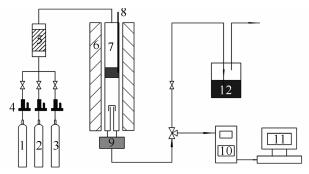
Catalytic performance of V2O5/Ti-Ce-PILC in the selective oxidation of H2S
-
摘要: 合成了TiO2-CeO2柱撑黏土负载V2O5催化剂,通过XRD、氮气吸附脱附、TG、FT-IR、H2-TPR、NH3-TPD、XPS等方法对其物理化学性质进行了表征,研究了该催化剂在H2S选择性催化氧化反应中的活性。结果表明,负载5%V2O5的TiO2-CeO2柱撑黏土在180℃下催化效果最好,且尾气中不含SO2。V2O5、TiO2和CeO2之间的相互作用提高了催化剂的活性,CeO2提高了催化剂的热稳定性,同时提供大量晶格氧,加强了V2O5的氧化还原作用,降低了反应温度;TiO2加强了VOx和CeOx的再氧化,降低了硫酸盐的覆盖率,从而降低了催化剂的失活速率。Abstract: A series of V2O5 doped Ti-Ce-pillared clays (V2O5/Ti-Ce-PILC) were prepared and characterized by XRD, nitrogen physisorption, FT-IR, XPS, H2-TPR, TG and NH3-TPD. The catalytic performance of V2O5/Ti-Ce-PILC in the selective oxidation of H2S was investigated in a fixed bed reactor. The results illustrated that 5%V2O5 doped Ti-Ce-pillared clays performs well at 180℃ in the selective oxidation of H2S, which can be ascribed to the interaction among V2O5, TiO2 and CeO2. CeO2 can enhance the catalyst thermostability and supply more lattice oxygen, which is of benefits to the redox of V2O5 and the decrease of reaction temperature. Meanwhile, the addition of TiO2 may promote the reoxidation of VOx and CeOx and reduce the sulfate coverage, which can then postpone the catalyst deactivation.
-
Key words:
- pillared clays /
- TiO2 /
- CeO2 /
- H2S /
- catalytic oxidation
-
表 1 催化剂的比表面积和孔结构
Table 1 BET specific surface areas total pore volume average pore diameter of various catalysts
Sample ABET/(m2·g-1) vp/(cm3·g-1) dp/nm Laponite 145 0.18 2.93 Ti-PILC 208 0.35 7.09 Ce-PILC 306 0.48 7.50 Ti-Ce-PILC 247 0.46 10.13 V2O5/Ti-PILC 172 0.25 5.19 V2O5/Ce-PILC 251 0.48 5.08 V2O5/Ti-Ce-PILC 179 0.27 6.77 表 2 样品的XPS分析
Table 2 XPS analysis data of various catalysts
Catalyst Relative content ratio Ce3+/Ce4+ V4+/V5+ lattice oxygen/adsorbed oxygen SO42-/Sn V2O5/Ti-Ce-PILC (fresh) 0.302 0.000 0.696 - V2O5/Ti-Ce-PILC (used) 0.325 0.674 0.631 1.223 V2O5/Ce-PILC (fresh) 0.262 0.000 0.508 - V2O5/Ce-PILC (used) 0.412 0.754 0.286 2.327 表 3 样品的XPS结合能
Table 3 XPS binding energy of various catalysts
Element Binding energy E/eV V2O5/Ti-Ce-PILC (fresh) V2O5/Ti-Ce-PILC (used) V2O5/Ce-PILC (fresh) V2O5/Ce-PILC (used) Ce4+ 3d5/2 883.2 882.2 882.7 882.7 Ce4+ 3d3/2 901.3 900.3 900.8 900.8 Ce4+ 3d5/2 898.9 897.8 898.5 898.4 Ce4+ 3d3/2 917.0 915.8 916.6 916.5 Ce4+ 3d5/2 888.1 888.1 887.9 889.0 Ce4+ 3d3/2 906.2 906.2 906.0 907.1 Ce3+ 3d5/2 886.0 885.4 885.8 886.0 Ce3+ 3d3/2 904.1 903.5 903.9 904.0 Ti4+ 2p5/2 458.2 458.7 - - Ti4+ 2p3/2 464.0 464.3 - - V5+ 2p3/2 517.8 518.0 518.1 518.2 V4+ 2p5/2 - 517.0 - 517.0 Adsorbed oxygen 531.4 531.8 531.2 531.5 Lattice oxygen 529.3 529.5 529.4 529.0 Sn - 162.9 - 163.2 SO42- - 169.1 - 168.5 -
[1] 陈赓良.克劳斯法硫磺回收工艺技术发展评述[J].天然气与石油, 2013, 31(4):23-28. http://www.cnki.com.cn/Article/CJFDTOTAL-TRYS201304010.htmCHEN Geng-liang. Review on development of claus process used for sulfur recovery[J]. Nat Gas Oil, 2013, 31(4):23-28. http://www.cnki.com.cn/Article/CJFDTOTAL-TRYS201304010.htm [2] JOHN S. Recovery of sulfur from sour acid gas:A review of the technology[J]. Environ Prog, 2002, 21(3):143-162. doi: 10.1002/(ISSN)1547-5921 [3] 郝郑平, 窦广玉, 张鑫, 曲思秋. H2S选择性催化氧化工艺及催化剂研究现状[J].环境科学, 2012, 33(8):2909-2916. http://www.cqvip.com/QK/91181X/201208/42830451.htmlHAO Zhi-ping, DOU Guang-yu, ZHANG Xin, QU Si-qiu. Current research situation of H2S selective catalytic oxidation technologies and catalysts[J]. Chin J Environ Sci, 2012, 33(8):2909-2916. http://www.cqvip.com/QK/91181X/201208/42830451.html [4] ZHANG X, DOU G Y, WANG Z, CHENG J, WANG H L, MA C Y, HAO Z P. Selective oxidation of H2S over V2O5 supported on CeO2-intercalated Laponite clay catalysts[J]. Cat Sci Technol, 2013, 3(10):2778-2785. doi: 10.1039/c3cy00431g [5] BINEESH K V, KIM D K, CHO H J, PARK D W. Synthesis of metal-oxide pillared montmorillonite clay for the selective catalytic oxidation of H2S[J]. J Ind Eng Chem, 2010, 16(4):593-597. doi: 10.1016/j.jiec.2010.03.014 [6] DAVYDOV A A, MARSHNEVA V I, SHEPOTKO M L. Metal oxides in hydrogen sulfide oxidation by oxygen and sulfur dioxide:I The comparison study of the catalytic activity. Mechanism of the interactions between H2S and SO2 on some oxides[J]. Appl Catal A:Gen, 2003, 244(1):93-100. doi: 10.1016/S0926-860X(02)00573-2 [7] BINEESH K V, KIM S Y, JERMY B R, PARK D W. Synthesis, characterization and catalytic performance of vanadia-doped delaminated zirconia-pillared montmorillonite clay for the selective catalytic oxidation of hydrogen sulfide[J]. J Mol Catal A:Chem, 2009, 308(1):150-158. https://www.researchgate.net/publication/229229769_Synthesis_characterization_and_catalytic_performance_of_vanadia-doped_delaminated_zirconia-pillared_montmorillonite_clay_for_the_selective_catalytic_oxidation_of_hydrogen_sulfide [8] LEÓN M, JIMÉNEZ-JIMÉNEZ J, JIMÉNEZ-LÓPEZ A, JIMÉNEZ-JIMÉNEZ J, LÓPEZ NIETO J M, RODRÍGUEZ-CASTELLÓN E. Vanadium oxide-porous phosphate heterostructure catalysts for the selective oxidation of H2S to sulphur[J]. Solid State Sci, 2010, 12(6):996-1001. doi: 10.1016/j.solidstatesciences.2009.08.009 [9] KALINKIN P, KOVALENKO O, LAPINA O, KHABIBULIN D, KUNDO N. Kinetic peculiarities in the low-temperature oxidation of H2S over vanadium catalysts[J]. J Mol Catal A:Chem, 2002, 178(1):173-180. [10] 郝郑平, 窦广玉, 张鑫, 张新艳, 曲思秋.负载型柱撑黏土催化材料在硫化氢选择氧化过程中的应用:中国, 102698595[P]. 2012-10-03.HAO Zhi-ping, DOU Guang-yu, ZHANG Xin, ZHANG Xin-yan, QU Si-qiu. Selective oxidation of H2S over supported pillared layered clays catalysts:CN, 102698595[P]. 2012-10-03. [11] ZHU H Y, ZHAO J C, LIU J W, YANG X Z, SHEN Y N. General synthesis of a mesoporous composite of metal oxide and silicate nanoparticles from a metal salt and laponite suspension for catalysis[J]. Chem Mater, 2006, 18(17):3993-4001. doi: 10.1021/cm060390+ [12] ZENG L, WANG S, PENG X, GENG J, CHEN C, LI M. Al-Fe PILC preparation, characterization and its potential adsorption capacity for aflatoxin B1[J]. Appl Clay Sci, 2013, 83:231-237. [13] 沈伯雄, 马宏卿, 杨晓燕, 姚燕. Mn-CeOx/Ti-PILC的制备、表征及脱硝性能研究[J].燃料化学学报, 2012, 40(5):615-620. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17952.shtmlSHEN Bo-xiong, MA Hong-qin, YANG Xiao-yan, YAO Yan. Study on preparation, characterization and de-NO activity of Mn-CeOx/Ti-PILC[J]. J Fuel Chem Technol, 2012, 40(5):615-620. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17952.shtml [14] CLARK P D, DOWLING N I, HUANG M. Role of Ti3+ in CS2 conversion over TiO2 claus catalyst[J]. Appl Catal A:Gen, 2015, 489:111-116. doi: 10.1016/j.apcata.2014.10.010 [15] YANG S X, ZHU W P, JIANG Z P, CHEN Z X, WANG J B. The surface properties and the activities in catalytic wet air oxidation over CeO2-TiO2 catalysts[J]. Appl Surf Sci, 2006, 252(24):8499-8505. doi: 10.1016/j.apsusc.2005.11.067 -





 下载:
下载: