Hydrodenitrogenation and hydrodesulfurization of coal tar on Ni-W catalysts with different metal loadings
-
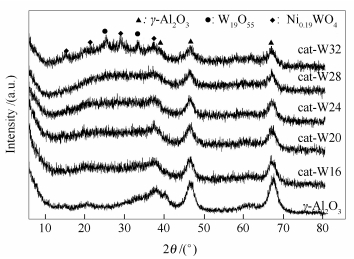
摘要: 以γ-Al2O3为载体, 采用等体积浸渍法制备了五种金属原子比相同而金属负载量不同的Ni-W基催化剂。通过X射线衍射(XRD)、光电子能谱(XPS)、程序升温脱附(NH3-TPD)、氮气吸附、高分辨透射电镜(HRTEM) 等技术对催化剂进行了表征。在固定床反应器中, 以中低温煤焦油为原料, 考察了催化剂的加氢脱氮(HDN) 和加氢脱硫(HDS) 性能。结果表明, 当Ni/W原子比为0.786时, 负载金属后, 催化剂的总酸量减少, 且以中强酸为主。随着金属负载量的增加, 催化剂的硫化程度逐渐增加, HDN活性先增加后降低, 当WO3负载量为24%时达到最优值, 而HDS活性逐渐增强。Abstract: The γ-Al2O3 supported Ni-W catalysts with different metal contents for the hydroprocessing of low and middle-temperature coal tar were prepared and characterized by the X-ray diffractogram (XRD), X-ray photoelectron spectroscopy (XPS), temperature programmed desorption of NH3 (NH3-TPD), N2 adsorption and high resolution transmission electron microscopy (HRTEM). The hydrodenitrogenation (HDN) and hydrodesulfurization (HDS) performances of the catalysts were evaluated by using low and middle temperature coal tar. The results show that the sulfidation degree of the catalysts increases with increasing metal content within a certain range, while the amount of total acid that mainly consists of intermediate acid decreases. The HDN activity of the catalysts with Ni/W atomic ratio of 0.786 increases initially and reaches an optimum value at 24% WO3, and then decreases, while the HDS activity increases gradually.
-
Key words:
- coal tar /
- hydrodenitrogenation and hydrodesulfurization /
- Ni-W /
- catalyst
-
表 1 催化剂的金属含量及配比
Table 1 Metal content and atomic ratio of the catalysts
WO3 w/% NiO w/% Ni/W Ni/(Ni+W) Cat-W16 16 4.05 0.786 0.44 Cat-W20 20 5.06 0.786 0.44 Cat-W24 24 6.07 0.786 0.44 Cat-W28 28 7.09 0.786 0.44 Cat-W32 32 8.10 0.786 0.44 表 2 中低温煤焦油的元素分析
Table 2 Chemical compositions of low and middle-temperature coal tar
Contentw/% C H N S O* 83.05 8.81 0.88 0.27 6.99 *: the oxygen content was calculated by the subtraction method 表 3 载体及催化剂的孔结构
Table 3 Pore structure parameters of the support and catalysts
Catalyst Al2O3 cat-W16 cat-W20 cat-W24 cat-W28 cat-W32 BET surface Area A/(m2·g-1) 256.86 208.46 193.04 182.34 164.56 147.38 Pore volume v/(cm3·g-1) 0.62 0.48 0.44 0.41 0.36 0.33 Average pore diameter d/nm 9.71 9.14 9.04 8.92 8.82 7.81 表 4 载体及催化剂的BJH孔结构分析
Table 4 Pore size distributions of the support and catalysts
d/nm Al2O3 cat-W16 cat-W20 cat-W24 cat-W28 cat-W32 V/cm3 w/% V/cm3 w/% V/cm3 w/% V/cm3 w/% V/cm3 w/% V/cm3 w/% >20 0.126 19.968 0.097 20.083 0.089 20.045 0.085 20.531 0.075 20.270 0.070 21.148 13-20 0.067 10.618 0.053 10.973 0.050 11.261 0.046 11.111 0.041 11.081 0.037 11.178 10-13 0.097 15.372 0.067 13.872 0.057 12.838 0.057 13.768 0.041 11.081 0.035 10.574 7-10 0.163 25.832 0.122 25.259 0.106 23.874 0.095 22.947 0.086 23.243 0.076 22.961 4-7 0.129 20.444 0.097 20.083 0.097 21.847 0.086 20.773 0.086 23.243 0.077 23.263 2-4 0.047 7.448 0.044 9.110 0.042 9.459 0.042 10.097 0.038 10.270 0.034 10.272 <2 0.002 0.317 0.003 0.621 0.003 0.676 0.003 0.725 0.003 0.811 0.002 0.604 表 5 载体及催化剂的酸性分布
Table 5 Acid distributions of the support and catalysts
Catalyst 100-200 ℃ w/% Relative content 200-350 ℃ w/% Relative content 350-550 ℃ w/% Relative content Total relative content Al2O3 4.74 1 43.67 1 51.58 1 1 Cat-W16 7.08 1.18 56.71 1.03 36.21 0.56 0.79 Cat-W20 5.77 0.77 60.42 0.87 33.81 0.41 0.63 Cat-W24 3.25 0.44 55.19 0.81 41.56 0.52 0.64 Cat-W28 5.61 0.58 57.49 0.80 36.90 0.45 0.61 Cat-W32 4.51 0.72 48.27 0.83 47.22 0.69 0.75 表 6 硫化态催化剂W和Ni的结合能和硫化程度
Table 6 Binding energy and sulfidation degree of W and Ni of the catalysts after sulfidation
Catalyst cat-W16 cat-W20 cat-W24 cat-W28 cat-W32 WS2 EB 4f7/2/eV 32.50 32.50 32.50 32.50 32.50 relative content w/% 34.51 38.59 47.77 50.27 51.97 WO3 EB 4f7/2/eV 35.90 35.90 35.90 35.90 35.90 relative content w/% 35.01 22.81 18.27 16.52 10.54 NiS EB 2p3/2(eV) 854.2 854.2 854.2 854.2 854.2 relative content w/% 20.27 26.48 28.51 39.61 43.36 NiO EB 2p3/2/eV 856.2 856.2 856.2 856.2 856.2 relative content w/% 79.73 73.52 71.49 60.39 56.64 表 7 GC-MS对煤焦油原料及加氢产物的分析
Table 7 Analysis of coal tar and hydrogenation products with GC-MS
Composition Coal tar cat-W16 cat-W20 cat-W24 cat-W28 cat-W32 Alkane 22.93 25.08 23.78 26.92 27.25 24.79 Cycloparaffin 0.64 18.27 19.62 14.44 19.14 19.57 Benzene 3.04 17.70 15.33 14.82 13.66 19.08 Phenol 31.45 0.59 0.57 0.53 0.71 1.62 Naphthalenes 18.71 23.34 23.09 22.55 23.04 19.36 Indene 2.62 7.25 8.45 11.51 9.34 8.06 Fluorene 2.67 1.99 2.02 1.94 2.34 2.00 Dibenzofuran 7.60 2.00 1.96 1.97 2.28 2.03 Anthracene phenanthrene pyrene 8.47 2.60 2.93 3.10 2.23 2.95 Else 1.83 1.18 2.25 2.22 0.01 0.54 Grand total 100.00 100.00 100.00 100.00 100.00 100.00 -
[1] 马宝岐, 任沛建, 杨占彪, 王树宽.煤焦油制燃料油[M].北京:化学工业出版社, 2011.MA Bao-qi, REN Pei-jian, YANG Zhan-biao, WANG Shu-kuan. Preparation of Fuel Oil from Coal Tar[M]. Beijing: Chemical Industry Press, 2011. [2] 李大东.加氢处理工艺与工程[M].北京:中国石化出版社, 2004.LI Da-dong. Hydrotreating Technology and Engineering[M]. Beijing: China Petrochemical Press, 2004. [3] 石垒, 张增辉, 邱泽刚, 郭芳, 张伟, 赵亮富. P改性对Mo-Ni/Al2O3煤焦油加氢脱氮性能的影响[J].燃料化学学报, 2015, 43(1): 74-80. doi: 10.1016/S1872-5813(15)60007-XSHI Lei, ZHANG Zeng-hui, QIU Ze-gang, GUO Fang, ZHANG Wei, ZHAO Liang-fu. Effect of phosphorus modification on the catalytic properties of Mo-Ni/Al2O3 in the hydrodenitrogenation of coal tar[J]. J Fuel Chem Technol, 2015, 43(1): 74-80. doi: 10.1016/S1872-5813(15)60007-X [4] GRANGE P, VANHAEREAN X. Hydrotreating catalysts, an old story with new chanllenges[J]. Catal Today, 1997, 36(4): 375-391. doi: 10.1016/S0920-5861(96)00232-5 [5] 王永刚, 张海永, 张培忠, 许德平, 赵宽, 王芳杰. NiW/γ-Al2O3催化剂的低温煤焦油加氢性能研究[J].燃料化学学报, 2012, 40(12): 1492-1497. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18088.shtmlWANG Yong-gang, ZHANG Hai-yong, ZHANG Pei-zhong, XU De-ping, ZHAO Kuan, WANG Fang-jie. Hydroprocessing of low temperature coal tar on NiW/γ-Al2O3 catalyst[J]. J Fuel Chem Technol, 2012, 40(12): 1492-1497. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18088.shtml [6] LAREDO S G C, DELOSREYES H J A, LUISCANO D J. Inhibition effects of nitrogen compounds on the hydrodesulfurization of dibenzothiophene[J]. Appl Catal A: Gen, 2001, 207(1/2): 103-112. https://www.researchgate.net/profile/Georgina_Laredo/publication/223685716_Inhibition_Effects_of_Nitrogen_Compounds_on_the_Hydrodesulfurization_of_Dibenzothiophene/links/53d10f7a0cf2fd75bc5d6555.pdf?origin=publication_detail [7] LAREDO S G C, MONTESINOS A, ANTONIO DE LOS REYES J. Inhibition effects observed between dibenzothiophene and carbazole during the hydrotreating process[J]. Appl Catal A: Gen, 2004, 265(2): 171-183. doi: 10.1016/j.apcata.2004.01.013 [8] 张海永, 王永刚, 张培忠, 林雄超, 朱豫飞. NiW/Al2O3-Y催化剂的制备及其对煤焦油加氢处理的研究[J].燃料化学学报, 2013, 41(9): 1085-1091. doi: 10.1016/S1872-5813(13)60046-8ZHANG Hai-yong, WANG Yong-gang, ZHANG Pei-zhong, LIN Xiong-chao, ZHU Yu-fei. Preparation of NiW catalysts with alumina and zeolite Y for hydroprocessing of coal tar[J]. J Fuel Chem Technol, 2013, 41(9): 1085-1091. doi: 10.1016/S1872-5813(13)60046-8 [9] LAREDOS G C, ALTAMIRANO E, DELOSREYES J. Inhibition effects of nitrogen compounds on the hydrodesulfurization of dibenzothiophene: Part 2[J]. Appl Catal A: Gen, 2003, 243(2): 207-214. doi: 10.1016/S0926-860X(02)00321-6 [10] QIAN E W, ABE S, KAGAWA Y, IKEDA H. Hydrodenitrogenation of porphyrin on Ni-Mo based catalysts[J]. Chin J Catal, 2013, 34(1): 152-158. doi: 10.1016/S1872-2067(11)60514-7 [11] LEWANDOWSKI M, SARBAK Z. The effect of boron addition on hydrodesulfurization and hydrodenitrogenation activity of NiMo/Al2O3 catalysts[J]. Fuel, 2000, 79(5): 487-495. doi: 10.1016/S0016-2361(99)00151-9 [12] 石垒. Mo-Ni/Al2O3催化剂对煤焦油加氢脱氮性能的研究[D].太原:中国科学院山西煤炭化学研究所, 2014.SHI Lei. Studies on the performance of Mo-Ni/Al2O3 catalyst in the hydrodenitrogenation of coal tar[D]. Tauyuan: Institute of Coal Chemistry Chinese Aacdemy of Sciences, 2014. [13] ZHANG L, AFANASIEV P, LI D D, LONG X Y, VRINAT M.Solution synthesis of the unsupported Ni-Wsulfide hydrotreating catalysts[J]. Catal Today, 2007, 130(1): 24-31. http://www.cqvip.com/qk/90650x/201309/47435291.html [14] KARAKONSTANTIS L, MATRALIS H, KORDULIS C. Tungsten-oxo-species deposited on alumina. 2. Characterization and catalytic activity of unpromoted W-(Ⅵ)/gamma-Al2O3catalysts prepared by equilibrium deposition filtration (EDF) at various pH's and non-dry impregnation (NDI)[J]. J Catal, 1996, 162(2): 306-319. doi: 10.1006/jcat.1996.0288 [15] 郑禄彬.新固体酸和碱及其相互作用[M].北京:化学工业出版社, 1992.ZHENG Lu-bin. New Solid Acid and Alkali as Well as Their Interaction[M]. Beijing: Chemical Industry Press, 2011. [16] DING L H, ZHENG Y, ZHANG Z S, RING Z, CHEN J W. Hydrotreating of light cycle oil using WNi catalysts containing hydrothermally and chemically treated zeolite Y[J]. Catal Today, 2007, 125(3): 229-238. https://www.researchgate.net/publication/229101775_Hydrotreating_of_light_cycle_oil_using_WNi_catalysts_containing_hydrothermally_and_chemically_treated_zeolite_Y [17] DUGULAN A I, HENSEN E J M, VAN VEEN J A R. Effect of pressure on the sulfidation behavior of NiW catalysts: A 182W Mossbauer spectroscopy study[J]. Today, 2010, 150(3/4): 224-230. https://www.researchgate.net/publication/244323097_Effect_of_pressure_on_the_sulfidation_behavior_of_NiW_catalysts_A_182W_Mossbauer_spectroscopy_study [18] HENSENE J M, VAN DER MEER Y, VAN VEEN J A R. Insight into the formation of the active phases in supported NiW hydrotreating catalysts[J]. Appl Catal A: Gen, 2007, 322: 16-32. doi: 10.1016/j.apcata.2007.01.003 [19] YU F, HAN X, GANG S, LIU H Y, QIAN Y, WANG T H, GONG G B, BAO X J. Citrc acid-assisted hydrothermal method for preparing NiW/USY-Al2O3 ultradeep hydrodesulfurization catalysts[J]. J Catal, 2011, 279(5): 27-35. https://www.researchgate.net/publication/256737599_Citric_acid-assisted_hydrothermal_method_for_preparing_NiWUSY-Al2O3_ultradeep_hydrodesulfurization_catalysts [20] 聂红, 龙湘云, 刘清河, 李大东.柠檬酸对NiW/Al2O3加氢脱硫催化剂硫化行为的影响[J].石油学报(石油加工), 2010, 26(3): 329-335. http://www.cqvip.com/qk/94167x/201003/34293150.htmlNIE Hong, LONG Xiang-yun, LIU Qing-he, LI Da-dong. Effect of citric acid on sulfidation behavior of NiW/Al2O3 hydrodesulfurization catalyst[J]. Acta Pet Sin (Pet Process Sect), 2010, 26(3): 329-335. http://www.cqvip.com/qk/94167x/201003/34293150.html [21] ENGELEN C W R, WOLTHUIZEN J P, VANHOOFF J H C. Reactions of propane over a bifunctional Pt/H-ZSM-5 catalyst[J]. Appl Catal, 1985, 19(1): 153-163. doi: 10.1016/S0166-9834(00)82677-9 [22] BRAGIN O V, SHPIRO E S, PREOBRAZGHENSKY A V, PALISHKINA N V, VASINA TV, DYUSENBINA B B, ANTOSHIN G V, MINACHEV K M. The state of platinum in high-silica zeolites and its catalytic activity in ethane aromatization[J]. Bull Acad Sci USSR Div Chem Sci, 1986, 35(1): 10-15. doi: 10.1007/BF00952835 [23] SATTERFIELD C N, MODELL M, MAYER J F. Interactions between catalytic hydrodenitrogenation of Hydrodesulfurization thiopene and hydrodenitrogenation of Pyridine[J]. AIChE J, 1975, 21(6): 1100-1107. doi: 10.1002/(ISSN)1547-5905 [24] STANISLAUS A, MARAFI A, RANA M S. Recent advances in the science and technology of ultra low sulfur diesel (ULSD) production[J]. Catal Today, 2010, 153(1/2): 1-68. http://www.sciencedirect.com/science/article/pii/S0920586110003299 [25] EGOROVA M, PRINS R. Competitive hydrodesulfurization of 4, 6-dimethyl dibenzothiophene, hydrodenitrogenation of 2-methylpyridine, and hydrogenation of naphthalene over sulfided NiMo/γ-Al2O3[J]. J Catal, 2004, 244(2): 278-287. [26] EGOROVA M, ROEL P. Mutual influence of the HDS of dibenzothiophene and HDN of 2-methylpyridine[J]. J Catal, 2004, 221(1): 11-19. doi: 10.1016/S0021-9517(03)00264-1 [27] SUNDARAMURTHY V, DALAI AK, ADJAYE J. The effect of phosphorus on hydrotreating property of NiMo/γ-Al2O3 nitride catalyst[J]. Appl Catal A: Gen, 2008, 335(2): 204-210. doi: 10.1016/j.apcata.2007.11.024 [28] JIAN M, PRINS R. Existence of different catalytic sites in HDN catalysts[J]. Catal Today, 1996, 30(1/3): 127-134. http://www.ingentaconnect.com/content/els/09205861/1996/00000030/00000001/art00326 [29] HOUALLA M, NAG N K, SAPRE A V. Hydrodesulfurization of dibenzothiophenes catalyzed by sulfided CoMo/γ-Al2O3[J]. React Net, 1978, 24(4): 1015-1021. -





 下载:
下载: