Preparation of DESs/SG catalyst and its performance in the oxidative desulfurization
-
摘要: 通过溶胶-凝胶法将脯氨酸基低共熔溶剂负载到硅胶上制得DESs/SG型催化剂。采用FT-IR、XRD、SEM/EDS及N2吸附-脱附等手段对催化剂的结构进行表征。结果发现,低共熔溶剂可以成功负载到硅胶中,硅胶的比表面积和孔体积有所下降,而孔径增大。以DESs/SG为吸附剂和催化剂,H2O2为氧化剂,研究其对模拟油中的二苯并噻吩的脱除性能,考察了低共熔溶剂负载量、反应温度、n(H2O2)/n(S)比、催化剂用量、含硫化合物的类型以及催化剂循环使用次数对脱硫效率的影响。结果表明,在最优脱硫条件下,DESs/SG对二苯并噻吩、4,6-二甲基二苯并噻吩和苯并噻吩的脱硫率分别为97%、96.5%和46.4%;催化剂循环使用九次后,催化脱硫效率仍高达89.4%。Abstract: DESs/SG catalyst was prepared by supporting the proline-based deep eutectic solvent (DES) on the silica-gel (SG) matrix using a sol-gel method; the catalyst structure was characterized by FT-IR, XRD, SEM/EDS and N2 adsorption-desorption. The results showed that the DESs were successfully incorporated into the silica-gel matrix, leading to a decrease in the surface area and pore volume, but an increase in the pore diameter. With hydrogen peroxide as oxidant, the catalytic performance of DESs/SG in the oxidative desulfurization of a model oil containing dibenzothiophene (DBT) was investigated; the effects of deep eutectic solvent loading, reaction temperature, n(H2O2)/n(S) ratio, catalyst amount, sulfur compound type and catalyst recycle times on the desulfurization efficiency were considered. The results indicated that under optimum desulfurization conditions, the desulfurization degrees for DBT, 4, 6-dimethyl-dibenzothiophene (4, 6-DMDBT) and benzothiophene (BT) over the DESs/SG catalyst were 97%, 96.5% and 46.4%, respectively; after recycling for 9 times, the DESs/SG catalyst still displayed a desulfurization degree of above 89.4%.
-
Key words:
- silica gel /
- oxidation desulfurization /
- deep eutectic solvents /
- sol-gel /
- dibenzothiophene
-
表 1 催化剂的比表面积和孔结构
Table 1 Surface area and pore structure of the DESs/SG catalysts with various DES loadings
Sample Specific surface area A/(m2·g-1) Pore volume v/(cm3·g-1) Pore diameter d/nm SG 664 0.7677 2.315 4%-DESs/SG 544 0.6936 3.617 8%-DESs/SG 335 0.3780 3.130 12%-DESs/SG 269 0.3266 3.614 表 2 不同催化剂的脱硫性能
Table 2 Desulfurization performances of different catalysts
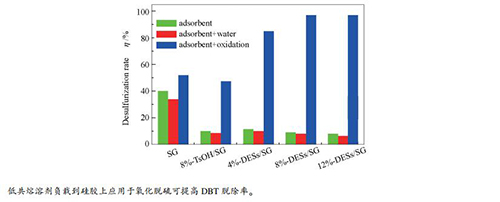
Catalyst Oxidant Desulphurization rate η/% SG H2O2 52 8%-TsOH/SG H2O2 47.6 8%-DESs/SG H2O2 97 - H2O2 1 reaction condition: 70 ℃, 5 mL model oil, 0.2 g catalyst, n(H2O2)/n(S)= 6, 3 h -
[1] MA X, ZHOU A, SONG C. A novel method for oxidative desulfurization of liquid hydrocarbon fuels based on catalytic oxidation using molecular oxygen coupled with selective adsorption[J]. Catal Today, 2007, 123(1/4):276-284. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d68b61c8bff99a02cd2cfbe83cc76f22 [2] 李旭贺, 方磊, 杨浩, 张健, 梁飞雪, 王海彦, 王彦娟.石墨相氮化碳负载磷钨酸杂化材料的制备及其氧化脱硫催化性能[J].燃料化学学报, 2019, 47(2):174-182. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201902006LI Xu-he, FANG Lei, YANG Hao, ZHANG Jian, LIANG Fei-xue, WANG Hai-yan, WANG Yan-juan. Preparation of g-C3N4 supported phosphotungstate hybrid materials and their catalytic performance in the oxidative desulfurization[J]. J Fuel Chem Technol, 2019, 47(2):174-182. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201902006 [3] YU M, LI Z, JI Q, WANG S, SU D, LIN Y S. Effect of thermal oxidation of activated carbon surface on its adsorption towards dibenzothiophene[J]. Chem Eng J, 2009, 148(2/3):242-247. doi: 10.1016-j.cej.2008.08.018/ [4] LIU W, LI T, YU G, WANG J, ZHOU Z. One-pot oxidative desulfurization of fuels using dual-acidic deep eutectic solvents[J]. Fuel, 2020, 265:116967-116981. http://cn.bing.com/academic/profile?id=27b6049b0aa9ea52ec99f6f1b72d1794&encoded=0&v=paper_preview&mkt=zh-cn [5] YANG Y, LV G, DENG L, LU B, LI J, ZHANG J, SHI J Y, DU S J. Ultra-deep desulfurization of diesel fuel via selective adsorption over modified activated carbon assisted by pre-oxidation[J]. J Cleaner Prod, 2017, 161:422-430. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=2c0fad38985479ba5c5efabf595806a0 [6] SAFA M, MOKHTARANI B, MORTAHEB H R. Deep extractive desulfurization of dibenzothiophene with imidazolium or pyridinium-based ionic liquids[J]. Chem Eng Res Des, 2016, 111:323-331. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb200703008 [7] JIANG B, YANG H, ZHANG L, ZHANG Y Y, SUN Y L, HUANG Y. Efficient oxidative desulfurization of diesel fuel using amide-based ionic liquids[J]. Chem Eng J, 2016, 283:89-96. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=898a1c51d9e07eb9c842f3aecb70f821 [8] JIANG W, ZHU W, LI H, WANG X, YIN S, CHANG Y H, LI H M. Temperature-responsive ionic liquid extraction and separation of the aromatic sulfur compounds[J]. Fuel, 2015, 140:590-596. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=2204a419319a1784a1f3a6286ff71f7a [9] YIN J, WANG J, LI Z, LI D, YANG G, CUI Y N, WANG A L, LI C P. Deep desulfurization of fuels based on an oxidation/extraction process with acidic deep eutectic solvents[J]. Green Chem, 2015, 17(9):4552-4559. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=ccc32cfe3913990cb62ac508bc92249a [10] ALI E, HADJ-KALI M K, MULYONO S, ALNASHEF I, FAKEEHA A, MJALLI F, HAYYAN A. Solubility of CO2 in deep eutectic solvents:Experiments and modelling using the Peng-Robinson equation of state[J]. Chem Eng Res Des, 2014, 92(10):1898-1906. https://www.sciencedirect.com/science/article/abs/pii/S0263876214000720 [11] LIU P, HAO J W, MO L P, ZHANG Z H. Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions[J]. RSC Adv, 2015, 5(60):48675-48704. http://cn.bing.com/academic/profile?id=c1752d69d74b471d4c4e873d5cd62431&encoded=0&v=paper_preview&mkt=zh-cn [12] NKUKU C A, LESUER R J. Electrochemistry in deep eutectic solvents[J]. J Phys Chem B, 2007, 111(46):13271-13277. http://d.old.wanfangdata.com.cn/Periodical/zngydxxb201809003 [13] SMEETS S, LIU L, DONG J, MCCUSKER L B. Cheminform abstract:Ionothermal synthesis and structure of a new layered zirconium phosphate[J]. Inorg Chem, 2015, 46(42):7953-7958 http://med.wanfangdata.com.cn/Paper/Detail/PeriodicalPaper_PM26234954 [14] 唐晓东, 张晓普, 李晶晶, 王治宇, 杨柳, 王春.低共熔溶剂在车用燃料脱硫中的研究进展[J].化工进展, 2018, 37(11):82-89. http://d.old.wanfangdata.com.cn/Periodical/hgjz201811010TANG Xiao-dong, ZHANG Xiao-pu, LI Jing-jing, WANG Zhi-yu, YANG Liu, WANG Chun. Research progress of deep eutectic solvents in desulfurization of vehicle fuel[J]. Chem Ind Eng Prog, 2018, 37(11):82-89. http://d.old.wanfangdata.com.cn/Periodical/hgjz201811010 [15] LÜ H, LI P, DENG C, REN W Z, WANG S N, LIU P, ZHANG H. Deep catalytic oxidative desulfurization (ODS) of dibenzothiophene (DBT) with oxalate-based deep eutectic solvents (DESs)[J]. Chem Commun, 2015, 51(53):10703-10706. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=5a93bdbc705be5b419e573e26c25079a [16] LÜ H, LI P, LIU Y, HAO L W, REN W Z, ZHU W J, DENG C L, YANG P. Synthesis of a hybrid Anderson-type polyoxometalate in deep eutectic solvents (DESs) for deep desulphurization of model diesel in ionic liquids (ILs)[J]. Chem Eng J, 2017, 313:1004-1009. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d2cfdb6e08b43b0e896463b319cfa677 [17] ZHANG Q, KARINE D O V, ROYER S, JEROME F. Deep eutectic solvents:Syntheses, properties and applications[J]. Chem Soc Rev, 2012, 41(21):7108-7146. http://d.old.wanfangdata.com.cn/Periodical/sdhg201411022 [18] GARCÍA-GUTIÉRREZ J L, FUENTES G A, HERNÁNDEZ-TERÁN M E, MURRIETA F, NAVARRETE J, JIMÉNEZ-CRUZ F. Ultra-deep oxidative desulfurization of diesel fuel with H2O2 catalyzed under mild conditions by polymolybdates supported on Al2O3[J]. Appl Catal A:Gen, 2006, 305(1):15-20. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6776437ea0e0c9549eb8d8c4feed9cc6 [19] YANG C, ZHAO K, CHENG Y, ZENG G M, ZHANG M M, SHAO J J, LU L. Catalytic oxidative desulfurization of BT and DBT from n-octane using cyclohexanone peroxide and catalyst of molybdenum supported on 4A molecular sieve[J]. Sep Purif Technol, 2016, 163:153-161. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=c989782e4a44439a0100a3cdbad60231 [20] LI S, MOMINOU N, WANG Z, WANG L. Ultra-deep desulfurization of gasoline with CuW/TiO2-go through photocatalytic oxidation[J]. Energy Fuels, 2016, 30(2):962-967. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=3dd28a0cddc63487f3fd74328cb0e6ea [21] 颜学敏, 苏高申, 熊麟.银离子修饰的介孔磷钨酸/二氧化硅催化剂氧化脱硫性能研究[J].燃料化学学报, 2009, 37(3):318-323. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb200903011YAN Xue-ming, SU Gao-shen, XIONG Lin. Oxidative desulfurization of diesel oil over Ag-modified mesoporous HPW/SiO2 catalyst[J]. J Fuel Chem Technol, 2009, 37(3):318-323. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb200903011 [22] SELVAM T, MACHOKE A, SCHWIEGER W. Supported ionic liquids on non-porous and porous inorganic materials-A topical review[J]. Appl Catal A:Gen, 2012, 445:92-101. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=94c940eb46ee5e982f59a9621d8b7b65 [23] AZIZI N, EDRISI M, ABBASI F. Mesoporous silica SBA-15 functionalized with acidic deep eutectic solvent:A highly active heterogeneous N-formylation catalyst under solvent-free conditions[J]. Appl Organomet Chem, 2018, 32(1):3901-3910. doi: 10.1002/aoc.3901 [24] AZIZI N, EDRISI M. Deep eutectic solvent immobilized on SBA-15 as a novel separable catalyst for one-pot three-component Mannich reaction[J]. Microporous Mesoporous Mater, 2017, 240:130-136. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=e95230d22101a4ace36e3e6686af99b8 [25] HAO L, WANG M, SHAN W, DENG C L, REN W Z, SHI Z Z, LÜ H Y. L-proline-based deep eutectic solvents (DESs) for deep catalytic oxidative desulfurization (ODS) of diesel[J]. J Hazard Mater, 2017, 339:216-222. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=56fbbb34f9f6cbbd9295113cc26ce2ce [26] ZHAO R, LI X, SU J, GAO X H. Preparation of WO3/g-C3N4 composites and their application in oxidative desulfurization[J]. Appl Surf Sci, 2016, 392:810-816. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=1ed09bc53c4bc5d519a61fe0bb0b7c20 [27] PIRZADA T, SHAH S S. Water-resistant poly (vinyl alcohol)-silica hybrids through sol-gel processing[J]. Chem Eng Technol, 2014, 37(4):620-626. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ0232439734/ [28] MATOS M C, ILHARCO L M, ALMEIDA R M. The evolution of TEOS to silica gel and glass by vibrational spectroscopy[J]. J Non-Cryst Solids, 1992, 147:232-237. http://cn.bing.com/academic/profile?id=6cf17d2990eb949af04abbb6780798b6&encoded=0&v=paper_preview&mkt=zh-cn [29] WU Y, LI Z, XIA C. Silica-gel-supported dual acidic ionic liquids as efficient catalysts for the synthesis of polyoxymethylene dimethyl ethers[J]. Ind Eng Chem Res, 2016, 55(7):1859-1865. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=a1d29c96fd35e234c4dfa82c2d6fd0a6 [30] AZIZI N, EDRISI M, ABBASI F. Mesoporous silica SBA-15 functionalized with acidic deep eutectic solvent:A highly active heterogeneous N-formylation catalyst under solvent-free conditions[J]. Appl Organomet Chem, 2018, 32(1):3901-3910. http://cn.bing.com/academic/profile?id=b6753d224632abbc75f45994b8eda142&encoded=0&v=paper_preview&mkt=zh-cn [31] SAFA M, MOKHTARANI B, MORTAHEB H R, HEIDAR K T, SHARIFI A, MIRZAEI M. Oxidative desulfurization of diesel fuel using a Brønsted acidic ionic liquid supported on silica gel[J]. Energy Fuels, 2017, 31(9):10196-10205. [32] WEI J, ZHU W, LI H, WANG X, YIN S, CHANGY H, LI H M. Temperature-responsive ionic liquid extraction and separation of the aromatic sulfur compounds[J]. Fuel, 2015, 140:590-596. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=2204a419319a1784a1f3a6286ff71f7a [33] YANG W X, GUO G, MEI Z, YU Y H. Deep oxidative desulfurization of model fuels catalysed by immobilized ionic liquid on MIL-100(Fe)[J]. RSC Adv, 2019, 9(38):21804-21809. http://cn.bing.com/academic/profile?id=402c1c39f2681024d4e4d26c5437cf31&encoded=0&v=paper_preview&mkt=zh-cn [34] YANG C, ZHAO K, CHENG Y, ZENG G M, ZHANG M M, SHAO J J. Catalytic oxidative desulfurization of BT and DBT from n-octane using cyclohexanone peroxide and catalyst of molybdenum supported on 4A molecular sieve[J]. Sep Purif Technol, 2016, 163:153-161. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=c989782e4a44439a0100a3cdbad60231 [35] 于凤丽, 王睿.有机-无机型杂多酸相转移催化氧化脱硫性能研究[J].化学学报, 2014, 72(1):105-113. http://d.old.wanfangdata.com.cn/Periodical/hxxb201401014YU Feng-li, WANG Rui. Study on oxidative desulfurization catalyzed by organic-inorganic heteropoiyacids as phase transfer catalyst[J]. Acta Chim Sin, 2014, 72(1):105-113. http://d.old.wanfangdata.com.cn/Periodical/hxxb201401014 [36] MAO C, ZHAO R, LI X. Phenylpropanoic acid-based DESs as efficient extractants and catalysts for the removal of sulfur compounds from oil[J]. Fuel, 2017, 189:400-407. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=291862f6f76379646c35817b390c1e4c [37] SHI C, WANG W, LIU N, XUEYAN XU, WANG D H, ZHANG M H, SUN P C, CHEN T H. Low temperature oxidative desulfurization with hierarchically mesoporous titaniumsilicate Ti-SBA-2 single crystals[J]. Chem Commun, 2015, 51(57):11500-11503. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=61722593b40f714b12fd0911da16c14d [38] VIDAL L, RIEKKOLA M L, CANALS A. Ionic liquid-modified materials for solid-phase extraction and separation:A review[J]. Anal Chim Acta, 2012, 715:0-41. http://cn.bing.com/academic/profile?id=a1ec0fa3a518242111c8382676079f15&encoded=0&v=paper_preview&mkt=zh-cn [39] ZHANG B Y, JIANG Z X, LI J, ZHANG Y, LIN F, LIU Y, LI C. Catalytic oxidation of thiophene and its derivatives via dual activation for ultra-deep desulfurization of fuels[J]. J Catal, 2012, 287(3):5-12. http://cn.bing.com/academic/profile?id=317c327d8605f12ed81d9adb66df3b96&encoded=0&v=paper_preview&mkt=zh-cn [40] WANG S, LI P, HAO L, DENG C L, REN W Z, LÜ H Y. Oxidative desulfurization of model diesel using a fenton-like catalyst in the ionic liquid[Dmim]BF4[J]. Chem Eng Technol, 2017, 40(3): 555-560. [41] MAGGI R, PISCOPO C G, SARTORI G, STORAROB L, MORETTI E. Supported sulfonic acids:Metal-free catalysts for the oxidation of hydroquinones to benzoquinones with hydrogen peroxide[J]. Appl Catal A:Gen, 2012, 411(2):146-152. http://cn.bing.com/academic/profile?id=d395816349610de26a9775f6166a0827&encoded=0&v=paper_preview&mkt=zh-cn [42] LI N, YUAN G, ZHANG X, YU Z J, SHI L, SUN Q. Oxidation of styrene to benzaldehyde by p-toluenesulfonic acid using hydrogen peroxide in the presence of activated carbon[J]. Chin J Catal, 2015, 36(5):721-727. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cuihuaxb201505006 -





 下载:
下载: