Promoting effect of SO42- functionalization on the performance of Fe2O3 catalyst in the selective catalytic reduction of NOx with NH3
-
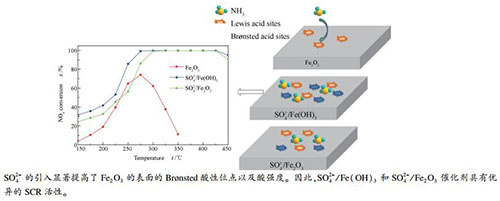
摘要: 采用沉淀法制备了Fe(OH)3和Fe2O3。通过硫酸化处理得到SO42-/Fe(OH)3和SO42-/Fe2O3两种催化剂,并将其应用于氨选择性催化还原NOx(NH3-SCR)反应,研究了SO42-功能化处理对Fe2O3催化剂上NH3-SCR脱硝性能的促进机理。结果表明,与纯的Fe2O3相比,硫酸化处理得到的催化剂上SCR活性得到显著提升;其中,SO42-/Fe(OH)3表现出更加优异的催化性能,在250-450 ℃时NOx转化率高于80%,且具有优异的稳定性和抗H2O + SO2性能。XRD、Raman、TG、FT-IR、H2-TPR、NH3-TPD和in situ DRIFTS等表征结果显示,硫酸功能化处理能抑制Fe2O3的晶粒生长,同时SO42-与Fe3+结合形成硫酸盐复合物,提高了催化剂表面酸性位点的数量和酸强度,抑制了Fe2O3上的氨氧化反应,从而提高了其脱硝催化性能。Abstract: Fe(OH)3 and Fe2O3 were first prepared by a precipitation method and then sulfated to obtain the SO42--functionalized SO42-/Fe(OH)3 and SO42-/Fe2O3 catalysts for the selective catalytic reduction of NOx with NH3 (NH3-SCR); the promoting effect of SO42- functionalization on the performance of Fe2O3 catalyst in NH3-SCR was then investigated. The results indicate that the SCR activity of the SO42--functionalized Fe2O3 catalysts is significantly improved in comparison with that of unmodified Fe2O3. In particular, the SO42-/Fe(OH)3 catalyst exhibits excellent performance in NH3-SCR, with the NOx conversion of over 80% at 250-450 ℃; besides, it also display high catalytic stability and resistance towards H2O + SO2. A series of characterization results including XRD, Raman spectroscopy, TG analysis, FT-IR spectroscopy, H2-TPR, NH3-TPD and in situ DRIFTS reveal that the functionalization with sulfuric acid can inhibit the growth of Fe2O3 grains; moreover, SO42- combines with Fe3+ to form the sulfate complex, leading to an increase in the number of surface acid sites and the acid strength, which can inhibit the ammonia over-oxidation on Fe2O3 and enhance the deNOx performance of Fe2O3.
-
表 1 三种样品的比表面积、平均孔径和孔容
Table 1 Surface area, average pore size and pore volume of various catalysts
Sample Surface area A/(m 2·g -1) Pore diameter d/nm Pore volume v/(cm 3·g -1) Fe 2O 3 41.12 20.01 0.201 SO 4 2-/Fe(OH) 3 49.98 17.12 0.298 SO 4 2-/Fe 2O 3 37.25 25.88 0.189 表 2 三种样品的Fe2O3晶粒粒径和NH3脱附峰面积
Table 2 Crystallite size of Fe2O3 and NH3 desorption peak areas of various catalysts
Catalyst Fe2O3 crystallite size /nm Area of NH3 desorption peaks /(10-9) Fe2O3 28 3.9 SO42-/Fe(OH)3 16 80.0 SO42-/Fe2O3 20 5.6 -
[1] SUN C Z, LIU H, CHEN W, CHEN D Z, YU S H, LIU A N, DONG L, FENG S. Insights into the Sm/Zr co-doping effects on N2 selectivity and SO2 resistance of a MnOx-TiO2 catalyst for the NH3-SCR reaction[J]. Chem Eng J, 2018, 347:27-40. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=38afac51aa0a94f072055c97b0a8cef6 [2] LIU Z M, SU H, LI J H, LI Y. Novel MoO3/CeO2-ZrO2 catalyst for the selective catalytic reduction of NOx by NH3[J]. Catal Commun, 2015, 65:51-54. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=934b07f85c2fae2fe29c92b536393996 [3] MARBERGERA A, FERRI D, ELSENER M, SAGAR A, ARTNER C, SCHERMANZ K, KROCHER O. Relationship between structures and activities of supported metal vanadates for the selective catalytic reduction of NO by NH3[J]. Appl Catal B:Environ, 2017, 218:731-742. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=618e7719739bb4fe3850054ce5f62918 [4] 闫春迪, 程昊, 王树东. Cu交换量对Cu-SAPO-34催化剂上NH3选择性催化还原NOx反应的影响[J].燃料化学学报, 2014, 42(6):743-750. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18438.shtmlYAN Chun-di, CHENG Hao, WANG Shu-dong. Effects of copper content in Cu-SAPO-34 on its catalytic performance in NH3-SCR of NOx[J]. J Fuel Chem Technol, 2014, 42(6):743-750. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18438.shtml [5] PUTLURU S S R, SCHILL L, GODIKSEN A, POREDDY R, MOSSIN S, JENSEN A D, FEHRMANN R. Promoted V2O5/TiO2 catalysts for selective catalytic reduction of NO with NH3 at low temperatures[J]. Appl Catal B:Environ, 2016, 183:282-290. https://www.sciencedirect.com/science/article/abs/pii/S0926337315302253 [6] YANG Y J, LIU J, LIU F, WANG Z, DING J Y, HUANG H. Reaction mechanism for NH3-SCR of NOx over CuMn2O4 catalyst[J]. Chem Eng J, 2019, 361:578-587. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=2e26455081e8cccaf5a132ea5e602f2a [7] YAO X J, KONG T T, CHEN L, DING S M, YANG F M, DONG L, Enhanced low-temperature NH3-SCR performance of MnOx/CeO2 catalysts by optimal solvent effect[J]. Appl Surf Sci, 2017, 420:407-415. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=0168781038fd5fba2e3f024f1658fd85 [8] ZHANG T, CHANG H Z, LI K Z, PENG Y, LI X, LI J H. Different exposed facets VOx/CeO2 catalysts for the selective catalytic reduction of NO with NH3[J]. Chem Eng J, 2018, 349:184-191. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=985fd851e8ec24c1062c6453242b948e [9] ANDREOLI S, DEORSOLA F A, PIRONE R, MnOx-CeO2 catalysts synthesized by solution combustion synthesis for the low-temperature NH3-SCR[J]. Catal Today, 2015, 253:199-206. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=8dd95d7f44044dfdeab436bc1721bdb5 [10] 刘昕, 宁平, 李昊, 宋忠贤, 王艳彩, 张金辉, 唐小苏, 王明智, 张秋林.水相法制备Ce-W@TiO2催化剂的氨选择性催化还原NO(NH3-SCR)活性和抗SO2性能研究[J].燃料化学学报, 2016, 44(2):225-231. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18783.shtmlLIU Xin, NING Ping, LI Hao, SONG Zhong-xian, WANG Yan-cai, ZHANG Jin-hui, TANG Xiao-su, WANG Ming-zhi, ZHANG Qiu-lin. Probing NH3-SCR catalytic activity and SO2 resistance over aqueous-phase synthesized Ce-W@TiO2 catalyst[J]. J Fuel Chem Technol, 2016, 44(2):225-231. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18783.shtml [11] KANG L, HAN L P, HE J B, LI H R, YAN T T, CHEN G R, ZHANG J P, SHI L Y, ZHANG D S. Improved NOx reduction in the presence of SO2 by using Fe2O3-promoted halloysite-supported CeO2-WO3 catalysts[J]. Environ Sci Technol, 2019, 53:938-945. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6e8cb2b704428cf152b94e0a9b75711f [12] YE D, REN X Y, QU R Y, LIU S J, ZHENG C H, GAO X. Designing SO2-resistant cerium-based catalyst by modifying with Fe2O3 for the selective catalytic reduction of NO with NH3[J]. Mol Catal, 2019, 462:10-18. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=61fd54225a5bc30afa0c3606a40f92df [13] LIU Z M, SU H, CHEN B C, LI J H, WOO S I. Activity enhancement of WO3 modified Fe2O3 catalyst for the selective catalytic reduction of NOx by NH3[J]. Chem Eng J, 2016, 299:255-262. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=eb6c4a62c51458cc19520dfac87b9efb [14] XIONG Z B, NING X, ZHOU F, YANG B, TU Y W, JIN J, LU W, LIU Z H. Environment-friendly magnetic Fe-Ce-W catalyst for the selective catalytic reduction of NOx with NH3:Influence of citric acid content on its activity-structure relationship[J]. RSC Adv, 2018, 8:21915-21925. http://pubs.rsc.org/-/content/articlelanding/2018/ra/c8ra03131b [15] 王继封, 王慧敏, 张亚青, 张秋林, 宁平. WO3的引入对MnOx-Fe2O3催化剂上NH3-SCR反应中N2选择性的促进作用[J].燃料化学学报, 2019, 47(7):814-822. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract29417.shtmlWANG Ji-feng, WANG Hui-min, ZHANG Ya-qing, ZHANG Qiu-lin, NING Ping. Promotion effect of tungsten addition on N2 selectivity of MnOx-Fe2O3 for NH3-SCR[J]. J Fuel Chem Technol, 2019, 47(7):814-822. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract29417.shtml [16] SHAN W P, LIU F D, HE H, SHI X Y, ZHANG C B. A superior Ce-W-Ti mixed oxide catalyst for the selective catalytic reduction of NOx with NH3[J]. Appl Catal B:Environ, 2012, 115/116:100-106. https://www.sciencedirect.com/science/article/abs/pii/S0926337311005893 [17] LIU Z M, ZHANG S X, LI J H, MA L L. Promoting effect of MoO3 on the NOx reduction by NH3 over CeO2/TiO2 catalyst studied with in situ DRIFTS[J]. Appl Catal B:Environ, 2014, 144:90-95. https://www.sciencedirect.com/science/article/abs/pii/S0926337313004190 [18] CHENG K, LIU B, SONG W Y, LIU J, CHEN Y S, ZHAO Z, WEI Y C. Effect of Nb promoter on the structure and performance of iron titanate catalysts for the selective catalytic reduction of NO with NH3[J]. Ind Eng Chem Res, 2018, 57(23):7802-7810. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=92a52406e4535d98fc1bcc0826384451 [19] ZEB F, ISHAQUE M, NADEEM K, KAMRAN M, KRENN H, SZABO D V, BROSSMANN U, LETOFSKY-PAPST I. Reduced surface effects in weakly interacting ZrO2 coated MnFe2O4 nanoparticles[J]. J Magn Magn Mater, 2019, 469:580-586. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=ededfaaca2e9559b4c55b83a5619d9b8 [20] 牛茹洁, 王成章, 刘晓越, 易春雄, 陈梁, 米铁, 吴正舜.负载Fe2O3的Zr基钙钛矿催化剂对逆水煤气反应的影响[J].燃料化学学报, 2019, 47(3):92-97. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb201901012NIU Ru-jie, WANG Cheng-zhang, LIU Xiao-yue, YI Chun-xiong, CHEN Liang, MI Tie, WU Zheng-shun. Preparation of Zr-based perovskite supported Fe2O3 catalyst and its performance in the reverse water gas shift reaction[J]. J Fuel Chem Technol, 2019, 47(3):92-97. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb201901012 [21] MANSOUR H, BARGOUGUI R, AUTRET-LAMBERT C, GADRI A, AMMAR S. Co-precipitation synthesis and characterization of tin-doped α-Fe2O3 nanoparticles with enhanced photocatalytic activities[J]. J Phys Chem Solids, 2018, 114:1-7. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=ccc7bcbdd4cb21d0f175665f7156b10c [22] GUO S, WANG H J, YANG W, FIDA H, YOU L M, ZHOU K. Scalable synthesis of Ca-doped α-Fe2O3 with abundant oxygen vacancies for enhanced degradation of organic pollutants through peroxymonosulfate activation[J]. Appl Catal B:Environ, 2020, 262:118250-118261. https://www.sciencedirect.com/science/article/abs/pii/S092633731930997X [23] SONG L Y, CHAO J D, FANG Y J, HE H, LI J, QIU W G, ZHANG G Z. Promotion of ceria for decomposition of ammonia bisulfate over V2O5-MoO3/TiO2 catalyst for selective catalytic reduction[J]. Chem Eng J, 2016, 303:275-281. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=3c6e2d58684e930fc2571a5038943185 [24] GAO S, CHEN X B, WANG H Q, MO J S, WU Z B, LIU Y, WENG X L. Ceria supported on sulfated zirconia as a superacid catalyst for selective catalytic reduction of NO with NH3[J]. J Colloid Interface Sci, 2013, 394:515-521. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=4dd2a41bf7d2d7ec506b236348b54d33 [25] CHANG H Z, MA L L, YANG S J, LI J H, CHEN L, WANG W, HAO J M. Comparison of preparation methods for ceria catalyst and the effect of surface and bulk sulfates on its activity toward NH3-SCR[J]. J Hazard Mater, 2013, 262:782-788. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=04a8e043b2c0a6bdd24abb82b1032c3e [26] QU R Y, GAO X, CEN K F, LI J H. Relationship between structure and performance of a novelcerium-niobium binary oxide catalyst for selective catalytic reduction of NO with NH3[J]. Appl Catal B:Environ, 2013, 142-143:290-297. https://www.sciencedirect.com/science/article/abs/pii/S0926337313003299 [27] ZIC M, RISTIC M, MUSIC S. 57Fe Mössbauer, FT-IR and FE SEM investigation of the formation of hematite and goethite at high pH values[J]. J Mol Struct, 2007, 834/836:141-149. doi: 10.1016-j.molstruc.2006.10.030/ [28] YAN Z X, XU Z H, YANG Z H, YUE L, HUANG L Y. Graphene oxide/Fe2O3 nanoplates supported Pt for enhanced room-temperature oxidation of formaldehyde[J]. Appl Surf Sci, 2019, 467/468:277-285. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=de63141fa111869a01b2e2f1efd5fdbc [29] YAMAGUCHI T, JIN T, ISHIDA I, TANABE K. Structural identification of acid sites of sulfur-promoted solid super acid and construction of its structure on silica support[J]. Mater Chem Phys, 1986, 17(1/2):3-19. https://www.sciencedirect.com/science/article/abs/pii/0254058487900459 [30] KAYO A, YAMAGUCHI T, TANABE K, The effect of preparation method on the acidic and catalytic properties of iron oxide[J]. J Catal, 1983, 83(1):99-106. https://www.sciencedirect.com/science/article/pii/0021951783900337 [31] ZIELINSKI J, ZGLINICKA I, ZNAK L, KASZKUR Z, Reduction of Fe2O3 with hydrogen[J]. Appl Catal A:Gen, 2010, 381(1/2):191-196. http://d.old.wanfangdata.com.cn/OAPaper/oai_doaj-articles_fe66a93bbec32aeec53f90b2866831bc [32] LI H J, SONG H L, CHEN L W, XIA C G, Designed SO42-/Fe2O3-SiO2 solid acids for polyoxymethylene dimethyl ethers synthesis:the acid sites control and reaction pathways[J]. Appl Catal B:Environ, 2015, 165:466-476. https://www.sciencedirect.com/science/article/abs/pii/S0926337314006468 [33] MENG D M, XU Y, GUO Y L, GUO Y, WANG L L, LU G Z, ZHAN W C. Spinel structured CoaMnbOx mixed oxide catalyst for the selective catalytic reduction of NOx with NH3[J]. Appl Catal B:Environ, 2018, 221:652-663. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=be59ea36246427c2d3f503e88e26fc84 [34] WANG X X, SHI Y, LI S J, LI W. Promotional synergistic effect of Cu and Nb doping on a novel Cu/Ti-Nb ternary oxide catalyst for the selective catalytic reduction of NOx with NH3[J]. Appl Catal B:Environ, 2018, 220:234-250. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=1b4501cf82aedb05bb7fbb2545171004 [35] MU J C, LI X Y, SUN W B, FAN S Y, WANG X Y, WANG L, QIN M C, GAN G Q, YIN Z F, ZHANG D K. Inductive effect boosting catalytic performance of advanced Fe1-xVxOδ catalysts in low-temperature NH3 selective catalytic reduction:Insight into the Structure, Interaction, and Mechanisms[J]. ACS Catal, 2018, 8:6760-6774. https://pubs.rsc.org/-/content/articlelanding/2018/ra/c8ra03131b [36] GAO C, SHI J W, FAN Z Y, WANG B R, WANG Y, HE C, WANG X B, LI J, NIU C M. "Fast SCR" reaction over Sm-modified MnOx-TiO2 for promoting reduction of NOx with NH3[J]. Appl Catal A:Gen, 2018, 564:102-112. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=71948277ab1004a5307804c341edf21b [37] MA S B, ZHAO X Y, LI Y S, ZHANG T R, YUAN F L, NIU X Y, ZHU Y J. Effect of W on the acidity and redox performance of the Cu0.02Fe0.2WaTiOx (a=0.01, 0.02, 0.03) catalysts for NH3-SCR of NO[J]. Appl Catal B:Environ, 2019, 248:226-238. https://www.sciencedirect.com/science/article/abs/pii/S0926337319301262 [38] GUO M Y, LIU Q L, ZHAO P P, HAN J F, LI X, HA Y, FU Z C, SONG C F, JI N, LIU C X, MA D G, LI Z G. Promotional effect of SO2 on Cr2O3 catalysts for the marine NH3-SCR reaction[J]. Chem Eng J, 2019, 361:830-838. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=2dbfbc5e646dfdaefef41a547c7d2ed5 -





 下载:
下载: