Effect of support properties on the performance of supported Pd catalysts in hydrodesulfurization
-
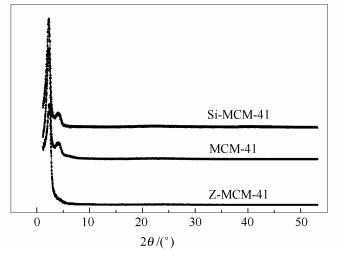
摘要: 以SiO2、全硅MCM-41(Si-MCM-41)、通过机械混合Si-MCM-41与ZSM-5得到的Z-MCM-41-M以及通过在ZSM-5外部包覆MCM-41制备得到的Z-MCM-41四种材料为载体,制备了四种负载型Pd催化剂。采用XRD、HRTEM、N2吸附-脱附、NH3-TPD手段对Pd催化剂进行了表征;以二苯并噻吩(DBT)为模型化合物,在固定床反应器上对四种催化剂的加氢脱硫(HDS)活性、加氢路径选择性和加氢裂化活性进行了考察,研究了不同类型载体对Pd催化剂加氢脱硫性能的影响。结果表明,载体的性质会显著影响负载型Pd催化剂的加氢脱硫性能。载体的比表面积对负载型Pd催化剂加氢脱硫活性影响不大,但是HYD路径的选择性与载体的孔道结构有关;具有介孔孔道结构有利于加氢路径选择性的提高。酸性载体负载的Pd催化剂表现出较好加氢脱硫活性和加氢选择性,这与氢溢流有关。介孔材料的孔道结构与微孔沸石的酸性有机结合,所得到的Z-MCM-41复合材料是是潜在的贵金属Pd加氢脱硫催化剂优良载体,可有效提升其加氢脱硫活性。Abstract: SiO2, siliceous MCM-41 (Si-MCM-41), physical mixture of Si-MCM-41 and ZSM-5 (Z-MCM-41-M), and Z-MCM-41 composite obtained by coating MCM-41 on ZSM-5 zeolite particles were prepared and characterized by XRD, HRTEM, N2 adsorption-desorption and NH3-TPD. The performances of the supported Pd catalysts in hydrodesulfurization (HDS) were evaluated in a fixed-bed reactor with dibenzothiophene (DBT) as the model sulfur-containing molecule; the effect of support properties on the performance of supported Pd catalysts in HDS was then investigated. The results indicated that the performance in HDS of supported Pd catalysts are significantly influenced by the support properties. Although the specific surface has a minor influence on the HDS activity of the supported Pd catalysts, the hydrogenation (HYD) selectivity is probably related to the pore structure of support; the mesoporous structure is beneficial to the improvement of HYD selectivity. The acid supports can give the Pd catalysts higher HDS activity, owing to the hydrogen spillover effect. The Z-MCM-41 composite is an excellent support for noble metal Pd catalyst in HDS, which can be ascribed to the synergy of the mesoporous structure of MCM-41 and acidic properties of ZSM-5.
-
Key words:
- MCM-41 /
- ZSM-5 /
- composite zeolite /
- Pd /
- hydrodesulfurization
-
表 1 载体的孔结构参数
Table 1 Textural properties of various supports

表 2 负载Pd催化剂的HDS转化率、HYD和DDS路径选择性、HYC转化率
Table 2 HDS conversion the selectivities to HYD and DDS pathways, and HYC conversion in DBT HDS on supported Pd catalysts

-
[1] SONG C S. An overview of new approaches to deep desulfurization for ultra-clean gasoline, diesel fuel and jet fuel[J]. Catal Today, 2003, 86: 211-263. doi: 10.1016/S0920-5861(03)00412-7 [2] SHYAMAL K B, MAITY S K, TURAGA U T. Search for an Efficient 4, 6-DMDBT Hydrodesulfurization Catalyst: A Review of Recent Studies[J]. Energ Fuel, 2004, 18(5): 1227-1237. doi: 10.1021/ef030179+ [3] KUNISADA N, CHOI K H, KORAI Y, MOCHIDA I, NAKANO K. Novel zeolite based support for NiMo sulfide in deep HDS of gas oil[J]. Appl Catal A, 2004, 269: 43-51. doi: 10.1016/j.apcata.2004.03.051 [4] NIQUILLE-RÖTHLISBERGER A, PRINS R. Hydrodesulfurization of 4, 6-dimethyldibenzothiophene and dibenzothiophene over alumina-supported Pt, Pd, and Pt-Pd catalysts[J]. J Catal, 2006, 242(1): 207-216. doi: 10.1016/j.jcat.2006.06.009 [5] SUN Y Y, PRINS R. Hydrodesulfurization of 4, 6-Dimethyldibenzothiophene over Noble Metals Supported on Mesoporous Zeolites[J]. Angew Chem Int Edit, 2008, 47: 8478-8481. doi: 10.1002/anie.v47:44 [6] STAKHEEV A Y, KUSTOV L M. Effects of the support on the morphology and electronic properties of supported metal cluster: modern concepts and progress in 1990s[J]. Appl Catal A, 1999, 188(1/2): 3-35. https://www.researchgate.net/publication/222238954_Effects_of_The_Support_on_The_Morphology_and_Electronic_Properties_of_Supported_Metal_Clusters_Modern_Concepts_and_Progress_in_1990s [7] 杨晓东. 介孔复合分子筛负载Pt、Pd催化剂的加氢脱硫性能[D]. 辽宁: 大连理工大学, 2009. http://cdmd.cnki.com.cn/Article/CDMD-10141-2009116304.htmYANG Xiao-dong. Hydrodesulfurization Performance of Pt or Pd Catalysts Supported on Mesoporous Molecule Sieves[D]. Liaoning: Dalian University of Technology, 2009. http://cdmd.cnki.com.cn/Article/CDMD-10141-2009116304.htm [8] WANG A J, RUAN L F, TENG Y, LI X, LU M H, REN J, WANG Y, HU Y K. Hydrodesulfurization of dibenzothiophene over siliceous MCM-41 supported nickel phosphide catalysts[J]. J Catal, 2005, 229(2): 314-321. doi: 10.1016/j.jcat.2004.09.022 [9] WANG A J, WANG Y, KABE T, CHEN Y Y, ISHHARA A, QIAN W H. Hydrodesulfurization of dibenzothiophene over siliceous MCM-41-supported catalysts I Sulfided Co-Mo catalysts[J]. J Catal, 2001, 199(1): 19-29. doi: 10.1006/jcat.2000.3148 [10] WANG A J, WANG Y, KABE T, CHEN Y Y, ISHHARA A, QIAN W H. Hydrodesulfurization of dibenzothiophene over siliceous MCM-41-supported catalysts Ⅱ Sulfided Ni-Mo catalysts[J]. J Catal, 2002, 210(2): 319-327. doi: 10.1006/jcat.2002.3674 [11] BAI X L, SACHTLER W M H. Methylcyclopentane conversion catalysis by zeolite encaged palladium clusters and palladium-proton adducts[J]. J Catal, 1991, 129(1): 121-129. doi: 10.1016/0021-9517(91)90016-W [12] SACHTLER W M H. Metal clusters in zeolites: an intriguing class of catalysts[J]. Acc Chem Res, 1993, 26(7): 383-387. doi: 10.1021/ar00031a005 [13] KLOETSTRA K R, ZANDBERGEN H W, JANSEN J C, BEKKUM H V. Overgrowth of mesoporous MCM-41 on faujasite[J]. Micro Mater, 1996, 6(5/6): 287-293. https://www.researchgate.net/publication/235693158_Overgrowth_of_Mesoporous_MCM-41_on_Faujasite [14] HARTMANN M. Hierarchical Zeolites: A Proven Strategy to Combine Shape Selectivity with Efficient Mass Transport[J]. Angew Chem Int Edit, 2004, 43(44): 5880-5582. doi: 10.1002/(ISSN)1521-3773 [15] FANG Y M, HU H Q. An ordered mesoporous aluminosilicate with completely crystalline zeolite wall structure[J]. J Am Chem Soc, 2006, 128(33): 10636-10637. doi: 10.1021/ja061182l [16] 王林英, 王安杰, 李翔, 周峰, 胡永康. ZSM-5/MCM-41介孔硅铝分子筛担载Pd和Pt制备加氢脱硫催化剂[J].石油学报(石油加工), 2012, 28(3): 380-387. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG201203008.htmWANG Lin-ying, WANG An-jie, LI Xiang, ZHOU Feng, HU Yong-kang. Preparation of Pd and Pt Hydrodesulfurization Catalysts Supported on ZSM-5/MCM-41[J]. Acta Petrolei Sinica, 2012, 28(3): 380-387. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG201203008.htm [17] WANG A J, KABE T. Fine-tuning of pore size of MCM-41 by adjusting the initial pH of the synthesis mixture[J]. Chem Comm, 1999, 2067-2068. https://www.mendeley.com/research-papers/finetuning-pore-size-mcm41-adjusting-initial-ph-syntheis-mixture/ [18] REN J, WANG A J, LI X, HU Y Y. Hydrodesulfurization of dibenzothiophene catalyzed by Ni-Mo sulfides supported on a mixture of MCM-41 and HY zeolite[J]. Appl Catal A, 2008, 344(1/2): 175-182. https://www.researchgate.net/publication/244108974_Hydrodesulfurization_of_dibenzothiophene_catalyzed_by_Ni-Mo_sulfides_supported_on_a_mixture_of_MCM-41_and_HY_zeolite [19] CORMA A, MARTÍNEZ A, MARTÍNEZ-SORIAV. Hydrogenation of Aromatics in Diesel Fuels on Pt/MCM-41 Catalysts[J]. J Catal, 1997, 169(2): 480-489. doi: 10.1006/jcat.1997.1737 [20] WANG J, HUANG L M, LI Q Z. Influence of different diluents in Pt/Al2O3 catalyst on the hydrogenation of benzene, toluene and o-xylene[J]. Appl Catal A, 1998, 175(1/2): 191-199. http://www.sciencedirect.com/science/article/pii/S0926860X98002166 [21] WANG J, LI Q Z, YAO J D. The effect of metal-acid balance in Pt-loading dealuminated Y zeolite catalysts on the hydrogenation of benzene[J]. Appl Catal A, 1999, 184(2): 181-188. doi: 10.1016/S0926-860X(98)00369-X [22] MENDES P S F, GREGÓRIO A F C, DAUDIN A, BOUCHY C, SILVA J M, RIBEIRO M F. Elucidation of the zeolite role on the hydrogenating activity of Pt-catalysts[J]. Catal Comm, 2017, 89: 152. doi: 10.1016/j.catcom.2016.11.006 -





 下载:
下载: