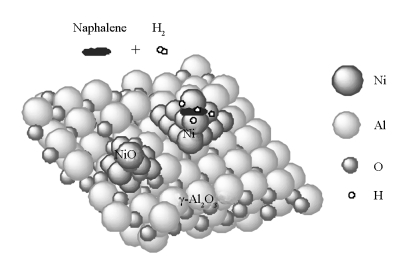
Effect of reaction conditions on the hydrogenation of naphthalene to decalin over Ni/Al2O3 catalyst
-
摘要: 采用Ni/Al2O3催化剂,在高压固定床反应器中考察了反应温度、压力、空速和氢油体积比比等因素对萘饱和加氢反应行为的影响,尤其是反应条件对反式十氢萘和顺式十氢萘选择性的影响。研究表明,反式十氢萘和顺式十氢萘的选择性与反应操作条件密切相关;反式十氢萘与顺式十氢萘的比例随着氢油比和温度的升高而增加,而随着压力和空速的增加而减小。在反应温度260-290℃、反应压力为5-7 MPa、空速为1-1.5 h-1及氢油体积比大于250时,十氢萘的选择性最高可达99%以上,萘的转化率接近100%,产物中反式和顺式十氢萘的比例最高,可达4.0左右。对Ni/γ-Al2O3催化剂稳定性进行了考察,初步发现催化活性组分的烧结或流失是催化剂失活和影响产物中反式十氢萘和顺式十氢萘比例的主要原因。
-
关键词:
- 反应条件 /
- 萘加氢 /
- Ni/Al2O3催化剂 /
- 十氢萘
Abstract: The effect of reaction conditions, including temperature, pressure, space velocity and hydrogen to oil ratio, on the hydrogenation of naphthalene to decalin over Ni/Al2O3 catalyst was investigated in a high pressure fixed bed reactor. The results indicate that the conversion of naphthalene and the selectivity to tran-decalin and cis-decalin are closely related to the reaction conditions. The ratio of tran-decalin to cis-decalin increases with an increase in the hydrogen to oil ratio and reaction temperature, but decreases with an increase in the liquid hourly space velocity (LHSV) and reaction pressure. Under a temperature 260-290℃, 5-7 MPa, a LHSV 1-1.5 h-1, and a hydrogen to oil ratio higher than 250, the conversion of naphthalene is 100% and the selectivity to decalin is close to 100%, with a tran-decalin to cis-decalin ratio of about 4.0. Meanwhile, it was found that the sintering and/or loss of active component are the main factors that cause the deactivation of Ni/Al2O3 catalyst in naphthalene hydrogenation and influence the ratio of tran-decalin to cis-decalin in the products.-
Key words:
- reaction conditions /
- naphthalene hydrogenation /
- Ni/Al2O3 /
- decalin
-
表 1 催化剂表征仪器表
Table 1 A list of instruments for the catalyst characterization
Name Model Manufacturer XRD Rigaku Max-2600 Japan Science Corp. SEM Quanta 400F Thermo Fisher Scientific Inc. BET Quadrasor SI Quantachrome Ins H2-TPR Chemisorb 2750 Micromeritics Instrument Corp. 表 2 Ni/γ-Al2O3催化剂反应200 h前后的比表面积和孔道结构参数
Table 2 Specific surface area and pore volumes of Ni/γ-Al2O3 catalysts before and after reaction for 200 h
Catalyst Surface area A/(m2·g-1) Pore volume v/(mL·g-1) Pore width d/nm Before 172.5 0.38 5.64 After 171.9 0.41 5.16 -
[1] 陈和平, 包存宽.我国化学工业中清洁生产技术的研究进展[J].化工进展, 2013, 32(6):1407-1414. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgjz201306039CHEN He-ping, BAO Cun-kuan. Progress of cleaner production technologies in chemical industry in China[J]. Chem Ind Eng Prog, 2013, 32(6):1407-1414. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgjz201306039 [2] 梁朝林, 谢颖, 黎广贞.绿色化工与绿色环保[M].北京:中国石化出版社, 2002. [3] 佟瑞利, 王永刚, 张旭, 张海永, 戴谨泽, 林雄超, 许德平.改性NiW/γ-Al2O3的低温焦油芳烃组分加氢性能研究[J].燃料化学学报, 2015, 43(12):1461-1469 doi: 10.3969/j.issn.0253-2409.2015.12.009TONG Rui-li, WANG Yong-gang, ZHANG Xu, ZHANG Hai-yong, DAI Jin-ze, LIN Xiong-chao, XU De-ping. Effect of phosphorus modification on the catalytic properties of NiW/γ-Al2O3 in the hydrogenation of aromatics from coal tar[J]. J Fuel Chem Technol, 2015, 43(12):1461-1469. doi: 10.3969/j.issn.0253-2409.2015.12.009 [4] 郑修新, 赵甲, 孙国方, 高鹏, 费亚南, 刘有鹏, 于海斌.萘加氢催化剂的研究进展[J].化工进展, 2015, 34(5):1295-1299. http://www.docin.com/p-1554935897.htmlZHENG Xiu-xin, ZHAO Jia, SUN Guo-fang, GAO Peng, FEI Ya-nan, LIU You-peng, YU Hai-bin. Research progress in catalysts for the hydrogenation of naphthalene[J]. Chem Ind Eng Prog, 2015, 34(5):1295-1299. http://www.docin.com/p-1554935897.html [5] HODOSHIMA S, ARAI H, TAKAIWA S, SAITO Y. Catalytic decalin dehydrogenation naphthalene hydrogenation pair as a hydrogen source for fuel-cell vehicle[J]. Inter J Hydrogen Energy, 2003, 28(11):1255-1262. doi: 10.1016/S0360-3199(02)00250-1 [6] PARK K, YIM D, IHM S. Characteristics of Al-MCM-41 supported Pt catalysts:Effect of Al distribution in Al-MCM-41 on its catalytic activity in naphthalene hydrogenation[J]. Catal Today, 2002, 74(3/4):281-290. http://www.sciencedirect.com/science/article/pii/S092058610200024X [7] 谭凤宜. 固定床法萘催化加氢合成十氢萘工艺研究[D]. 南京: 南京工业大学, 2006. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=D023631TAN Feng-yi. Synthesis of decalin by catalytic hydrogenation of naphthalene in fixed bed reactor[D]. Nanjing: Nanjing University of Technology, 2006. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=D023631 [8] 宋会. 工业萘两步法催化加氢制十氢萘的研究[D]. 大连: 大连理工大学, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10141-1015357580.htmSONG Hui. Two-step catalytic hydrogenation of industrial naphthalene to decalin[D]. Dalian: Dalian University of Technology, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10141-1015357580.htm [9] HIYOSHI N, OSADA M, RODE C V, SATO O, SHIRAI M. Hydrogenation of benzothiophene-free naphthalene over charcoal-supported metal catalysts in supercritical carbon dioxide solvent[J]. Appl Catal, 2007, 331:1-7. doi: 10.1016/j.apcata.2007.05.020 [10] WEITKAMP A W. Stereochemistry and mechanism of hydrogenation of naphthalenes on transition metal catalysts and comformational analysis of the products[J]. Adv Synth Catal, 1968, (18):1-110. https://www.researchgate.net/publication/279972721_Stereochemistry_and_Mechanism_of_Hydrogenation_of_Naphthalenes_on_Transition_Metal_Catalysts_and_Conformational_Analysis_of_the_Products [11] SCHMITZ A D, BOWERS G, SONG C. Shape-selective hydrogenation of naphthalene over zeolite-supported Pt and Pd catalysts[J]. Catal Today, 1996, 31(1/2):45-56. http://www.sciencedirect.com/science/article/pii/0920586196000338 [12] SAPRE A V, GATES B C. Hydrogenation of aromatic hydrocabrons catalyzed by sulfided cobalt monoxide-molybdenum trioxide/Y-aluminum oxide:Reactivities reaction newtokrs and kinetics[J]. J Am Chem Soc, 1979, 25(l):66-77. http://www.researchgate.net/publication/231294259_Hydrogenation_of_aromatic_hydrocarbons_catalyzed_by_sulfided_CoO-MoO3Al2O3._Reactivities_and_reaction_networks [13] RUATANEN P A, AITTAMAA J R, KRAUSE A O I. Liquid-phase hydrogenation of tetralin on Ni/Al2O3[J]. Chem Eng Sci, 2001, 56:1247-1254. doi: 10.1016/S0009-2509(00)00346-8 [14] RUATANEN P A, LYLYKANGAS M S, AITTAMAA J R. Liquid-phase hydrogenation of naphthalene and tertalin on Ni/Al2O3:Kinetic modeling[J]. Ind Eng Chem Res, 2002, 41(24):5966-5975. doi: 10.1021/ie020395q [15] RUATANEN P A, LYLYKANGAS I, KRAUSE A O. Liquid-phase hydrogenation kinetics of multicomponent aromatic mixtures on Ni/Al2O3[J]. Ind Eng Chem Res, 2002, 41:5632-5639. doi: 10.1021/ie0202930 [16] DOKJAMPA S, RIRKSOMBOON T, OSUWAN S, JONGPATIWUT S, RESASCO D E. Comparative study of the hydrogenation of tetralin on supported Ni Pt and Pd catalysts[J]. Catal Today, 2007, 123(4):218-223. http://www.sciencedirect.com/science/article/pii/S0920586107000090 [17] SCHUCKER R. Chemial equilibria in condensed-ring systems & isomerization equilibria of cis-and trans-decalin[J]. J Chem Eng Data, 1981, 26(3):239-241. doi: 10.1021/je00025a002 [18] COSTA P D, LEMBERTON J L, POTVIN C, MANOLI J M, PEROT G, BREYSSE M, DJEGA-MARIADASSOU G. Tetralin hydrogenation catalyzed by MO2C/Al2O3 and WC/Al2O3 in the presence of H2S[J]. Catal Today, 2001, 65:195-200. doi: 10.1016/S0920-5861(00)00593-9 [19] OYAMA S T. Novel catalysts for advanced hydroprocessing:Transition metal phosphides[J]. J Catal, 2003, 216(1/2):343-352. http://www.sciencedirect.com/science/article/pii/S0021951702000696 [20] ROBINSON W R A M, VAN GESTEL J N M, KORÁNYI T I, ROBINSON W R A M, VAN GESTEL J N M, KORÁNYI T I, EIJSBOUTS S, VAN der KRAAN A M, VAN VEEN J A R, DE BEER V H J. Phosphorus promotion of Ni(Co)-containing Mo-free catalysts in quinoline hydrogenation[J]. J Catal, 1996, 161(2):539-550. doi: 10.1006/jcat.1996.0216 [21] DUTTA R E, SCHOBART H H. Hydrogenation/dehydrogenation of polycyclic aromatic hydrocarbons using ammonium tetrathiomolybdate as catalyst precursor[J]. Catal Today, 1996, 31:65-77. doi: 10.1016/0920-5861(96)00084-3 [22] FRYE C Q, WEITKAMP A W. Equilibrium hydrogenation of multi-ring aromatics[J]. J Chem Eng Data, 1969, 14:372-376. doi: 10.1021/je60042a026 [23] 鞭学梅, 张毓萤, 胡志海, 王丽新, 李大东. Ni Mo加氢催化剂上1-甲基萘的饱和反应规律[J].石油学报(石油加工), 2012, 28(4):539-543. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=syxb-syjg201204002JU Xue-yan, ZHANG Yu-ying, HU Zhi-hai, WANG Li-xin, LI Da-dong. Hydrogenation saturation discipline of 1-methyl naphthalene over Ni-Mo catalyst[J]. Acta Pet Sin (Pet Process Sect), 2012, 28(4):538-543. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=syxb-syjg201204002 [24] MORI S, HANWAA M. Hydrogenation catalyst: Japan, 51121495[P]. 1976-10-23. [25] 曹袓宾, 邱建国.四氢萘加氢裂化反应网络的热力学分析[J].抚顺石油学院学报, 1992, 13(4):1-5. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=syhg199904007CAO Zu-bin, QIU Jian-guo. Analysis of the themdynatics of tetralin hydrocracking reaction network[J]. J Fushun Petrol Inst, 1992, 13(4):1-5. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=syhg199904007 [26] HUANG T C, KANG B C. Kinetic study of naphthalene hydrogenation over Pt/Al2O3 catalyst[J]. Ind Eng Chem Res, 1995, 34(4):1140-1148. doi: 10.1021/ie00043a016 [27] 米星, 杨索和, 何广湘, 罗国华, 徐新, 靳海波. Ni/γ-Al2O3催化剂制备条件的优化及对萘饱和加氢反应的影响[J].石油化工, 2017, 414-421. http://www.shiyouhuagong.com.cn/CN/abstract/abstract2471.shtmlMI Xing, YANG Suo-he, HE Guang-xiang, LUO Guo-hua, XU Xin, JIN Hai-bo. Effects of preparation conditions for Ni/γ-Al2O3 catalyst on saturated hydrogenation of naphthalene[J]. Petrochem Technol, 2017, 46(4):414-421. http://www.shiyouhuagong.com.cn/CN/abstract/abstract2471.shtml [28] LIN H P, WONG S T, MOU C Y. Extensive void defects in mesoporous aluminosilicate MCM-41[J]. J Phy Chem B, 2000, 104(38):8967-8975. doi: 10.1021/jp001569p [29] MARINO F, BARONETTI G, JOBBAGY M. Cu-Ni-K/γ-Al2O3 Supported catalysts reforming formation of hydrotalcite-type compounds as a result of metal-support interaction[J]. Appl Catal A:Gen, 2003, 238(1):41-54. doi: 10.1016/S0926-860X(02)00113-8 [30] OCAMPO F, LOUIS B, ROGER A C. Methanation of carbon dioxide over nickel-based Ce(0.72)Zr(0.28)O(2) mixed oxide catalysts prepared by sol-gel method[J]. Appl Catal A:Gen, 2009, 369(1/2):90-96. http://www.sciencedirect.com/science/article/pii/S0926860X09006279 [31] MARINO F, BARONETTI G, JOBBAGY M. Cu-Ni-K/γ-Al2O3 supported catalysts reforming formation of hydrotalcite-type compounds as a result of metal-support interaction[J]. Appl Catal A:Gen, 2003, 238(1):41-54. doi: 10.1016/S0926-860X(02)00113-8 -





 下载:
下载: