Molecular dynamics-quantum model simulation of pyrolysis reactivity of kerogen in oil shale from Fushun
-
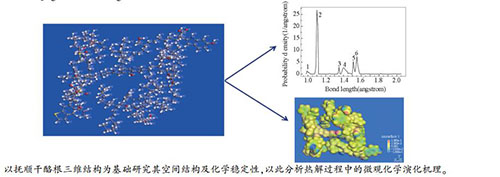
摘要: 采用MS(Materials Studio 2017)软件中Forcite模块,对自主构建的抚顺油页岩干酪根二维结构模型进行能量最小化分子动力学模拟,通过能量最优化过程得到干酪根初始优化结构。在此基础上进行分子动力学退火模拟,获得全局能量最优化构型,即油页岩干酪根分子三维结构模型。基于密度泛函理论的量子力学模拟方法,计算分析干酪根三维结构模型的动力学、键能、键级、电荷密度等参数,分析化学活性位点,探讨了干酪根热解微观化学演化机理,进而预测了反应性。Abstract: Using the Forcite module in MS (Materials Studio 2017) software, the energy minimization molecular dynamics simulation was performed on the self-constructed two-dimensional structural model of Fushun oil shale kerogen, and the initial optimized structure of kerogen was obtained through the energy optimization process. Then, molecular dynamics annealing simulations were performed to obtain a global energy optimization configuration, ie a three-dimensional structural model of oil shale kerogen molecules. Based on the density functional theory of quantum mechanics simulation method, a three-dimensional structural model of kerogen dynamics, bond energy, bond level, charge density and other parameters were calculated, and the chemical active sites were analyzed. The microchemical evolution mechanism of kerogen pyrolysis was discussed. And then, the reactivity was predicted.
-
Key words:
- oil shale /
- kerogen /
- molecular simulation /
- structural model
-
表 1 抚顺结构模型结构优化前后的势能变化
Table 1 Changes of potential energy before and after optimization of Fushun structural model
Structure Total energy/ (kcal·mol-1) Valence electron energy/(kcal·mol-1) Non-bond energy/(kcal·mol-1) bond energy angle energy torsion energy inversion energy van der Waals energy electrostatic energy hydrogen bond energy Initial structure 13441.84 2802.15 197.48 130.84 7.33 10286.74 17.31 0.00 Optimized structure 652.29 89.184 219.50 51.43 0.82 352.87 -56.27 -5.25 表 2 抚顺干酪根化学键键级
Table 2 Chemical bond level of Fushun kerogen
Chemical bond Bond order Chemical bond Bond order Chemical bond Bond order C118-S119 0.6185 C265-O266 0.9054 C150-C151 0.9140 S119-C120 0.6319 C74-C200 0.9058 C85-C172 0.9146 S177-C178 0.7123 C97-C196 0.9062 C169-C260 0.9146 C176-S177 0.7638 C75-C76 0.9063 C255-C256 0.9156 C263-O264 0.8726 C223-O270 0.9064 C209-C210 0.9159 C144-C145 0.8743 C82-C183 0.9066 C126-C127 0.9161 C267-O268 0.8774 C253-C254 0.9070 C87-C153 0.9163 C116-C117 0.8876 C175-C176 0.9091 C105-C242 0.9164 C164-C165 0.8923 C214-C215 0.9093 C131-C189 0.9166 C140-C260 0.8963 C113-C114 0.9107 C86-C87 0.9170 C82-C137 0.9027 C159-C160 0.9120 C144-C211 0.9172 C82-C161 0.9027 C144-C261 0.9127 C83-C188 0.9180 C131-C132 0.9030 C197-C198 0.9130 C148-C149 0.9186 C105-C106 0.9053 C164-C263 0.9137 C196-C250 0.9187 -
[1] 战金辉, 赖登国, 许光文.油页岩:固体石油[J].科学世界, 2016, (12):68-73. http://www.bookask.com/book/1793020.htmlZHAN Jin-hui, LAI Deng-guo, XU Guang-wen. Oil shale:Solid petroleum[J]. Sci World, 2016, (12):68-73. http://www.bookask.com/book/1793020.html [2] 朱煜凯, 张宇哲.国外油页岩资源的利用分析[J].化工设计通讯, 2016, 42(1):152-152. http://www.cnki.com.cn/Article/CJFDTOTAL-SYZW201403005.htmZHU Yu-kai, ZHANG Yu-zhe. Analysis of utilization of foreign oil shale resources[J]. Chem Des Commun, 2016, 42(1):152-152. http://www.cnki.com.cn/Article/CJFDTOTAL-SYZW201403005.htm [3] 王忠鑫, 赵丹丹.油页岩资源开发潜力评价研究与实践[J].露天采矿技术, 2016, 31(5):83-87. http://www.cqvip.com/QK/98598X/201605/668892401.htmlWANG Zhong-xin, ZHAO Dan-dan. Research and practice of oil shale resource development potential evaluation[J]. Ope Min Technol, 2016, 31(5):83-87. http://www.cqvip.com/QK/98598X/201605/668892401.html [4] 王擎, 许祥成, 迟铭书, 张宏喜, 崔达, 柏静儒.干酪根组成结构及其热解生油特性的红外光谱研究[J].燃料化学学报, 2015, 43(10):1158-1166. doi: 10.3969/j.issn.0253-2409.2015.10.002WANG Qing, XU Xiang-cheng, CHI Ming-shu, ZHANG Hong-xi, CUI Da, BAI Jing-ru. FT-IR study on composition of oil shale kerogen and its pyrolysis oil generation characteristics[J]. J Fuel Chem Technol, 2015, 43(10):1158-1166. doi: 10.3969/j.issn.0253-2409.2015.10.002 [5] 王越, 高燕, 白向飞, 武琳琳.桦甸油页岩有机岩相特征及其富集特性[J].燃料化学学报, 2016, 44(3):321-327. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18796.shtmlWANG Yu, GAO Yan, BAI Xiang-fei, WU Lin-lin. Characteristics and enrichment characteristics of organic lithofacies of huadian oil shale[J]. J Fuel Chem Technol, 2016, 44(3):321-327. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18796.shtml [6] KUMAR R, BANSAL V, BADHE R M, MADHIRA I S S, SUGUMARAN V, AHMED S, CHRISTOPHER J, PATEL M B, BASU B. Characterization of Indian origin oil shale using advanced analytical techniques[J]. Fuel, 2013, 113:610-616. doi: 10.1016/j.fuel.2013.05.055 [7] LⅡV S, KAASIK M. Trace metals in mosses in the estonian oil shale processing region[J]. J Atmos Chem, 2004, 49(1):563-578. [8] ZHOU B, SHI L, LIU Q, LIU Z. Examination of structural models and bonding characteristics of coals[J]. Fuel, 2016, 184:799-807. doi: 10.1016/j.fuel.2016.07.081 [9] RU X, CHENG Z Q, SONG L H, WANG H Y, LI J F. Experimental and computational studies on the average molecular structure of Chinese Huadian oil shale kerogen[J]. J Mol Struct, 2012, 1030(4):10-18. [10] 王擎, 黄宗越, 迟铭书, 石聚欣, 王智超, 隋义.油页岩干酪根化学结构特性分析[J].化工学报, 2015, 66(5):1861-1866. http://www.cqvip.com/QK/92960X/201304/46812833.htmlWANG Qing, HUANG Zong-yue, CHI Ming-shu, SHI Ju-yi, WANG Zhi-chao, SUI Yi. Analysis of chemical structure of oil shale kerogen[J]. CIESC J, 2015, 66(5):1861-1866. http://www.cqvip.com/QK/92960X/201304/46812833.html [11] GUAN X H, LIU Y, WANG D, WANG Q, CHI M S, LIU S, LIU C G. Three-dimensional structure of huadian oil shale kerogen model:An experimental and theoretical study[J]. Energy Fuels, 2015, 29(7):4122-4136. doi: 10.1021/ef502759q [12] FAULON J L, VANDENBROUCKE M, DRAPPIER J M, BEHAR F, ROMERO M. 3D chemical model for geological macromolecules[J]. Org Geochem, 1990, 16(4):981-993. [13] ORENDT A M, PIMIENTA S O, BADU S R, SOLUM M S, PUGMIRE R J, FACELLI J C. Three-dimensional structure of the siskin green river oil shale kerogen model:A comparison between calculated and observed properties[J]. Energy Fuels, 2013, 27(2):702-710. doi: 10.1021/ef3017046 [14] COLLELL J, UNGERER P, GALLIERO G, YIANNOURAKOU M, MONTEL F, PUJOL M. Molecular simulation of bulk organic matter in type Ⅱ shales in the middle of the oil formation window[J]. Energy Fuels, 2014, 28(12):7457-7466. doi: 10.1021/ef5021632 [15] COLLELL J, GALLIERO G, VERMOREL R, UNGERER P, YIANNOURAKOU M, MONTEL F, PUJOL M. Transport of multicomponent hydrocarbon mixtures in shales organic matter by molecular simulations[J]. J Phys Chem C, 2015, 119(39):22587-22595. doi: 10.1021/acs.jpcc.5b07242 [16] UNGERER P, COLLELL J, YIANNOURAKOU M. Molecular modeling of the volumetric and thermodynamic properties of kerogen:In fluence of organic type and maturity[J]. Energy Fuels, 2015, 29(1):91-105. doi: 10.1021/ef502154k [17] GAO Y, ZOU Y R, LIANG T, PENG P. Jump in the structure of Type Ⅰ kerogen revealed from pyrolysis and 13C DP MAS NMR[J]. Org Geochem, 2017, 112:105-118. doi: 10.1016/j.orggeochem.2017.07.004 [18] 王擎, 程枫, 潘朔.油页岩干酪根化学键浓度与能量密度研究[J].燃料化学学报, 2017, 45(10):1209-1218. doi: 10.3969/j.issn.0253-2409.2017.10.008WANG Qing, CHENG Feng, PAN Shuo. Study on chemical bond concentration and energy density of oil shale kerogen[J]. J Fuel Chem Techol, 2017, 45(10):1209-1218. doi: 10.3969/j.issn.0253-2409.2017.10.008 [19] ZHANG Z, JAMILI A. Modeling the Kerogen 3D Molecular Structure[C]//SPE/CSUR Unconventional Resources Conference. 2015. [20] 马延平. 柳林3#煤的超分子构建及分子模拟[D]. 太原: 太原理工大学, 2012.MA Yan-ping. The supramolecular construction and molecular simulation of Liulin 3# coal[D]. Taiyuan: Taiyuan University of Technology, 2012. [21] 张世良, 戚力, 高伟, 冯士东, 刘日平.分子模拟中常用的结构分析与表征方法综述[J].燕山大学学报, 2015, 39(3):213-220. http://staff.ustc.edu.cn/~zyli/download/ESTC.pdfZHANG Shi-liang, WEI Li, GAO Wei, FENG Shi-dong, LIU Ri-ping. A Summary of structural analysis and characterization methods commonly used in molecular simulation[J]. J Yanshan Univ, 2015, 39(3):213-220. http://staff.ustc.edu.cn/~zyli/download/ESTC.pdf [22] GUAN X H, WANG D, WANG Q, CHI M S, LIU C G. Estimation of various chemical bond dissociation enthalpies of large-sized kerogen molecules using DFT methods[J]. Mol Phys, 2016, 114(11):1705-1755. doi: 10.1080/00268976.2016.1143983 [23] WANG Q, LIU Q, WANG Z C, LIU H P, BAI J R, YE J B. Characterization of organic nitrogen and sulfur in the oil shale kerogens[J]. Fuel Process Technol, 2017, 160:170-177. doi: 10.1016/j.fuproc.2017.02.031 [24] 王擎, 张岩, 迟铭书.干酪根热解过程中的热解特性分析[J].石油学报(石油加工), 2017, 33(3):507-514. https://www.researchgate.net/profile/Ansong_Geng/publication/281350986_The_effects_of_minerals_and_water_on_hydrocarbon_generation_from_kerogen_III_Steranes_and_triterpane_generation_and_maturation/links/56b01d2d08ae9ea7c3adb2e9.pdf?origin=publication_detailWANG Qing, ZHANG Yan, CHI Ming-shu. Analysis of pyrolysis characteristics in pyrolysis of kerogen[J]. Acta Pet Sin(Pet Process Sect), 2017, 33(3):507-514. https://www.researchgate.net/profile/Ansong_Geng/publication/281350986_The_effects_of_minerals_and_water_on_hydrocarbon_generation_from_kerogen_III_Steranes_and_triterpane_generation_and_maturation/links/56b01d2d08ae9ea7c3adb2e9.pdf?origin=publication_detail [25] 李慧莉, 邱楠生, 金之钧, 何治亮, 朱映康.利用干酪根自由基浓度反演碳酸盐岩地层热历史[J].石油与天然气地质, 2005, 26(3):337-343. doi: 10.11743/ogg20050312LI Hui-li, QIU Nan-sheng, JIN Zhi-jun, HE Zhi-liang, ZHU Ying-kang. Inversion of thermal history of carbonate formation using kerogen radical concentration[J]. Oil Gas Geol, 2005, 26(3):337-343. doi: 10.11743/ogg20050312 -





 下载:
下载: