Preparation of CoMo/γ-Al2O3 catalyst for hydrodesulfurization by impregnation with pulsed electromagnetic fields
-
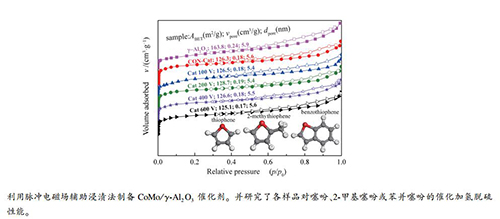
摘要: 利用脉冲电磁场(PEMF)辅助浸渍法和常规等体积浸渍法制备了一系列CoMo/γ-Al2O3加氢脱硫催化剂样品。以噻吩、2-甲基噻吩或苯并噻吩为模型化合物,在微反装置上评价了催化剂的加氢脱硫活性,经200 V脉冲电磁场处理的催化剂上各硫化物的转化率均比常规催化剂明显提高。采用XRD、BET、H2-TPR和TEM等方法表征了催化剂样品的表面状态和物化性质。结果表明,适当强度的脉冲电磁场与制备体系内带电粒子的交互作用有助于催化剂活性组分的分散,使活性组分在γ-Al2O3载体表面分布更为均匀,同时削弱了活性组分MoO3同载体间的较强相互作用,降低了催化剂表面钼物种的还原温度,促进了CoMoS活性相的形成。
-
关键词:
- 脉冲电磁场 /
- CoMo/γ-Al2O3 /
- 负载型催化剂 /
- 浸渍法 /
- 加氢脱硫
Abstract: CoMo/γ-Al2O3 catalyst for hydrodesulfurization (HDS) were prepared by equal volume impregnation method with and without the presence of pulsed electronmagnetic field (PEMF). Experimental results revealed that the catalyst prepared by PEMF with a voltage of 200 V exhibited higher catalytic activity for hydrodesulfurization of thiophene, 2-methythiophene and benzothiophene than the catalyst prepared by conventional impregnation. The surface morphology and physico-chemical properties were characterized by using BET, XRD, H2-TPR and TEM techniques, respectively. The results showed that appropriate PEMF treatment promotes the active component dispersion on the γ-Al2O3 surface by interacting with the charged particles in reaction system. The interaction between the support and the active species MoO3 is weakened and thus facilitates the reduction of the catalyst and the formation of CoMoS active phase.-
Key words:
- pulsed electronmagnetic field /
- CoMo/γ-Al2O3 /
- supported catalyst /
- impregnation /
- hydrodesulfurization
-
表 1 不同催化剂样品对噻吩、2-甲基噻吩和苯并噻吩加氢脱硫的催化活性
Table 1 Catalytic activity for hydrodesulfurization (HDS) of thiophene, 2-methythiophene and benzothiophene
Catalyst Thiophene conversion x /% 2-methythiophene conversion x /% Benzothiophene conversion x /% 60 min 90 min 120 min 60 min 90 min 120 min 60 min 90 min 120 min CON-Cat 82.97 54.79 39.43 75.68 50.73 35.82 69.52 51.64 32.86 Cat 100 V 83.36 65.38 41.94 73.58 48.96 37.63 71.35 53.48 33.62 Cat 200 V 91.50 74.09 51.59 86.25 71.59 48.26 80.67 71.95 43.55 Cat 400 V 83.78 72.65 43.79 71.45 46.65 32.79 70.12 48.85 36.78 Cat 600 V 78. 82 63.18 35.68 68.95 43.26 31.54 68.56 43.98 33.59 T203 78.34 57.84 47.62 78.63 56.26 46.65 65.25 52.31 42.96 表 2 噻吩、2-甲基噻吩和苯并噻吩的非氢原子净电荷
Table 2 Non-hydrogen atom charges for thiophene, 2-methythiophene and benzothiophene
Atom Thiophene 2-methythiophene Benzothiophene C1 -0.0951 -0.0991 -0.052 C2 -0.0747 -0.0776 -0.0503 C3 -0.0747 -0.0825 -0.0174 C4 -0.0951 -0.0321 -0.0267 S 0.0786 0.0690 0.0738 C6 - -0.0965 -0.0574 C7 - - -0.0533 C8 - - -0.0704 C9 - - -0.0813 Molecular structure 


-
[1] 刘丽, 郭蓉, 孙进, 丁莉, 杨成敏, 段为宇, 姚运海.柴油加氢脱硫催化剂的研究进展[J].化工进展, 2016, 35(11):3503-3510. http://d.old.wanfangdata.com.cn/Periodical/hgjz201611018LIU Li, GUO Rong, SUN Jin, DING Li, YANG Cheng-min, DUAN Wei-yu, YAO Yun-hai. The research development of diesel hydrodesulfurization catalysts[J]. Chem Ind Eng Prog, 2016, 35(11):3503-3510. http://d.old.wanfangdata.com.cn/Periodical/hgjz201611018 [2] 李翠清, 潘雅妹, 李继文, 王虹, 靳广州, 孙桂大.含镍WP/MCM-41催化剂二苯并噻吩加氢脱硫性能[J].燃料化学学报, 2011, 39(12):930-935. doi: 10.3969/j.issn.0253-2409.2011.12.009LI Cui-qing, PAN Ya-mei, LI Ji-wen, WANG Hong, JIN Guang-zhou, SUN Gui-da. Performance of dibenzothiophene hydrodesulfurization for WP/MCM-41 catalyst containing promoter nickel[J]. J Fuel Chem Technol, 2011, 39(12):930-935. doi: 10.3969/j.issn.0253-2409.2011.12.009 [3] ASADI A A, ALAVI S M, ROYAEE S J, BAZMI M. Ultradeep hydrodesulfurization of feedstock containing cracked gasoil through NiMo/γ-Al2O3 catalyst pore size optimization[J]. Energy Fuels, 2018, 32(2):2203-2212. doi: 10.1021/acs.energyfuels.7b03461 [4] 王海涛, 徐学军, 王继锋, 刘东香, 冯小萍.钨、钼、镍含量的改变对体相催化剂物化性质、加氢活性影响[J].燃料化学学报, 2018, 46(3):337-345. doi: 10.3969/j.issn.0253-2409.2018.03.011WANG Hai-tao, XU Xue-jun, WANG Ji-feng, LIU Dong-xiang, FENG Xiao-ping. Effects of tungsten, molybdenum and nickel content change on physicochemical properties and hydrogenation activity of bulk catalysts[J]. J Fuel Chem Technol, 2018, 46(3):337-345. doi: 10.3969/j.issn.0253-2409.2018.03.011 [5] 陈茂森, 宋华, 李锋, 陈彦广, 张建.制备方法对稀土金属钇改性Ni2P催化剂结构及其加氢脱硫性能的影响[J].燃料化学学报, 2017, 45(2):213-219. doi: 10.3969/j.issn.0253-2409.2017.02.011CHEN Mao-sen, SONG Hua, LI Feng, CHEN Yan-guang, ZHANG Jian. Effects of preparation method on the structure of rare earth metal Y modified Ni2P catalysts and its HDS performance[J]. J Fuel Chem Technol, 2017, 45(2):213-219. doi: 10.3969/j.issn.0253-2409.2017.02.011 [6] ABRAHAMSON J P, WINCEK R T, ESER S. Effects of catalyst properties on hydrodesulfurization activity for sulfur removal from fluid catalytic cracking decant oils[J]. Energy Fuels, 2016, 30(9):7173-7179. doi: 10.1021/acs.energyfuels.6b01441 [7] 庞伟伟.负载型加氢催化剂载体对中间馏分油HDS活性的影响[D].北京: 中国石油大学, 2008. http://cdmd.cnki.com.cn/article/cdmd-10425-2009221568.htmPANG Wei-wei. Study of support affection on catalysts in HDS of middle distillate oil[D]. Beijing: China University of Petroleum, 2008. http://cdmd.cnki.com.cn/article/cdmd-10425-2009221568.htm [8] 曾永康, 曾利辉, 潘丽娟, 杨乔森, 文永忠, 张之翔.超声浸渍法制备Pd/Al2O3催化剂及其催化硝基苯加氢性能[J].稀有金属材料与工程, 2008, 37(4):674-676. doi: 10.3321/j.issn:1002-185X.2008.04.025ZENG Yong-kang, ZENG Li-hui, PAN Li-juan, YANG Qiao-sen, WEN Yong-zhong, ZHANG Zhi-xiang. Preparation of supported Pd/Al2O3 catalysts by ultrasonic impregnation and their catalytic performance for nitrobenzene hydrogenation[J]. Rare Metal Mater Eng, 2008, 37(4):674-676. doi: 10.3321/j.issn:1002-185X.2008.04.025 [9] 刘学芬, 张乐, 石亚华, 聂红, 龙湘云.超声法-微波法制备NiW/Al2O3加氢脱硫催化剂[J].催化学报, 2004, 25(9):748-752. doi: 10.3321/j.issn:0253-9837.2004.09.016LIU Xue-fen, ZHANG Le, SHI Ya-hua, NIE Hong, LONG Xiang-yun. Preparation of NiW/Al2O3 hydrodesulfurization catalyst by ultrasound-microwave treatment[J]. Chin J Catal, 2004, 25(9):748-752. doi: 10.3321/j.issn:0253-9837.2004.09.016 [10] GROBAS J, CARMELO BOLIVAR A, SCOTT C E. Hydrodesulfurization of benzothiophene and hydrogenation of cyclohexene, biphenyl, and quinoline, assisted by ultrasound, using formic acid as hydrogen prcursor[J]. Energy Fuels, 2007, 21(1):19-22. doi: 10.1021/ef0603939 [11] 刘文洁, 张庆军, 隋宝宽, 袁胜华.微波辅助双功能渣油加氢催化剂的研究[J].石油炼制与化工, 2016, 47(9):57-61. doi: 10.3969/j.issn.1005-2399.2016.09.011LIU Wen-jie, ZHANG Qing-jun, SUI Bao-kuan, YUAN Sheng-hua. Study of microwave-assisted dual functional residue hydrogenation catalysi[J]. Pet Process Petrochem, 2016, 47(9):57-61. doi: 10.3969/j.issn.1005-2399.2016.09.011 [12] 甘丹丹.不同晶型氧化铝载体制备及其催化剂对汽油加氢脱硫性能研究[D].北京: 中国石油大学(北京), 2016.GAN Dan-dan. Synthesis of different crystal structures of alumina and the hydrodesulfurization performance of the corresponding catalyst. Beijing: China University of Petroleum, 2016. [13] 白庆伟, 麻永林, 邢淑清, 冯艳飞, 鲍鑫宇, 陈重毅.表面脉冲电磁场处理下7A04铝合金凝固组织演变[J].材料导报, 2018, 32(6):2021-2027. http://d.old.wanfangdata.com.cn/Periodical/cldb201812015BAI Qing-wei, MA Yong-lin, XING Shu-qing, FENG Yan-fei, BAO Xin-yu, CHEN Zhong-yi. Solidified microstructure evolution of 7A04 alloy under surface electromagnetic pulse treatment[J]. Mater Rev, 2018, 32(6):2021-2027. http://d.old.wanfangdata.com.cn/Periodical/cldb201812015 [14] DU H, WANG J, WANG B, CANG D. Preparation of cobalt oxalate powders with the presence of a pulsed electromagnetic field[J]. Powder Technol, 2010, 199(2):149-153. doi: 10.1016/j.powtec.2009.12.015 [15] 彭淑静, 唐立丹, 王冰, 王建中, 林静.脉冲电磁场对用湿法制备的草酸镍盐形貌和粒度的影响[J].材料研究学报, 2017, 31(12):947-954. doi: 10.11901/1005.3093.2017.161PENG Shu-jing, TANG Li-dan, WANG Bing, WANG Jian-zhong, LIN Jing. Effect of pulsed electromagnetic field on the morphology and power size during nickel oxalate salt produced by wet chemical method[J]. Chin J Mater Res, 2017, 31(12):947-954. doi: 10.11901/1005.3093.2017.161 [16] MICHÈLE B, GÉRALD D M, STÉPHANIE P, GEANTET C, VRINAT M, PÉROT G, LEMAIRE M. Deep desulfurization:Reactions, catalysts and technological challenges[J]. Catal Today, 2003, 84:129-138. doi: 10.1016/S0920-5861(03)00266-9 [17] SAKASHITA Y. Effects of surface orientation and crystallinity of alumina supports on the microstructures of molybdenum oxides and sulfides[J]. Surf Sci, 2001, 489(1/3):45-48. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6cc76feabd520bb9240f4d3f9477e63f [18] 李自夏, 常晓昕, 孙伟. CoMo/Al2O3催化剂上FCC汽油加氢脱硫活性研究[J].计算机与应用化学, 2016, 33(8):920-924. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jsjyyyhx201608014LI Zi-xia, CHANG Xiao-xin, SUN Wei. Research of hydrodesulfurization acitivity of FCC gasoline on CoMo/Al2O3 catalysits[J]. Comput Appl Chem, 2016, 33(8):920-924. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jsjyyyhx201608014 [19] 胡亚杰, 徐流杰, 周玉成, 李继文, 刘伟, 魏世忠.水热法制备Al2O3掺杂钼合金[J].材料热处理学报, 2015, 36(6):15-20. http://d.old.wanfangdata.com.cn/Periodical/jsrclxb201506004HU Ya-jie, XU Liu-jie, ZHOU Yu-cheng, LI Ji-wen, LIU Wei, WEI Shi-zhong. Preparation of molybdenum alloy doped Al2O3 by hydrothermal synthesis method[J]. Trans Mater Heat Treat, 2015, 36(6):15-20. http://d.old.wanfangdata.com.cn/Periodical/jsrclxb201506004 [20] KALUŽA L, ZDRAŽIL M. Relative activity of niobia-supported CoMo hydrodesulphurization catalyst prepared with NTA:A kinetic approach[J]. Catal Commun, 2018, 107:62-67. doi: 10.1016/j.catcom.2018.01.020 [21] OKAMOTO Y, OCHIAI K, KAWANO M, KOBAYASHI K, KUBOTA T. Effects of supports on the activity of Co-Mo sulfide model catalysts[J]. Appl Catal A:Gen, 2002, 226:115-127. doi: 10.1016/S0926-860X(01)00893-6 [22] 周同娜, 尹海亮, 柳云骐, 韩姝娜, 柴永明, 刘晨光.磷含量对NiMo/γ-Al2O3催化剂活性相结构的影响[J].燃料化学学报, 2010, 38(1):69-74. doi: 10.3969/j.issn.0253-2409.2010.01.013ZHOU Tong-na, YIN Hai-liang, LIU Yun-qi, HAN Shu-na, CHAI Yong-mign, LIU Chen-guang. Effects of phosphorus content on the active phase structure of NiMo/γ-Al2O3 catalyst[J]. J Fuel Chem Technol, 2010, 38(1):69-74. doi: 10.3969/j.issn.0253-2409.2010.01.013 [23] REARDON J, DATYE A K, SAULT A G. Tailoring alumina surface chemistry for efficient use of supported MoS2[J]. J Catal, 1998, 173(1):145-156. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=a79de7bcdd326d3770b1d7c60199c194 [24] LI P, CHEN Y, ZHANG C, HUANG B, LIU X, LIU T, JIANG Z, LI C. Highly selective hydrodesulfuization of gasoline on unsupported Co-Mo sulfide catalysts:Effect of MoS2 morphology[J]. Appl Catal A:Gen, 2017, 533:99-108. doi: 10.1016/j.apcata.2017.01.009 [25] 施尔畏.水热结晶学[M].北京:科学出版社, 2004.SHI Er-wei. Hydrothermal Crystallization[M]. Beijing:Science Press, 2004. [26] OKAMOTO Y, KAWANO M, KAWABATA T, KUBOTA T, HIROMITSU I. Structure of the active sites of Co-Mo hydrodesulfurization catalysts as studied by magnetic susceptibility measurement and NO adsorption[J]. J Phys Chem B, 2005, 109(1):288-296. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=f1cab5b5551fcd287ebd0d6c1e695413 [27] DONPHAI W, PIRIYAWATE N, WITOON T, JANTARATANA P, VARABUNTOONVIT V, CHAREONPANICH M. Effect of magnetic field on CO2 conversion over Cu-ZnO/ZrO2 catalyst in hydrogenation reaction[J]. J CO2 Util, 2016, 16:204-211. doi: 10.1016/j.jcou.2016.07.007 -





 下载:
下载: