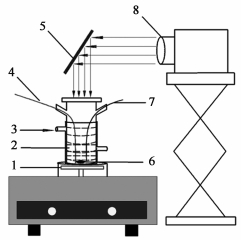
Synergy of photocatalysis and adsorption on TiO2-CeO2 for the removal of organosulfur compounds from diesel fuel
-
摘要: 采用共沉淀法制备TiO2-CeO2光催化吸附脱硫材料,通过低温N2吸附脱附和X射线衍射等技术对TiO2-CeO2的物理化学性质进行了表征。结果表明,紫外光辐射显著提高了TiO2-CeO2的吸附脱硫性能;柴油中有机硫在TiO2-CeO2表面发生了光催化氧化转化为极性较强的砜类,可选择性地吸附在材料表面而被脱除。当TiO2-CeO2材料中钛铈物质的量比为9:1、煅烧温度为500℃时,其光催化吸附协同脱硫效果最好;在紫外光辐射下反应5 h,油品中DBT的脱除率高达99.6%。TiO2-CeO2光催化吸附协同脱硫工艺可有效解决吸附脱硫工艺中芳烃竞争吸附导致吸附脱硫选择性低的问题;在模拟油品中添加质量分数为25%的甲苯,反应7 h后油品脱硫率仍高达96.6%。TiO2-CeO2对不同硫化物的光催化吸附协同脱硫效果顺序为:4,6-DMDBT> DBT> BT。TiO2-CeO2经四次再生循环使用后,脱硫率没有明显降低。Abstract: TiO2-CeO2 mixed oxides were prepared by urea gelation and co-precipitation method and then characterized by N2 sorption and X-ray diffraction (XRD). The synergy of photocatalysis and adsorption on TiO2-CeO2 adsorbents for the removal of organosulfur compounds from diesel fuel was investigated. The results show that the UV irradiation can greatly enhance the adsorption of organosulfur in model fuel on TiO2-CeO2; the organosulfur compounds is first photocatalytically oxidized to polar sulfoxides and sulfones over TiO2-CeO2, which are then selectively adsorbed on the bifucntional TiO2-CeO2 material due to their much higher polarities than the original oranosulfur compounds and other organic compounds in the diesel fuel. The TiO2-CeO2 material with a Ti/Ce molar ratio of 9:1 and calcined at 500℃ exhibits the highest synergistic photocatalysis-adsorption desulfurization performance; over it the sulfur removal rate reaches 99.6% for a model fuel after reaction for 5 h under UV irradiation. The low desulfurization selctivity because of the strongly competitive adsorption of aromatics in the diesel fule could be greatly improved by employing the synergistic photocatalysis-adsorption desulfurization process; the sulfur removal rate is still higher than 96.6% for the model fuel containing 25% toluene after reaction for 7 h under UV irradiation. The desulfurization performance of TiO2-CeO2 for different organosulfur compounds in the diesel fuel follows the order of 4, 6-DMDBT > DBT > BT. Moreover, TiO2-CeO2 can be well regenrated by washing with acetonitrile followed by heat treatment in air; it still gives a high synergistic photocatalysis-adsorption desulfurization performance even after four regeneration cycles.
-
Key words:
- adsorptive desulfurization /
- photocatalysis /
- diesel /
- adsorption selectivity
-
图 3 模拟柴油在不同脱硫条件的GC
Figure 3 SCD chromatograms of model fuels treated at different desulfurization conditions
(a): initial model diesel fuel (50 μg·g-1S); (b): Ti0.9Ce0.1O2-500, dark (29.4 μg·g-1S); (c): No Ti0.9Ce0.1O2-500, UV irradiation 1 h (50 μg·g-1S); (d): Ti0.9Ce0.1O2-500, UV irradiation 1 h (10.8 μg·g-1S); (e): Ti0.9Ce0.1O2-500, UV irradiation 3 h (2.1 μg·g-1S); (f): Ti0.9Ce0.1O2-500, UV irradiation 5 h (0.2 μg·g-1S)
图 7 TixCe(1-x)O2-500中Ti/Ce物质的量比对其光催化吸附协同脱硫性能的影响
Figure 7 Effect of Ti/Ce molar ratio in TixCe(1-x)O2-500 on its synergistic photocatalysis-adsorption desulfurization performance
●: Ti0.9Ce0.1O2-500; ▼: Ti0.5Ce0.5O2-500; ▶: Ti0.1Ce0.9O2-500; ■: TiO2-500; ▲: Ti0.7Ce0.3O2-500; ◀: Ti0.3Ce0.7O2-500; ◆: CeO2-500
表 1 TixCe(1-x)O2材料的孔结构参数
Table 1 Textural properties of various TixCe(1-x)O2 materials
Sample BET surface area
A/(m2·g-1)Average pore size
d/nmTotal pore volume
v/(cm3·g-1)TiO2-500 98.6 7.08 0.234 9 Ti0.9Ce0.1O2-500 159.4 10.83 0.553 1 Ti0.7Ce0.3O2-500 193.5 4.36 0.300 4 Ti0.5Ce0.5O2-500 126.4 5.71 0.242 2 Ti0.3Ce0.7O2-500 145.7 4.62 0.227 2 Ti0.1Ce0.9O2-500 132.6 3.88 0.169 1 CeO2-500 105.3 3.93 0.120 8 Ti0.9Ce0.1O2-400 251.8 8.41 0.422 0 Ti0.9Ce0.1O2-600 58.9 21.09 0.378 8 -
[1] SONG C S. An overview of new approaches to deep desulfurization for ultra-clean gasoline, diesel fuel and jet fuel[J]. Catal Today, 2003, 86(1/4): 211-263. [2] 宋红艳, 何静, 李春喜. 燃料油深度脱硫的技术策略及研究进展[J]. 石油化工, 2015, 44(3): 279-286. http://www.cnki.com.cn/Article/CJFDTOTAL-SYHG201503003.htmSONG Hong-yan, HE Jing, LI Chun-xi. Technical strategies and recent advances for deep desulfurization of fuel oils[J]. Petrochem Technol, 2015, 44(3): 279-286. http://www.cnki.com.cn/Article/CJFDTOTAL-SYHG201503003.htm [3] 李文秀, 崔安磊, 范俊刚, 孙向乐, 张志刚. 载铜球形活性炭的制备及其吸附脱硫性能的研究[J]. 燃料化学学报, 2013, 41(5): 613-618. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18186.shtmlLI Wen-xiu, CUI An-lei, FAN Jun-gang, SUN Xiang-le, ZHANG Zhi-gang. Synthesis of spherical activated carbon supported copper catalyst and its performance for adsorptive desulfurization[J]. J Fuel Chem Technol, 2013, 41(5): 613-618. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18186.shtml [4] 王晓静, 刘超, 董悦, 李发堂, 赵君, 李玉佩. 四氟硼酸改性活性炭的制备及其吸附脱除二苯并噻吩性能[J]. 燃料化学学报, 2015, 43(5): 607-613. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18630.shtmlWANG Xiao-jing, LIU Chao, DONG Yue, LI Fa-tang, ZHAO Jun, LI Yu-pei. Modification of activated carbon with tetrafluoroboric acid and its performance in adsorption desulfurization of dibenzothiophene[J]. J Fuel Chem Technol, 2015, 43(5): 607-613. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18630.shtml [5] SUN H Y, SUN L P, LI F, ZHANG L. Adsorption of benzothiophene from fuels on modified NaY zeolites[J]. Fuel Process Technol, 2015,134: 284-289. doi: 10.1016/j.fuproc.2015.02.010 [6] 宋丽娟, 胡月婷, 秦玉才, 于文广, 张晓彤. NiY分子筛表面酸性影响其选择性吸附脱硫性能的机理研究[J]. 燃料化学学报, 2016, 44(9): 1082-1088. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18896.shtmlSONG Li-juan, HU Yue-ting, QIN Yu-cai, YU Wen-guang, ZHANG Xiao-tong. Mechanism of effects of surface acidity on performance of adsorption desulfurization of NiY zeolites[J]. J Fuel Chem Technol, 2016, 44(9): 1082-1088. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18896.shtml [7] WU L M, XIAO J, WU Y, XIAN S K, MIAO G, WANG H H, LI Z. A combined experimental/computational study on the adsorption of organosulfur compounds over metal-organic frameworks from fuels[J]. Langmuir, 2014, 30(4): 1080-1088. doi: 10.1021/la404540j [8] QIN L, SHI W P, LIU W F, YANG Y Z, LIU X G, XU B S. Surface molecularly imprinted polymers grafted on ordered mesoporous carbon nanospheres for fuel desulfurization[J]. Rsc Adv, 2016, 6(15): 12504-12513. doi: 10.1039/C5RA23582K [9] 胡廷平, 张宴铭, 郑立辉, 范国枝. 硅胶表面苯并噻吩分子印迹聚合物的分子识别与吸附性能[J]. 燃料化学学报, 2010, 38(6): 722-729. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17658.shtmlHU Ting-ping, ZHANG Yan-ming, ZHENG Li-hui, FAN Guo-zhi. Molecular recognition and adsorption performance of benzothiophene imprinted polymer on silica gel surface[J]. J Fuel Chem Technol, 2010, 38(6): 722-729. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17658.shtml [10] 张伟, 李鑫, 童靖予, 胡雨, 李翠清. 吸附法燃油超深度脱硫的研究进展[J]. 石油化工, 2016, 45(11): 1396-1402. doi: 10.3969/j.issn.1000-8144.2016.11.019ZHANG Wei, LI Xin, TONG Jing-yu, HU Yu, LI Cui-qing. Progresses in ultra-deep desulfurization of fuel oil through adsorption[J]. Petrochem Technol, 2016, 45(11): 1396-1402. doi: 10.3969/j.issn.1000-8144.2016.11.019 [11] BHANDARI V M, HYUN KO C, GEUN PARK J, HAN S S, CHO S H, KIM J N. Desulfurization of diesel using ion-exchanged zeolites[J]. Chem Eng Sci, 2006, 61(8): 2599-2608. doi: 10.1016/j.ces.2005.11.015 [12] KOBAYASHI M, FLYTZANI-STEPHANOPOULOS M. Reduction and sulfidation kinetics of cerium oxide and Cu-modified cerium oxide[J]. Ind Eng Chem Res, 2002, 41(13): 3115-3123. doi: 10.1021/ie010815w [13] ZHANG W, LIU H Y, XIA Q B, LI Z. Enhancement of dibenzothiophene adsorption on activated carbons by surface modification using low temperature oxygen plasma[J]. Chem Eng J, 2012, 209: 597-600. doi: 10.1016/j.cej.2012.08.050 [14] MA X, SPRAGUE M, SONG C. Deep desulfurization of gasoline by selective adsorption over nickel-based adsorbent for fuel cell applications[J]. Ind Eng Chem Res, 2005, 44(15): 5768-5775. doi: 10.1021/ie0492810 [15] OTSUKI S, NONAKA T, TAKASHIMA N, QIAN W H, ISHIHARA A, IMAI T, KABE T. Oxidative desulfurization of light gas oil and vacuum gas oil by oxidation and solvent extraction[J]. Energy Fuels, 2000, 14(6): 1232-1239. doi: 10.1021/ef000096i [16] XIAO J, SONG C S, MA X L, LIT Z. Effects of aromatics, diesel additives, nitrogen compounds, and moisture on adsorptive desulfurization of diesel fuel over activated carbon[J]. Ind Eng Chem Res, 2012, 51(8): 3436-3443. doi: 10.1021/ie202440t [17] WANG L F, SUN B D, YANG F H, YANG R T. Effects of aromatics on desulfurization of liquid fuel by π-complexation and carbon adsorbents[J]. Chem Eng Sci, 2012, 73: 208-217. doi: 10.1016/j.ces.2012.01.056 -





 下载:
下载: