Thermal dissolution of Huozhou and Xinghe lignites and the occurrence forms of organic oxygen in them
-
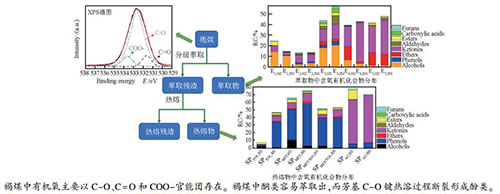
摘要: 依次采用石油醚、二硫化碳、甲醇、丙酮和等体积的丙酮/二硫化碳混合溶剂对霍州(HZ)和兴和(XH)褐煤进行分级萃取得到萃取物(E1-E5)和萃取残渣(ER1-ER5),利用甲醇、甲苯、等体积甲醇/甲苯混合溶剂和丙酮等溶剂分别对ER5在320℃进行热溶得到热溶物(SPs)。HZ和XH的累积萃取物产率分别为7.03%和7.86%,其中,E3的产率最高;萃取残渣在甲醇/甲苯混合溶剂中的热溶物产率最高,ER5,HZ和ER5,XH的热溶物产率分别为45.76%和40.14%。E1-E5的傅里叶变换红外光谱(FT-IR)谱图中均有较强的脂肪C-H的伸缩振动峰,而SPs的FT-IR谱图中具有较强的C=C、C=O和缔合羟基的吸收峰。气相色谱质谱联用(GC/MS)分析表明,E1-E5中含氧化合物(OCOCs)以醇、醚和酮类化合物为主,而SPs中OCOCs以醇、酚和酮类化合物为主,且极性较强的溶剂有利于褐煤中OCOCs的溶出。与原煤FT-IR谱图相比,萃取残渣和热溶残渣的FT-IR谱图中缔合羟基、C=O和C-O-C的伸缩振动峰强度明显减弱。X射线光电子能谱(XPS)分析表明,两种褐煤中不同形态氧的相对含量为C-O > C=O > COO-,并且HZ具有更高的C-O和COO-含量。Abstract: Huozhou (HZ)and Xinghe (XH) lignites were extracted in turn with petroleum ether, carbon disulfide, methanol, acetone and isometric acetone/carbon disulfide mixed solvent to obtain the extracts (E1-E5) and the extraction residues (ER1-ER5), then ER5 was thermally dissolved at 320℃ using methanol, toluene, isometric methanol/toluene mixed solvent and acetone to gain the soluble portions (SPs). The total yields of extraction for HZ and XH are 7.03% and 7.86%, respectively, in which the yield of E3 is the highest. The SPs yield of extraction residue with isometric methanol/toluene mixed solvent is the highest, and the SPs of ER5, HZ and ER5, XH with isometric methanol/toluene mixed solvent reaches 45.76% and 40.14%, respectively. There exist strong adsorption peaks ascribed to aliphatic C-H in the Fourier transform infrared (FT-IR) spectra of E1-E5, while the intensity of adsorption peaks ascribed to C=C, C=O and O-H in the FT-IR spectra of SPs is obviously higher than that of extracts. The gas chromatography/mass spectrometer (GC/MS) analyses show that the oxygen containing organic compounds (OCOCs) in E1-E5 are dominated with alcohols, ethers and ketones, while it is mainly composed of alcohols, phenols and ketones in the SPs, and the strong polar solvents contribute to dissolving the OCOCs in lignites. The adsorption peaks ascribed to associated O-H, C=O and C-O-C in the FT-IR spectra of extraction residues and thermal dissolution residues are weaker than those of raw coals. The X-ray photoelectron spectroscopy (XPS) analyses indicate that the relative content of oxygen forms in XH and HZ is C-O > C=O > COO-, and the relative content of C-O and COO-in HZ is higher than that in XH.
-
Key words:
- lignite /
- thermal dissolution /
- organic oxygen /
- GC/MS
-
表 1 褐煤的工业分析和元素分析
Table 1 Proximate and ultimate analyses of coal samples
Sample Proximate analysis w/% Ultimate analysis wdaf/% Mad Ad Vdaf C H N S Oa HZ 18.49 30.01 53.36 55.03 4.05 1.41 0.62 38.44 XH 13.26 13.74 48.14 60.22 6.23 0.81 2.67 30.07 a: by difference 表 2 E1-E5的GC/MS可检测化合物分布
Table 2 Distribution of group components in E1-E5 identified by GC/MS
Component Relative content w/% HZ XH E1 E2 E3 E4 E5 E1 E2 E3 E4 E5 AHs 59.15 51.72 8.91 46.12 2.95 49.21 48.71 0.80 43.41 2.44 ARs 9.87 29.50 21.15 6.93 40.45 34.42 33.27 13.14 10.06 40.92 OCOCs 24.50 13.31 43.34 38.97 41.24 14.48 13.36 56.74 42.54 47.43 NCOCs 4.98 2.89 17.56 6.69 13.80 1.39 1.87 20.40 3.99 8.16 OCs 1.50 2.58 9.04 1.29 1.56 0.50 2.79 8.92 0.00 1.05 表 3 SPs的GC/MS可检测化合物分布
Table 3 Distribution of group components in SPs identified by GC/MS
Component Relative content w/% HZ XH SPTOL SPMET SPTOL/MET SPAC SPTOL SPMET SPTOL/MET SPAC AHs 40.76 2.79 8.43 1.68 5.81 4.44 8.01 1.86 ARs 49.04 17.97 25.51 15.82 45.52 12.17 29.94 22.65 OCOCs 7.53 66.34 52.36 80.03 46.95 76.00 53.69 71.11 NCOCs 2.44 10.83 12.83 1.91 1.17 5.75 6.72 3.63 OCs 0.23 2.07 0.87 0.56 0.55 1.64 1.64 0.75 表 4 HZ、XH、ERs和TDRs有机氧形态(O 1s)及其相对含量
Table 4 Relative content of organic oxygen forms (O 1s) in HZ, XH, ERs and TDRs
Sample Relative content w/% C=O C-O COO- HZ 9.60 79.04 11.36 ER5, HZ 30.94 52.50 16.56 TDRMET/TOL, HZ 21.28 56.15 22.57 TDRAC, HZ 22.38 46.56 31.06 XH 25.99 63.26 10.78 ER5, XH 33.98 47.99 18.03 TDRMET/TOL, XH 16.11 52.96 30.93 TDRAC, XH 16.69 34.58 48.73 -
[1] 谢克昌.煤的结构与反应性[M].北京:科学出版社, 2002, 243-268. http://d.old.wanfangdata.com.cn/Periodical/zgysjsxb200203042XIE Ke-chang. Structure and Reactivity of Coal[M]. Beijing:Science Press, 2002, 243-268. http://d.old.wanfangdata.com.cn/Periodical/zgysjsxb200203042 [2] WANG J, LI C, SAKANISHI K, NAKAZATO T, TAO H, TAKANOHASHI T, TAKARADA T, SAITO I. Investigation of the remaining major and trace elements in clean coal generated by organic solvent extraction[J]. Fuel, 2005, 84(12/13):1487-1493. http://www.sciencedirect.com/science/article/pii/S0016236105000426 [3] RAHMAN M, SAMANTA A, GUPTA R. Production and characterization of ash-free coal from low-rank Canadian coal by solvent extraction[J]. Fuel Process Technol, 2013, 115(11):88-98. http://www.sciencedirect.com/science/article/pii/S0378382013001604 [4] SIMSEK E H, KARADUMAM A, CALⅡSKAN S, TOGRUL T. The effect of preswelling and/or pretreatment of some Turkish coals on the supercritical fluid extract yield[J]. Fuel, 2002, 81(4):503-506. doi: 10.1016/S0016-2361(01)00191-0 [5] BRAND S, SUSANTI R F, KIM S K, LEE H S, KIM J, SANG B I. Supercritical ethanol as an enhanced medium for lignocellulosic biomass liquefaction:Influence of physical process parameters[J]. Energy, 2013, 59(9):173-182. [6] 杜姣姣, 赵云鹏, 田由甲, 张迪.白音华褐煤可溶有机质的组成和结构特征[J].燃料化学学报, 2017, 45(1):9-14. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18955.shtmlDU Jiao-jiao, ZHAO Yun-peng, TIAN You-jia, ZHANG Di. Composition and structural characteristics of soluble organic species in Baiyinhua lignite[J]. J Fuel Chem Technol, 2017, 45(1):9-14. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18955.shtml [7] OKUYAMA N, KOMATSU N, SHIGEHISA T, KANEKO T, TSURUYA S. Hyper-coal process to produce the ash-free coal[J]. Fuel Process Technol, 2004, 85(8/10):947-967. [8] MATHEWS J P, BURGESS-CLIFFORD C, PAINTER P. Interactions of Illinois No. 6 bituminous coal with solvents:A review of solvent swelling and extraction literature[J]. Energy Fuels, 2015, 29(3):1279-1294. doi: 10.1021/ef502548x [9] NIEKERK D V, HALLECK P M, MATHEWS J P. Solvent swelling behavior of permian-aged south african vitrinite-rich and inertinite-rich coals[J]. Fuel, 2010, 89(1):19-25. doi: 10.1016/j.fuel.2009.06.028 [10] LIU F J, WEI X Y, LI W T, GUI J, LI P, WANG Y G, XIE R L, ZONG Z M. Methanolysis of extraction residue from Xianfeng lignite with NaOH and product characterizations with different spectrometries[J]. Fuel Process Technol, 2015, 136(8):8-16. http://www.sciencedirect.com/science/article/pii/S037838201400294X [11] HAO P, BAI Z, HOU R, XU J, BAI J, GUO Z, KONG L, LI W. Effect of solvent and atmosphere on product distribution, hydrogen consumption and coal structural change during preheating stage in direct coal liquefaction[J]. Fuel, 2018, 211(1):783-788. http://www.deepdyve.com/lp/elsevier/effect-of-solvent-and-atmosphere-on-product-distribution-hydrogen-ctuFY11lmO?impressionId=5a638a6e5fdd0&i_medium=docview&i_campaign=recommendations&i_source=recommendations [12] DING M, ZHAO Y P, DOU Y Q, WEI X Y, FAN X, CAO J P, WANG Y L, ZONG Z M. Sequential extraction and thermal dissolution of Shengli lignite[J]. Fuel Process Technol, 2015, 135(7):20-24. http://www.sciencedirect.com/science/article/pii/S0378382014004032 [13] WANG Z, SHUI H, PAN C, LI L, REN S, LEI Z, KANG S, WEI C, HU J. Structural characterization of the thermal extracts of lignite[J]. Fuel Process Technol, 2014, 120(4):8-15. [14] WEI X Y, WANG X H, ZONG Z M. Extraction of organonitrogen compounds from five Chinese coals with methanol[J]. Energy Fuels, 2009, 23(10):4848-481. doi: 10.1021/ef900086h [15] SHI K, GUI X, TAO X, LONG J, JI Y. Macromolecular structural unit construction of Fushun nitric-acid-oxidized coal[J]. Energy Fuels, 2015, 29(6):3566-3572. doi: 10.1021/ef502859r -





 下载:
下载: