Effects of low-temperature hydrothermal pretreatment of high-protein Chlorella sp.on N distribution and thermal degradation of solid residue
-
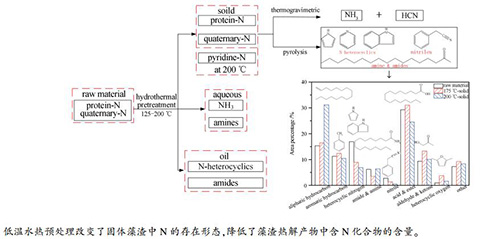
摘要: 利用水热釜研究了低温水热预处理过程中高蛋白小球藻的水热降解特性,考察了125-200℃各相产物产率、元素组成、能量回收率和关键元素C、N的分布规律。结果表明,大量的C、N元素富集在水相中,且提高温度(>175℃)有利于脱氨反应进而提高水相中NH3的相对含量。随着预处理温度升高,N在油相产物中分布逐渐增加且在175℃后快速增加;固相产率及能量回收率逐渐降低,但同时其N/C和O/C也降低,表明水热预处理是一个提质过程。借助FT-IR、XPS、TG-FTIR-MS和Py-GC-MS对比分析了原料和藻渣的官能团结构和热降解特性。结果表明,水热预处理过程改变了C、N不同官能团的相对含量,与原料相比,藻渣中C-C相对含量增加,而C-N、C-O等相对含量降低;除了蛋白质-N和季胺类-N,预处理后藻渣中出现了少量吡啶-N。固体藻渣官能团组分含量和结构变化降低了热失重过程NH3和HCN的释放、及快速热解产物中含N杂环化合物的含量。Abstract: Decomposition behavior of Chlorella sp. during low-temperature hydrothermal pretreatment (HTP) were studied. Distribution of various product yields, element components, energy recovery ratio and key elements (i.e., C and N) along with temperature (125-200℃) were investigated. The results show that amounts of C and N are enriched into aqueous phase, and high content of NH3-N is detected due to deamination reaction above 175℃. During the HTP process, N distribution in oil product first increases gradually and then rapidly increases above 175℃. During the whole low-temperature HTP process, the yield and energy recovery ratio of solid residue decreases continuously. The N/C and O/C ratio of the solid residue also decreases, indicating HPT would promote property of the solid residue. The functional structure and thermal-degradation of Chlorella sp. and its solid residue are then comparatively examined by various techniques including FT-IR, XPS, TG-FTIR-MS and Py-GC/MS. The results show that functional structure of the solid residue is distinguished from that of the raw sample. The relative content of C-C bond increases while that of C-N and C-O bonds decreases. In addition to protein-N and quaternary-N, a low fraction of pyridine-N is also detected in the solid residue. Compared with these from raw material, less NH3 and HCN are released from solid residue, and less N-containing heterocyclic compounds are generated during rapid pyrolysis.
-
表 1 小球藻的工业分析、组成和元素分析
Table 1 Proximate, component and ultimate analyses of Chlorella (dry basis)
Proximate analysis w/% Component w/% Ultimate analysis w/% Atomic ratio QHHV/(MJ·kg-1) V FC* A protein lipids carbohydrates* C H O* N S H/C N/C O/C 80.80 12.06 7.14 42.77 8.01 42.08 52.45 7.12 23.84 8.57 0.88 1.63 0.14 0.34 23.62 *by difference 表 2 不同预处理温度下固体藻渣和油相产物的元素分析、热值和能量回收率
Table 2 Ultimate analysis, QHHV and energy recovery of oil and solids
Operating condition Ultimate analysis w/% Ash w/% Atomic ratio QHHV /(MJ·kg-1) Energy recovery η/% N C H S O* H/C N/C O/C Temp. t /℃ solid obtained at holding time of 30 min 125 8.48 50.41 6.90 1.04 28.88 4.29 1.642 0.144 0.430 22.63 69.49 137.5 8.20 50.57 7.12 0.86 29.24 4.01 1.689 0.139 0.434 22.90 61.94 150 7.52 50.93 7.26 0.79 29.38 4.12 1.710 0.127 0.433 23.17 55.14 162.5 6.49 51.87 7.30 0.68 29.41 4.25 1.690 0.107 0.425 23.55 40.78 175 6.07 53.45 7.52 0.64 28.11 4.22 1.689 0.097 0.394 24.50 33.68 187.5 5.70 56.96 7.66 0.50 24.77 4.41 1.614 0.086 0.326 26.22 25.20 200 5.34 60.22 7.69 0.31 20.82 5.61 1.533 0.076 0.259 27.77 21.48 Temp. t /℃ oil obtained at holding time of 30 min 125 1.60 64.39 9.33 0.90 23.78 - 1.739 0.021 0.277 31.08 6.44 137.5 1.91 66.41 9.90 0.56 21.22 - 1.788 0.025 0.240 32.68 5.80 150 1.98 67.20 9.68 0.42 20.72 - 1.728 0.025 0.231 32.73 8.17 162.5 3.42 66.89 9.78 0.42 19.49 - 1.754 0.044 0.219 32.85 13.48 175 3.46 68.18 9.96 0.40 18.00 - 1.752 0.043 0.198 33.66 12.96 187.5 5.76 66.37 9.16 0.19 18.52 - 1.656 0.074 0.209 31.98 13.54 200 6.59 65.15 8.88 0.51 18.88 - 1.635 0.087 0.217 31.21 30.90 *: by difference 表 3 C和N的XPS相对峰面积
Table 3 Relative peak area of C and N by XPS analysis
Peak Relative peak area A/% C1 C2 C3 C4 C5 N1 N2 N3 Raw material 39.82 23.92 21.66 9.47 5.13 96.17 3.83 175 ℃-solid 58.37 18.22 13.58 6.97 2.87 90.78 3.28 5.94 200 ℃-solid 70.41 15.81 6.75 5.50 1.54 88.80 5.30 5.90 -
[1] ALBA L G, TORRI C, SAMORI C, VAN DER SPEK J, FABBRI D, KERSTEN S R A, BRILMAN D W F. Hydrothermal treatment (HTT) of microalgae:Evaluation of the process as conversion method in an algae biorefinery concept[J]. Energy Fuels, 2012, 26(1):642-657. doi: 10.1021/ef201415s [2] SABER M, NAKHSHINIEV B, YOSHIKAWA K. A review of production and upgrading of algal bio-oil[J]. Renewable Sustainble Energy Rev, 2016, 58:918-930. doi: 10.1016/j.rser.2015.12.342 [3] AKHTAR J, AMIN N A S. A review on process conditions for optimum bio-oil yield in hydrothermal liquefaction of biomass[J]. Renewable Sustainble Energy Rev, 2011, 15(3):1615-1624. doi: 10.1016/j.rser.2010.11.054 [4] DIMITRIADIS A, BEZERGIANNI S. Hydrothermal liquefaction of various biomass and waste feedstocks for biocrude production:A state of the art review[J]. Renewable Sustainable Energy Rev, 2017, 68:113-125. doi: 10.1016/j.rser.2016.09.120 [5] XU Y P, DUAN P G, WANG F. Hydrothermal processing of macroalgae for producing crude bio-oil[J]. Fuel Process Technol, 2015, 130:268-274. doi: 10.1016/j.fuproc.2014.10.028 [6] SINGH R, BHASKAR T, BALAGURUMURTHY B. Effect of solvent on the hydrothermal liquefaction of macro algae Ulva fasciata[J]. Process Saf Environ Protect, 2015, 93:154-160. doi: 10.1016/j.psep.2014.03.002 [7] GUO Q J, WU M, WANG K, ZHANG L, XU X F. Catalytic hydrodeoxygenation of algae bio-oil over bimetallic Ni-Cu/ZrO2 catalysts[J]. Ind Eng Chem Res, 2015, 54(3):890-899. doi: 10.1021/ie5042935 [8] DUAN P, WANG B, XU Y. Catalytic hydrothermal upgrading of crude bio-oils produced from different thermo-chemical conversion routes of microalgae[J]. Bioresour Technol, 2015, 186:58-66. doi: 10.1016/j.biortech.2015.03.050 [9] SINGH R, BALAGURUMURTHY B, BHASKAR T. Hydrothermal liquefaction of macro algae:Effect of feedstock composition[J]. Fuel, 2015, 146:69-74. doi: 10.1016/j.fuel.2015.01.018 [10] BILLER P, ROSS A B. Potential yields and properties of oil from the hydrothermal liquefaction of microalgae with different biochemical content[J]. Bioresour Technol, 2011, 102(1):215-225. doi: 10.1016/j.biortech.2010.06.028 [11] JAZRAWI C, BILLER P, ROSS A B, MONTOYA A, MASCHMEYER T, HAYNES B S. Pilot plant testing of continuous hydrothermal liquefaction of microalgae[J]. Algal Res, 2013, 2(3):268-277. doi: 10.1016/j.algal.2013.04.006 [12] COLE A, DINBURG Y, HAYNES B S, HE Y, HERSKOWITZ M, JAZRAWI C, LANDAU M, LIANG X, MAGNUSSON M, MASCHMEYER T, MASTERS A F, MEIRI N, NEVEUX N, DE NYS R, PAUL N, RABAEV M, VIDRUK-NEHEMYA R, YUEN A K L. From macroalgae to liquid fuel via waste-water remediation, hydrothermal upgrading, carbon dioxide hydrogenation and hydrotreating[J]. Energy Environ Sci, 2016, 9(5):1828-1840. doi: 10.1039/C6EE00414H [13] CHIARAMONTI D, PRUSSI M, BUFFI M, RIZZO A M, PARI L. Review and experimental study on pyrolysis and hydrothermal liquefaction of microalgae for biofuel production[J]. Appl Energy, 2017, 185:963-972. doi: 10.1016/j.apenergy.2015.12.001 [14] GUO Y, YEH T, SONG W, XU D, WANG S. A review of bio-oil production from hydrothermal liquefaction of algae[J]. Renewable Sustainable Energy Rev, 2015, 48:776-790. doi: 10.1016/j.rser.2015.04.049 [15] ARVINDNARAYAN S, SIVAGNANA PRABHU K K, SHOBANA S, KUMAR G, DHARMARAJA J. Upgrading of micro algal derived bio-fuels in thermochemical liquefaction path and its perspectives:A review[J]. Int Biodeterior Biodegrad, 2017, 119:260-272. doi: 10.1016/j.ibiod.2016.08.011 [16] TERI G, LUO L G, SAVAGE P E. Hydrothermal treatment of protein, polysaccharide, and lipids alone and in mixtures[J]. Energy Fuels, 2014, 28(12):7501-7509. doi: 10.1021/ef501760d [17] JAZRAWI C, BILLER P, HE Y Y, MONTOYA A, ROSS A B, MASCHMEYER T, HAYNES B S. Two-stage hydrothermal liquefaction of a high-protein microalga[J]. Algal Res, 2015, 8:15-22. doi: 10.1016/j.algal.2014.12.010 [18] DU Z, MOHR M, MA X, CHENG Y, LIN X, LIU Y, ZHOU W, CHEN P, RUAN R. Hydrothermal pretreatment of microalgae for production of pyrolytic bio-oil with a low nitrogen content[J]. Bioresour Technol, 2012, 120:13-18. doi: 10.1016/j.biortech.2012.06.007 [19] HUANG Z, WUFUER A, WANG Y, DAI L. Hydrothermal liquefaction of pretreated low-lipid microalgae for the production of bio-oil with low heteroatom content[J]. Process Biochem, 2018, 69:136-143. doi: 10.1016/j.procbio.2018.03.018 [20] GOLLAKOTA A R K, KISHORE N, GU S. A review on hydrothermal liquefaction of biomass[J]. Renewable Sustainable Energy Rev, 2018, 81:1378-1392. doi: 10.1016/j.rser.2017.05.178 [21] XU D H, SAVAGE P E. Effect of reaction time and algae loading on water-soluble and insoluble biocrude fractions from hydrothermal liquefaction of algae[J]. Algal Res, 2015, 12:60-67. doi: 10.1016/j.algal.2015.08.005 [22] MIAO C, CHAKRABORTY M, CHEN S. Impact of reaction conditions on the simultaneous production of polysaccharides and bio-oil from heterotrophically grown Chlorella sorokiniana by a unique sequential hydrothermal liquefaction process[J]. Bioresour Technol, 2012, 110:617-627. doi: 10.1016/j.biortech.2012.01.047 [23] PETERSON A A, VOGEL F, LACHANCE R P, FROLING M, ANTAL M J, TESTER J W. Thermochemical biofuel production in hydrothermal media:A review of sub-and supercritical water technologies[J]. Energy Environ Sci, 2008, 1(1):32-65. doi: 10.1039/b810100k [24] DOTE Y, INOUE S, OGI T, YOKOYAMA S. Studies on the direct liquefaction of protein-contained biomass:The distribution of nitrogen in the products[J]. Biomass Bioenergy, 1996, 11(6):491-498. doi: 10.1016/S0961-9534(96)00045-1 [25] DOTE Y, INOUE S, OGI T, YOKOYAMA S. Distribution of nitrogen to oil products from liquefaction of amino acids[J]. Bioresour Technol, 1998, 64(2):157-160. doi: 10.1016/S0960-8524(97)00079-5 [26] ZOU S, WU Y, YANG M, KALEEM I, CHUN L, TONG J. Production and characterization of bio-oil from hydrothermal liquefaction of microalgae dunaliella tertiolecta cake[J]. Energy, 2010, 35(12):5406-5411. doi: 10.1016/j.energy.2010.07.013 [27] CHEN Y, WU Y L, ZHANG P L, HUA D R, YANG M D, LI C, CHEN Z, LIU J. Direct liquefaction of Dunaliella tertiolecta for bio-oil in sub/supercritical ethanol-water[J]. Bioresour Technol, 2012, 124:190-198. doi: 10.1016/j.biortech.2012.08.013 [28] HUANG Y Q, CHEN Y P, XIE J J, LIU H C, YIN X L, WU C Z. Bio-oil production from hydrothermal liquefaction of high-protein high-ash microalgae including wild Cyanobacteria sp and cultivated Bacillariophyta sp[J]. Fuel, 2016, 183:9-19. doi: 10.1016/j.fuel.2016.06.013 [29] CHEN Y, WU Y, ZHANG P, HUA D, YANG M, LI C, CHEN Z, LIU J. Direct liquefaction of Dunaliella tertiolecta for bio-oil in sub/supercritical ethanol-water[J]. Bioresour Technol, 2012, 124:190-198. doi: 10.1016/j.biortech.2012.08.013 [30] WANG S, JIANG D, CAO B, HU Y M, YUAN C, WANG Q, HE Z X, HUI C W, ABOMOHRA A, LIU X L, FENG Y Q, ZHANG B. Study on the interaction effect of seaweed bio-coke and rice husk volatiles during co-pyrolysis[J]. J Anal Appl Pyrolysis, 2018, 132:111-122. doi: 10.1016/j.jaap.2018.03.009 [31] TIAN Y, ZHANG J, ZUO W, CHEN L, CUI Y N, TAN T. Nitrogen conversion in relation to NH3 and HCN during microwave pyrolysis of sewage sludge[J]. Environ Sci Technol, 2013, 47(7):3498-3505. doi: 10.1021/es304248j [32] MACKINNON S L, HILTZ D, UGARTE R, CRAFT C A. Improved methods of analysis for betaines in Ascophyllum nodosum and its commercial seaweed extracts[J]. J Appl Phycol, 2010, 22(4):489-494. doi: 10.1007/s10811-009-9483-0 [33] RIZZI G P. Free radicals in the maillard reaction[J]. Food Rev Int, 2003, 19(4):375-395. doi: 10.1081/FRI-120025481 [34] WANG S, WANG Q, HU Y M, XU S N, HE Z X, JI H S. Study on the synergistic co-pyrolysis behaviors of mixed rice husk and two types of seaweed by a combined TG-FTIR technique[J]. J Anal Appl Pyrolysis, 2015, 114:109-118. doi: 10.1016/j.jaap.2015.05.008 [35] LI R, ZHONG Z, JIN B, ZHENG A. Selection of temperature for bio-oil production from pyrolysis of algae from lake blooms[J]. Energy Fuels, 2012, 26(5):2996-3002. doi: 10.1021/ef300180r [36] SEBESTYEN Z, BARTA-RAJNAI E, CZEGENY Z, BHASKAR T, KRISHNA B B, MAY Z, BOZI J, BARTA Z, SINGH R, JAKAB E. Thermoanalytical characterization and catalytic conversion of deoiled micro algae and jatropha seed cake[J]. Energy Fuels, 2016, 30(10):7982-7993. doi: 10.1021/acs.energyfuels.6b01024 [37] YANG H, YAN R, CHEN H, LEE D H, ZHENG C. Characteristics of hemicellulose, cellulose and lignin pyrolysis[J]. Fuel, 2007, 86(12/13):1781-1788. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=3fa85712db16f40b571417e71fe15255 [38] SINGH S, WU C, WILLIAMS P T. Pyrolysis of waste materials using TGA-MS and TGA-FTIR as complementary characterisation techniques[J]. J Anal Appl Pyrolysis, 2012, 94:99-107. doi: 10.1016/j.jaap.2011.11.011 [39] HANSSON K M, SAMUELSSON J, TULLIN C, AMAND L E. Formation of HNCO, HCN, and NH3 from the pyrolysis of bark and nitrogen-containing model compounds[J]. Combust Flame, 2004, 137(3):265-277. doi: 10.1016/j.combustflame.2004.01.005 [40] CHIAVARI G, GALLETTI G C. Pyrolysis-gas chromatography mass-spectrometry of amino-acids[J]. J Anal Appl Pyrolysis, 1992, 24(2):123-137. doi: 10.1016/0165-2370(92)85024-F [41] GAUTAM R, VARMA A K, VINU R. Apparent kinetics of fast pyrolysis of four different microalgae and product analyses using pyrolysis-FTIR and pyrolysis-GC/MS[J]. Energy Fuels, 2017, 31(11):12339-12349. doi: 10.1021/acs.energyfuels.7b02520 [42] MIAO X, WU Q. High yield bio-oil production from fast pyrolysis by metabolic controlling of Chlorella protothecoides[J]. J Biotechnol, 2004, 110(1):85-93. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=c14b75b846385f5109c3c6782cf780cc [43] ROSS A B, JONES J M, KUBACKI M L, BRIDGEMAN T. Classification of macroalgae as fuel and its thermochemical behavior[J]. Bioresour Technol, 2008, 99(14):6494-6504. doi: 10.1016/j.biortech.2007.11.036 -





 下载:
下载: