Preparation and characterization of colloidal dispersions of graphene-like structures from different ranks of coals
-
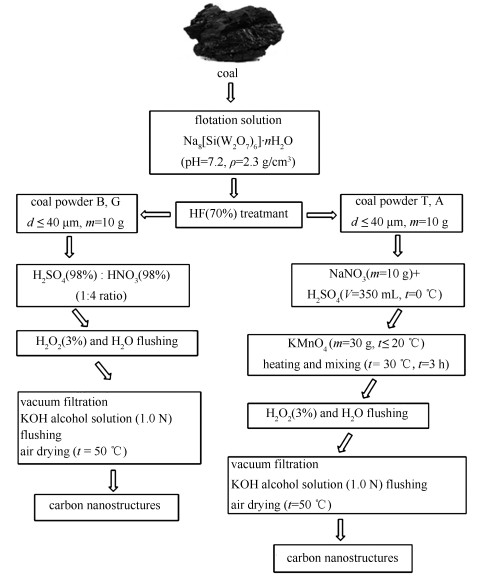
Abstract: This paper focuses on preparation of colloidal solution of graphene-like structures from different ranks of coals: brown coal, bituminous coal, low-volatile bituminous coal, anthracite. It was found that brown coal thermo-oxidative destruction leads to formation of small d=32 nm (V=17%) and large d=122 nm (V=11%) fractions of nanoparticles. The thermo-oxidative destruction of bituminous coal leads to formation of nanoparticles d=50 nm (V=5.2%) and d=164 nm (V=16%). Thermo-oxidative destruction of low-volatile bituminous coal and anthracite leads to formation of nanoparticles, predominantly, d=122-190 nm. Carbon nanostructures obtained from coal are negatively charged at pH=2-12. Colloidal solution of carbon nanostructures at dispersed phase concentration 0.01 mg/mL is stable for 1 month. Electron diffraction patterns and X-ray analysis of carbon nanostructures showed that nanostructure from brown coal is amorphous and nanostructure from anthracite is crystalline. Results of coal macromolecules modeling and graphene-like structures obtained from them are presented.
-
Table 1 Proximate and ultimate analyses of coal

Table 2 Structural and sorption characteristics of coal

-
[1] RAO C N R, SOOD A K. Graphene: Synthesis, Properties, and Phenomena[M]. Wiley, 2012. [2] ZHAO J, LIU L, LI F. Graphene Oxide: Physics and Applications[M]. Springer, 2015. [3] HU Y, LI F, HAN D, LI N. Biocompatible Graphene for Bioanalytical[M]. Springer, 2015. [4] GEOGARKILAS V, TIWARI J N, KEMP KC, PERMAN J A, BOURLINOS A B, KIM K S, ZBORIL R. Noncovalent functionalization of graphene and graphene oxide for energy materials, biosensing, catalytic, and diomedical applications[J]. Chem Rev, 2016, 116(9): 5464-5519. doi: 10.1021/acs.chemrev.5b00620 [5] KUILAA T, BOSEA S, MISHRAB A K, KHANRAA P, HOON K N, LEE J H. Chemical functionalization of graphene and its applications[J]. Prog Mater Sci, 2012, 57(7): 1061-1105. doi: 10.1016/j.pmatsci.2012.03.002 [6] GRAYFER E D, MAKOTCHENKO V G, NAZAROV A S, KIM S J, FEDOROV V E. Graphene: chemical approaches to the synthesis and modification[J]. Russ Chem Rev, 2011, 80(8): 751-770. doi: 10.1070/RC2011v080n08ABEH004181 [7] BOEHM H P. Surface oxides on carbon and their analysis: A critical assessment[J]. Carbon, 2002, 40(2): 145-149. doi: 10.1016/S0008-6223(01)00165-8 [8] BOTAS C, A'LVAREZ P, BLANCO C. The effect of the parent graphite on the structure of graphene oxide[J]. Carbon, 2012, 50(1): 275-282. doi: 10.1016/j.carbon.2011.08.045 [9] POTTS J R, DREYER D R, BIELAWSKI C W, RUOFF R S. Graphene-based polymer nanocomposites[J]. Polymer, 2011, 52(1): 5-25. doi: 10.1016/j.polymer.2010.11.042 [10] JOHNSON D W, DOBSON B P, COLEMAN K S. A manufacturing perspective on graphene dispersions[J]. Curr Opin Colloid In, 2015, 20(5/6): 367-382. http://www.sciencedirect.com/science/article/pii/S135902941500093X [11] REHBINDER P A, IZBRANNYE T. Poverhnostnye Yavleniya v Dispersnyh Sistemah[M]. Moscow: Nauka, 1978. [12] MORARU V, LEBOVKA N, SHEVCHENKO D. Structural transitions in aqueous suspensions of natural graphite[J]. Colloids and Surf A, 2004, 242(1/3): 181-187. http://www.sciencedirect.com/science/article/pii/S0927775704002985 [13] NAPPER D H. Polymeric Stabilization of Volloidal Dispersions[M]. Academic Press Incorp, 1983. [14] MATHEWS J P, CHAFFEE A L. The molecular representations of coal-A review[J]. Fuel, 2012, 96(1): 1-14. http://www.sciencedirect.com/science/article/pii/S0016236111007198 [15] FEDYAEVA O N, PATRAKOV Y F. The structure and properties of fragments of coal organic matter[J]. Solid Fuel Chem, 2004, 5: 21-27. https://www.researchgate.net/publication/288696793_The_structure_and_properties_of_fragments_of_coal_organic_matter [16] van KREVELEN D W. Coal: Typology, Chemistry, Physics, Constitution[M]. Elsevier, 1961. [17] RUDAKOV E S, SAPUNOV V A, KUCHERENKO V A. The mechanism of oxidation high level metamorphism coal with gas phase nitric acid[J]. Solid Fuel Chem, 1991, 2: 41-48. [18] FORSMAN W C, VOGEL F L, CARL D E, HOFFMAN J. Chemistry of graphite intercalation by nitric acid[J]. Carbon, 1978, 16(4): 269-271. doi: 10.1016/0008-6223(78)90040-4 -





 下载:
下载: