Preparation and properties of Ni/KIT-6 catalysts modified with different metals for methanation of CO2
-
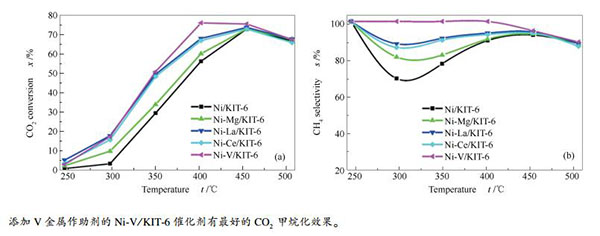
摘要: 采用共浸渍法分别制备了用Mg、Ce、V、La金属改性的Ni/KIT-6催化剂,用于CO2甲烷化反应的研究。利用N2吸附-脱附、X射线衍射、H2程序升温还原、H2程序升温脱附、透射电镜手段对催化剂进行了表征,考查了不同金属助剂对Ni/KIT-6的影响。结果表明,在KIT-6载体上活性金属和助剂的分散度都非常高,Ni粒子的分散度主要取决于KIT-6载体高度有序的介孔结构的限域作用,不受助剂金属添加的影响。各助剂金属的加入几乎不影响Ni/KIT-6催化剂的表面形貌,但对Ni金属还原的难易程度和还原度有影响。在研究的几种金属中,V金属使催化剂中Ni金属的还原最容易,还原度更高,且V金属的氧化物具有改变CO2反应机理的作用,使得甲烷化反应进行的最好。用V改性后的催化剂与未改性的催化剂相比,CO2的转化率提高了3.7%,CH4的选择性提高了11.6%,CH4的选择性达到了100%。Abstract: Ni/KIT-6 catalysts modified by Mg, Ce, V and La for CO2 methanation were prepared by co-impregnation method.The catalysts were characterized by N2 absorption-desorption, XRD, H2-TPD and TEM.The effects of different promoters on structure and properties of Ni/KIT-6 catalysts were investigated. The results show that the dispersity of Ni and promoters are very high. The dispersion of Ni particles depends primarily on the confinement effect of the well-ordered mesoporous structure of KIT-6, and is not affected by the addition of metal promoters. The addition of promoters does not affect the surface morphology of Ni/KIT-6 catalyst, but has effects on the difficulty and reduction degree of Ni reduction. Among the metal promoters studied, V addition makes the reduction of NiO in the catalyst easily and results in a high reduction degree. The oxide of V can change the reaction mechanism of CO2 methanation, resulting in the best methanation performance. Compared with the unmodified catalyst, the catalyst modified by V makes the conversion of CO2 and the selectivity of CH4 increase by 3.7% and 11.6% respectively. The selectivity of CH4 is 100%.
-
Key words:
- Ni catalyst /
- KIT-6 carrier /
- carbon dioxide /
- methanation
-
表 1 各催化剂的理化性质
Table 1 Physicochemical properties of the catalysts
Catalyst Specific surface area A/(m2·g-1) Pore volume v/(cm3·g-1) Average pore size d/nm Ni nanoparticle size d/nm H2 uptake /(μmol·g-1) Degree of dispersion /% KIT-6 724 1.08 5.85 - - - Ni/KIT-6 552 0.87 5.28 2.5 106.1 15.9 Ni-V/KIT-6 582 0.85 5.49 2.0 177.7 26.6 Ni-Ce/KIT-6 585 0.84 5.44 2.1 145.6 21.7 Ni-La/KIT-6 589 0.85 5.47 2.4 146.6 22.0 Ni-Mg/KIT-6 563 0.83 5.35 2.1 141.1 21.1 -
[1] QIU M, TAO, H L, LI Y, ZHANG Y F. Insight into the mechanism of CO2 and CO methanation over Cu(100) and Co-modified Cu(100) surfaces:A DFT study[J]. Appl Surf Sci, 2019, 495:143457. doi: 10.1016/j.apsusc.2019.07.199 [2] FALBO L, VISCONTI C G, LIETTI L, SZANYI J. The effect of CO on CO2 methanation over Ru/Al2O3 catalysts:A combined steady-state reactivity and transient DRIFT spectroscopy study[J]. Appl Catal B:Environ, 2019, 256. [3] CABRERO-ANTONINO M, REMIRO-BUENAMAÑANA S, SOUTO M, VALDIVIA A G, CHOQUESILLO-LAZARTE D, NAVALÓN S, RODRIGUEZ-DIÉGUEZ A, MINGUEZ ESPALLARGAS G, GARCÍA H. Design of cost-efficient and photocatalytically active Zn-based MOFs decorated with Cu2O nanoparticles for CO2 methanation[J]. Catal Commun, 2019, 55(73):10932-10935. https://www.researchgate.net/publication/335005724_Design_of_Cost-Efficient_and_Photocatalytically_Active_Zn-Based_MOFs_Decorated_with_Cu2O_Nanoparticles_for_CO2_Methanation [4] HUANG J, LI X, WANG X, FANG X ZH, WANG H M, XU X L.New insights into CO2 methanation mechanisms on Ni/MgO catalysts by DFT calculations:Elucidating Ni and MgO roles and support effects[J]. J CO2 Util, 2019, 33:55-63. doi: 10.1016/j.jcou.2019.04.022 [5] 张志磊.氧化锆负载镍催化剂的制备及其二氧化碳甲烷化反应性能研究[D].太原: 太原理工大学, 2019. http://cdmd.cnki.com.cn/Article/CDMD-10112-1019871788.htmZHANG Zhi-lei. Preparation and catalytic performance of nickel-based zirconia catalysts for carbon dioxide methanation[D]. Taiyuan: Taiyuan University of Technology, 2019. http://cdmd.cnki.com.cn/Article/CDMD-10112-1019871788.htm [6] 孙漪清, 金保昇, 董新新, 张文杰, 王金德. ZrO2-Al2O3复合载体负载Ni基催化剂COx甲烷化性能[J].化工进展, 2019, 38(7):3176-3184. http://d.old.wanfangdata.com.cn/Periodical/hgjz201907019SUN Yi-qing, JIN Bao-sheng, DONG Xin-xin, ZHANG Wen-jie, WANG Jin-de. Cox methanation over nickel-based catalysts supported on ZrO2-Al2O3 composite[J]. Chem Ind Eng Prog, 2019, 38(7):3176-3184. http://d.old.wanfangdata.com.cn/Periodical/hgjz201907019 [7] 韩阳.合成气甲烷化反应Ni基催化剂的研究[D].石河子: 石河子大学, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10759-1017729125.htmHAN Yang. Study on Ni-based catalyst for methanation of syngas[D]. Shihezi: Shihezi University, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10759-1017729125.htm [8] 路怀良. Ni-M(La, Ce, Fe, Co)/ZrO2-蒙脱土复合催化剂制备及其用于甲烷化反应的催化性能研究[D].呼和浩特: 内蒙古大学, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10126-1014267820.htmLU Huai-liang. Preparation of catalysts of Ni-Mn(La, Ce, Fe, Co)/ZrO2-montmorillonite nanocomposites and study on their methanation performance[D]. Hohhot: Inner Mongolia University, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10126-1014267820.htm [9] ZHI G J, GUO X N, WANG Y Y, JIN G Q. Effect of La2O3 modification on the catalytic performance of Ni/SiC for methanation of carbon dioxide[J]. Catal Commun, 2011, 16(1):56-59. doi: 10.1016/j.catcom.2011.08.037 [10] WANG W, CHU W, WANG N, YANG W, JIANG C F. Mesoporous nickel catalyst supported on multi-walled carbon nanotubes for carbon dioxide methanation[J]. Int J Hydrogen Energy, 2016, 41(2):967-975. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=16309f8cf92babb1f69325f0cb98d8ce [11] GUO M, LU G X. The effect of impregnation strategy on structural characters and CO2 methanation properties over MgO modified Ni/SiO2catalysts[J]. Catal Commun, 2014, 54:55-60. doi: 10.1016/j.catcom.2014.05.022 [12] LU X P, GU F N, LIU Q, GAO J J, LIU Y J, LI H F, JIA L H, ZHONG Z Y, SU F B. VOx promoted Ni catalysts supported on the modified bentonite for CO and CO2 methanation[J]. Fuel Process Technol, 2015, 135:34-46. doi: 10.1016/j.fuproc.2014.10.009 [13] 程凯, 白龙律, 朴文香. KIT-6介孔材料的研究进展[J].高分子通报, 2018, (7):58-68. http://d.old.wanfangdata.com.cn/Periodical/hljkjxx201718080CHENG Kai, BAI Long-lu, PIAO Wen-xiang. Research progress of mesoporous material KIT-6[J]. Chin Polym Bull, 2018, (7):58-68. http://d.old.wanfangdata.com.cn/Periodical/hljkjxx201718080 [14] 梅德均. Beta/KIT-6复合微/介孔分子筛的制备、改性及吸附CO2研究[D].桂林: 桂林理工大学, 2019. http://cdmd.cnki.com.cn/Article/CDMD-10596-1019627730.htmMEI De-jun. Preparation, modification and CO2 adsorption of Beta/KIT-6 micro/mesoporous composite molecular sieves[D]. Guilin: Guilin University of Technology, 2019. http://cdmd.cnki.com.cn/Article/CDMD-10596-1019627730.htm [15] 李白滔, 罗歆, 黄静, 王秀军, 梁振兴.含铜有序介孔Cu-KIT-6的合成及其苯乙烯环氧化催化性能:pH值的影响(英文)[J].催化学报, 2017, 38(3):518-528. http://d.old.wanfangdata.com.cn/Periodical/cuihuaxb201703012LI Bai-tao, LUO Xin, HUANG Jing, WANG Xiu-jun, LIANG Zhen-xing. One-pot synthesis of ordered mesoporous Cu-KIT-6 and its improved catalytic behavior for the epoxidation of styrene:Effects of the pH value of the initial gel[J]. Chin J Catal, 2017, 38(3):518-528. http://d.old.wanfangdata.com.cn/Periodical/cuihuaxb201703012 [16] 曹红霞. CO/CO2甲烷化催化剂的制备、表征及性能研究[D].徐州: 中国矿业大学, 2018.CAO Hong-xia. Study on preparation, Characterization and performance of catalyst for CO/CO2 methanation[D]. Xuzhou: China University of Mining and Technology, 2018. [17] 黄进.立方ZrO2、MgO负载Ni催化剂CO2甲烷化反应机理的理论与实验研究[D].南昌: 南昌大学, 2019.HUANG jin. CO2 methanation mechanismon cubic-ZrO2 and MgO supported Ni catalysts: A combined theoretical and experimental study[D]. Nanchang: Nanchang University, 2019. [18] LU B W, KAWAMOTO K. Direct synthesis of highly loaded and well-dispersed NiO/SBA-15 for producer gas conversion[J]. RSC Adv, 2012, 2(17):6800-6805. doi: 10.1039/c2ra20344h [19] QIU S B, ZHANG X, LIU Q Y, WANG T J, ZHANG Q, MA L L. A simple method to prepare highly active and dispersed Ni/MCM-41 catalysts by co-impregnation[J]. Catal Commun, 2013, 42(23):73-78. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=76144143874e277af52011edbadc05f4 [20] ZHANG Q, WANG T J, LI B, JIANG T, MA L L, ZHANG X H, LIU Q Y. Aqueous phase reforming of sorbitol to bio-gasoline over Ni/HZSM-5 catalysts[J]. Appl Energy, 2012, 97(3):509-513. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=f86e309e594b194e224bd9a491ac45eb -





 下载:
下载: