油焦在工业应用高温工况下的热解过程以及硫的析出特性,本研究采用高温固定床对青岛高硫石油焦进行了高温(900-1500℃)热解实验,考察了高温热解下热解气体释放规律,热解过程中焦的物理孔隙结构以及化学特性的演变,并对热解过程中硫的析出与演变特性进行了研究。结果表明,随着热解温度的升高,石油焦热解气中的H2含量逐渐增加,CO含量变化不大,CH4与CO2含量则逐渐下降;热解焦的比表面积与平均孔隙均随热解温度的升高有所增加,颗粒的表面形态则受温度影响较小;热解温度的升高会降低石油焦中含有的非定型碳比例,提高其微晶结构的有序性以及石墨化程度;热解焦的气化活性随热解温度的升高先降低后升高,在1100℃附近有最小值;1500℃高硫石油焦硫元素析出率达81.34%,仅少量硫醇类有机硫和噻吩环内的硫元素得以残存。Abstract: In order to understand the pyrolysis process and sulfur transformation property of high sulfur petroleum coke at high temperature, the pyrolysis experiment of Qingdao high sulfur petroleum coke at high temperature (900-1500℃) was carried out in a high temperature fixed bed. The release rule of pyrolysis gas and the evolution of physical pore structure and chemical characteristics of coke during pyrolysis were investigated. At the same time, the content and existing mode of sulfur in the samples before and after pyrolysis were studied. The results show that with the increase in pyrolysis temperature, the content of H2 in the pyrolysis gas of petroleum coke increases gradually; the content of CO changes little; while the content of CH4 and CO2 decreases gradually. Moreover, as the pyrolysis temperature is increased, the specific surface area and average porosity of pyrolysis coke increase; the surface morphology of particles is less affected; the content of amorphous carbon in petroleum coke reduces; and the order and graphitization degree of microcrystalline structure increase gradually. However, with the increase in pyrolysis temperature, the gasification activity of pyrolysis coke is first decreased and then increased, with the minimum value around 1100℃. It is found that the sulfur release rate in high sulfur petroleum coke pyrolysis at 1500℃ reaches 81.34%, and only a small amount of organic sulfur in the form of mercaptans and thiophene rings is retained in solid.
-
表 1 青岛高硫石油焦的工业分析和元素分析
Table 1 Proximate analysis and ultimate analysis of Qingdao high sulfur petroleum coke
Sample Proximate analysis
wd/%Ultimate analysis
w/%FC A V C H N S SYJ 85.62 0.11 14.27 86.76 4.05 0.15 6.97 表 2 不同温度热解石油焦的比表面积和平均孔径
Table 2 BET surface and average pore size of petroleum coke made at different pyrolysis temperatures
Sample SYJ-900 SYJ-1100 SYJ-1300 SYJ-1500 SBET/(m2·g-1) 4.464 5.005 15.973 19.285 Average pore size
d/nm4.137 5.628 9.433 11.571 表 3 拉曼光谱分峰拟合数据
Table 3 Parameters of curve-fitted Raman spectra
Sample IG/IAll ID1/IG ID3/IG ID4/IG SYJ-900 0.20166 2.05958 0.44862 0.56203 SYJ-1100 0.21468 2.03658 0.41839 0.56091 SYJ-1300 0.23931 1.85613 0.34716 0.49604 SYJ-1500 0.25810 1.81919 0.30466 0.46628 表 4 高温热解石油焦元素组成和硫、氮元素析出率
Table 4 Ultimate analysis of different samples and the corresponding removal rate of nitrogen and sulfur
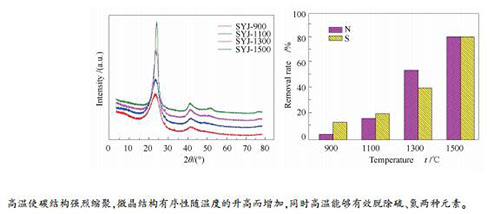
Sample Utinate andysis w/% Removal rate η/% C H N S N S SYJ 86.76 4.05 1.27 6.97 - - SYJ-900 90.14 1.20 1.21 5.99 4.72 14.06 SYJ-1100 92.41 0.90 1.05 5.52 17.32 20.80 SYJ-1300 94.70 0.73 0.57 4.09 55.12 41.32 SYJ-1500 96.59 0.78 0.23 1.30 81.89 81.34 -
[1] 曲展鸿, 刘咏杭.煅烧焦市场分析与预测[J].轻金属, 2015, (7):5-7. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=qjs201507001QU Zhan-hong, LIU Yong-hang. Analysis and predication of calcined petroleum coke market[J]. Light Metals, 2015, (7):5-7. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=qjs201507001 [2] TRIPATHI N, SINGH R S, HILLS C D. Microbial removal of sulphur from petroleum coke (petcoke)[J]. Fuel, 2019, 235:1501-1505. doi: 10.1016/j.fuel.2018.08.072 [3] XIAO J, LI F, ZHONG Q F, HUANG J D, WANG B J, ZHANG Y B. Effect of high-temperature pyrolysis on the structure and properties of coal and petroleum coke[J]. J Anal Appl Pyrolysis, 2015, 117:64-71. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=2fc25cb97fe8fa86e1ad3f5f16819dfe [4] ZHONG Q F, MAO Q Y, ZHANG L Y, XIANG J H, XIAO J. Structural features of Qingdao petroleum coke from HRTEM lattice fringes:Distributions of length, orientation, stacking, curvature, and a large-scale image-guided 3D atomistic representation[J]. Carbon, 2018, 129:790-802. doi: 10.1016/j.carbon.2017.12.106 [5] EDWARDS L C, NEYREY K J, LOSSIUS L P. A review of coke and anode desulfurization[J]. TMS Light Metals, 2007:895-900. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=CC026120397 [6] 郭慧卿, 付琦, 王鑫龙, 刘粉荣, 胡瑞生, 张浩. CO2气氛对煤热解过程中硫逸出的影响[J].燃料化学学报, 2017, 45(5):523-528. doi: 10.3969/j.issn.0253-2409.2017.05.002GUO Hui-qing, FU Qi, WANG Xin-long, LIU Fen-rong, HU Rui-Sheng, ZHANG Hao. Effect of CO2 atmosphere on sulfur release during coal pyrolysis[J]. J Fuel Chem Technol, 2017, 45(5):523-528. doi: 10.3969/j.issn.0253-2409.2017.05.002 [7] MILENKOVA K S, BORREGO A G, ALVAREZ D, XIBERTA J, MENENDZE R. Devolatilisation behaviour of petroleum coke under pulverised fuel combustion conditions[J]. Fuel, 2003, 82(15):1883-1891. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=82f855af1335ab5ee9c8ab735b50fe0e [8] IRFAN M F, USMAN M R, KUSAKABE K. Coal gasification in CO2 atmosphere and its kinetics since 1948:A brief review[J]. Energy, 2011, 36(1):12-40. https://www.academia.edu/31799677/Coal_gasification_in_CO2_atmosphere_and_its_kinetics_since_1948_A_brief_review [9] 梁永煌, 游伟, 章卫星.我国洁净煤气化技术现状与存在的问题及发展趋势(上)[J].化肥工业, 2013, 40(6):30-36. http://d.old.wanfangdata.com.cn/Periodical/hfgy201306011LIANG Yong-huang, YOU Wei, ZHANG Wei-xing. The current situation and development trend of clean coal gasification technology in china (Ⅰ)[J]. Chem Fert Ind, 2013, 40(6):30-36. http://d.old.wanfangdata.com.cn/Periodical/hfgy201306011 [10] 胡启静, 刘鑫, 周志杰, 于广锁.氯化铁对高硫石油焦-CO2气化的催化作用[J].石油学报(石油加工), 2012, 28(3):463-469. doi: 10.3969/j.issn.1001-8719.2012.03.018HU Qi-jing, LIU Xin, ZHOU Zhi-jie, YU Guang-suo. Catalytic activity of ferric chloride for high-sulfur petroleum coke-carbon dioxide gasification[J].Acta Pet Sin (Pet Process Sect), 2012, 28(3):463-469. doi: 10.3969/j.issn.1001-8719.2012.03.018 [11] GUO Z, FU Z, WAMG S. Sulfur distribution in coke and sulfur removal during pyrolysis[J]. Fuel Process Technol, 2007, 88(10):935-941. doi: 10.1016/j.fuproc.2007.05.003 [12] 袁帅, 陈雪莉, 李军, 代正华, 周志杰, 王辅臣.煤快速热解固相和气相产物生成规律[J].化工学报, 2011, 62(5):204-210. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgxb201105030YUAN Shuai, CHEN Xue-li, LI Jun, DAI Zheng-hua, ZHOU Zhi-jie, WANG Fu-chen. Formations of solid and gas phase products during rapid pyrolysis of coal[J]. CIESC J, 2011, 62(5):204-210. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgxb201105030 [13] 吴幼青.不同热解过程产物炭的理化性质及石油焦催化气化反应特性研究[D].上海: 华东理工大学, 2011. http://d.wanfangdata.com.cn/thesis/Y1859294WU You-qing. Studies on physico-chemical properties of resultant carbons from different pyrolysis processes and catalytic gasification reaction characteristics of petroleum coke[D]. Shanghai: East China University of Science and Technology, 2011. http://d.wanfangdata.com.cn/thesis/Y1859294 [14] SADEZKY A, MUCKENHUBER H, GROTHE H, NIESSNER R, POSCHL U. Raman microspectroscopy of soot and related carbonaceous materials:Spectral analysis and structural information[J]. Carbon, 2005, 43(8):1731-1742. doi: 10.1016/j.carbon.2005.02.018 [15] 刘冬冬, 高继慧, 吴少华, 秦裕琨.热解过程煤焦微观结构变化的XRD和Raman表征[J].哈尔滨工业大学学报, 2016, 48(7):39-45. http://d.old.wanfangdata.com.cn/Periodical/hebgydxxb201607006LIU Dong-dong, GAO Ji-hui, WU Shao-hua, QIN Yu-kun. XRD and Raman characterization of microstructure changes of char during pyrolysis[J]. J Harbin Inst Technol, 2016, 48(7):39-45. http://d.old.wanfangdata.com.cn/Periodical/hebgydxxb201607006 [16] 朱亚明, 赵雪飞, 高丽娟, 程俊霞, 吕君, 赖仕全.煤系针状焦微晶结构的XRD与Raman分峰拟合定量研究[J].光谱学与光谱分析, 2017, 37(6):1919-1924. http://d.old.wanfangdata.com.cn/Periodical/gpxygpfx201706049ZHU Ya-ming, ZHAO Xue-fei, GAO Li-juan, CHENG Jun-xia, LV Jun, LAI Shi-quan. Quantitative study of the microcrystal structure on coal based on needle coke with curve-fitted of XRD and Raman spectrum[J]. Spectrosc Spectral Anal, 2017, 37(6):1919-1924. http://d.old.wanfangdata.com.cn/Periodical/gpxygpfx201706049 [17] LU L, SAHAJWALLA V, HARRIS D. Characteristics of chars prepared from various pulverized coals at different temperatures using drop-tube furnace[J]. Energy Fuels, 2000, 14(4):869-876. doi: 10.1021/ef990236s [18] WU Y Q, WU S Y, GAO J S. Differences in physical properties and CO2 gasification reactivity between coal char and petroleum coke[J]. Process Saf Environ Prot, 2009, 87(5):323-330. doi: 10.1016/j.psep.2009.05.001 [19] ZHONG Q F, XIAO J, DU H J, YAO Z. Thiophenic sulfur transformation in a carbon anode during the aluminum electrolysis process[J]. Energy Fuels, 2017, 31(4):4539-4547. doi: 10.1021/acs.energyfuels.6b03018 [20] 陈喜平, 周孑民, 李旺兴.阳极石油焦的煅烧脱硫研究[J].轻金属, 2007:93-96. http://d.old.wanfangdata.com.cn/Conference/7292590CHEN Xi-ping, ZHOU Jie-min, LI Wang-xing. Study on desulphurization by calcination of anode petroleum coke[J]. Light Met, 2007:93-96. http://d.old.wanfangdata.com.cn/Conference/7292590 [21] 杨彦成, 陶秀祥, 许宁, 罗来芹.煤中含硫基团FTIR表征的可行性分析[J].中国科技论文, 2015, 10(18):2110-2116. doi: 10.3969/j.issn.2095-2783.2015.18.003YANG Yan-cheng, TAO Xiu-xiang, XU Ning, LUO Lai-qin. Feasibility study on the FTIR characterization of sulfur-containing groups in coal[J]. China Sciencepaper, 2015, 10(18):2110-2116. doi: 10.3969/j.issn.2095-2783.2015.18.003 -





 下载:
下载: