Catalytic performance of La1-xRbxMnO3 perovskite in the simultaneous removal of NO and soot
-
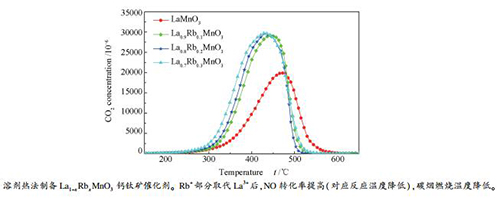
摘要: 采用溶剂热法制备了La1-xRbxMnO3(x=0、0.1、0.2、0.3)钙钛矿型复合金属氧化物催化剂,通过XRD、FT-IR、SEM、XPS和H2-TPR等手段对催化剂进行表征,在微型固定床反应器上评价了其同时消除NO和碳烟的催化性能。结果表明,La1-xRbxMnO3催化剂具有单一的钙钛矿结构,样品中Mn物种以Mn3+和Mn4+的形式存在。与LaMnO3催化剂相比,Rb+部分取代La3+,催化剂体系中形成较多的高价Mn4+和氧空位,其氧化还原性能提高,催化性能得到改善。随着Rb+取代量的增加,NO转化率升高,碳烟燃烧温度降低。当x=0.3时,La0.7Rb0.3MnO3催化剂上CO2浓度峰值温度tmax为430 ℃,CO2的选择性为99.0%;反应温度为429 ℃,NO转化率达到最大,为59.7%。Abstract: A series of La1-xRbxMnO3 perovskite catalysts (x=0, 0.1, 0.2, 0.3) were prepared by solvothermal method and characterized by means of XRD, FT-IR, SEM, XPS and H2-TPR; their catalytic performance in the simultaneous removal of NO and soot was investigated in a fixed-bed micro-reactor. The results show that the La1-xRbxMnO3 catalysts have a single perovskite structure and the Mn species exist in the form of Mn3+ and Mn4+. In comparison with LaMnO3, when La3+ is partially substituted by Rb+, more high-valence Mn4+ and oxygen vacancies are formed in La1-xRbxMnO3, which can improve the redox properties and enhance the catalytic performance of La1-xRbxMnO3. With the increase of Rb+ content in La1-xRbxMnO3, the conversion of NO increases and the soot combustion temperature decreases. For NO and soot removal over La0.7Rb0.3MnO3, x=0.3, the peak CO2 formation temperature (tmax) is 430 ℃, with a selectivity of 99.0% to CO2; at 429 ℃, the maximum conversion of NO reaches 59.7%.
-
Key words:
- Mn-based perovskite /
- Rb /
- La /
- NO /
- soot
-
表 1 La1-xRbxMnO3催化剂的XPS表征
Table 1 XPS data of the La1-xRbxMnO3 catalysts
Sample Binding energy E/eV Oads/Olat Binding energy E/eV Mn4+/Mn3+ Olat Oads Mn3+ Mn4+ LaMnO3 529.32 531.27 0.661 641.38 642.88 0.592 La0.9Rb0.1MnO3 529.00 530.84 0.701 641.21 642.49 0.668 La0.8Rb0.2MnO3 529.05 530.94 0.845 641.11 642.12 0.714 La0.7Rb0.3MnO3 528.99 530.81 0.976 641.05 642.21 0.824 -
[1] LIU J, ZHAO Z, XU C M, DUAN A J, JIANG G Y. Simultaneous removal of soot and NOx over the (La1.7Rb0.3CuO4)x/nmCeO2 nanocomposite catalysts[J]. Ind Eng Chem Res, 2010, 49(7):3112-3119. doi: 10.1021/ie100004m [2] URAN L, GALLEGO J, LI W Y, SANTAMARIA A. Effect of catalyst preparation for the simultaneous removal of soot and NOx[J]. Appl Catal A:Gen, 2019, 569:157-169. doi: 10.1016/j.apcata.2018.10.029 [3] YOSHIDA K, MAKINO S, SUMIYA S, MURAMATSU G, HELFERICH R. Simultaneous reduction of NOx and particulate emissions from diesel engine exhaust[J]. SAE paper no. 892046, 1989. [4] COOPER B J, THOSS J E. Role of NO in diesel particulate emission contral[J]. SAE paper no. 890404, 1989. [5] 王虹, 王军利, 李翠清, 丁福臣, 宋永吉, 迟姚玲.柴油机尾气碳颗粒燃烧中La-Mn-Fe-Cu/HZSM-5的催化性能[J].燃料化学学报, 2012, 40(3):374-379. doi: 10.3969/j.issn.0253-2409.2012.03.021WANG Hong, WANG Jun-li, LI Cui-qing, DING Fu-chen, SONG Yong-ji, CHI Yao-ling. Catalytic performance of La-Mn-Fe-Cu/HZSM-5 catalysts for soot combustion from diesel exhaust[J]. J Fuel Chem Technol, 2012, 40(3):374-379. doi: 10.3969/j.issn.0253-2409.2012.03.021 [6] LI Z Q, MENG M, DAI F F, HU T D, XIE Y N, ZHANG J. Performance of K and Ni substituted La1-xKxCo1-yNiyO3-δ perovskite catalysts used for soot combustion, NOx storage and simultaneous NOx-soot removal[J]. Fuel, 2012, 93:606-610. doi: 10.1016/j.fuel.2011.10.040 [7] YOON D Y, LIM E, KIM Y J, KIM J H, RYU T, LEE S, K. CHO B K, NAM I S, CHOUNG J W, YOO S. NO oxidation activity of Ag-doped perovskite catalysts[J]. J Catal, 2014, 319:182-193. doi: 10.1016/j.jcat.2014.09.007 [8] LI Z Q, MENG M, ZHA Y Q, DAI F F, HU T D, XIE Y N, ZHANG J. Highly efficient multifunctional dually-substituted perovskite catalysts La1-xKxCo1-yCuyO3-δ used for soot combustion, NOx storage and simultaneous NOx-soot removal[J]. Appl Catal B:Environ, 2012, 121-122:65-74. doi: 10.1016/j.apcatb.2012.03.022 [9] 王虹, 赵震, 徐春明, 刘坚, 吕志校.纳米La-Mn-O钙钛矿型氧化物催化剂上柴油机尾气碳颗粒催化燃烧性能的研究[J].科学通报, 2005, 50(4):336-339. doi: 10.3321/j.issn:0023-074X.2005.04.006WANG Hong, ZHAO Zhen, XU Chun-ming, LIV Jian, LV Zhi-xiao. Catalytic performance of nanometric La-Mn-O perovskite-type oxides catalysts for soot combustion from diesel exhaust[J]. Chin Sci Bull, 2005, 50(4):336-339. doi: 10.3321/j.issn:0023-074X.2005.04.006 [10] WANG H, ZHAO Z, XU C M, LIU J. Nanometric La1-xKxMnO3 perovskite-type oxides-highly active catalysts for the combustion of diesel soot particle under loose contact conditions[J]. Catal Lett, 2005, 102(3/4):251-256. doi: 10.1007/s10562-005-5864-4 [11] LI K B, LI X J, ZHU K G, ZHU J S, ZHANG Y H. Infrared absorption spectra of manganese oxides La1-x-yRyCaxMnO3-δ[J]. J Appl Phys, 1997, 81(10):6943-6947. doi: 10.1063/1.365234 [12] ROYER S, DUPREZ D, CAN F, COURTOIS X, BATIOT-DUPEYRAT C, LAASSIRI S, ALAMDARI H. Perovskites as substitutes of noble metals for heterogeneous catalysis:Dream or reality[J]. Chem Rev, 2014, 114(20):10292-10368. doi: 10.1021/cr500032a [13] MACHOCKI A, IOANNIDES T, STASINSKA B, GAC W, AVGOUROPOULOS G, DELIMARIS D, GRZEGORCZYK W, PASIECZNA S. Manganese-lanthanum oxides modified with silver for the catalytic combustion of methane[J]. J Catal, 2004, 227(2):282-296. doi: 10.1016-j.jcat.2004.07.022/ [14] CHEN J H, SHEN M Q, WANG X Q, WANG J, SU Y G, ZHAO Z. Catalytic performance of NO oxidation over LaMeO3 (Me=Mn, Fe, Co) perovskite prepared by the sol-gel method[J]. Catal Commun, 2013, 37:105-108. doi: 10.1016/j.catcom.2013.03.039 [15] XU J F, LIU J, ZHAO Z, XU C M, ZHENG J X, DUAN A J, JIANG G Y. Easy synthesis of three-dimensionally ordered macroporous La1-xKxCoO3 catalysts and their high activities for the catalytic combustion of soot[J]. J Catal, 2011, 282(1):1-12. http://www.sciencedirect.com/science/article/pii/S0021951711000960 [16] WANG H, LIU J, ZHAO Z, WEI Y, XU C M. Comparative study of nanometric Co-, Mn- and Fe-based perovskite-type complex oxide catalysts for the simultaneous elimination of soot and NOx from diesel engine exhaust[J]. Catal Today, 2012, 184(1):288-300. doi: 10.1016/j.cattod.2012.01.005 [17] YANG J P, ZHANG M G, LI H L, QU W Q, ZHAO Y C, ZHANG J Y. Simultaneous NO reduction and Hg0 oxidation over La0.8Ce0.2MnO3 perovskite catalysts at low temperature[J]. Ind Eng Chem Res, 2018, 57(29):9374-9385. doi: 10.1021/acs.iecr.8b01431 [18] TENG F, HAN W, LIANG S H, GAUGEU B, ZONG R L, ZHU Y F. Catalytic behavior of hydrothermally synthesized La0.5Sr0.5MnO3 single-crystal cubes in the oxidation of CO and CH4[J]. J Catal, 2007, 250(1):1-11. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d9db6893075abf72a5f5ea3a110f980d [19] LU Y J, DAI Q G, WANG X Y. Catalytic combustion of chlorobenzene on modified LaMnO3 catalysts[J]. Catal Commun, 2014, 54:114-117. doi: 10.1016/j.catcom.2014.05.018 [20] LIU Y X, DAI H X, DENG J G, DU Y C, LI X W, ZHAO Z X, WANG Y, GAO B Z, YANG H G, GUO G S. In situ poly(methyl methacrylate)-templating generation and excellent catalytic performance of MnOx/3DOM LaMnO3 for the combustion of toluene and methanol[J]. Appl Catal B:Environ, 2013, 140-141:493-505. doi: 10.1016/j.apcatb.2013.04.051 [21] ZHU Y J, SUN Y Q, NIU X Y, YUAN F L, FU H G. Preparation of La-Mn-O perovskite catalyst by microwave irradiation method and its application to methane combustion[J]. Catal Lett, 2010, 135(1/2):152-158. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=a3c1866172809e38c8e0181e0d25ccfb [22] ARANDIYAN H, DAI H X, DENG J G, LIU Y X, BAI B Y, WANG Y, LI X W, XIE S H, LI J H. Three-dimensionally ordered macroporous La0.6Sr0.4MnO3 with high surface areas:Active catalysts for the combustion of methane[J]. J Catal, 2013, 307:327-339. doi: 10.1016/j.jcat.2013.07.013 [23] SHEN M Q, ZHAO Z, CHEN J H, SU Y G, WANG J, WANG X Q. Effects of calcium substitute in LaMnO3 perovskites for NO catalytic oxidation[J]. J Rare Earth, 2013, 31(2):119-123. doi: 10.1016/S1002-0721(12)60244-0 [24] ZHANG-STEENWINKEL Y, BECKERS J, BLIEK A. Surface properties and catalytic performance in CO oxidation of cerium substituted lanthanum-manganese oxides[J]. Appl Catal A:Gen, 2002, 235(1/2):79-92. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=44b5eb2b10dd8d7fe85829eeb7c4853b [25] WEN Y X, ZHANG C B, HE H, YU Y B, TERAOKA Y. Catalytic oxidation of nitrogen monoxide over La1-xCexCoO3 perovskites[J]. Catal Today, 2007, 126(3/4):400-405. http://www.sciencedirect.com/science/article/pii/S0920586107003598 [26] WANG Y Z, XIE S H, DENG J G, DENG S X, WANG H, YAN H, DAI H X. Morphologically controlled synthesis of porous spherical and cubic LaMnO3 with high activity for the catalytic removal of toluene[J]. ACS Appl Mater Inter, 2014, 6(20):17394-17401. doi: 10.1021/am500489x [27] DING Y, WANG S, ZHANG L, CHEN Z Q, WANG M Z, WANG S D. A facile method to promote LaMnO3 perovskite catalyst for combustion of methane[J]. Catal Commun, 2017, 97:88-92. doi: 10.1016/j.catcom.2017.04.022 [28] LIU J, ZHAO Z, XU C M, DUAN A J, MENG T, BAO X J. Simultaneous removal of NOx and diesel soot particulates over nanometric La2-xKxCuO4 complex oxide catalysts[J]. Catal Today, 2007, 119(1/4):267-272. doi: 10.1016-j.cattod.2006.08.040/ -





 下载:
下载: