Hydrogenation of coal tar on NiMoP/γ-Al2O3 catalyst
-
摘要: 采用分步浸渍法制备了一系列NiMoP/γ-Al2O3催化剂, 并用电感耦合等离子体光谱、N2吸附脱附、透射电镜、H2程序升温还原等技术对NiMoP/γ-Al2O3催化剂进行了表征。在固定床反应器中, 进行了模型化合物的加氢实验, 确定了催化剂活性组分Ni的最佳含量(4%, 质量分数), 将最佳Ni含量的催化剂用于实际的煤焦油加氢研究, 并对比分析了酚油的切除对加氢效果的影响, 结果表明, 酚油的切除可以促进S、N原子的脱除以及芳烃的饱和。Abstract: The NiMoP/γ-Al2O3 catalyst was prepared through a multi-step impregnation method and characterized by inductively coupled plasma mass spectrometry (ICP-MS), N2 adsorption-desorption, transmission electron microscope (TEM) and H2 temperature programmed reduction (H2-TPR). The catalyst with 4%Ni is determined to be the best one with optimum component proportion by the tests with model compounds in a fixed-bed reactor. The coal tar hydrogenation was conducted over the optimum catalyst. The results reveal that the removal of carbolic oil can promote the removal of S and N and the saturation of aromatics.
-
Key words:
- coal tar /
- hydrogenation /
- carbolic oil
-
表 1 NiMoP/γ-Al2O3催化剂的活性组分含量
Table 1 Content of active component in NiMoP/γ-Al2O3 catalysts
Catalyst Theoretical results w/% Test results w/% Ni Mo P Ni Mo P HT1 1 10 1.5 1.01 9.71 1.65 HT2 2 10 1.5 1.95 9.56 1.46 HT3 4 10 1.5 3.99 9.65 1.54 HT4 6 10 1.5 5.87 9.92 1.63 表 2 不同Ni含量的NiMoP/γ-Al2O3催化剂的物理特性
Table 2 Physical properties of NiMoP/γ-Al2O3 catalyst with different Ni content
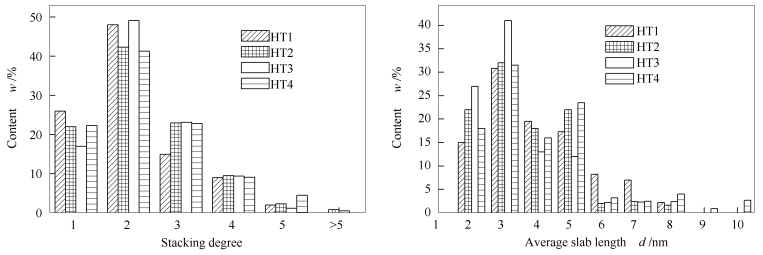
Catalyst BET surface area A/(m2·g-1) Pore volume v/(cm3·g-1) Average pore diameter d/nm γ-Al2O3 283.30 0.49 19.15 HT1 145.07 0.24 3.83 HT2 146.31 0.25 3.84 HT3 147.48 0.27 3.82 HT4 143.54 0.21 3.84 表 3 不同Ni含量的NiMoP/γ-Al2O3催化剂中MoS2平均片层长度与堆叠层数
Table 3 Average slab length and stacking degree of MoS2 in NiMoP/γ-Al2O3 catalyst with different Ni content
Catalyst Average slab length d/nm Average stacking degree HT1 4.02 2.11 HT2 3.61 2.28 HT3 3.45 2.28 HT4 4.00 2.27 表 4 煤焦油馏分油模型化合物的组成
Table 4 Model compound of distillate oil
Model compound Content w/% Type Dibenzothiophene 1 S-containing compound Quinoline 5 N-containing compound o-xylene 10 mononuclear aromatic 1-methylnaphthalene 25 diaromatics Phenanthrene 10 triaromatics Hexadecane 10 long chain hydrocarbon Hexane 39 chain hydrocarbon 表 5 1-甲基萘的加氢产物物质的量比
Table 5 Molar ratio of hydrogenated products of 1-methylnaphthalene
Catalyst Methyl tetralin/methyl decalin HT1 8.05 HT2 1.50 HT3 1.67 HT4 2.30 表 6 馏分油加氢前后物质类别分布
Type Distillate oil
(phenol oil excision)Distillate oil Hydrogenation products
(phenol oil excision)Hydrogenation product Alkene 1.02 0.87 0.00 0.00 Alkane 4.79 4.09 13.98 11.46 Cycloalkane 0.00 0.00 10.66 9.40 Bicyclic alkane 3.91 3.33 2.76 2.30 Cycloolefine 0.00 0.00 0.11 0.23 Alkylbenzene 3.91 3.33 13.00 13.25 Hydrogenated aromatic 8.33 7.11 43.54 43.52 Other di-aromatics 9.65 8.23 1.38 1.96 Alkylnaphthalene 45.09 38.49 4.57 6.98 Biphenyl 6.05 5.17 2.89 4.92 Triaromatic 1.85 1.58 1.56 1.50 Oxygen containing compound 8.48 7.24 5.57 4.48 Phenol 6.97 20.59 0.00 0.00 表 7 馏分油以及加氢产物的S、N含量
Table 7 Content of S and N in distillate oil and hydrogenation products
Distillate oil
(phenol oil excision)Distillate oil Hydrogenation products
(phenol oil excision)Hydrogenation product S/10-6 12 150.00 12 000.00 76.18 220.41 N/10-6 4 300.00 4 900.00 5.00 21.13 -
[1] BALSTER L M, CORPORAN E, DEWITT M J. Development of an advanced, thermally stable, coal-based jet fuel[J]. Fuel Process Technol, 2008, 89(4): 364-378. doi: 10.1016/j.fuproc.2007.11.018 [2] 燕京, 吕才山, 刘爱华, 达建文.高温煤焦油加氢制取汽油和柴油[J].石油化工, 2006, 35(1): 33-36. http://www.cnki.com.cn/Article/CJFDTOTAL-SYHG200601008.htmYAN Jing, LÜ Cai-shan, LIU Ai-hua, DA Jian-wen. Production of gsoline and desel ol by hdrogenation of hgh tmperature cal tar[J]. Petrochem Technol, 2006, 35(1): 33-36. http://www.cnki.com.cn/Article/CJFDTOTAL-SYHG200601008.htm [3] 潘海涛, 高歌, 雷振, 陆江银. Co-Mo/γ-Al2O3催化剂上煤焦油加氢裂化工艺[J].石油化工, 2014, 43(5): 517-522. http://www.cqvip.com/qk/92242x/20145/49666146.htmlPAN Hai-tao, GAO Ge, LEI Zhen, LU Jiang-yin. Hydrocracking pocess of cal tar over Co-Mo/γ-Al2O3 catalysts[J]. Petrochem Technol, 2014, 43(5): 517-522. http://www.cqvip.com/qk/92242x/20145/49666146.html [4] 李大东.加氢处理工艺与流程[M].北京:中国石化出版社. 2004.LI Da-dong. Hydroteating Technology and Engineering[M]. Beijing: China Petrochemical Press, 2004. [5] 周同娜, 尹海亮, 韩妹娜, 柴永明, 柳云骐, 刘晨光.不同磷含量对NiMoP/Al2O3加氢处理催化剂的影响[J].燃料化学学报, 2009, 37(3): 330-334. doi: 10.1016/S1872-5813(09)60023-2ZHOU Tong-na, YIN Hai-liang, HAN Mei-na, CHAI Yong-ming, LIU Yun-qi, LIU Chen-guang. Influences of different phosphorus contents on NiMoP/Al2O3 hydrotreating catalysts[J]. J Fuel Chem Technol, 2009, 37(3): 330-334. doi: 10.1016/S1872-5813(09)60023-2 [6] 石垒, 张增辉, 邱泽刚, 郭芳, 张伟, 赵亮富. P改性对Mo-Ni/Al2O3煤焦油加氢脱氮性能的影响[J].燃料化学学报, 2015, 43(1): 74-80. doi: 10.1016/S1872-5813(15)60007-XSHI Lei, ZHANG Zeng-hui, QIU Ze-gang, GUO Fang, ZHANG Wei, ZHAO Liang-fu. Effect of phosphorus modification on the catalytic properties of Mo-Ni/Al2O3 in the hydrodenitrogenation of coal tar[J]. J Fuel Chem Technol, 2015, 43(1): 74-80. doi: 10.1016/S1872-5813(15)60007-X [7] FERDOUS D, DALAI A K, ADJAYE J. A series of NiMo/Al2O3 catalysts containing boron and phosphorus: Part Ⅰ. Synthesis and characterization[J]. Appl Catal A: Gen, 2004, 260(2): 137-151. doi: 10.1016/j.apcata.2003.10.010 [8] 王永刚, 张海永, 张培忠, 许德平, 赵宽, 王芳杰. NiW/γ-Al2O3催化剂的低温煤焦油加氢性能研究[J].燃料化学学报, 2012, 40(12): 1492-1497. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18088.shtmlWANG Yong-gang, ZHANG Hai-yong, ZHANG Pei-zhong, XU De-ping, ZHAO Kuan, WANG Fang-jie. Hydroprocessing of low temperature coal tar on NiW/γ-Al2O3 catalyst[J]. J Fuel Chem Technol, 2012, 40(12): 1492-1497. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18088.shtml [9] 范建锋, 张忠清, 姚春雷, 全辉.中温煤焦油加氢生产清洁燃料油试验研究[J].煤炭学报, 2013, 38(10): 1868-1872. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201310029.htmFAN Jian-feng, ZHANG Zhong-qing, YAO Chun-lei, QUAN Hui. Study on hydrogenation of medium temperature coal tar to clean fuel[J]. J Chin Coal Soc, 2013, 38(10): 1868-1872. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201310029.htm [10] 战风涛, 吕志凤, 王洛秋, 苏贻勋, 阙国和.催化柴油中的酚类化合物及其对柴油安定性的影响[J].燃料化学学报, 2000, 28(1): 59-62. http://www.cnki.com.cn/Article/CJFDTOTAL-RLHX200001013.htmZHAN Feng-tao, LÜ Zhi-feng, WANG Luo-qiu, SU Yi-xun, QUE Guo-he. Identification of phenolic compounds in LCO and their effects on stability of diesels[J]. J Fuel Chem Technol, 2000, 28(1): 59-62. http://www.cnki.com.cn/Article/CJFDTOTAL-RLHX200001013.htm [11] 吴世逵, 梁朝林, 黄克明, 胡智华, 谢涛.催化裂化汽油组成对其储存安定性的影响[J].石油学报, 2008, 4: 478-483. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG200804024.htmWU Shi-kui, LIANG Cao-lin, HUANG Ke-ming, HU Zhi-hua, XIE Tao. Effects of components of FCC gasoline on its storage stability[J]. Acta Pet Sin, 2008, 4: 478-483. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG200804024.htm [12] PHILIPPE M, RICHARD F, HUDEBINE D, BRUNET S. Inhibiting effect of oxygenated model compounds on the HDS of dibenzothiophenes over CoMoP/Al2O3 catalyst[J]. Appl Catal A: Gen, 2010, 383(1): 14-23. http://www.sciencedirect.com/science/article/pii/S0926860X10003285 [13] 夏良燕, 夏芝香, 方梦祥, 唐巍, 王勤辉, 骆仲泱.煤焦油中芳烃(萘) 的加氢饱和试验[J].浙江大学学报(工学版), 2015, 49(3): 578-584. http://www.cnki.com.cn/Article/CJFDTOTAL-ZDZC201503025.htmXIA Liang-yan, XIA Zhi-xiang, FANG Meng-xiang, TANG Wei, WANG Qin-hui, LUO Zhong-yang. Hydrogenation saturation of aromatic compounds (naphthalene) in coal tar[J]. J Zhejiang Univ (Eng Sci), 2015, 49(3): 578-584. http://www.cnki.com.cn/Article/CJFDTOTAL-ZDZC201503025.htm [14] WANDAS R, SURYGALA J, OLLIWKA E. Conversion of cresols and naphthalene in the hydroprocessing of three-component model mixtures simulating fast pyrolysis tars[J]. Fuel, 1996, 75(6): 687-694. doi: 10.1016/0016-2361(96)00011-7 [15] SALERNO P, MENDIOROZ S, AGUDO A L. Al-pillared montmorillonite-based NiMo catalysts for HDS and HDN of gas oil: Influence of the method and order of Mo and Ni impregnation[J]. Appl Catal A: Gen, 2004, 259(1): 17-28. doi: 10.1016/j.apcata.2003.09.019 [16] FERDOUS D, DALAI A K, ADJAYE J. A series of NiMo/Al2O3 catalysts containing boron and phosphorus: Part Ⅰ. Synthesis and characterization[J]. Appl Catal A: Gen, 2004, 260(2): 137-151. doi: 10.1016/j.apcata.2003.10.010 [17] FERDOUS D, DALAI A K, ADJAYE J. A series of NiMo/Al2O3 catalysts containing boron and phosphorus: Part Ⅱ. Hydrodenitrogenation and hydrodesulfurization using heavy gas oil derived from Athabasca bitumen[J]. Appl Catal A: Gen, 2004, 260(2): 153-162. doi: 10.1016/j.apcata.2003.10.009 [18] KNUDSEN K G, COOPER B H, TOPSØE H. Catalyst and process technologies for ultra low sulfur diesel[J]. Appl Catal A: Gen, 1999, 189(2): 205-215. doi: 10.1016/S0926-860X(99)00277-X [19] JIAN M, PRINS R. Mechanism of the hydrodenitrogenation of quinoline over NiMo (P)/Al2O3 catalysts[J]. Ind Eng Chem Res, 1998, 179(1): 18-27. [20] LU M, WANG A, LI X, DUAN X P, TENG Y, WANG Y, SONG C S, HU Y K. Hydrodenitrogenation of quinoline catalyzed by MCM-41-supported nickel phosphides[J]. Energy Fuels, 2007, 21(2): 554-560. doi: 10.1021/ef060467g [21] ZUO D, LI D, NIE H, SHI Y, LACROIX M, VRINAT M. Acid-base properties of NiW/Al2O3 sulfided catalysts: Relationship with hydrogenation, isomerization and hydrodesulfurization reactions[J]. J Mol Catal A: Chem, 2004, 211(1/2): 179-189. http://www.sciencedirect.com/science/article/pii/S1381116903007301 [22] MIKI Y, SUGIMOTO Y. Hydrocracking of polycyclic aromatic compounds. 1. Methylnaphthalenes[J]. Fuel Process Technol, 1995, 43(2): 137-146. doi: 10.1016/0378-3820(95)00005-R [23] 李会峰, 刘锋, 刘泽龙, 李明丰, 聂红.菲在不同加氢催化剂上的转化[J].石油学报, 2011, 27(1): 20-25. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG201101007.htmLI Hui-feng, LIU Feng, LIU Ze-long, LI Ming-feng, NIE Hong. Hydrogenation of phenanthrene over different catalysts[J]. Acta Pet Sin, 2011, 27(1): 20-25. http://www.cnki.com.cn/Article/CJFDTOTAL-SXJG201101007.htm [24] COOPER B H, DONNIS B. Aromatic saturation of distillates: An overview[J]. Appl Catal A: Gen, 1996, 137(2): 203-223. doi: 10.1016/0926-860X(95)00258-8 -





 下载:
下载: