Improvement of stability of nano-SiO2/HPAM/SDS dispersion systems and its effect on oil displacement performances
-
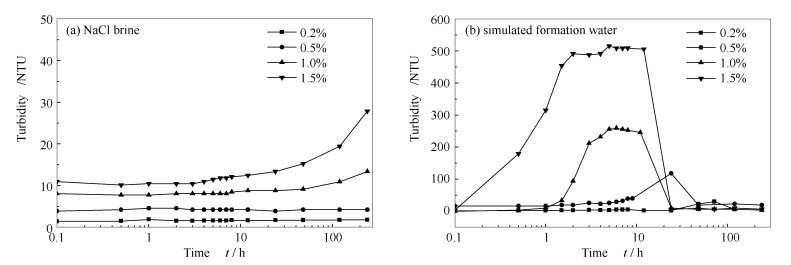
摘要: 分析了60℃、1.0×104 mg/L氯化钠盐水和模拟地层水中纳米SiO2/HPAM/SDS分散体系的浊度实验及Zeta电位,发现Ca2+、Mg2+离子是体系失去稳定性的主要原因。根据沉降实验及Zeta电位分析仪探讨了降低pH值和添加络合剂对模拟地层水中纳米SiO2/HPAM/SDS体系稳定性的改善效果及机理,同时利用流变仪及界面张力仪分析了两种方法对体系驱油性能的影响。结果表明,pH值降低,体系的Zeta电位绝对值降低,但SiO2周围H+保护层的形成及水化作用力的增强改善了体系的稳定性;络合剂Na2EDTA、ATMP和Na4EDTA均能增强体系的稳定性,Na2EDTA和ATMP络合Ca2+、Mg2+的同时降低了体系的pH值,而体系的黏度随pH值的降低急剧下降;Na4EDTA加入后,体系的pH值增大,稳定配位化合物的形成使体系的Zeta电位绝对值、黏度、储能模量和损耗模量增加,降低界面张力的能力增强。因此,在SiO2质量分数为0.5%的体系中加入质量分数为0.4%的Na4EDTA(最佳质量分数),采收率提高了3.1%。Abstract: The turbidity and Zeta potential experiments of nano-SiO2/HPAM/SDS dispersion systems at 60 ℃, 1.0×104 mg/L NaCl brine and simulated formation water were first investigated and the results indicated that Ca2+ and Mg2+ ions were responsible for the instability of dispersion system. Then, the methods of reducing pH value and adding chelating agents were used to improve the stability of nano-SiO2/HPAM/SDS dispersion system in simulated formation water, and the improvement effects and mechanisms were discussed based on sedimentation experiments and Zetasizer. The influences of these two methods on the oil displacement performances of dispersion system were analyzed by rheometer and interfacial tensiometer. The experimental results suggested that with the decrease of pH value, the stability of dispersion system was enhanced by the protection of H+ in the SiO2 double electric layer and the enhancement of hydration forces between particles although the absolute value of Zeta potential(|ζ|) of dispersion system decreased. All of the chelating agents including Na2EDTA, ATMP and Na4EDTA can improve the stability of dispersion system. Ca2+ and Mg2+ ions were complexed and pH value of dispersion system was decreased with the addition of Na2EDTA and ATMP, however, the decrease of pH value resulted in a sharp deterioration in the viscosity of dispersion systems. With the addition of Na4EDTA, the pH value of dispersion systems increased. The|ζ|, viscosity, storage modulus and loss modulus of dispersion systems all increased, which were benefited from the formation of stable complexes between Na4EDTA, and Ca2+ and Mg2+. Meanwhile, the ability of dispersion system to reduce oil and water interfacial tension was enhanced. Thus, after addition of Na4EDTA with mass ratio of 0.4% (optimal mass ratio)in dispersion system with nano-SiO2 mass ratio of 0.5%, the oil recovery was increased by 3.1%.
-
Key words:
- nano-SiO2 /
- pH value /
- chelating agents /
- stability /
- oil displacement performance
-
表 1 纳米SiO2/HPAM/SDS分散体系的Zeta电位
Table 1 Zeta potential of nano-SiO2/HPAM/SDS dispersion systems
w (SiO2)/% 0.2 0.5 1.0 1.5 Formation water -29.7 -24.2 -21.1 -18.8 NaCl solution -45.3 -39.1 -37.6 -33.5 表 2 岩心参数及各阶段采收率
Table 2 Physical parameters of core samples and oil recovery at each production stage
Pore volume V/mL Permeability /(10-3 μm2) Original oil saturation /% Chemical formula Recovery /% water flood recovery tertiary recovery final recovery 11.0 1358.0 76.4 SiO2/HPAM/SDS 41.0 23.9 64.9 10.8 1345.0 77.2 SiO2/HPAM/SDS + Na4EDTA 40.8 27.0 67.8 -
[1] 刘潇潇.聚驱后聚/表二元驱驱油效果及流度控制研究[D].大庆: 东北石油大学, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10220-1015362756.htmLIU Xiao-xiao. Study on displacement effect factors of surfactant-polymer system and method of mobility control after polymer[D]. Daqing: Northeast Petroleum University, 2015. http://cdmd.cnki.com.cn/Article/CDMD-10220-1015362756.htm [2] THOMAS S, ALI S M F, SCOULAR J R, VERKOCZY B. Chemical methods for heavy oil recovery[J]. J Can Pet Technol, 2001, 40(3):56-61. http://www.researchgate.net/publication/266663654_chemical_methods_for_heavy_oil_recovery [3] 李美蓉, 柳智, 宋新旺, 马宝东, 张文.金属阳离子对聚丙烯酰胺溶液黏度的影响及其降黏机理研究[J].燃料化学学报, 2012, 40(1):43-47. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb201201007LI Mei-rong, LIU Zhi, SONG Xin-wang, MA Bao-dong, ZHANG Wen. Effect of metal ions on the viscosity of polyacrylamide solution and the mechanism of viscosity degradation[J]. J Fuel Chem Technol, 2012, 40(1):43-47. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb201201007 [4] ZHANG L, ZHANG D, JIANG B. The rheological behavior of salt tolerant polyacrylamide solutions[J]. Chem Eng Technol, 2006, 29:395-400. doi: 10.1002/ceat.200500306 [5] 孙宠.纳米SiO2颗粒稳定泡沫体系的研制及性能评价[D].大庆: 东北石油大学, 2018. http://cdmd.cnki.com.cn/Article/CDMD-10220-1018163133.htmSUN Chong. Preparation and performance evaluation of modified nano silica stabilized foam system[D]. Daqing: Northeast Petroleum University, 2018. http://cdmd.cnki.com.cn/Article/CDMD-10220-1018163133.htm [6] 王瑶.纳米流体在储层岩芯表面的铺展及其驱油机理研究[D].西安: 西安石油大学, 2015. http://d.wanfangdata.com.cn/thesis/Y2796652WANG Yao. A study on nanofluids spreading on the reservoir cores surface and its displacement mechanism[D]. Xian: Xi'an Shiyou University, 2015. http://d.wanfangdata.com.cn/thesis/Y2796652 [7] CORREDOR R L M, SARAPARDEH A H, HUSEIN M M, DONG P M.Rheological behavior of surface modified silica nanoparticles dispersed in partially hydrolyzed polyacrylamide and xanthan gum solutions:Experimental measurements, mechanistic understanding, and model development[J].Energy Fuels, 2018, 32(10):10628-10638. doi: 10.1021/acs.energyfuels.8b02658 [8] MOHAMMED B A, KOUROSH R, RADZUAN J, ALI E B.Appraising the impact of metal-oxide nanoparticles on rheological properties of HPAM in different electrolyte solutions for enhanced oil recovery[J]. J Pet Sci Eng, 2019, 172:1057-1068. doi: 10.1016/j.petrol.2018.09.013 [9] BINKS B P, RODRIGUES J A, FRITH W J. Synergistic interaction in emulsions stabilized by a mixture of silica nanoparticles and cationic surfactant[J]. Langmuir, 2007, 23(14):3626-3636. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6a1d23b02d38902303096e6876a206f6 [10] KHALILINEZHAD S S, CHERAGHIAN G, ROAYAEI E, TABATABAEE H, KARAMBEIGI M S. Improving heavy oil recovery in the polymer flooding process by utilizing hydrophilic silica nanoparticles[J]. Energy Source Part A, 2017, 12:1-10. doi: 10.1080/15567249.2014.881931 [11] SAHA R, UPPALURI R V S, TIWARI P. Silica nanoparticle assisted polymer flooding of heavy crude oil:Emulsification, rheology, and wettability alteration characteristics[J]. Ind Eng Chem Res, 2018, 57:6364-6376. doi: 10.1021/acs.iecr.8b00540 [12] 覃孝平.改性纳米SiO2/AA/AM共聚物提高波及效率的可行性研究[D].成都: 西南石油大学, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10615-1014419507.htmTAN Xiao-ping. The feasible research of improving sweep efficiency using the modified nano-SiO2/AA/AM copolymer[D]. Chengdu: Southwest Petroleum University, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10615-1014419507.htm [13] 郑超.表面改性纳米二氧化硅的制备及其对HPAM驱油性能的影响[D].开封: 河南大学, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10475-1017227144.htmZHENG Chao.Preparation of surface modified nanosilica and its impact on enhanced oil recovery of HPAM solution[D]. Kaifeng: Henan University, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10475-1017227144.htm [14] 王海英, HPAM溶液黏度影响因素研究及络合剂的选择[D].大庆: 东北石油大学, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10220-1014384402.htmWANG Hai-ying. Study in the factors that influence HPAM solution viscosity and select the complexing agent[D]. Daqing: Northeastern Petroleum University, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10220-1014384402.htm [15] AMIRI A, ØYE G, SJOBLOM J. Influence of pH, high salinity and particle concentration on stability and rheological properties of aqueous suspensions of fumed silica[J]. Colloid Surface A, 2009, 349:43-54. doi: 10.1016/j.colsurfa.2009.07.050 [16] 陈五花, 王业飞, 何臻培, 丁名臣.纳米SiO2/HPAM/NaCl分散体系的稳定性、流变性及驱油性能研究[J].燃料化学学报, 2020, 48(5):568-576. http://www.ccspublishing.org.cn/article/id/75fab549-3c5c-4c15-9aa9-aaaadaa26470CHEN Wu-hua, WANG Ye-fei, HE Zhen-pei, DING Ming-chen.Stability, rheology and displacement performance of nano-SiO2/HPAM/SDS dispersion systems[J]. J Fuel Chem Technol, 2020, 48(5):568-576. http://www.ccspublishing.org.cn/article/id/75fab549-3c5c-4c15-9aa9-aaaadaa26470 [17] 沈钟, 赵振国, 康万利.胶体与表面化学[M].北京:化学工业出版社, 2012, 5.SHEN Zhong, ZHAO Zhen-guo, KANG Wan-li. Colloid and Surface Chemistry[M]. Beijing:Chemical Industry Press, 2012, 5. [18] AHUALLI S, IGLESIAS G R, WACHTER W. Adsorption of anionic and cationic surfactants on anionic colloids:Supercharging and destabilization[J]. Langmuir, 2011, 27(15):9182-9192. doi: 10.1021/la201242d [19] 李慧.莠灭净悬浮剂的制备及其稳定性研究[D].泰安: 山东农业大学, 2012. http://cdmd.cnki.com.cn/Article/CDMD-10434-1012487277.htmLI Hui. Study on the preparation and stability of Ametryne suspension concentrate[D]. Taian: Shandong Agultural University, 2012. http://cdmd.cnki.com.cn/Article/CDMD-10434-1012487277.htm [20] 彭陈亮, 闵凡飞, 赵晴, 李宏亮.微细矿物颗粒表面水化膜研究现状及进展综述[J].矿物学报, 2012, 32(4):515-522. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=kwxb201204009PENG Chen-liang, MIN Fan-fei, ZHAO Qing, LI Hong-liang. A review:Research status and process on hydration layers near fine mineral particles[J]. Acta Mineral Sin, 2012, 32(4):515-522. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=kwxb201204009 [21] 张云.分析化学[M].上海:同济大学出版社, 2003, 9.ZHAN Yun. Analytical Chemistry[M]. Shanghai:Tongji University Press, 2003, 9. [22] 张荣华, 朱志良, 邓守权, 倪亚明. ATMP对钙、镁离子阻垢作用机理的配位化学研究[J].工业水处理, 2003, 23(7):25-27. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=gyscl200307008ZHANG Rong-hua, ZHU Zhi-liang, DENG Shou-quan, NI Ya-ming. Study on the coordination chemistry of ATMP with Ca2+, Mg2+ in aqueous solution[J]. Ind Water Treat, 2003, 23(7):25-27. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=gyscl200307008 [23] 李晓阳. Ca(EDTA)2-络合物及有机酸钙的量化计算研究[D].北京: 中国石油大学(北京), 2016. http://cdmd.cnki.com.cn/Article/CDMD-11414-1018700792.htmLI Xiao-yang. The quantum chemical calculation of Ca(EDTA)2- complex and organic acid calcium compound[D]. Beijing: China University of Petroleum(Beijing), 2015. http://cdmd.cnki.com.cn/Article/CDMD-11414-1018700792.htm [24] 孟祥灿.胱氨酸盐型Gemini表面活性剂的合成及驱油性能研究[D].青岛: 中国石油大学(华东), 2013. http://cdmd.cnki.com.cn/Article/CDMD-10425-1015025005.htmMENG Xiang-can. Study on synthesis and oil displacement property of cystine Gemini surfactants[D]. Qingdao: China University of Petroeum(East China), 2013. http://cdmd.cnki.com.cn/Article/CDMD-10425-1015025005.htm [25] MAHMOUD M, ATTIA A, AL-HASHIN H. EDTA chelating agent/seawater solution as enhanced oil recovery fluid for sandstone reservoirs[J]. J Pet Sci Eng, 2017, 152:275-283 doi: 10.1016/j.petrol.2017.03.019 -





 下载:
下载: