Calibration of solid state NMR carbon structural parameters and application in coal structure analysis
-
摘要: 为消除13C CP/MAS/TOSS NMR测试中碳核NOE效应,获得相对准确的碳结构参数,考察了不同模型化合物的碳核NOE效应强度。结果表明,不同模型化合物碳谱分峰拟合的测试值与样品碳结构参数的理论值之间存在明显误差,其中,脂肪碳在25%-125%、芳香碳为4%-50%,NOE效应在固体核磁碳谱测试中影响显著。为此,将模型化合物脂肪碳和芳香碳的实测值和理论值进行回归分析,得到非线性回归方程。用该方程对9,10-二甲基蒽进行碳结构修正,发现修正后脂肪碳实测值与理论值之间误差由不修正时的119.60%减小至7.84%,芳香碳误差为由不修正时的-17.10%到1.11%,修正后误差均在10%以内;同时用该回归方程修正了不同煤的碳结构参数,发现不同煤未修正的H/C原子比与其元素分析H/C原子比误差在45%-53%,修正后误差只有4%-13%,与元素分析结果具有一致性,表明非线性回归方程能够方便、准确地消除固体核磁NOE效应,为煤中碳结构分析提供新的技术支撑。Abstract: Carbon structure parameters were obtained accurately, if the NOE effect of carbon nucleus has been eliminated in13C CP/MAS/TOSS NMR test. This study has investigated different model compounds NOE effect intensity. And the results show that there is an obvious carbon structure error between the fitting test value and theoretical value in the different model compounds. The aliphatic carbon error is 25%-125%, and the aromatic carbon is 4%-50%, the NOE effect has great influence in solid state NMR. Therefore, the nonlinear regression equation was obtained by regression analysis of the measured and theoretical values of the model compound aliphatic and aromatic carbon. And this equation was used to calibrate the carbon structure of 9, 10-dimethylanthracene. It is found that after calibration the error between the measured value and the theoretical value of the modified aliphatic is reduced from 119.60% to 7.84%. The error of the aromatic carbon is reduced from -17.10% to 1.11%. And the error is within 10%. Then, the carbon structural parameters of different coal are calibrated by the regression equation. It is found that the H/C error of different coal is 45%-53% compared with elements analysis, and the calibration error is only 4%-13%, which is consistent with the results of elemental analysis. It indicated that the nonlinear regression equation could easily and precisely correct the solid state NMR carbon nuclear NOE effects and also provides new technical support for analysis of carbon structure in coal.
-
Key words:
- coal structure /
- model compounds /
- NOE /
- NMR /
- carbon structural parameters
1) 本文的英文电子版由Elsevier出版社在ScienceDirect上出版(http://www.sciencedirect.com/science/journal/18725813). -
表 1 不同煤种的工业分析与元素分析
Table 1 Proximate and ultimate analysis of different coals
Sample Proximate analyses w/% Ultimate analyses wdaf/% H/Ce O/Cf Mada Adb Vdafc FCdaf C H N S Od XLT 16.68 10.21 55.62 44.38 68.94 4.72 1.86 1.21 23.26 0.822 0.253 NMSL 9.58 14.41 46.48 53.52 72.99 4.65 1.16 1.40 19.80 0.764 0.203 SM 3.78 8.63 37.97 62.03 84.24 4.99 1.08 0.32 9.37 0.711 0.083 a: ad, air dry basis;b: d, dry basis;c: daf, dry ash-free basis;d: by difference;e: atomic ratio, H/C=(H%×12)/C%;f: atomic ratio, O/C= (O%×12)/(C%×16) 表 2 13C NMR中不同类型碳对应的化学位移
Table 2 Main characters for different structural carbons in solid-state13C NMR spectra
Assignment Location Chemical shift δ Characters Aliphatic methyl R-CH3 14-22 fal3 Aromatic methyl 
22-26 fala Methylene -CH2, 26-37 fal2 Quaternery sp3 C -CH--C 37-50 fal1, fal* Oxygen aliphatic carbon R-O-R, 50-95 falO Protonated aromatic carbon 
95-129 faH Aromatic bridgehead carbon 
129-137 faB Aliphatic substituted aromatic carbon 
137-149 faS Oxygen aromatic carbon 
149-164 faO Carboxyl RCOOH 164-190 faCC1 Quinone and carbonyl carbon RCOR 190-220 faCC2 abbreviations in table 2 are as follows: fal3: fraction of aliphatic methyl carbon; fala: fraction of aromatic methyl carbon; fal2: fraction of methylene carbon; fal1: fraction of methine carbon; fal*: fraction of carbon that is aliphatic and either quaternary, methyl, or mobile methylene; falO: fraction of total carbon associated with aliphatic ethers and alcohols; faH: protonated aromatic carbon; faB: bridgehead aromatic carbon; faS: alkyl substituted aromatic carbon; faO: oxygenated aromatic carbon; faCC1: fraction of carboxyl, faCC2: fraction of quinone and carbonyl carbons. 表 3 不同模型化合物不同类型有机碳分布
Table 3 Carbon structure distributions of the different model compounds
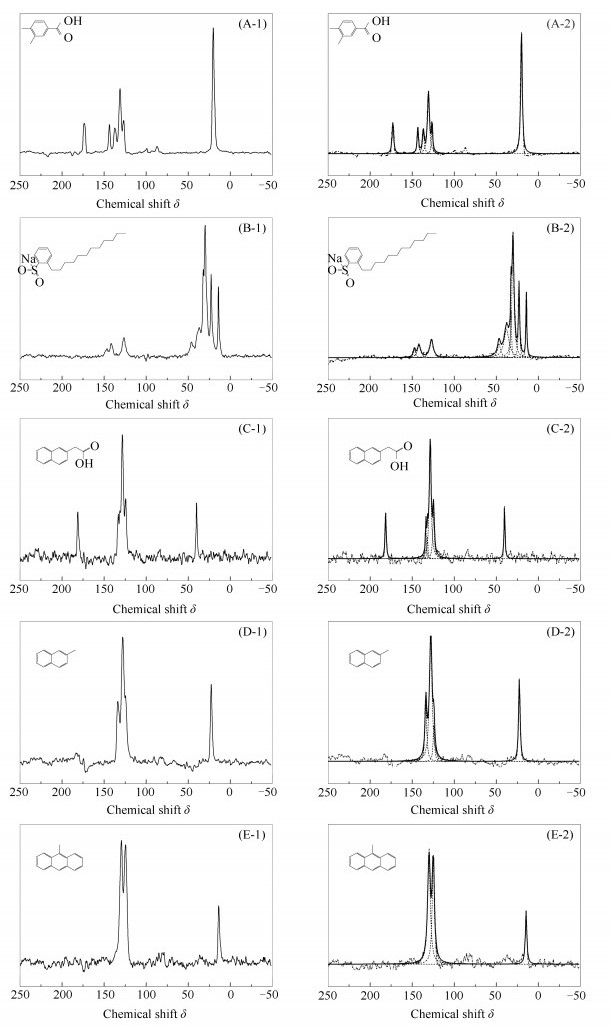
Sample Structure parameter distribution /% fal3 fala fal2 fal1, fal* falO fal faH faB+ faS+ faO faCC1+ faCC2 fa 3, 4-dimethyl benzoic acid 0.00 40.42 0.00 0.00 0.00 40.42 33.42 15.27 0.00 59.58 sodium dodecyl benzene sulfonic acid 7.71 0.00 75.40 0.00 0.00 83.11 8.89 8.00 0.00 16.89 2-naphthalene acetic acid 0.00 0.00 11.92 0.00 0.00 11.92 64.80 11.24 12.04 88.08 2-methyl naphthalene 0.00 20.42 0.00 0.00 0.00 20.42 62.34 27.28 0.00 79.58 9-methyl anthracene 0.00 13.60 0.00 0.00 0.00 13.60 37.83 48.57 0.00 86.40 表 4 不同模型化合物碳结构理论值与实测值比较
Table 4 Coal structure comparison of different models compound error theory and measured values
Sample Aliphatic(fal) Aromatic(fa) measured value theoretical value relativeerror /% measured value theoretical value relative error /% 3, 4-dimethyl benzoic acid 40.42 22.23 +81.80 59.58 77.77 -23.41 sodium dodecyl benzene sulfonic acid 83.11 66.67 +24.70 16.89 33.33 -49.30 2-naphthalene acetic acid 11.92 8.33 +43.10 88.08 91.67 -3.90 2-methyl naphthalene 20.42 9.09 +124.60 79.58 90.91 -12.50 9-methyl anthracene 13.60 6.67 +103.90 86.40 93.33 -7.40 表 5 9, 10-二甲基蒽不同类型有机碳分布
Table 5 Carbon structure distributions of the 9, 10-dimethyl anthracene
Sample Structure parameter distribution /% fal3 fala fal2 fal1, fal* falO fal faH faB+ faS+ faO faCC1+ faCC2 fa 9, 10-methyl anthracene 0.00 27.45 0.00 0.00 0.00 27.45 35.85 36.70 0.00 72.55 表 6 9, 10-二甲基蒽中碳结构实测值和理论值误差
Table 6 9, 10-dimethyl anthracene error theory and measured value
Sample Aliphatic(fal) Aromatic(fa) measured value theoretical value relative error /% measured value theoretical value relative error /% 9, 10-methyl anthracene 27.45 12.50 +119.60 72.55 87.50 -17.10 表 7 9, 10-二甲基蒽修正后实测值和理论值误差
Table 7 9, 10-dimethyl anthracene error theory and calibrated values
Sample Aliphatic(fal) Aromatic(fa) calibrated value theoretical value relative error /% calibrated value theoretical value relative error /% 9, 10-methyl anthracene 13.48 12.50 7.84 86.53 87.50 -1.11 表 8 修正后9, 10-二甲基蒽不同类型有机碳分布
Table 8 Carbon structure distributions of the 9, 10-dimethyl anthracene after calibration
Sample Structure parameter distribution /% fal3 fala fal2 fal1, fal* falO fal faH faB+ faS+ faO faCC1+ faCC2 fa 9, 10-methyl anthracene 0.00 13.48 0.00 0.00 0.00 13.48 42.76 43.77 0.00 86.53 表 9 不同煤种不同类型有机碳分布
Table 9 Carbon structure distributions of the different coals
Sample Structure parameter distribution /% fal3 fala fal2 fal1, fal* falO fal faH faB faS faO faCC1 faCC2 fa XLT 11.95 12.43 14.45 3.95 3.77 46.55 17.1 12.26 8.97 5.91 4.71 4.50 53.45 NMSL 4.93 15.61 11.53 2.78 6.58 41.43 23.80 10.48 7.57 8.24 5.76 2.72 58.57 SM 12.03 9.56 7.64 5.58 1.78 36.59 29.24 24.77 5.96 3.26 0.00 0.18 63.41 表 10 修正后不同煤种不同类型有机碳分布
Table 10 Calibration of carbon structure distributions of the different coals
Sample Structure parameter distribution /% fal3 fala fal2 fal1, fal* falO fal faH faB faS faO faCC1 faCC2 fa XLT 6.80 7.07 8.22 2.26 2.14 26.49 23.52 16.86 12.34 8.13 6.47 6.19 73.51 NMSL 2.70 8.53 6.29 1.57 3.37 22.46 31.51 13.87 10.02 10.91 7.62 3.61 77.54 SM 6.25 4.97 3.97 2.90 0.93 19.02 37.34 31.63 7.61 4.16 0.00 0.24 80.98 表 11 不同煤种元素分析H/C原子比和NMR修正前后H/C原子比
Table 11 Different types of H/C before and after calibrated in ultimate analyses and NMR
Sample Ultimate analyses H/C NMR H/C Error of ultimate analyses and NMR H/C /% NMR calibrated H/C Error of ultimate analyses and NMR calibrated H/C /% XLT 0.822 1.196 45.46 0.856 4.15 NMSL 0.764 1.166 52.56 0.862 12.95 SM 0.711 1.061 49.19 0.772 8.69 表 12 不同煤种修正前后的芳香度
Table 12 Different types of aromaticity before and after calibration
Sample Tested aromaticity Calibrated aromaticity XLT 0.535 0.735 NMSL 0.586 0.775 SM 0.634 0.810 -
[1] 虞继舜.煤化学[M].北京:冶金工业出版社, 2000.YU Ji-shun. Coal Chemistry[M]. Beijing:Metallurgical Industry Press, 2000. [2] 张蓬洲, 李丽云, 叶朝辉.用固体高分辨核磁共振研究煤结构[J].燃料化学学报, 1993, 21(3):310-316. http://www.cnki.com.cn/Article/CJFDTOTAL-RLHX199303012.htmZHANG Peng-zhou, LI Li-yun, YE Chao-hui. Study of structural feature of coal by solid state13C NMR spectroscopy. J Fuel Chem Technol, 1993, 21(3):310-316. http://www.cnki.com.cn/Article/CJFDTOTAL-RLHX199303012.htm [3] 刘传福, 孙润仓, 叶君.固体核磁CP/MAS13C NMR在植物纤维原料研究中的应用[J].中国造纸学报, 2005, 20(2):184-188.LIU Chuan-fu, SUN Run-cang, YE Jun. Application of CP/MAS13C-NMR Spectroscopy in the Study of Lignocellulosic Materials[J]. Trans China Pulp Pap, 2005, 20(2):184-188. [4] 马志茹, 张蓬洲, 李丽云.核磁共振技术在煤化学研究中的应用[J].煤炭转化, 1996, 19(1):33-39. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH601.003.htmMA Zhi-ru, ZHANG Peng-zhou, LI Li-yun. The progress of NMR techniques applied to coal chemistry[J]. Coal Convers, 1996, 19(1):33-39. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH601.003.htm [5] MICHAEL A W, RONALD J P. New solid state NMR techniques in coal analysis[J]. TrAC Trends Anal Chem, 1984, 3(6):144-147. doi: 10.1016/0165-9936(84)88007-3 [6] MARK S S, RONALD J P, DAVID M G.13C Solid-state NMR of Argonne Premium Coals[J]. Energy Fuels, 1989, 3(2):187-193. doi: 10.1021/ef00014a012 [7] 李振广.干酪根结构中碳分布特征及其与生烃潜力的关系-13C NMR C P/MAS与DD技术的应用[J].地球化学, 1995, 24(2):101-109.LI Zheng-guang. Carbon distribution of kerogen and it's relationship to hydrocarbon generation potential-application of13C NMR CP/MAS and DD techniques[J]. Geochimica, 1995, 24(2):101-109. [8] 陈明秀.用固体交叉极化魔角自旋(CP/MAS)和偶极去相核磁共振光谱对煤的研究[J].煤炭分析及利用, 1989, 4(3):1-7. http://www.cnki.com.cn/Article/CJFDTOTAL-MTFX198903000.htmCHEN Ming-xiu. The application of solid cross polarization magic angle spinning(CP/MAS) and dipole to nuclear magnetic resonance(NMR) spectroscopy to study coal[J]. Coal Anal Util, 1989, 4(3):1-7. http://www.cnki.com.cn/Article/CJFDTOTAL-MTFX198903000.htm [9] XIAO Y C, MARK A C, MARIA M, MAO J. Chemical structure changes in kerogen from bituminous coal in response to dike intrusions as investigated by advanced solid-state13C NMR spectroscopy[J]. Int J Coal Geol, 2013, 108:53-64. doi: 10.1016/j.coal.2012.05.002 [10] MAO J, CAO X, NAN C O, KLAUS S. Advanced solid-state NMR spectroscopy of natural organic matter[J]. Prog Nucl Magn Reson Spectrosc, 2017, 100:17-22. doi: 10.1016/j.pnmrs.2016.11.003 [11] STACEY M A, KANMI M, GORDON J K, MAREK P. Solid-state NMR studies of fossil fuels using one-and two-dimensional methods at high magnetic field[J]. Energy Fuels, 2012, 26(7):4405-4412. doi: 10.1021/ef3004637 [12] CHENG H N, LYNDA H W, KLASSON K T, JOHN C E. Solid-state NMR and ESR studies of activated carbons produced from pecan shells[J]. Carbon, 2010, 48(9):2455-2469. doi: 10.1016/j.carbon.2010.03.016 [13] MICHAEL A W, ANTHONY M V. Developments in high-solid-state13C NMR spectroscopy of coals[J]. Org Geochem, 1985, 8(5):299-312. doi: 10.1016/0146-6380(85)90009-9 [14] JAMES A F, ROBERTO G, JOHN C L. Single-pulse excitation13C NMR measurements on the argonne premium coal samples[J]. Energy Fuels, 1992, 6(5):598-602. doi: 10.1021/ef00035a009 [15] ROBERT V L, DAVID C S, COLIN E S. Quantitative solid state13C NMR studies of highly cross-linked poly(divinylbenzene) resins[J]. Macromolecules, 1997, 30(10):2868-2875. doi: 10.1021/ma9616470 [16] ZHANG R, KAMAL H M. Hybridizing cross-polarization with NOE or refocused-INEPT enhances the sensitivity of MAS NMR spectroscopy[J]. J Magn Reson, 2016, 266:59-66. doi: 10.1016/j.jmr.2016.03.006 [17] KOH K, SATORU M, MASAKASTSU N. Studies on the chemical structural change during carbonization process[J]. Energy Fuels, 1996, 10(3):672-678. doi: 10.1021/ef9501096 [18] 王立英, 冯继文, 叶朝辉.典型高分子材料的固体核磁共振研究[J].波谱学杂志, 2006, 23(4):547-549. http://cdmd.cnki.com.cn/Article/CDMD-80020-2006155703.htmWANG Li-ying, FENG Ji-wen, YE Chao-hui. Typical polymers studied by solid-state NMR[J]. Chin J Magn Reson, 2006, 23(4):547-549. http://cdmd.cnki.com.cn/Article/CDMD-80020-2006155703.htm [19] 徐大江, 舒婕.高分辨固体核磁共振技术在材料化学研究中的应用[J].分析仪器, 2015, 1:25-34. doi: 10.3969/j.issn.1001-232x.2015.01.005XU Da-jiang, SHU Jie. Application of high-resolution solid-state nuclear magnetic resonance techniques in chemical materials. Anal Instrum, 2015, 1:25-34. doi: 10.3969/j.issn.1001-232x.2015.01.005 [20] 潘铁英, 张玉兰, 苏克曼.波谱解析法[M].上海:华东理工大学出版社. 2009.PAN Tie-ying, ZHANG Yu-lan, SU Ke-man. Spectrum Analytical Method[M]. Shanghai:East China University of Science and Technology Press, 2009. [21] 相建华, 曾凡桂, 李彬.成庄无烟煤大分子结构模型及其分子模拟[J].燃料化学学报, 2013, 41(4):391-399. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18157.shtmlXIANG Jian-hua, ZENG Fan-gui, LI Bin. Construction of macromolecular structural model of anthracite from Chengzhuang coal mine and its molecular simulation[J]. J Fuel Chem Technol, 2013, 41(4):391-399. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18157.shtml [22] 相建华, 曾凡桂, 梁虎珍.兖州煤大分子结构模型构建及其分子模拟[J].燃料化学学报, 2011, 39(7):481-488. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17764.shtmlXIANG Jian-hua, ZENG Fan-gui, LIANG Hu-zhen. Model construction of the macro molecular structure of Yanzhou coal and its molecular simulation[J]. J Fuel Chem Technol, 2011, 39(7):481-488. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17764.shtml [23] ANDREAS G. Aspects of solid state13C CPMAS NMR spectroscopy in coals from the Balkan peninsula[J]. J Serb Chem Soc, 2003, 68(8/9):599-605. [24] 蔺华林, 李克健, 章序文.上湾煤及其惰质组富集物的结构表征与模型构建[J].燃料化学学报, 2013, 41(6):641-648. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18190.shtmlLIN Hua-lin, LI Ke-jian, ZHANG Xu-wen. Structure characterization and model construction of Shangwan coal and it's inertinite concentrated[J]. J Fuel Chem Technol, 2013, 41(6):641-648. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18190.shtml [25] WEI Z, GAO X, ZHANG D, DA J. Assessment of thermal evolution of kerogen geopolymers with their structural parameters measured by solid-state13C NMR spectroscopy[J]. Energy Fuels, 2005, 19(1):240-250. doi: 10.1021/ef0498566 [26] LIU P, WANG L L, ZHOU Y, PAN T Y, LU X L, ZHANG D X. Effect of hydrothermal treatment on the structure and pyrolysis product distribution of Xiaolongtan lignite[J]. Fuel, 2016, 164:110-118. doi: 10.1016/j.fuel.2015.09.092 [27] 刘鹏, 王岚岚, 张德祥.先锋褐煤在水热处理过程中的结构演绎[J].燃料化学学报, 2016, 44(2):129-137. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18771.shtmlLIU Peng, WANG Lan-lan, ZHANG De-xiang. Structural evolution of Xianfe ng lignite during hydrothermal treatment[J]. J Fuel Chem Technol, 2016, 44(2):129-137. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18771.shtml [28] YAN J, BAI Z, BAI J, GUO Z, LI W. Effects of organic solvent treatment on the chemical structure and pyrolysis reactivity of brown coal[J]. Fuel, 2014, 128(4):39-45. [29] 张娉, 潘铁英, 张德祥.煤直接液化中油浆热溶产物的13C NMR研究[J].波谱学杂志, 2006, 23(1):41-47.ZHANG Ping, PAN Tie-ying, Zhang De-xiang.Thermally dissolved products of coal-oil slurry during direct coal liquefaction studied by NMR spectroscopy[J]. Chin J Magn Reson, 2006, 23(1):41-47. [30] LOVE G D, LAW R V, SNAPE C E. Determination of Nonprotonated Aromatic Carbon Concentrations in Coals by Single Pulse Excitation13C NMR[J]. Energy Fuels, 1993, 7(5):639-644. doi: 10.1021/ef00041a012 [31] MERCEDS M V, JOHN M AN, JOSE D R, COLIN E S. Quantitative solid-state13C NMR. measurements on cokes, chars and coal tar pitch fractions[J]. Fuel, 1996, 75(15):1721-1726. doi: 10.1016/S0016-2361(96)00151-2 [32] ROBERT A MEYERS. Coal Structure[M]. New York:Academic press. 1982. [33] FIDEL C M, VLADISLAV V L, JONATHAN P M. A molecular model for Illinois No. 6 Argonne Premium coal:Moving toward capturing the continuum structure[J]. Fuel, 2012, 95(1):35-49. [34] HIROYUKI K, YASUMASA Y, IKUO S. Studies on structural changes of coal density-separated components during pyrolysis by means of solid-state13C NMR spectra[J]. J Anal Appl Pyrolysis, 2000, 53(1):35-50. doi: 10.1016/S0165-2370(99)00058-3 -





 下载:
下载: