Preparation and thermal stability of C-doped zirconia tetragonal particles by the methanol-thermal method
-
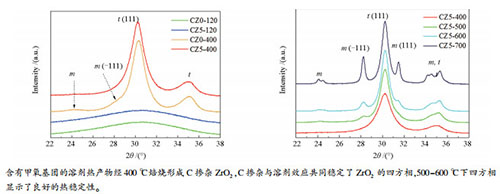
摘要: 在ZrO(NO3)2·2H2O-CO(NH2)2-CH3OH溶剂热过程中,水的缺乏使得甲醇通过其甲氧基与Zr4+发生亲核取代或以分子配位,直接参与锆盐的水解-缩聚反应,形成具有[ZrOz(OH)p(OCH3)q·rCH3OH]n结构的无机聚合物;同时,甲醇对聚合物低的溶解能力强烈抑制了Ostwald熟化过程,阻碍了溶剂热产物的晶化与热力学支持的结构重排。尿素通过其水解作用与锆盐竞争体系中的水及锆物种骨架上的羟基,这不仅导致无机聚合物中Zr-O-Zr键合相对Zr-OH键合的比例增加,使得溶剂热产物发生结构重排的几率进一步下降;而且也一定程度上增加了溶剂热产物中甲氧基的含量。含有大量甲氧基团的溶剂热产物经400 ℃焙烧后,形成C掺杂ZrO2。C掺杂与溶剂效应共同稳定了ZrO2的四方相。在500-600 ℃中等温度、空气气氛焙烧过程中,C掺杂ZrO2四方相结构显示了良好的热稳定性;提高焙烧温度至700 ℃,游离于颗粒表面的C被完全氧化去除,固溶于晶格中的C也部分脱溶,导致了部分四方相失稳转变成单斜相。Abstract: In the solvothermal process of ZrO(NO3)2·2H2O-CO(NH2)2-CH3OH system, methanol can act as both solvent and a reactant. Due to the lack of water, methanol is directly involved in the hydrolysis-condensation reaction of zirconium salt, through the nucleophilic substitution between its methoxy groups and Zr4+ as well as the coordination as a molecular state, to form inorganic polymers with[ZrOz(OH)p(OCH3)q·rCH3OH]n structure. At the same time, the low solubility of methanol to the polymers strongly inhibits the Ostwald ripening process, thus hindering the crystallization of solvothermal products and also reducing the probability of the thermodynamically supported structural rearrangement. Urea competes with zirconium salt for water in the system and the hydroxyl groups on the skeleton of zirconium species by its hydrolysis reaction, which not only leads to an increase in the amount of Zr-O-Zr bonds in polymers and then a further decrease in the probability of structural rearrangement of the solvothermal products, but also an increase in the content of methoxy group in solvothermal products. When calcined at 400℃, the solvothermal products containing a large amount of methoxy groups transformed into C-doped zirconia. Carbon doping, together with the solvent effect, stabilized the tetragonal phase of zirconia. The tetragonal phase in C-doped zirconia showed comparatively high thermal stability during calcination in air and at the medium temperature range of 500-600℃. Increasing the calcination temperature to 700℃, the free carbon species on the surface of particles was completely removed by oxidation, and the C dissolved in the lattice was also partially removed, resulting in some tetragonal phases lost stability and turned into monoclinic phases.
-
Key words:
- zirconia /
- carbon-doped zirconia /
- monoclinic phase /
- tetragonal phase /
- solvothermal synthesis
-
图 5 典型样品(初始溶液中尿素与锆盐物质的量比为5:1, 400 ℃焙烧)的XPS谱图
Figure 5 XPS analysis results of the typical sample (molar ratio of urea to zirconium in the initial solution of 5:1, calcined at 400 ℃)
(a)full range spectrum, (b), (c), (d) high-resolution spectra of the electron-binding energy peaks of C 1s, Zr 3d, O 1s
表 1 基于XRD分析计算的四方与单斜相ZrO2的体积分数及平均晶粒粒径
Table 1 Volume fraction and mean crystalline size data of the tetragonal and monoclinic ZrO2 derived from XRD analysis results
Sample CZ5-400 CZ5-500 CZ5-600 CZ5-700 CZ0-400 φt/% 100 93.5 89.0 71.8 94.8 φm/% 0 6.5 11.0 28.2 5.2 dt/nm 5.3 7.9 9.8 11.3 5.5 dm/nm - - 16.1 26.6 - 表 2 样品的元素定量分析与计算结果
Table 2 Mass fraction of the elements in the samples
Sample CZ5-120 CZ5-400 CZ5-700 CZ0-400 Mass fraction of C w/% 4.40 0.34 0.17 0.12 Mass fraction of H w/% 1.94 0.27 0.07 0.29 Mass fraction of N w/% 0.04 0.03 0.03 0.05 Mass fraction of S w/% 0.02 0.02 0.04 0.02 Mass fraction of Zr w/% 66.42 73.02 73.97 71.51 Mass fraction of O w/% 27.18 26.32 25.72 28.01 NC:NO:NZr 0.50:2.33:1 0.04:2.05:1 0.02:1.98:1 0.01:2.23:1 NC:NO:NZr is the atomic number ratio of C, O and Zr derived from the elemental analysis and TG analysis results 表 3 样品的XPS定量分析
Table 3 The quantitative analysis results of XPS
Sample CZ5-120 CZ5-400 CZ5-700 CZ0-400 Atom fraction of C w/% 56.36 45.76 33.32 31.87 Atom fraction of O w/% 36.41 42.36 49.60 53.18 Atom fraction of Zr w/% 7.23 11.88 17.08 14.94 Atom fraction of N w/% <1.0 <1.0 <1.0 <1.0 NC:NO:NZr 7.79:5.04:1 3.85:3.57:1 1.95:2.90:1 2.13:3.56:1 NC:NO:NZr is the atomic number ratio of C, O and Zr derived from XPS analysis results -
[1] JUNG K T, BELL A T. The effects of synthesis and pretreatment conditions on the bulk structure and surface properties of zirconia[J]. J Mol Catal A-Chem, 2000, 163:27-42. doi: 10.1016/S1381-1169(00)00397-6 [2] CHARY K V R, RAMESH K, NARESH D, RAO P V R, RAO R A, RAO V V. The effect of zirconia polymorphs on the structure and catalytic properties of V2O5/ZrO2 catalysts[J]. Catal Today, 2009, 141(1/2):187-194. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=bf86126d9411e862291753bc3c12ca55 [3] RUPPERT A M, NIEWIADOMSKI M, GRAMS J, KWAPIŃSKI W. Optimization of Ni/ZrO2 catalytic performance in thermochemical cellulose conversion for enhanced hydrogen production[J]. Appl Catal B:Environ, 2014, 145(1):85-90. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=0af078c48bfc0d7ddef75ede6669e2e0 [4] MIRANDA M C D, RAMÍREZ S A E, JURADO S G, VERA C R. Superficial effects and catalytic activity of ZrO2-SO42- as a function of the crystal structure[J]. J Mol Catal A-Chem, 2015, 398:325-335. doi: 10.1016/j.molcata.2014.12.015 [5] LI W Z, ZHAO Z K, JIAO Y H, WANG G R. Morphology effect of zirconia support on the catalytic performance of supported Ni catalysts for dry reforming of methane[J]. Chin J Catal, 2016, 37(12):2122-2133. doi: 10.1016/S1872-2067(16)62540-8 [6] FAN Y Q, CHENG S J, WANG H, TIAN J, XIE S H, PEI Y, QIAO M H, ZONG B N. Pt-WOx on monoclinic or tetrahedral ZrO2:Crystal phase effect of zirconia on glycerol hydrogenolysis to 1, 3-propanediol[J]. Appl Catal B:Environ, 2017, 217:331-341. doi: 10.1016/j.apcatb.2017.06.011 [7] HERNÁNDEZ S, GIONCO C, HUSAK T, CASTELLINO M, MUÑOZ-TABARES J A, TOLOD K, GIAMELLO E, PAGANINI M C, RUSSO N. Insights into the sunlight-driven water oxidation by Ce and Er-doped ZrO2[J]. Front Chem Sci Eng, 2018, 6:368. [8] LI W Z, HUANG H, LI H J, ZHANG W, LIU H C. Facile synthesis of pure monoclinic and tetragonal zirconia nanoparticles and their phase effects on the behavior of supported molybdena catalysts for methanol-selective oxidation[J]. Langmuir, 2008, 24:8358-8366. doi: 10.1021/la800370r [9] 李为臻, 刘海超.溶剂热法合成纯单斜和四方晶相氧化锆中的溶剂效应[J].物理化学学报, 2008, 24(12):2172-2178. doi: 10.3866/PKU.WHXB20081205LI Wei-zhen, LIU Hai-chao. Solvent effects on the solvothermal synthesis of pure monoclinic and tetragonal zirconia nanoparticles[J]. Acta Phy-Chim Sin, 2008, 24(12):2172-2178. doi: 10.3866/PKU.WHXB20081205 [10] 李亚伟, 田彩兰, 赵雷, 李远兵, 金胜利, 李淑静.碳包纳米氧化锆粉体的制备及其晶型转变[J].硅酸盐学报, 2009, 37(8):1273-1276. doi: 10.3321/j.issn:0454-5648.2009.08.001LI Ya-wei, TIAN Cai-lan, ZHAO Lei, LI Yuan-bing, JIN Sheng-li, LI Shu-jing. Preparation and phase transformation of nano-sized zirconia powder surrounded by carbon[J]. J Chin Ceram Soc, 2009, 37(8):1273-1276. doi: 10.3321/j.issn:0454-5648.2009.08.001 [11] 廖宁, 李亚伟, 桑绍柏, 金胜利, 李华军. ZrO2-C质耐火材料中碳对氧化锆的稳定作用[J].耐火材料, 2013, 47(5):329-333. doi: 10.3969/j.issn.1001-1935.2013.05.002LIAO Ning, LI Ya-wei, SANG Shao-bo, JIN Sheng-li, LI Hua-jun. Stabilization of carbon to zirconia in ZrO2-C refractories[J]. Refractories, 2013, 47(5):329-333. doi: 10.3969/j.issn.1001-1935.2013.05.002 [12] 张国芳, 翟亭亭, 侯忠辉, 许剑轶, 武悦, 葛启录.纳米CeO2-xNx固溶体的光谱特征对其催化性能影响研究[J].光谱学与光谱分析, 2018, 38(10):3192-3198. http://d.old.wanfangdata.com.cn/Periodical/gpxygpfx201810036ZHANG Guo-fang, ZHAI Ting-ting, HOU Zhong-hui, XU Jian-yi, WU Yue, GE Qi-lu. Research on the influence of spectrum characteristics on the catalysis effect of nanosized CeO2-xNx solid solutions[J]. Spectrosc Spect Anal, 2018, 38(10):3192-3198. http://d.old.wanfangdata.com.cn/Periodical/gpxygpfx201810036 [13] FOCA N, LISA G, RUSU I. Synthesis and characterization of some Cr(Ⅲ), Fe(Ⅲ) and Zr(Ⅳ) compounds with substituted o-hydroxy benzophenone[J]. J Therm Anal Calorim, 2004, 78:239-249. doi: 10.1023/B:JTAN.0000042171.81071.e3 [14] 邱羽, 高濂.由硝酸盐尿素配合物前驱体制备过渡金属氮化物粉体的研究[J].无机材料学报, 2004, 19(1):63-68. doi: 10.3321/j.issn:1000-324X.2004.01.011QIU Yu, GAO Lian. Synthesis of transition metal nitride powders from metal-urea nitratecomplex precursors[J]. J Inorg Mater, 2004, 19(1):63-68. doi: 10.3321/j.issn:1000-324X.2004.01.011 [15] PRIOR T J, KIFT R L. Synthesis and crystal structures of two metal urea nitrates[J]. J Chem Crystallogr, 2009, 39(8):558-563. doi: 10.1007/s10870-009-9517-0 -





 下载:
下载: