-
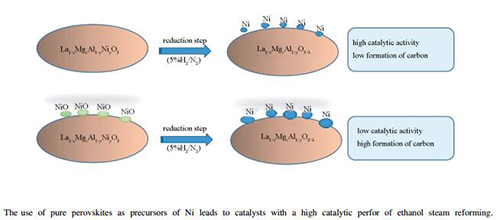
Abstract: Perovskites as host structures of cations were used in order to generate in situ active and stable catalysts for ethanol steam reforming. For this purpose, La1-xMgxAl1-yNiyO3 (x=0.1; y=0, 0.1, 0.2, 0.3) perovskites were synthetized by the citrate method. Ni segregation is evident for a substitution level higher than 0.2. The segregation of Ni as NiO generated species interacts with different metal-support after the reduction step. The y=0.1 catalyst presents the highest H2 yield value about 85% during reaction time, with low mean values of CH4 and CO selectivities of 3.4% and 11%, respectively and a low carbon formation. The better performance of y=0.1 catalyst could be attributed to the minor proportion of segregated phases, thus a controlled expulsion of Ni is successfully reached.
-
Key words:
- perovskites /
- catalyst design /
- ethanol /
- steam reforming
-
Table 1 BET specific surface area, H2 consumption from TPR, Ni content from ICP-OES and weight loss from TG
Catalyst ABET/(m2·g-1) H2 consumption/(μmol·mgNi-1) Ni content w/% Weight loss w/% LaAlO3 14 - - La0.9Mg0, 1AlO3 15 - - NiO/LaAlO3 17 0.63 3.3 (2.8)* LaMgNi1 13 2.16 2.2 (2.8) 6 LaMgNi2 10 2.04 3.4 (5.6) 7 LaMgNi3 8 1.39 5.2 (8.3) 15 *: (values between brackets correspond to nominal ones) Table 2 Structural parameters for the perovskites with nominal composition La0.9Mg0.1Al1-yNiyO3, refined in the rhombohedral R3c (no. 167) space group at 25 ℃ from NPD data
Ni contents y= 0.1 y= 0.2 y= 0.3 a/Å 5.3670(6) 5.3735(5) 5.3821(5) c/Å 13.139(2) 13.149(2) 13.149(2) v/Å3 327.75(8) 328.79(7) 329.86(6) La/Mg 6a (0 0 1/4) focc La/Mg 0.99(2)/0.01(2) 1.00(2)/0.00(2) 0.99(2)/0.01(2) B/Å2 0.36(4) 0.36(3) 0.31(3) (Al, Ni) 6b (0 0 0) focc Al/Ni 0.95(1)/0.05(1) 0.936(4)/0.064(1) 0.810(2)/0.190(2) B/Å2 0.49(6) 0.47(5) 0.88(3) O 18e (x 0 1/4) x 0.4751(2) 0.4737(2) 0.4717(2) focc 1.0 1.0 1.0 B/Å2 0.69(3) 0.77(2) 0.88(3) Fraction main phase 96.6(1) 93.8(1) 93.1(1) Fraction MgO 1.2(1) 0.6(1) 2.7(1) Fraction NiO 2.2(1) 5.6(1) 4.2(1) Reliability factors χ2 2.20 5.22 2.22 Rp/% 2.82 2.60 2.82 Rwp/% 2.27 3.61 2.12 RI/% 2.48 1.58 1.77 Table 3 Main interatomic distances (Å) and selected angles (°) for the perovskites with nominal composition La0.9Mg0.1Al1-yNiyO3
Ni content y=0.1 y= 0.2 y= 0.3 (Al, Ni)-O ×6 1.9018(7) 1.9044(7) 1.9073(8) O-(Ni, Mo)-O 90.16(4) 90.20(4) 90.28(5) (Ni, Mo)-O-(Ni, Mo) 171.94(2) 171.50(2) Table 4 XPS results of catalysts
Catalyst n(Ni)
/n(La+Mg+Al)n(Mg)
/n(La+Ni+Al)n(Oad)
/n(Ol)LaMgNi1 0.048 (0.05)* 0.05 (0.05) 0.15 LaMgNi2 0.091 (0.11) 0.07 (0.05) 0.18 LaMgNi3 0.131 (0.176) 0.095 (0.05) 0.22 *: values between brackets correspond to nominal ones -
[1] CHORKENDORFF I, NIEMANTSVERDRIET J W. Concepts of Modern Catalysis and Kinetics[M]. Wiley Editorial. 2003. [2] ARAMOUNI A K, TOUMA J G, TARBOUSH B A, ZEAITER J M. Catalyst design for dry reforming of methane:Analysis review[J]. Renewable Sustainable Energy Rev, 2017, 82(part 3):2570-2585. [3] CHOI S O, MOON S H. Performance of La1-xCexFe0.7Ni0.3O3 perovskite catalysts for methane steam reforming[J]. Catal Today, 2009, 146(1):148-153. http://www.sciencedirect.com/science/article/pii/S0920586109001187 [4] PALCHEVA R, OLSBYE U, PALCUT M, RAUWEL P, TYULIEV G, VELINOV N, FJELLVÄG H H, Rh promoted La0.75Sr0.25(Fe0.8Co0.2)1-xGaxO3-δ perovskite catalysts:Characterization and catalytic performance for methane partial oxidation to synthesis gas[J]. Appl Surf Sci, 2015, 357(part A):45-54. http://adsabs.harvard.edu/abs/2015ApSS..357...45P [5] NI M, LEUNG D, LEUNG M, A review on reforming bio-ethanol for hydrogen production[J]. Int J Hydrogen Energy, 2007, 32(15):3238-3247. doi: 10.1016/j.ijhydene.2007.04.038 [6] PEREIRA E B, RAMÍREZ D E L A, PISCINA P, MARTI S, HOMS N. H2 production by oxidative steam reforming of ethanol over K promoted Co-Rh/CeO2-ZrO2 catalysts[J]. Energy Environ Sci, 2010, 3:487-493. doi: 10.1039/b924624j [7] HAN X, YU Y, HE H, SHAN W. Hydrogen production from oxidative steam reforming of ethanol over rhodium catalysts supported on Ce-La solid solution[J]. Int J Hydrogen Energy, 2013, 38(25):10293-10304. doi: 10.1016/j.ijhydene.2013.05.137 [8] COSTA L O, VASCONCELOS S M, PINTO A L, SILVA A M, MATTOS L V, NORONHA F B. Rh/CeO2 catalyst preparation and characterization for hydrogen production from ethanol partial oxidation[J]. J Mater Sci, 2008, 43(2):440-449. doi: 10.1007/s10853-007-1982-2 [9] KRALEVA E, SOKOLOV S, NASILLO G, BENTRUP U, EHRICH H. Catalytic performance of CoAlZn and NiAlZn mixed oxides in hydrogen production by bio-ethanol partial oxidation[J]. Int J Hydrogen Energy, 2014, 39(1):209-220. doi: 10.1016/j.ijhydene.2013.10.072 [10] BEYHAN S, LÉGER J M, KADIRGAN F. Understanding the influence of Ni, Co, Rh and Pd additionto PtSn/C catalyst for the oxidation of ethanol by in situ Fourier transform infrared spectroscopy[J]. Appl Catal B:Environ, 2014, 144:66-74. doi: 10.1016/j.apcatb.2013.07.020 [11] CHEN H Q, YU H, YANG G X, PENG F, WANG H J, WANG J. Auto-thermal ethanol micro-reformer with a structural Ir/La2O3/ZrO2 catalyst for hydrogen production[J]. Chem Eng J, 2011, 167(1):322-327. doi: 10.1016/j.cej.2010.12.077 [12] SILVA A M, COSTA L O, BARANDAS A P, BORGES L E, MATTOS L V, NORONHA F B. Effect of the metal nature on the reaction mechanism of the partial oxidation of ethanol over CeO2-supported Pt and Rh catalysts[J]. Catal Today, 2008, 133-135:755-761. doi: 10.1016/j.cattod.2007.12.103 [13] FRUSTERI F, FRENI S. Bio-ethanol, a suitable fuel to produce hydrogen for a molten carbonate fuel cell[J]. J Power Sources, 2007, 173(1):200-209. doi: 10.1016/j.jpowsour.2007.04.065 [14] BION N, EPRON F, DUPREZ D. Bioethanol reforming for H2 production. A comparison with hydrocarbon reforming[J]. Catalysis, 2010, 22:1-55. http://pubs.rsc.org/en/Content/Chapter/9781847559630-00001/978-1-84755-963-0 [15] HARYANTO A, FERNANDO S, MURALI N, ADHIKARI S. Current status of hydrogen production techniques by steam reforming of ethanol:A Review[J]. Energy Fuels, 2005, 19(5):2098-2106. doi: 10.1021/ef0500538 [16] BENGAARD H S, NORSKOV J K, SEHESTED J, CLAUSEN B S, NIELSEN L P, MOLENBROEK A M, ROSTRUP-NIELSEN J R. Steam reforming and graphite formation on Ni catalysts[J]. J Catal, 2002, 209(2):365-384. doi: 10.1006/jcat.2002.3579 [17] BOROWIECKI T. Nickel catalysts for steam reforming of hydrocarbons; size of crystallites and resistance to coking[J]. Appl Catal, 1982, 4:223-231. doi: 10.1016/0166-9834(82)80104-8 [18] AGÜERO F, MORALES M R, LARREGOLA S, IZURIETA E, LOPEZ E, CADUS L E. La1-xCaxAl1-yNiyO3 perovskites used as precursors of nickel based catalysts for ethanol steam reforming[J]. Int J Hydrogen Energy, 2015, 40:15510. doi: 10.1016/j.ijhydene.2015.08.051 [19] COURTY P, AJOT H, MARCILLY C, DELMON B. Oxydes mixtes ou en solution solide sous forme très divisé e obtenus par décomposition thermique de précurseurs amorphes[J]. Powder Technol, 1973, 7(1):21-38. doi: 10.1016/0032-5910(73)80005-1 [20] RIETVELD H M, A profile refinement method for nuclear and magnetic structures[J]. Appl Crystallogr, 1969, 2:65-71. doi: 10.1107/S0021889869006558 [21] RODRIGUEZ CARVAJAL. Recent advances in magnetic-structure determination by neutron power diffraction[J]. Phys B, 1993, 192(1/2):55-69. http://adsabs.harvard.edu/abs/1993CCM....41..738B [22] WU Y J, DÍAZ ALVARADO F, SANTOS J C, GRACIA F, CUNHA A F, RODRIGUES A E. Sorption-enhanced steam reforming of ethanol:thermodynamic comparison of CO2 sorbents[J]. Chem Eng Technol, 2012, 35(5):847-858. doi: 10.1002/ceat.v35.5 [23] CUNHA A F, WU Y J, DÍAZ ALVARADO F A, SANTOS J C, VAIDYA P D, RODRIGUES A E. CAN, Steam reforming of ethanol on a Ni/Al2O3 catalyst coupled with a hydrotalcite-like sorbent in a multilayer pattern for CO2 uptake[J]. Can J Chem Eng, 2012, 90(6):1514-1526. doi: 10.1002/cjce.v90.6 [24] SÁNCHEZ-SÁNCHEZ M C, NAVARRO R M, FIERRO J L G. Ethanol steam reforming over Ni/MxOy-Al2O3 (M-Ce, La, Zr and Mg) catalysts:influence of support on the Hydrogen production[J]. Int J Hydrogen Energy, 2007, 32:1462. doi: 10.1016/j.ijhydene.2006.10.025 [25] HARDINI D, YOON C, HON J, YOON S, NAM S, LIM T. Influence of preparation methods and OSC on activity and stability[J]. Catal Lett, 2012, 142(2):205-212. doi: 10.1007/s10562-011-0746-4 [26] RIBEIRO N, NETO R, MOYA S, SOUZA M, SCHMAL M. Synthesis of NiAl2O4 with high surface area as precursor of Ni nanoparticles for hydrogen production[J]. Int J Hydrogen Energy, 2010, 35(21):11725-11732. doi: 10.1016/j.ijhydene.2010.08.024 [27] GALLEGO G S, MONDRAGÓN F, BARRAULT J, TATIBOUËT J-M, BATIOT-DUPEYRAT C. CO2 reforming of CH4 over La-Ni based perovskite precursors[J]. Appl Catal A:Gen, 2006, 311(1):164-171. [28] SIERRA GALLEGO G, MONDRAGON F, TATIBOUËT J-M, BARRAULT J BATIOT-DUPEYRAT, C, Carbon dioxide reforming of methane over La2NiO4 as catalyst precursor and characterization of carbon deposition[J]. Catal Today, 2008, 133-135:200-209. doi: 10.1016/j.cattod.2007.12.075 [29] JIRATOVA K, MIKULOVA J, KLEMPA J, GRYGAR T, BASTL Z, KOVANDA F. Modification of Co-Mn-Al mixed oxide with potassium and its effect on deep oxidation of VOC[J]. Appl Catal A:Gen, 2009, 361:106-116. doi: 10.1016/j.apcata.2009.04.004 -





 下载:
下载: